Abstract
Over 100 free-ranging adult California sea lions (Zalophus californianus) and one Northern fur seal (Callorhinus ursinus), predominantly adult females, were intoxicated by domoic acid (DA) during three harmful algal blooms between 1998 and 2000 in central and northern California coastal waters. The vector prey item was Northern anchovy (Engraulis mordax) and the primary DA-producing algal diatom was Psuedonitzschia australis. Postmortem examination revealed gross and histologic findings that were distinctive and aided in diagnosis. A total of 109 sea lions were examined, dying between 1 day and 10 months after admission to a marine mammal rehabilitation center. Persistent seizures with obtundation were the main clinical findings. Frequent gross findings in animals dying acutely consisted of piriform lobe malacia, myocardial pallor, bronchopneumonia, and complications related to pregnancy. Gross findings in animals dying months after intoxication included bilateral hippocampal atrophy. Histologic observations implicated limbic system seizure injury consistent with excitotoxin exposure. Peracutely, there was microvesicular hydropic degeneration within the neuropil of the hippocampus, amygdala, pyriform lobe, and other limbic structures. Acutely, there was ischemic neuronal necrosis, particularly apparent in the granular cells of the dentate gyrus and the pyramidal cells within the hippocampus cornu ammonis (CA) sectors CA4, CA3, and CA1. Dentate granular cell necrosis has not been reported in human or experimental animal DA toxicity and may be unique to sea lions. Chronically, there was gliosis, mild nonsuppurative inflammation, and loss of laminar organization in affected areas.
Keywords
Domoic acid (DA) is a neurotoxic analog of the excitatory neurotransmitter L-glutamate. DA is produced by marine algae of several genera and has been identified from the North Atlantic, Pacific, Gulf of Mexico, Sea of Japan, and European and New Zealand coastal waters. Toxic exposures to DA usually occur in the context of harmful algal blooms (HAB) created when local water conditions foster explosive growth of a toxigenic algal species. DA produced during a HAB may undergo trophic transfer by accumulating in algivorous or filter-feeding species such as anchovy, 13 Dungeness crab, razor clam, 36 or mussels. 19 Other algal toxins of concern in US waters include saxitoxin, brevetoxin, okadaic acid, ciguatoxin, and Pfeisteria-associated toxin. DA gained notoriety as the causative agent of amnesic shellfish poisoning in 1987, when human consumption of contaminated mussels led to illness and deaths. 19 DA may also pose a risk to marine wildlife and has caused mass mortalities of seabirds 25, 37 and marine mammals. 23
Initially described as the antihelmintic component of “domoi,” a Japanese folk medicine derived from the red algae Chondria armata, 28 DA is a potent agonist of the alpha-amino-3-hydroxyl-5-methyl-4-isoxazole propionate (AMPA) and kainate subclasses of the neuronal glutamate ionotropic receptors. Neuronal depolarization triggered by DA results in a cascade of unregulated events including endogenous glutamate release, 4 activation of N-methyl-D-aspartic acid (NMDA) glutamate receptors 3 and activation of voltage gated calcium channels. 38 The loss of homeostasis and resultant neuronal injury triggered by excessive glutamate receptor activation is termed excitotoxicity. Endogenous excitotoxicity has been implicated in the pathophysiology of diverse neurologic disease including ischemia, epilepsy, and head trauma. 17 Exogenous excitotoxic brain injury secondary to natural DA exposure has been described in humans 29 and experimentally in rats and macaques. 33, 34
In this study, we describe the pathologic findings in 84 California sea lions (Zalophus californianus, CSL) and one Northern fur seal (Callorhinus ursinus, NFS) involved in unusual stranding events. These animals were naturally exposed to DA during the course of three HAB in central and northern California. The vector prey item was Northern anchovy (Engraulis mordax) and the primary DA-producing algal diatom was Psuedonitzschia australis. Details of the confirmation of DA trophic transfer to CSLs during the HAB of May 1998 have been reported. 14, 23
Materials and Methods
Animals
CSL and NFS mortalities described in this report were associated with DA-producing HAB in May 1998 (N = 52), October 1998 (N = 12), and July 2000 (N = 45). Cases were defined as stranded animals with seizure activity as diagnosed by observation. The age and sex distribution was adult female (N = 96), adult male (N = 1), subadult female (N = 4), and subadult male (N = 8).
The overwhelming preponderance of adult female animals is most likely because of the foraging and migratory behavior of the population. 12 Adult male sea lions forage near the breeding rookeries from approximately April through August. They migrate and winter as far north as British Columbia, leaving the rookeries in the fall and returning in the spring. Adult females forage up to 100 km away from the rookeries during the summer and may stay in the vicinity year around. 20 The location of the spring and summer blooms was within the foraging area of the females, but well away from the rookeries. The males would also have migrated north before the October bloom.
Sea lions presented singly or in small groups during a period of several weeks surrounding the identification of the HAB. Stranded animals were usually first identified by the public.
Animals were captured and transported to The Marine Mammal Center (TMMC) rehabilitation center in Sausalito, CA, where clinical assessments and treatment were initiated. A full description of clinical signs and treatment has been reported. 10 Clinical signs included obtundation, head weaving, seizures, and coma. Animals were most often treated with anticonvulsants, such as barbiturates and benzodiazepines. Therapy for shock and bacterial infection was also administered. The fatality rate was approximately 70%. Sea lions dying in treatment, or euthanatized because of deteriorating clinical signs, were necropsied on-site by staff veterinarians, biologists, or veterinary pathologists. Postmortem interval was under 12 hours in most cases. Tissue samples from major organ systems including entire brain were fixed in 10% neutral buffered formalin and submitted to the UC Davis-Veterinary Medicine Teaching Hospital pathology service along with standard measurements and descriptions of gross findings. Eyes were not available from all animals. Tissues were also distributed to pathologists at Colorado State University and the Armed Forces Institute of Pathology.
The findings from 84 necropsies (83 CSL and 1 NFS) were compiled for this article. Although it is not known exactly when each individual was exposed to the toxin, cases were divided into groups on the basis of time of admission to the TMMC until death. Group 1 survived 0–1 days (n = 36), group 2 lived 2–7 days (n = 19), group 3 lived 8–14 days (n = 18), and group 4 lived for more than 15 days (n = 11). The animal surviving the longest in rehabilitation before being euthanatized lived approximately 10 months. The single NFS was a subadult female in group 2.
