Abstract
Multifocal submucosal stromal tumors were diagnosed in a 5.5-year-old rhesus macaque (Macaca mulatta) experimentally infected with simian immunodeficiency virus, strain SIVsmE660, and CD4+ T cell depleted. The animal was negative for simian retroviruses, SRV-1, -2, and -5. Polymerase chain reaction analysis of DNA from tumor and spleen tissue revealed abundant, preferential presence of retroperitoneal fibromatosis herpesvirus, the macaque homologue of the Kaposi sarcoma-associated herpesvirus (human herpesvirus-8), in the tumors. This was corroborated by demonstration of viral latent nuclear antigen-1 in the nuclei of a majority of the spindeloid tumor cells. Low levels of an additional macaque herpesvirus, rhesus rhadinovirus, were also detected in the spleen and tumor tissues. The spindeloid cells labeled positively for vimentin and CD117 but were negative for CD31, CD68, desmin, and smooth muscle cell actin. Collectively, these findings suggest a relation to but not absolute identity with simian mesenchymoproliferative disorders (MPD) or typical gastrointestinal stromal tumors (GISTs).
Keywords
Gastrointestinal tumors are, as are neoplasms in general, comparatively infrequent in non-human primates, perhaps in large part because of the relatively young age of most non-human primates used in research. 15 Intra-abdominal lesions with a spindle morphology, including leiomyomas, leiomyosarcomas, gastrointestinal stromal tumors (GISTs), inflammatory myofibroblastic tumors (IMFT) or inflammatory pseudotumors, mesenteric spindle cell–reactive lesions, peripheral nerve sheath tumors, and fibromatosis, are relatively uncommon in humans and constitute a differential diagnostic challenge. 1, 8, 25 Intra-abdominal fibromatosis is an uncommon tumor that primarily affects the mesentery or retroperitoneum but may encroach on, extend into, or even arise from the intestinal wall, mimicking a primary gastrointestinal neoplasm. 25 Retroperitoneal fibromatosis (RF) is one of several mesenchymoproliferative disorders (MPD) that have been described in macaques with simian-acquired immunodeficiency syndrome (SAIDS) associated with simian retrovirus type-D (SRV-2) infection. 10, 22 On the basis of morphological criteria, RF and the subcutaneous form (SF) were previously compared with Kaposi sarcoma (KS), 10 a multifocal proliferative lesion of supposed vascular origin, 2 and this contention was supported by the demonstration of the macaque herpesvirus homologue of KS-associated herpesvirus (human herpesvirus-8 [HHV-8]) and retroperitoneal fibromatosis herpesviruses (RFHV) in RF lesions of two macaque species (Macaca mulatta and M. nemestrina). 16 Subsequently, an additional herpesvirus, rhesus rhadinovirus (RRV), was identified in macaques and shown to be related to HHV-8 and RFHV. 4
It has been proposed that SAIDS-associated RF in macaques could constitute an appropriate animal model for acquired immunodeficiency syndrome–associated KS in humans. 16 However, although RF and other MPD have been identified in SAIDS cases resulting from SRV-2 infection, they have not been previously reported in animals with lentivirus-induced SAIDS. In this study, we describe a case of colonic submucosal stromal tumors, with similarities to macaque MPD, in an SRV-2–negative, simian immunodeficiency virus (SIV)–infected, and CD4+ T cell–depleted rhesus macaque and provide evidence for a latent infection of the tumor cells with RFHV.
