Abstract
Skin lesions are a frequent manifestation of Leishmania infantum infections in Mediterranean countries. This study demonstrates by real-time reverse transcriptase-polymerase chain reaction the local cytokine response in skin biopsies from Leishmania-infected dogs (n = 10). As controls, we investigated skin biopsies from healthy (n = 10) and fleabite hypersensitive dogs (n = 10). We established a quantitative PCR to determine the parasite burden in biopsies. The objective was to elucidate whether a correlation exists between parasite number, histologic response, and T helper-1 (TH1)/T helper-2 (TH2) cytokine expression in lesional skin of naturally infected dogs. In Leishmania-infected dogs, interleukin-4 (IL-4), tumor necrosis factor α (TNF-α) and interferon-γ (IFN-γ) messenger RNA production was significantly higher than controls. Furthermore, dogs with a high Leishmania burden had a significantly higher IL-4 expression, whereas no difference was noted with regard to expression of other cytokines. By comparing the pattern of inflammation and cytokine expression, a clear trend became evident in that levels of IL-4, TNF-α, and IFN-γ were elevated in biopsies with a periadnexal nodular pattern and in biopsies where the severity of the periadnexal infiltrate was equal to the perivascular to interstitial infiltrate. Expression of IL-4, IL-13, and TNF-α was slightly increased in biopsies where plasma cells prevailed on lymphocytes, whereas expression of IFN-γ was moderately higher when lymphocytes were predominating. In summary, the present study demonstrates that the local immune response in naturally occurring leishmaniasis includes TH1 as well as TH2 cytokine subsets. Furthermore, respective data suggest that increased expression of the TH2-type cytokine IL-4 is associated with both severe clinical signs and a high parasite burden in the skin lesions.
Leishmaniasis is an endemic zoonosis in the Mediterranean countries, parts of north and east Africa, India, China, as well as Central and South America. A protozoan of the genus Leishmania spp. causes the disease. In the Mediterranean, the causative agent is Leishmania infantum that is transmitted by sandflies of the subfamily Phlebotominae. 36, 39 Dogs have been identified to be an important host for the parasite. Infected dogs normally develop both visceral and cutaneous lesions, whereas restriction to the skin occurs in some forms of human leishmaniasis (oriental sore). But despite the viscerotropic nature of L. infantum, skin lesions are the most frequent manifestation of canine leishmaniasis. The clinical signs are caused by inflammatory processes and immune-mediated lesions, which are associated with the multiplication of amastigotes inside macrophages and other cells of the mononuclear phagocytic system. 16
In the Mediterranean, leishmaniasis is usually subclinical in humans and emerges preferentially in immunosuppressed individuals. In contrast, the prevalence of diseased dogs ranges from 2 to 20% and reaches 30–40% in certain regional populations. One study performed in Italy even revealed a 53.1% positivity in asymptomatic dogs. 4 Recent investigations demonstrated that in endemic regions a large population of Leishmania-infected but clinically healthy dogs is living. 3, 6, 35, 36 Reported histopathologic findings in canine cutaneous leishmaniasis consist of either a diffuse mononuclear dermatitis, a granulomatous dermatitis, an ulcerative dermatitis, or a sterile pustular dermatitis without acantholysis. 11 A perifollicular and perivascular mononuclear dermatitis has been described as well. 22 The number of parasites described in these histopathologic reports varies considerably. 11, 22 Parasites have been reported to occur most numerously in the nodular form of the disease and in immunosuppressed dogs. 11, 12
Several studies in humans and murine models indicated that the successful resolution of Leishmania infections depends on the ability of the host to mount a T cell–dependent response with activation of macrophages mediated by T cell–derived cytokines. 7, 18, 26 In murine models of Leishmania infections, the cytokine production by CD4+ T cells has been identified as a major factor in determining the outcome of the infection. In these models, T helper-1 (TH1) cells producing interferon-γ (IFN-γ) provide protection against the parasite whereas T helper-2 (TH2) cells producing interleukin (IL)–4 and IL-10 aggravate the disease. 13