Tissue sampling
For each case, representative samples of all submitted tissues were processed with additional sections taken from tissues with gross abnormalities. Brains were serially sectioned and examined for gross abnormalities. In 22 cases, 6 from group 1 and 5 from each of the other groups, systematic brain sampling and histologic processing of 5 large (6.6 cm2) coronal blocks per animal was undertaken. The canine brain was used as a model to delineate gross and microscopic neuroanatomical landmarks. 5 Minor gross differences were noted between the two species with the canine brain more elongated, with a relatively smaller cerebellum and pyriform lobe and a dorsally rotated hippocampal position. Tissue sections were taken at the level of and including the lateral olfactory tract (block 1), amygdala and hypothalamus (block 2), thalamus and head of the hippocampus (block 3), piriform lobe and tail of the hippocampus (block 4), and cerebellum and brain stem at the level of the pyramid tracts (block 5). In the remaining 62 cases, brain sampling was restricted to standard-sized cassettes with sections of olfactory tract, hippocampus, amygdala, brain stem, and cerebellum.
Tissues were routinely embedded in paraffin, sectioned at 7 μm, and stained with hematoxylin and eosin (HE). Selected blocks were evaluated with Luxol fast blue/Cresyl violet and by immunohistochemistry for glial fibrillary acidic protein. Archived histologic specimens from numerous CSLs dying of various causes were available as reference material. In addition, Fluoro-Jade (Histochem, Jefferson, AR), a fluorochrome marker for neuronal degeneration, 22 was used on paraffin sections from nine representative cases. Fluoro-Jade–stained slides were examined with a Nikon E600 epi-fluorescent microscope using a filter system suitable for fluorescein. It was hypothesized that Fluoro-Jade would identify lesions consistent with DA toxicity at peracute time points, before HE would identify ischemic necrotic neurons.
Results
Gross and histologic findings unique to animals in these unusual stranding events are summarized in Tables 1, 2. Diagnoses pertaining to chronic degenerative disease, neoplasia, parasitism, or other nonspecific lesions commonly noted in stranded sea lions were excluded. The vast majority of animals in groups 1 through 3 were in good body condition and either gravid or recently parturient.
Gross findings in California sea lions with domoic acid toxicity.

∗ Time to death after admission to TMMC.
Histologic findings in California sea lions with domoic acid toxicity.
∗ Time to death after admission to TMMC.
† Eyes were examined from Group 1 (9/36), Group 2 (9/19), Group 3 (9/18), and Group 4 (5/11).
Complete blood counts were normal, and serum chemistries revealed only elevated creatine kinase. Blood lead and brain cholinesterase were within normal limits. Virus neutralization assays for phocine distemper virus were consistent with exposure titers and with the known seropositivity for the population. 10
The temporal characteristics of the gross and histologic findings were consistent with lesions being created at the time of DA exposure and subsequent stranding with resolution while in the rehabilitation center.
Gross findings
Forty-three animals had no significant gross findings.
Myocardial lesions were present in animals from groups 1 through 3. Lesions consisted of myocardial pallor and hemorrhage and fibrinous epicarditis at earlier time points with fibrosis chronically.
Uterine lesions in gravid animals included placental hemorrhage, torsion and rupture with intraabdominal delivery.
The proximal gastrointestinal tract was usually empty in animals examined immediately after stranding. Gastrointestinal mucosal hemorrhage, pulmonary edema, and fibrinous ophthalmitis were also diagnosed in animals from groups 1 and 2.
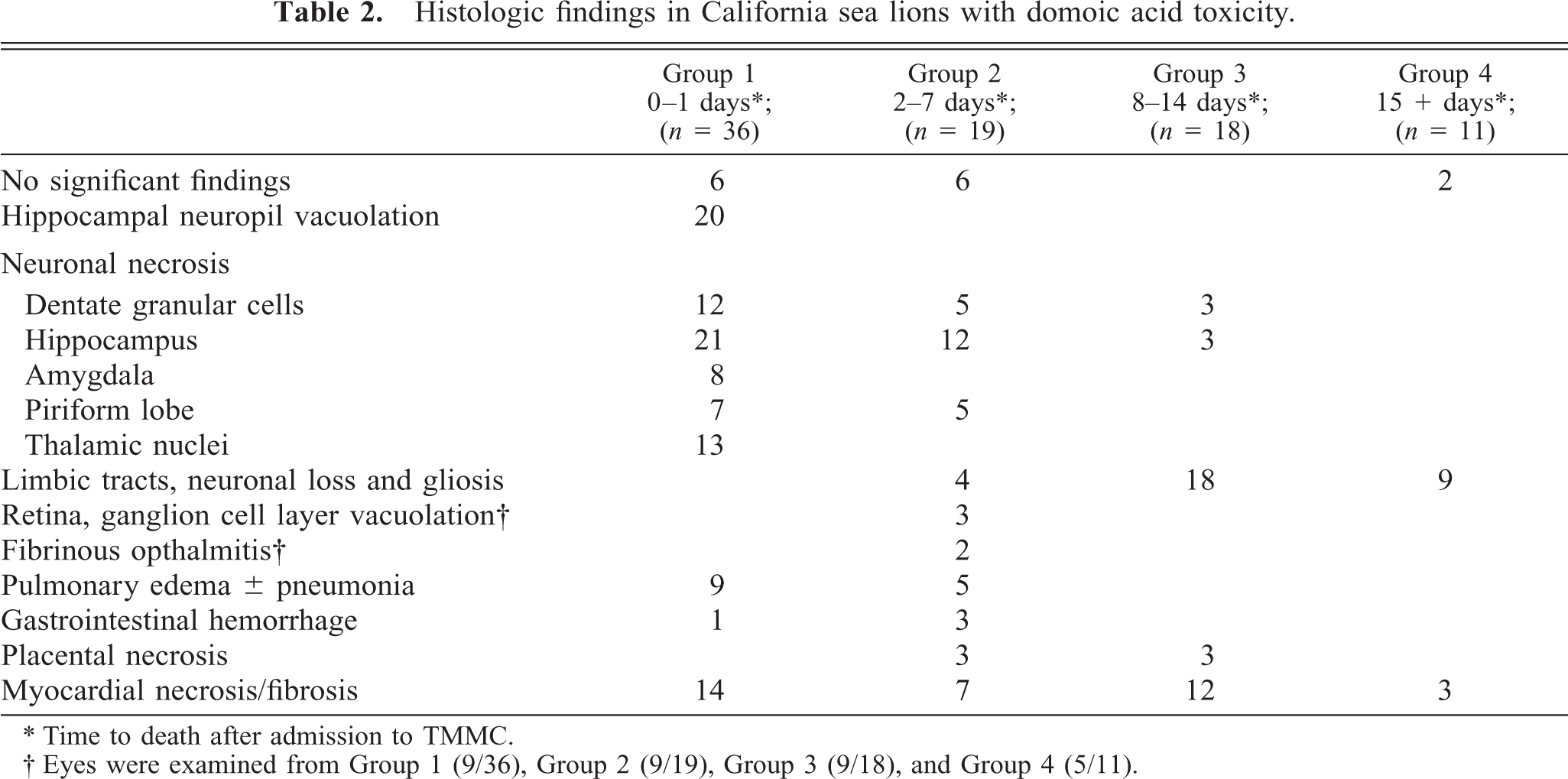
Cerebral lesions consisted of piriform lobe malacia in animals from groups 2 and 3 and hippocampal atrophy in animals from group 4. Hippocampal atrophy with relative enlargement of the inferior horn of the lateral ventricle (Fig. 1) was present in three animals dying 1 month, 5 months, and 10 months after stranding.