A young, healthy rhesus macaque (M. mulatta) was experimentally infected with SIVsmE660 in July of 1999 (study approved by the Harvard University and University of Washington Animal Care and Use Committees). It remained clinically healthy until November 2001 despite high plasma viral load (6–24 × 104 copies/ml), as determined by quantitative real-time reverse transcriptase–polymerase chain reaction (PCR), 11 and slowly declining peripheral blood CD4+ T-lymphocyte counts (down to 100 cells/ml in the last month before euthanasia), when an acute, severe prolapse of the rectum occurred. On physical examination, the animal was noted to be slightly dehydrated and, on palpation of the abdomen, several masses, localized to the lower abdomen, were diagnosed. In addition, the inguinal and axillary lymph nodes were enlarged. During the next 2 days, clinical intervention was attempted, but the prolapse recurred repeatedly, and the rectal mucosa on the prolapse was by then noted to be necrotic. Euthanasia was elected, and necropsy was immediately performed. Gross examination showed generalized enlargement of lymph nodes, both peripheral and visceral, as well as splenomegaly. The distal colon was intussuscepted and the rectal prolapse still patent with most of the rectal mucosa severely necrohemorrhagic. Multifocally in the submucosa of the distal colon and rectum were five large (up to 2 × 2 × 2.5 cm), well-circumscribed, firm masses that were uniformly tan to white in color. The only other abnormality found at necropsy was a large fibrin thrombus on the mitral valve.
Tissues were fixed in 10% neutral-buffered formaldehyde and routinely processed for histopathologic study. All the five tumors had similar light microscopic appearance, presenting as large nonencapsulated, but well demarcated by lamina muscularis mucosa and muscularis propria, expansively growing masses. The neoplasms were variably, although mostly densely cellular and composed of bundles of spindeloid cells with indistinct cell borders, moderate to abundant eosinophilic cytoplasm, and round to ovoid large nuclei with vesiculated chromatin and usually one large, acentric, magenta nucleolus. Mitoses were infrequent (< 1/20 hpf) and mainly seen in the periphery of the neoplasms, i.e., the advancing front. There were variable, in the larger masses usually abundant if not dominant, bundles of mature collagen separating the neoplastic cells. Vascularization was sparse to moderate (Fig. 1A–D). Multifocally, within the masses were clusters of epitheloid macrophages with abundant foamy cytoplasm; and diffusely, throughout the masses were large numbers of infiltrating eosinophilic granulocytes, lymphocytes, macrophages, fewer neutrophilic granulocytes (except in areas of necrosis of overlaying mucosa, in which case they were abundant), and rare plasma cells. No acid-fast bacterial rods were detected with Fite-Faraco stain. The mucosa overlying the smaller masses from the distal colon appeared within the normal range or with moderate crypt hyperplasia, whereas there was extensive necrosis of the overlaying mucosa, extending into the neoplastic masses, in those from the colorectal border and involved in the rectal prolapse. Many large vessels in the submucosa surrounding the masses had mild to moderately thickened intima because of the proliferation of subendothelial stromal cells and deposition of extracellular matrix. In the context of the two larger masses, the serosa was markedly thickened, myxomatous, and with pronounced vascular proliferation, in part involving the capsule of the retroperitoneal lymph nodes. Other histopathologic changes observed included follicular and paracortical hyperplasia of all peripheral and visceral lymph nodes and thrombotic endocardiopathy.