Hippocampus; California sea lion (group 4). Hippocampal atrophy. Note relative enlargement of the inferior horn of the lateral ventricle (arrow).
Emaciation was also present in animals from group 4.
Histologic findings
Fluoro-Jade–positive cells coincided with brightly eosinophilic neurons as identified with HE in the nine cases for which comparisons were made. This is consistent with cells undergoing ischemic necrosis. We were not successful in identifying neuronal soma or process injury at earlier time points than was possible with standard techniques.
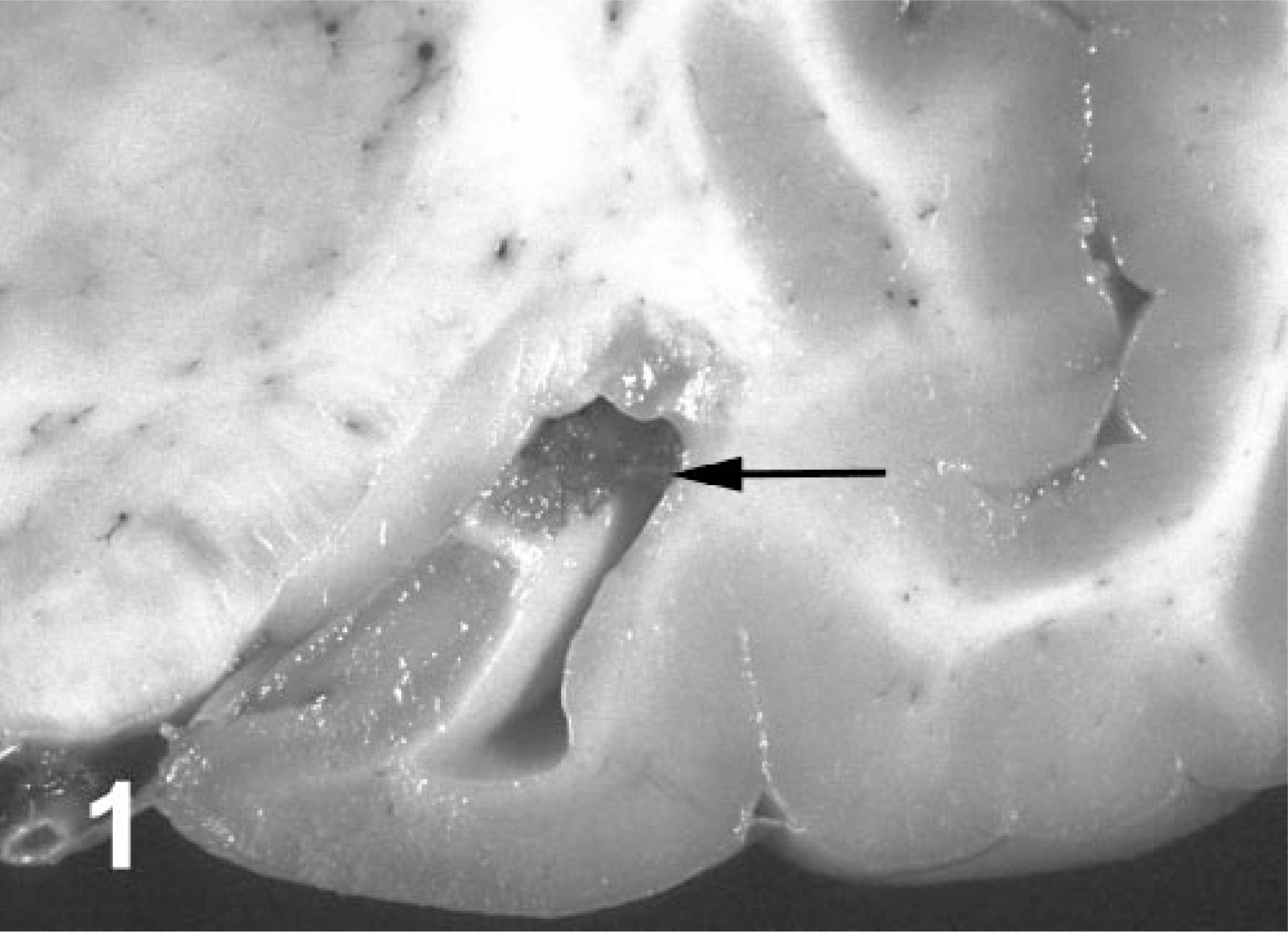
Brain lesions amongst chronologically grouped animals were consistent and distinctive. Neurologic lesions were diagnosed in every animal examined except for a few animals that died shortly after stranding. Lesions were topographically distributed within limbic system tracts (Fig. 2).
Brain, transverse sections; California sea lion. Topographic distribution of DA-induced brain lesions depicted with black dots. Bar = 1 cm.