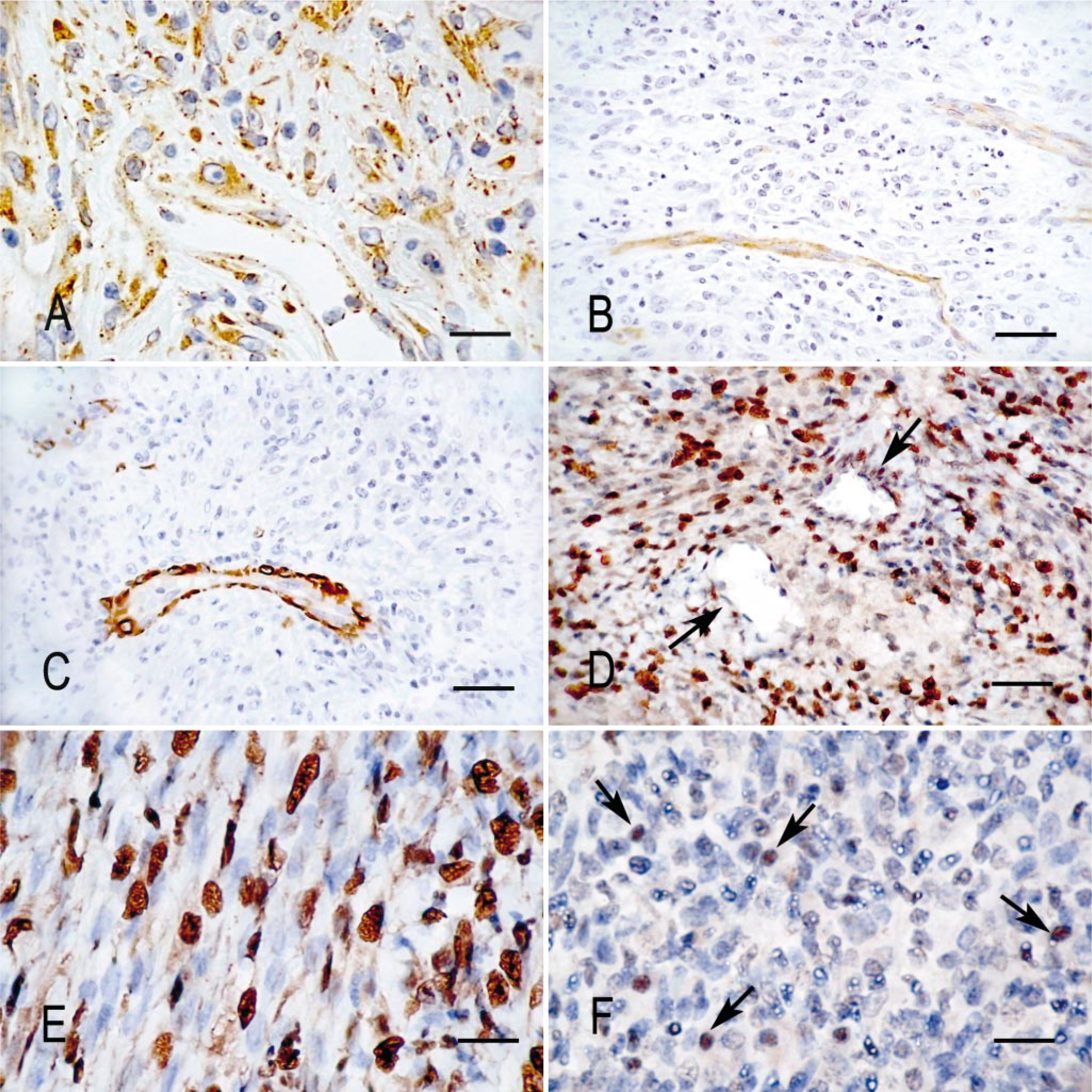
Intestinal stromal tumors and ileocecal lymph node; rhesus macaque. Immunohistochemical labeling for cluster determinants and viral antigen.