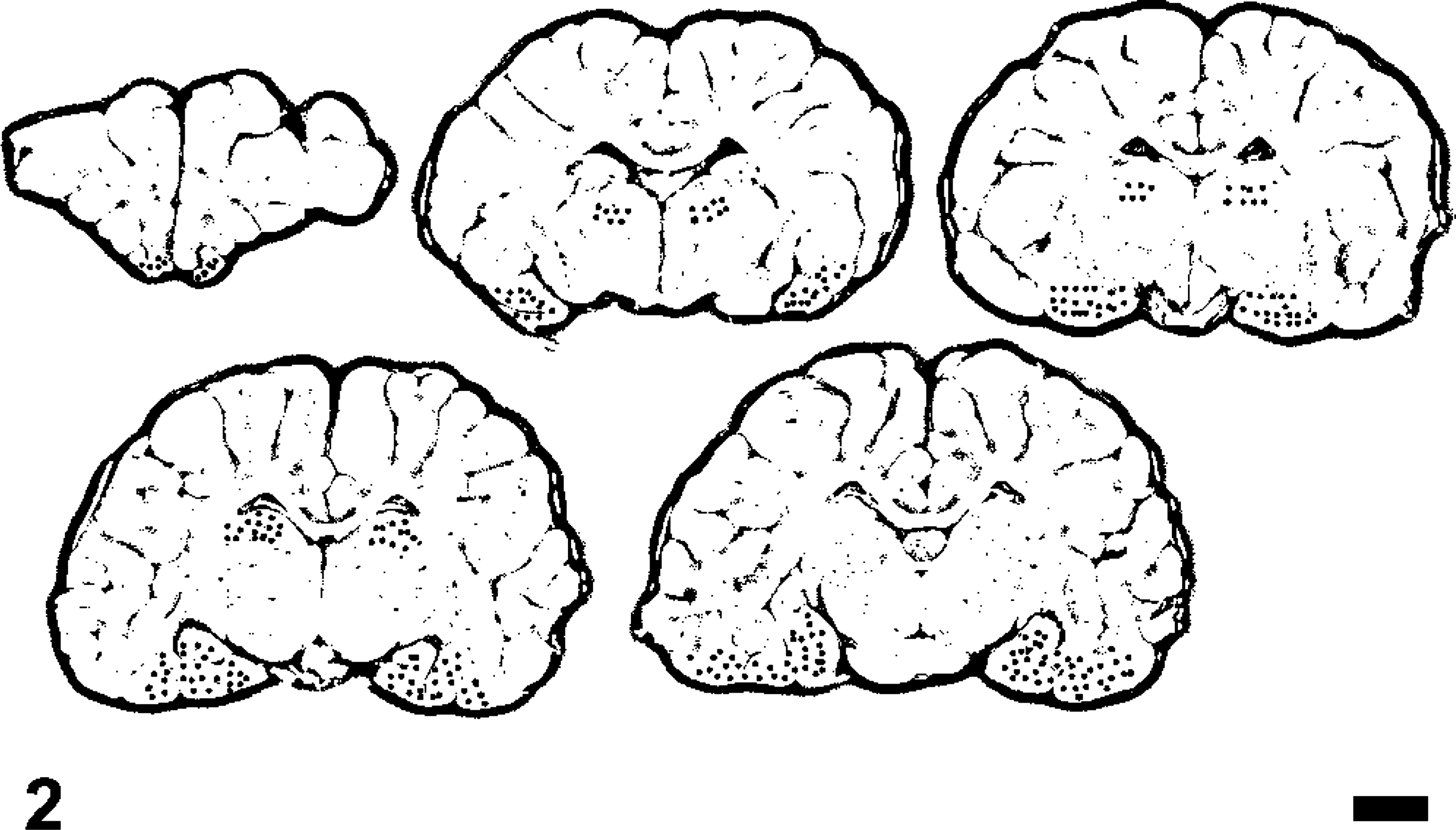
In 20 of 36 animals from group 1, a striking laminar microvesicular degeneration of the neuropil was present within the stratum lacunosummoleculare of the hippocampal formation (Figs. 3, 4). The vacuolation was present in the anterior ventral (head) aspect of the hippocampus most commonly, although it was noted throughout the hippocampal formation. Concurrent with this lesion was neuronal hyperchromasia and contraction in the granule cell layer of the dentate gyrus. Affected neurons were surrounded by a small to moderate amount of clear space.
Hippocampus; California sea lion (group 1). Arrows delineate a region of microvesicular hydropic degeneration within a dendrite rich region of the neuropil (stratum lacunosummoleculare). HE. Bar = 300 μm.
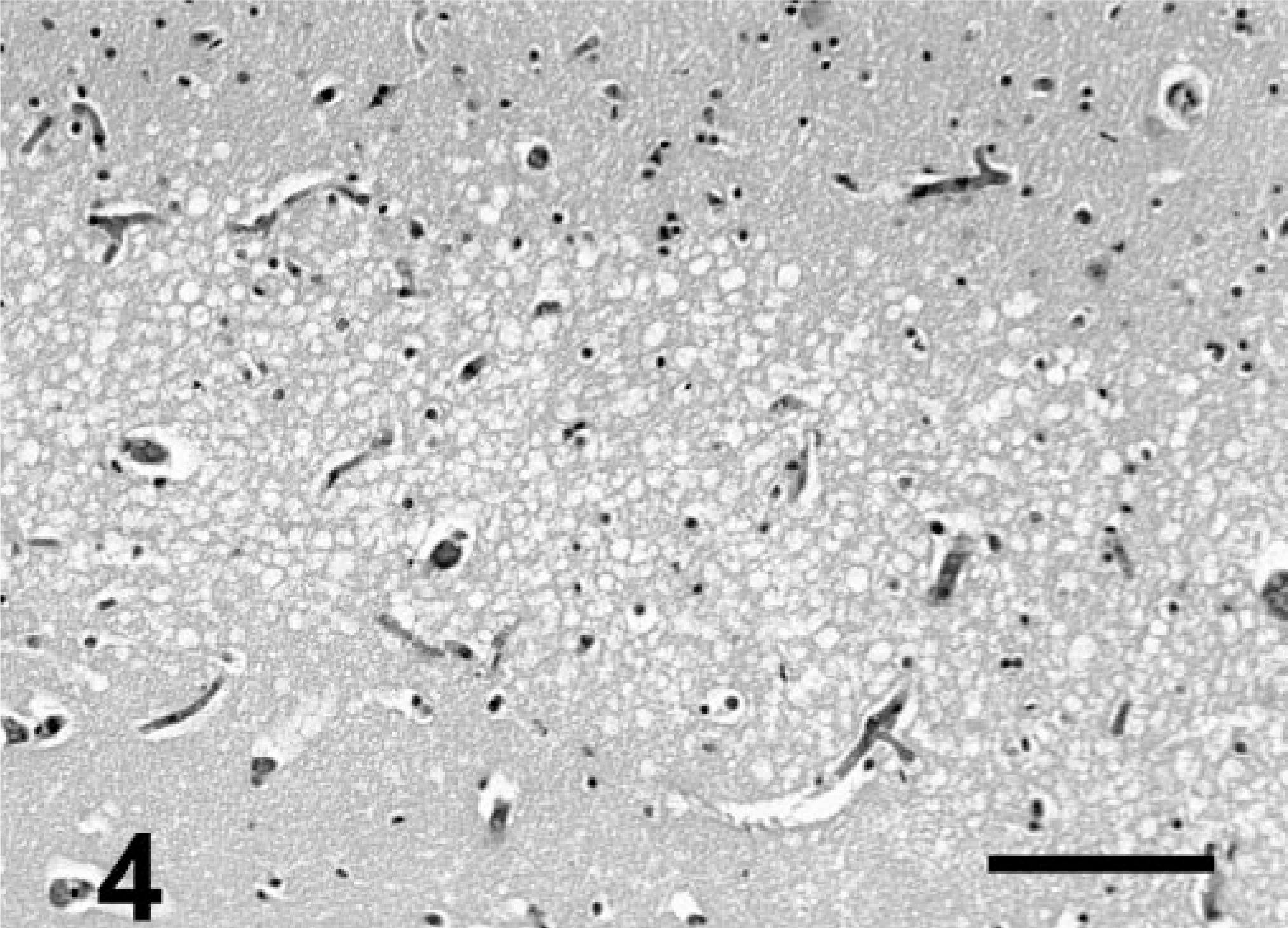
Hippocampus; California sea lion (group 1). Higher magnification of stratum lacunosummoleculare. Note laminar arrangement of clear round vacuoles within neuropil. HE. Bar = 200 μm.