For immunophenotyping of tumor cells and infiltrating leukocytes and immunolabeling for viral antigens, paraffin sections of the formaldehyde-fixed tissues were deparaffinized, subjected to antigen retrieval, and reacted with antibodies, as described. 3 The following mono- and polyclonal antibodies were used: rat monoclonal anti–HHV-8 latent nuclear antigen-1 (LANA-1), clone LN 53 (ABI Advanced Biotechnologies, Columbia, MD); mouse monoclonal anti–HHV-8 ORF59, clone 11D1; 24 mouse monoclonal anti–HHV-8 K8.1A/B, clone 4A4; 24 rabbit anti–SRV-2; 13 mouse monoclonal antibodies to HLA-DR, CD117, CD31, CD34, CD68, and rabbit polyclonal antibodies to vimentin, desmin, smooth muscle actin, cytokeratin, and S-100 (all from DakoCytomation, Carpinteria, CA). The neoplastic spindeloid cells were diffusely and intensely positive for vimentin and positive for CD117 (c-kit), with the labeling often being granular or distinctly membrane associated (Fig. 1A). The immunolabeling for desmin (Fig. 1C) and smooth muscle cell actin (not shown) were negative in the neoplastic cells, whereas vascular smooth muscle cells labeled intensely for both antigens. Vascular endothelial cells were positive for CD31, whereas the neoplastic cells were negative (Fig. 1B). Infiltrating macrophages were CD68 positive but expressed no or only low levels of major histocompatibility complex class II, and the spindeloid tumor cells were negative for both these markers (data not shown). The spindeloid tumor cells were also negative for cytokeratin and S-100. None of the antibodies to human CD34 tested appeared to cross-react with macaque cells in fixed tissues. Although no reaction was observed with antibodies to the HHV-8 ORF59 lytic nuclear protein or to the K8.1 virion-associated glycoprotein, a very intense reaction was detected in the nuclei of the spindeloid tumor cells, using a rat monoclonal antibody raised against the HHV-8 LANA-1, ORF73 (LANA-1) (Fig. 1D, E). This reactivity was similar to that detected for LANA-1 in human KS lesions 6 (data not shown) and in archival macaque RF and SF lesions (H. Bielefeldt-Ohmann, unpublished). A weaker signal was observed in scattered lymphocytes in the B-cell areas of spleen and lymph nodes (Fig. 1F). All other tissues and cell types were negative for LANA-1.
The animal was tested for simian T cell lymphotropic virus type-1–specific antibodies by enzyme-linked immunosorbent assay (ELISA) and for SRV-2 infection by ELISA (serum antibodies), Raji cell coculture, and PCR assays (virus in peripheral blood), as part of the routine WaNPRC infection status monitoring program and was negative for both viruses in these assays. DNA was also extracted from formaldehyde-fixed, paraffin-embedded sections of the sub-mucosal tumors and tested by heminested PCR for SRV-1, -2 and -5 5 and from fresh-frozen spleen and tested for SRV-2, all with negative results (data not shown). Furthermore, no reactivity was observed in the tumors when tested for the presence of SRV-2 antigens by immunohistochemistry (data not shown), corroborating the serological and virological assays.
The presence of RRV and RFHV in DNA samples prepared from fresh-frozen spleen or paraffin-embedded, formaldehyde-fixed tumor tissues from the animal was determined by semiquantitative PCR. For each sample, the level of RFHV and RRV DNA and a reference single copy genomic gene, oncostatin M (OSM), was determined by PCR. The rhadinovirus type 1 (RV-1) assay, targeting the RFHV genome, amplified a 370-bp amplicon from ORF8 (glycoprotein B) using RFHV-Gb-a (5′-TTAAAGACATCTACGC-CCTCC-3′) to RFHV-Gb-b (5′-GCAGTATGCCCTCGTG-GTGAG-3′). The rhadinovirus type 2 (RV-2) assay, targeting the RRV genome, amplified a 407-bp amplicon from the ORF59/60 locus using RDELa (5′-CTTGCCAACGATTAC-ATTTCCAGGGACGAGCT-3′) to N/R-59-b (5′-GGCCCG-GAAAATGAGTAACA-3′). The OSM assay amplified a 401-bp amplicon from exon 3 of macaque OSM using TLGCa (5′-GTGCAGGACGCAGCCCAGTGT-3′) to KGVRb (5′-GTCCTGCGCACCCCCTTC-3′). Reactions (50 μl) contained approximately 100 ng of template DNA, 1 μM forward and reverse primers, 200 μM each deoxynucleoside triphosphates, 20 mM Tris-HCl (pH 8.4), 50 mM KCl, 2.0 mM MgCl,2 and 2.5 U of Platinum Taq polymerase (Invitrogen, Carlsbad, CA). For the RV-1 assay, after activation of the polymerase by incubation for 1 minute at 95° C, reactions were run for 35 cycles of 95° C for 30 seconds, 69° C for 30 seconds, and 72° C for 30 seconds. The RV-2 and OSM reactions were run with the same protocol as the RV-1 reactions except that the annealing temperatures were 60° C and 68° C, respectively. In the spleen, a distinct amplification product for RRV (Fig. 2, lane B) was obtained at approximately 10% of the intensity of the OSM genomic DNA control (Fig. 2, lane A), whereas only a very weak amplification product was obtained with the RFHV assay (Fig. 2, lane C). In contrast, in the tumor, a prominent amplification product was observed with the RFHV assay (Fig. 2, lane F), which was approximately equivalent in intensity to that of OSM genomic control (Fig. 2, lane D). Only a faint amplification product was obtained with the RRV assay in the tumor sample (Fig. 2, lane E), but the ratio of RRV to OSM remained the same as that detected in the spleen sample, i.e., approximately 10%.
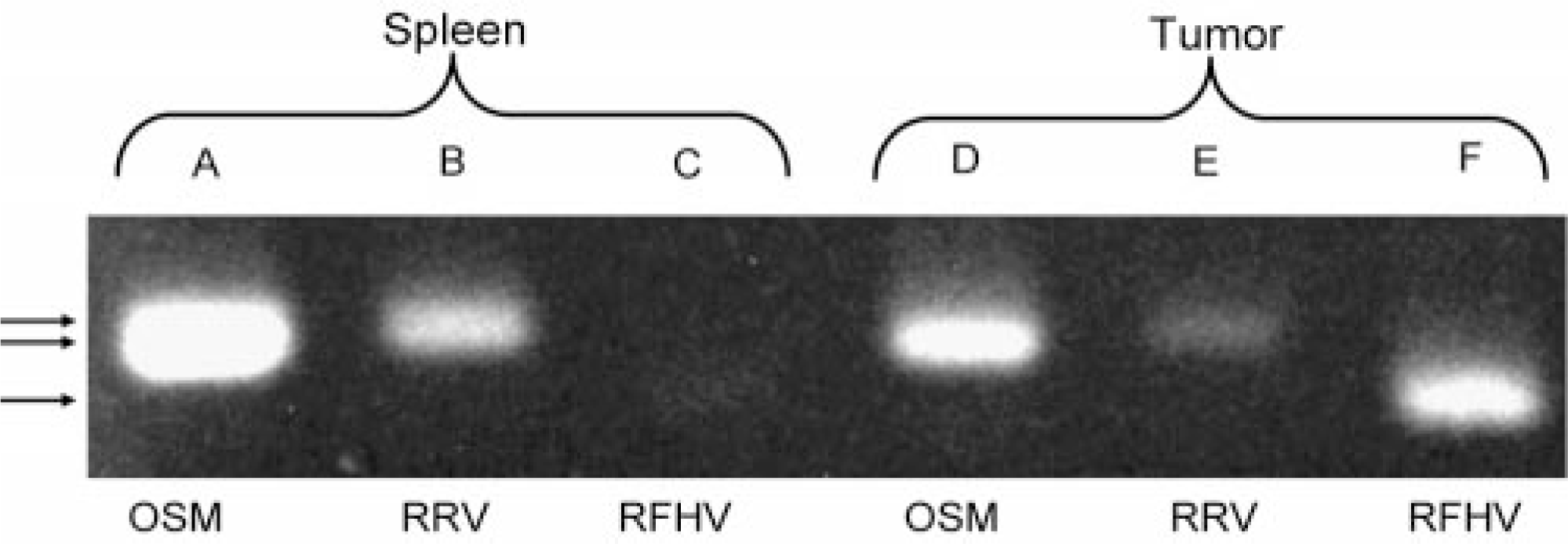
PCR amplification of ORF8 (370 bp) of RFHV (lanes C and F) and ORF59/60 (407 bp) of RRV (lanes B and E) in submucosal mesenchymal tumor and in spleen from an SRV-2–negative, SIV-positive rhesus macaque. As internal comparison, a 401-bp fragment of the single copy genomic gene (lanes A and D) was amplified from the same samples.