The majority of animals from group 1 also had ischemic neuronal necrosis localized to the limbic system. The hippocampus and amygdala complex were the most severely affected areas. Ischemic necrosis was widespread among granular cells of the dentate gyrus (Fig. 5) and pyramidal cells in the cornu ammonis (CA) sectors CA4, CA1, and CA3. The CA2 sector was relatively spared and often remained intact (Fig. 6). In some animals, necrotic neurons were present throughout much of the cortical and thalamic limbic system, including olfactory bulb, pyriform lobe, and rostral thalamic nuclei. A normal hippocampus is provided for reference (Fig. 7).
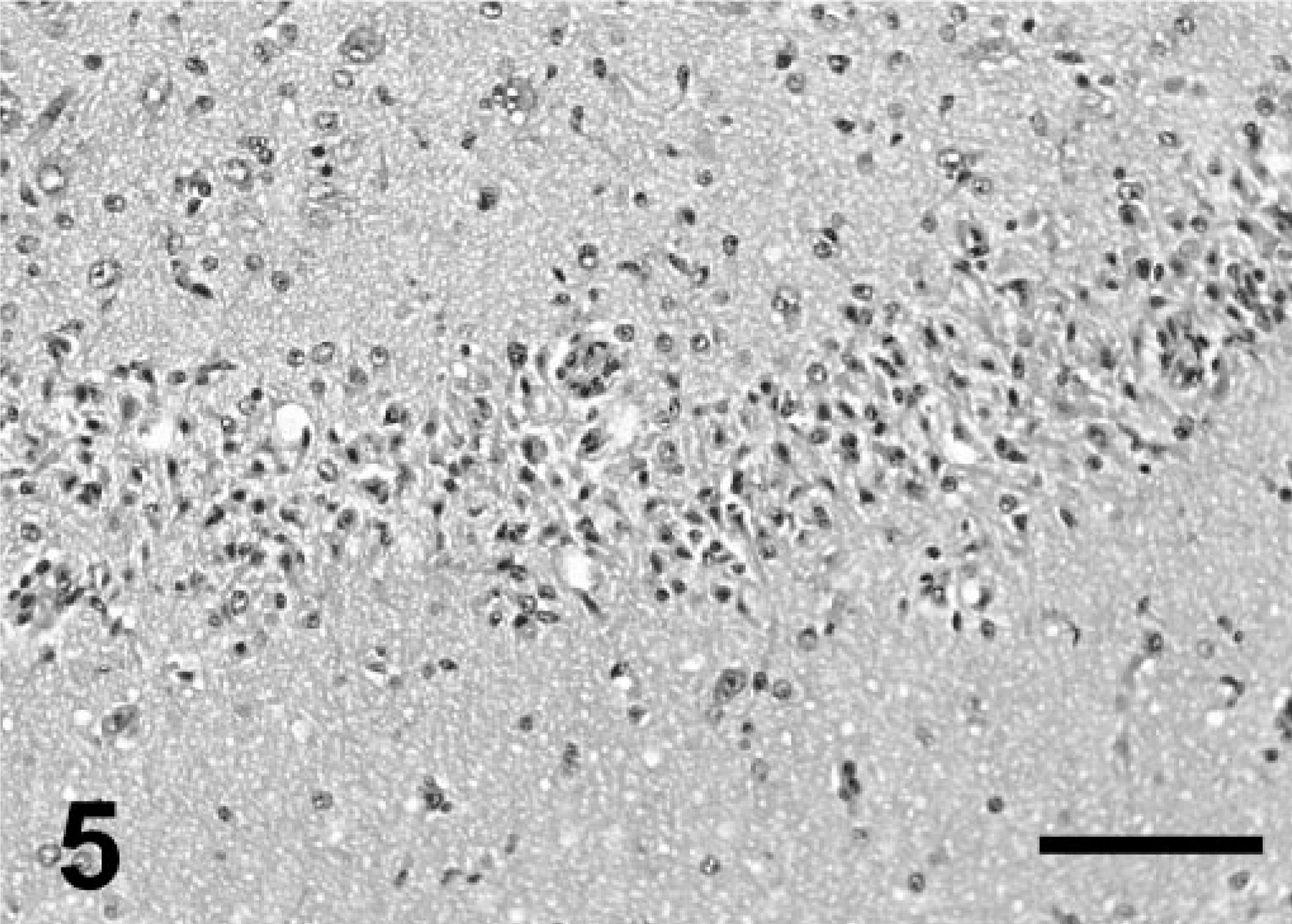
Dentate gyrus; California sea lion (group 2). Granular cell necrosis. HE. Bar = 200 μm.