Our results thus demonstrate selectively increased levels of RFHV in the intestinal stromal tumors of this rhesus macaque when compared with spleen and other tissues, as assessed by RFHV-specific PCR and immunohistochemistry. The antibody used in the latter test is specific for the repetitive protein domain of HHV-8 LANA-1, 7 which does not occur in RRV LANA-1, 18 and thus the strong nuclear signal detected is consistent with the preferential presence of RFHV. In contrast to the disparate RFHV levels in spleen and tumors, the levels of RRV in these two tissues were equivalent when compared with the cellular OSM control. These results may suggest an etiologic role, either directly or through the ensuing inflammation, for RFHV in the histiogenesis of the stromal tumors, similar to what has been surmised for RFHV in MPD, 16 of HHV-8 in KS 2, 6 and of Epstein-Barr virus and HHV-8 in, at least some cases of, IMFT and inflammatory fibroid polyps. 9
In non-human primates, RF is one of several MPD that histomorphologically bears superficial resemblance to KS, 10, 22 as well as to a range of other soft tissue neoplasms in humans, characterized by spindeloid cell morphology, notably GISTs, IMFT, inflammatory fibroid polyps, and fibrosarcoma. 1, 8, 25 However, the progenitor cell type or the types for macaque MPD remain unknown, as does the pathogenesis. The spindle cells in IFMT consistently label positively for vimentin, smooth muscle cell actin, and CD68, 9 whereas the spindeloid cells in the present macaque tumors and in archival MPD lesions are negative for the latter two markers (Fig. 1, and H. Bielefeldt-Ohmann unpublished). On the basis of immunophenotyping of cells cultured from RF lesions, it was previously suggested that the progenitor cell for macaque MDP is a multipotent myofibroblast, 23 and not an endothelial cell, the purported progenitor cell of KS. 2, 6 Contrary to that earlier report, we found that in this case the neoplastic cells in situ were negative for desmin, a marker of myogenic origin. This raises several, not mutually exclusive, possibilities, including 1) the previously described in vitro–cultured RF cells were not the actual tumor cells but overgrowth of the cultures by interstitial cells of the myofibroblast lineage, 2) the tumor cells had differentiated further during in vitro passage, 19 3) morphologically defined MPD in macaques represents more than one entity with several different progenitor cells, possibly even different types of myofibroblasts, 19 or 4) the particular microenvironment in which the neoplastic outgrowth of mesenchymal, pluripotent progenitor cells occurs, with its specific set of growth factors and inflammatory cytokines, may determine the expression of phenotypic markers. 9, 19 It is also possible that rhadinovirus infections may modulate host cell gene expression, as happens with HHV-8. 14 Notably, the tumor cells in this case expressed the product of the c-kit protooncogene, CD117. HHV-8 has been shown to induce the c-kit gene, 14 and by inference, RFHV and RRV may do the same. Alternatively, the CD117 expression could be taken to suggest an endothelial cell origin for the progenitor cells of these intestinal tumors, similar to the purported KS progenitor. 6 However, the spindeloid tumor cells in this case were negative for the vascular endothelial cell marker, CD31 (Fig. 1B), although CD31 is an almost invariable marker of KS tumor cells. 6, 8 Reportedly, tumor cells in early RF and SF lesions may express CD31, only to subsequently lose this marker. 10 Such disparate results may be reconciled if simian rhadinoviruses cause significant changes in expression of genes in the infected cells, as has been demonstrated for HHV-8 6, 14 and other herpesviruses. Upregulation of the c-kit gene was shown to be essential for the HHV-8–induced morphological transformation of endothelial cells. 14 CD117 expression and upregulation may play a similar role in other soft tissue tumors characterized by spindeloid tumor cells, most notably GISTs and inflammatory fibroid polyps, 9, 12, 17 and this role of CD117 may explain the morphological similarities between macaque MPD, KS, and several other stromal tumors, even if the etiology, progenitor cells, and final histogenesis for these various neoplasms in humans and macaques differ. 9
The CD34 membrane antigen, a very consistent marker of both GIST and KS cells 8 but absent in IMFT and most other stromal tumors, 9 is a heavily glycosylated transmembrane protein expressed mainly on hematopoietic and endothelial progenitor cells. 21 The large number of glycosylation sites on the antigen gives rise to a number of glycosylation-dependent epitopes, which are classified according to their sensitivity to different glycoproteases. 21 Although antibodies specific for the class-III epitopes of CD34 do cross-react with macaque CD34 on freshly isolated cells (H. Bielefeldt-Ohmann, unpublished), the antibodies used for immunohistochemical labeling of formaldehyde-fixed, paraffin-embedded human tissues are targeted to the class-II epitopes of human CD34 and do not cross-react with macaque cells 20 (H. Bielefeldt-Ohmann unpublished) thus precluding the application of this important phenotypic differential marker for elucidation of the question of the progenitor cell(s) of the present tumors and of macaque MPD, as well as their relationship to GISTs, IMFT, and KS, at least when examining archival samples.