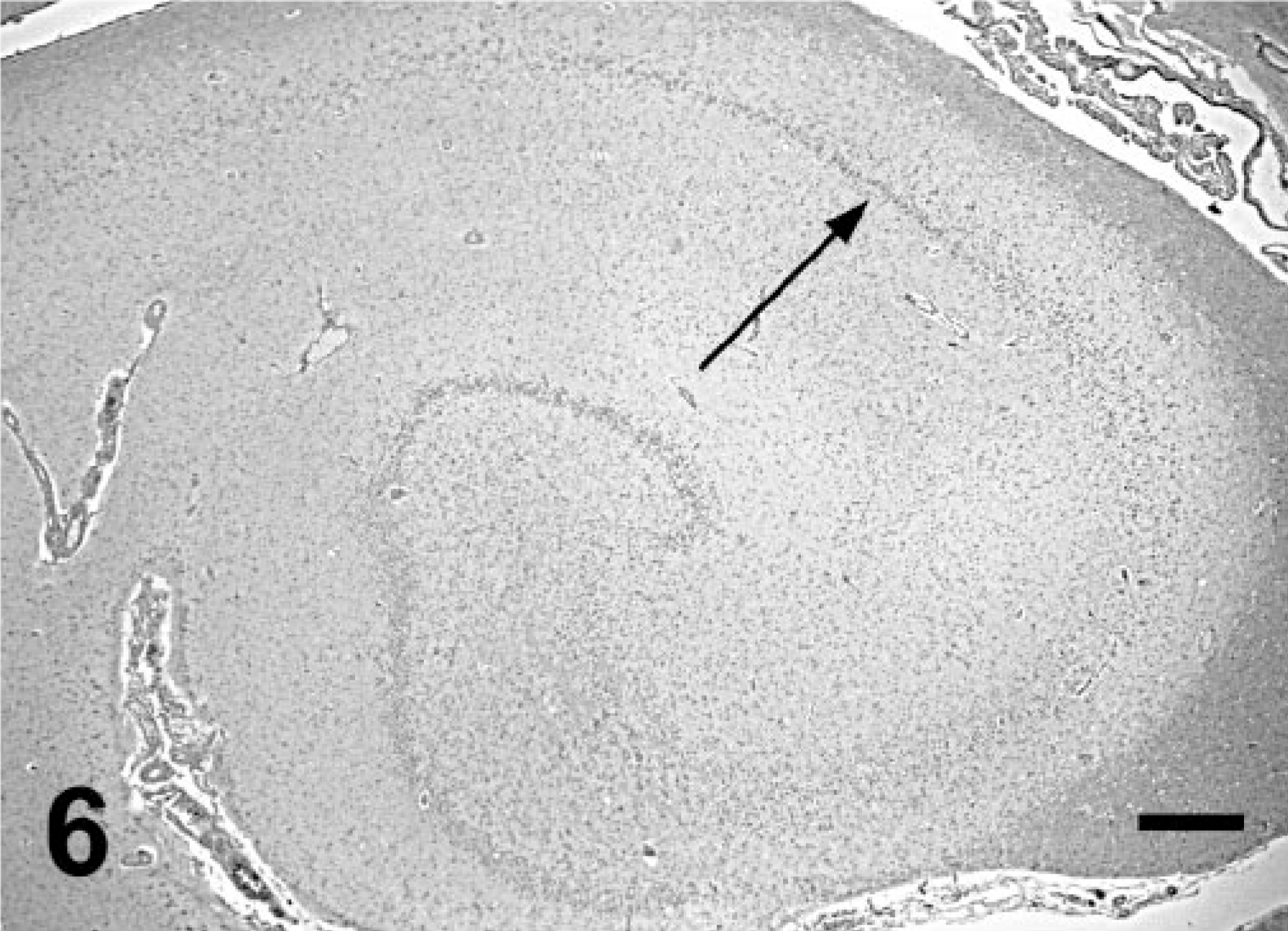
Hippocampus; California sea lion (group 2). Note loss of architecture because of necrosis and loss of pyramidal cells in sectors CA4, CA3, and CA1. Cells in CA2 (arrow) persist. HE. Bar = 500 μm. CA = cornu ammonis.
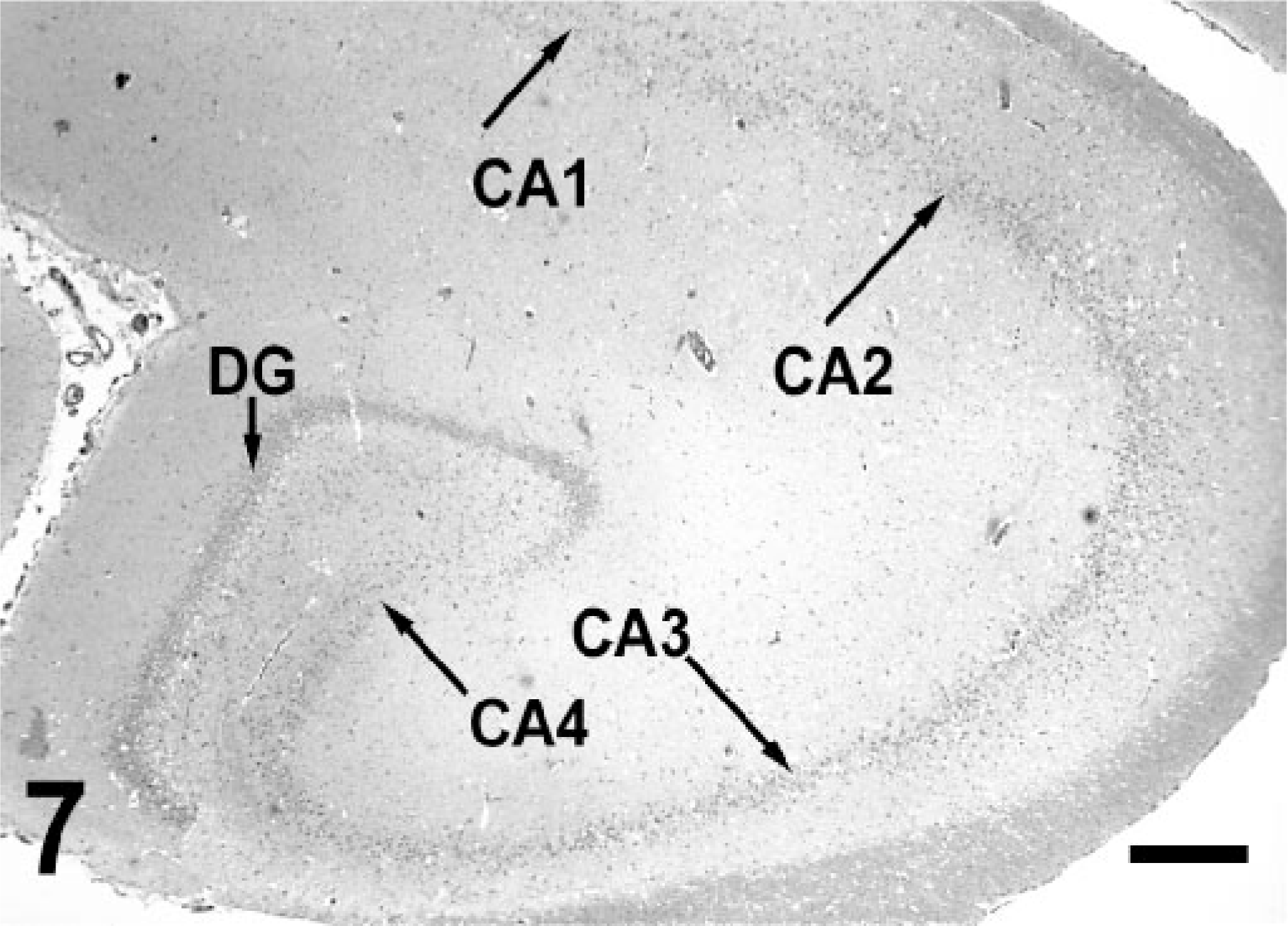
Hippocampus; California sea lion. Normal size and laminar organization. Necrosis of the dentate gyrus (DG) granular cells, and pyramidal cells of the cornu ammonis (CA) sectors CA1, CA3, and CA4 were frequent findings in DA toxicosis. HE. Bar = 500 μm.
Mild to moderate multifocal myofiber degeneration with contraction band formation, myofiber necrosis, and hemorrhage also was present in multiple animals with fibrinous epicarditis overlying the injured areas.
Mild widespread nonsuppurative meningitis was also present in many animals; however, this finding is a frequent background lesion of CSLs. The significance of this finding is unclear. Additional histologic findings in the peracute deaths included diffuse pulmonary edema and congestion, mild superficial gastrointestinal hemorrhages, and placental necrosis and hemorrhage.
Animals from group 2 were histologically similar to those in group 1 with the exception that the microvesicular hydropic degeneration of the neuropil was largely absent. In some animals, braininjured areas were marked by gliosis, perivascular lymphohistiocytic cuffing, and neocapillarization. Retinal ganglion layer vacuolation was noted in a few cases. Placental and uterine thrombosis and infarction with intralesional bacteria were present in animals suffering from uterine torsion and rupture.
Neuropathologic findings in groups 3 and 4 largely reflected continued resolution of the injured areas noted at earlier time points. In a few individuals with terminal seizures, there was virtually global acute cerebral laminar neuronal necrosis and edema with chronic lesions in the hippocampus. Neuronal dropout with hippocampal atrophy and marked gliosis (Fig. 8) was present in many animals. Binucleated hippocampal pyramidal cells were also noted in these animals. Affected neuropil had prominent neocapillarization and perivascular lymphohistiocytic cuffing. Myocardial fibrosis was also present. Other nonneuronal lesions included skeletal muscle atrophy and serous atrophy in adipose stores, reflecting the anorexia and emaciation noted clinically.
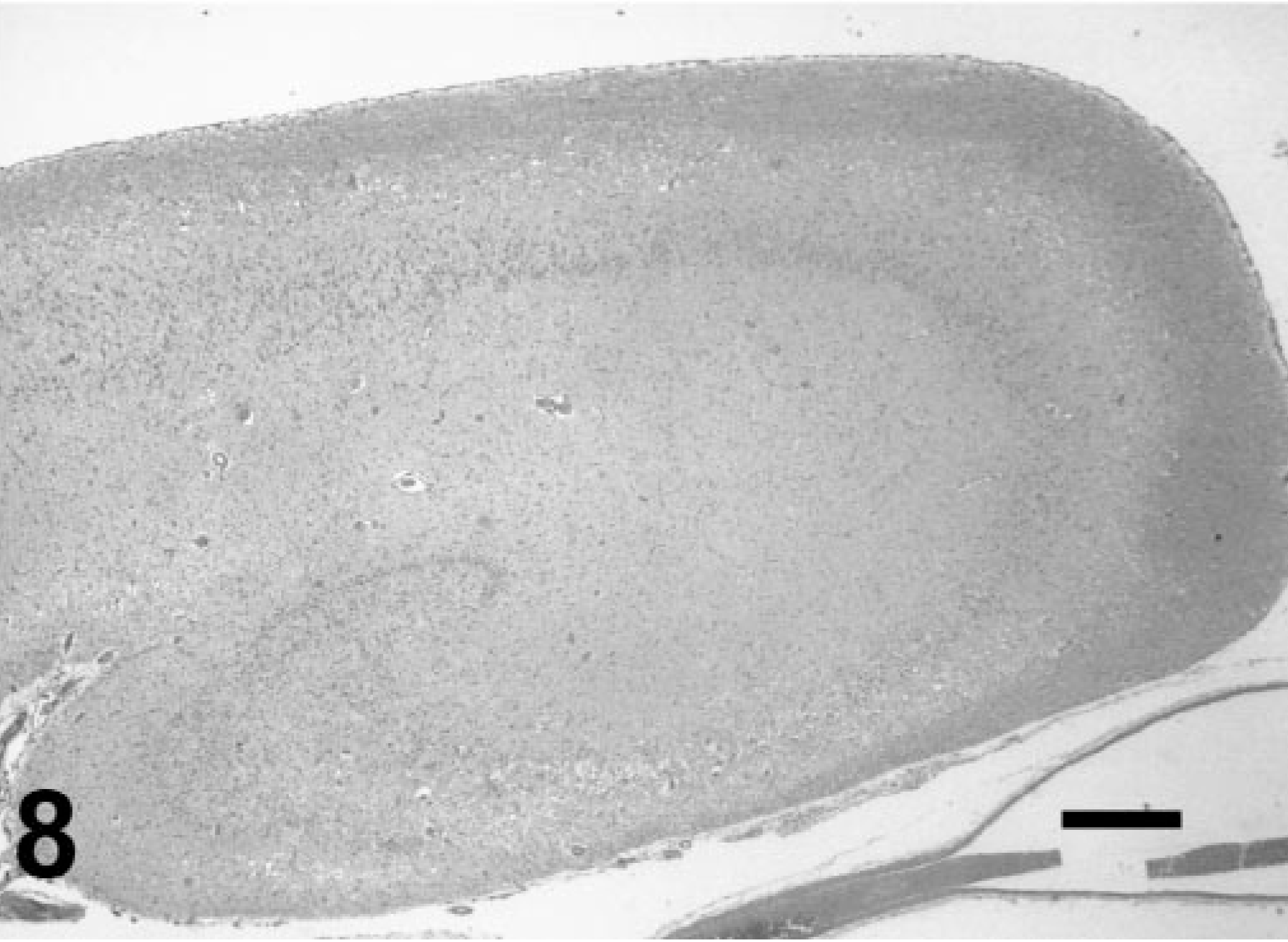
Hippocampus; California sea lion (group 4). Same animal as in Fig. 1. Loss of laminar architecture and marked neuronal dropout with parenchymal atrophy. HE. Bar = 500 μm.
Discussion
These findings, simultaneously occurring in dozens of sea lions, were consistent with intoxication by an excitotoxin. There was no clinical, gross, or histologic evidence to implicate an infectious agent. DA was subsequently identified in gastrointestinal content and urine in some of the afflicted animals. 14 Because of gastric clearance (vomiting), poor fat solubility, and rapid renal clearance, 27, 30 DA may be resident in body tissues for only a few hours after ingestion. Analytical confirmation requires obtaining gastrointestinal content, serum, or urine (or all) from acutely affected animals.
At the onset of the first mass stranding, a long list of differential diagnoses were considered given the epidemiologic, clinical, and laboratory data available at the time. The most probable etiologies included Morbillivirus infection and toxins. Potential toxins included algal toxins, heavy metals, and industrial chemicals. Clinical diagnostics were conducted by veterinarians at TMMC. 9 Simultaneous with the onset of the initial mass stranding, oceanologists were confirming the HAB of Pseudonitzchia spp. and the presence of DA in the coastal ecosystem. 23
Clinical and gross necropsy findings were consistent with acute onset of intermittent grand mal seizures or status epilepticus with somatic lesions most likely consequent to severe seizure activity. Histologic lesions identified the limbic system and the hippocampus in particular as the focus of the seizure activity. The microvesiculation in the stratum lacunosum-moleculare noted in peracute cases likely corresponded to dilated dendritic segments of hippocampal pyramidal cells. This finding was also noted 3–5 hours after intraperitoneal injection of the related excitotoxin kainate in rats. 24
The Fluoro-Jade technique had been reported to identify necrotic neuronal processes as early as 2 hours after administration of excitotoxins to rodents. 35 In the tissue sections of this study of naturally occurring toxicosis, we were not able to identify apparent dendritic lesions with Fluoro-Jade, but brightly eosinophilic (ischemic) neurons were positive. This apparent reduced sensitivity may be because of variability in the postmortem interval, time in formalin, immersion fixation, and paraffin processing in specimens in this study compared with the experimental data. Although successful use of the Fluoro-Jade technique has been reported with paraffin-embedded tissue, most of the reported results are with perfusion-fixed frozen sections.