Unlike the MPD cases identified in SRV-2–infected macaques in the 1970 and 1980, the present MPD-like intestinal submucosal tumors were identified in an SRV-2–negative rhesus macaque presenting with an immunodeficiency syndrome induced by experimental infection with an SIV variant, SIVsmE660. This is the first published case of an SRV-2–negative macaque diagnosed with an MPD-like condition. Although this finding might suggest that the role SRV-2 plays in the induction of MPD in macaques can be supplanted by a simian lentivirus, it is curious that SIV-associated MPD cases have so far not been identified in the vast number of macaque SIV studies, which have been initiated during the search for an HIV vaccine candidate over the past 20 years. This lack of SIV-associated MPD cases may be because of a low prevalence of an underlying RFHV or RRV (or both) infection within the primate colonies or unknown secondary factors. Because SIV provokes a more substantial immunodeficiency syndrome than SRV-2, the simple role of immunodeficiency as a cofactor in the induction of MPD seems insufficient. Rather, a time factor may be of decisive importance in combination with the immunodeficiency. Lentivirus-induced SAIDS tends to have a faster progression than SRV-2–induced SAIDS, resulting in euthanatization of the animals for humane reasons before the possible development of MPD and other neoplasms, with the notable exception of lymphoproliferative disorders and lymphoma. It is noteworthy that the macaque described in this report survived the SIV challenge for over 3 years, with slowly progressive CD4+ T cell decline, before finally developing clinical SAIDS, a course more reminiscent of SRV-2–induced SAIDS.
Further studies are required to elucidate the following: 1) the question about the progenitor cell(s) for MPD in macaques, 2) the relative importance of immunodeficiency and other regulatory effects of the different retroviruses in the development of MPD, and 3) the role of the various rhadinoviruses in the pathogenesis of macaque MPD.
Footnotes
Acknowledgements
We are indebted to Mr. Joel Ahrens and Dr. Stephen Kelley (WaNPRC) for veterinary clinical support of the animal, to Dr. Brian Rubin (Department of Pathology, UW) for providing KS-control tissue and antibodies, to Dr. C.-C. Tsai for access to archival RF samples, and to Dr. B. Chandran (University of Kansas Medical Center) for the gift of monoclonal antibodies. We thank Dr. JoAnn Schuh (Applied Veterinary Pathobiology, PLLC, WA) and Dr. David Fitzpatrick (Amgen Corporation) for critical reading of the manuscript. These studies were supported by the SIV Evaluation Unit (SVEU) contract from the National Institute of Allergy & Infectious Diseases, N01-AI-15431 (N. L. Letvin) and a grant from the National Institutes of Health, RO1-RR13154 (T. M. Rose). T. M. Rose is the recipient of an independent Scientist Award, K02-AI49275. The WaNPRC is supported by core-funding from the NIH NPRC (grant P51RR00166).