The pattern of neuronal necrosis noted in sea lions dying at later time points also correlated with the neuropathologic findings in a number of experimental studies with excitotoxins including DA and kainate. 16, 24, 32, 34 A significant difference in the lesions of the CSLs compared with experimental animal data is the necrosis of the dentate gyrus granule cells. The reason for this difference is not known. There are complex excitatory and inhibitory inputs to the granular cells involving the dentate polymorph region (mossy cell) and basket cells, as well as excitatory perforant path inputs from the entorhinal cortex. A feature in sea lion neurophysiology, perhaps related to the hypoxia tolerance demanded by deep-diving behavior, renders the granule cells sensitive to severe limbic seizures. Granule cell dispersion has been reported as an effect of recurrent temporal lobe epilepsy, 11 but widespread acute necrosis of granule cells is not a common feature of excitotoxic agents or idiopathic epilepsy in humans or experimental animals. The sparing of the CA2 sector in acute cases and the hippocampal atrophy noted chronically does bear remarkable similarity to the neuropathologic findings in human idiopathic temporal lobe epilepsy. This reinforces the hypothesis of endogenous excitotoxic mechanisms as a key feature in the development of the human lesions.
Neuropathologic findings from naturally occurring DA intoxications have only been described in humans. 29 Hippocampal and amygdala lesions were noted in four individuals dying between 9 and 98 days after intoxication. Positron emission tomography demonstrated decreased glucose metabolism in the mesial temporal lobes of surviving individuals. The cases in humans and experimental studies in rats and macaques support the conclusion that the limbic system is the primary neurologic target in mammals. 31 In rats, histologic lesions were present in the olfactory bulbs, pyriform lobe, entorhinal cortex, hippocampal pyramidal cells, amygdala, and several thalamic nuclei.
In macaques, the hypothalamus, hippocampal pyramidal cells, and area postrema were affected. Retinal lesions were also present. In the macaque study, seizure activity was induced in one animal that died approximately 3 hours after dosing. The remaining animals had less severe manifestations consisting of lip smacking, gagging, and vomiting. The neuronal injuries described consisted of vacuolation of the neuropil, neuronal shrinkage with hyperchromasia, and astrocytic swelling. These alterations were considered sub-lethal or prelethal to the affected neurons. It was hypothesized that the area postrema was affected by virtue of an incomplete blood-brain barrier and that other circumventricular structures may be potential targets for excitotoxins.
The pathogenesis of the neurologic lesions is unclear because it is difficult to distinguish direct DA-induced neuronal injury from injury secondary to seizure activity. Ionotropic glutamate agonists, such as DA, kainate, AMPA, and NMDA, have been shown to be neurotoxic to a wide variety of neuronal subtypes both in culture and after local intracerebral injection. 3, 15, 24
Observations of naturally occurring intoxication and experimental parenteral administration of glutamate agonists in mammals have resulted in induction of limbic seizure activity and injury. 18 Neuronal death is not seen in the absence of severe seizure activity even in the face of marked behavioral impairment. 1
Furthermore, neuronal damage secondary to parenteral excitotoxin administration of a dose known to cause cell death can be abolished by suppressing seizure activity with anticonvulsants. 2 These observations are consistent with the hypothesis that areas with low seizure threshold, such as the hippocampal hilar (CA4) pyramidal cells and the amygdala, are particularly sensitive to excessive excitation. The actions of excitotoxins such as DA on the ionotropic glutamate receptor–rich dendrites plus input from other stimulated populations, such as the entorhinal cortex, may lead to loss of ionic homeostasis and cell death. Hippocampal pyramidal cell death could lead to loss of regional network homeostasis. The resulting disinhibition of dentate granule cells leads to epileptiform activity and further seizure propagation to CA3 and CA1 pyramidal cells. 26 Damage is propagated both to and from the entorhinal cortex and olfactory tracts. This mechanism would explain the striking similarities in neuropathologic findings between disparate convulsants such as pilocarpine 6 and DA as well as idiopathic temporal lobe epilepsy. 26
Retinal lesions were noted in three animals from group 2; however, eyes were not examined in all animals. Lesions consisted of vacuolation of the ganglion cell layer in three animals from group 2. It is not known why the lesion differs from those described in the rat and macaque, which affected the inner nuclear area and outer plexiform layer.
The consistent observation of myocardial pallor grossly and multifocal myocardial necrosis histologically is interesting. There are multiple potential etiologies to explain this finding. Myocardial hypoxia secondary to ineffective ventilation and pulmonary edema secondary to status epilepticus may be implicated, although other hypoxia-sensitive tissues were not affected (i.e., cerebellar Purkinje cells). Second, brain-heart interactions have been shown to occur in excitotoxin-induced myocardial necrosis after intracerebral kainate and NMDA injection in rats. 21 Cardiac stimulation and injury were demonstrated to occur either by increased sympathetic secretion (“sympathetic storm”) or centrally mediated catecholamine release. A third potential pathway is through glutamate receptors located in peripheral tissues. These receptors have been demonstrated in heart, kidney, testis, and lung by molecular and immunohistochemical techniques. 7, 8 Although the function of peripheral glutamate receptors is not clear, their presence suggests the possibility of direct toxic effects of DA on various organs.
DA toxicity should be considered in marine mammals presenting with seizures and obtundation and demonstrating neurologic lesions centered on the limbic system. Confirmation requires demonstration of DA in tissues such as gastrointestinal content, serum, or urine (or all).
Footnotes
Acknowledgements
We thank the staff and volunteers at The Marine Mammal Center, Sausalito, CA; Drs. William McLellan and Sylvain de Guise for necropsy assistance; Teri Rowles, National Marine Fisheries Service; Drs. Bruce Lyeth and Robert Berman, UC Davis-School of Medicine, Department of Neurosurgery, for assistance with Fluoro-Jade staining; Cathy Lamm, Heather Schrader, and Rhonda Oates-O'Brian for tissue preparation, and the pathology residents and histotechnologists at the UC Davis-Veterinary Medicine Teaching Hospital. Support for PAS was provided by a training grant from NIEHS (Number ES07055). FMDG is supported by the Elaine and Arthur Court Nature Conservancy.
1Present address: W. L. Gore and Associates, Medical Products Division, Flagstaff, AZ.