In humans, a specific TH1 response with production of IFN-γ guarantees the control of the infection, whereas an impaired TH1 response and an increased production of IL-10 are responsible for the progression of the infection and the disease. Peripheral blood mononuclear cells (PBMCs) from individuals who have recovered from cutaneous leishmaniasis produce high levels of IFN-γ and tumor necrosis factor–α (TNF-α) but little or no IL-4 and IL-10 in response to stimulation with Leishmania antigens. 14 Thus, in humans, IFN-γ has been shown to be a mediator of resistance to the parasites because of its ability to induce killing of the parasite by macrophages. Furthermore, it has been shown that TNF-α also mediates host protection against leishmaniasis, and it mediates killing of L. major together with IFN-γ through the induction of nitric oxide. 19, 21 However, in another study, it has been reported that there is no difference in the Leishmania-specific proliferative response of PBMCs and in the secretion of IFN-γ, IL-10, IL-12p40, and IL-4 in humans with localized cutaneous leishmaniasis and asymptomatic individuals in an endemic area. 37 So far, only limited information on the pathogenesis of naturally occurring canine leishmaniasis is available. Much information has been extrapolated from human medicine and experimental infections in laboratory animals. In experimentally infected dogs and in dogs with natural Leishmania infection, it has been shown that in the supernatants of PBMCs from asymptomatic animals higher levels of TNF-α and IL-2 were found when compared with symptomatic and control uninfected dogs. 30 Furthermore, infected dogs with no clinical signs have a strong specific proliferative response with production of IL-2, TNF-α, and IFN-γ and low anti-Leishmania antibody titer, whereas diseased animals have a depressed T cell–mediated response and high levels of specific antibodies. 5, 29, 30 Thus, in the dogs, IL-2, TNF-α, and IFN-γ seem to play a protective role against the development of the clinical disease.
In a study investigating cytokine expression by PBMCs from experimentally infected dogs during a period of up to 23 months, Santos-Gomes et al. reported that in experimentally infected dogs only during a short prepatent phase, PBMCs had an increased expression of IFN-γ and IL-2. 33 In the patent phase, when dogs presented clinical signs, the expression of cytokines was reduced.
In humans, the immunologic environment of the skin plays a dominant role in determining the form of the dermatologic disorder and, more importantly, in signaling the type of the host immune response. 15, 24 To our knowledge, so far the cytokine environment in lesional skin of dogs with leishmaniasis has never been investigated.
Therefore, the goal of our study was to assess whether a correlation between the number of parasites, the histologic response and the expression of TH2 and TH1 cytokines in lesional skin of naturally infected dogs exists. To achieve this objective, we developed primers and probes specific for canine cytokines and evaluated by real-time reverse transcriptase–polymerase chain reaction (RT-PCR) the messenger RNA (mRNA) production of IL-4, IL-13, TNF-α, and IFN-γ. Furthermore, we developed a quantitative PCR to assess the number of Leishmania in paraffin-embedded tissue sections. These tools enabled us to investigate the cellular immune response in lesional skin biopsies from naturally infected dogs and compare it with the parasite burden.
Materials and Methods
Animals
Skin biopsies from 30 dogs were examined in this study. Ten biopsies of Leishmania-infected dogs were selected from archival material of the Institute of Animal Pathology, Vetsuisse Faculty, University of Berne, Switzerland. These biopsies had been examined in a previous study for the presence of Leishmania amastigotes by immunohistochemistry (IHC) and for the presence of Leishmania DNA by PCR. 25 As negative control, 10 skin biopsy specimens from healthy Beagles of the research dog colony, Novartis Centre de Recherche Santé Animale in St. Aubin, Switzerland, were used. As additional control for a disease with a TH2 response, 10 biopsies from Beagles with an experimentally induced fleabite hypersensitivity of the Novartis colony were used. The biopsies from both control groups had been taken in the context of another study, which has been reviewed by an Ethics Committee according to Principles of Laboratory Animal Care (NIH publication no. 86–23, revised 1985) and the Swiss Regulations on Animal Experiments (Schweizerisches Tierschutzgesetz).
Histopathologic evaluation
Biopsies were fixed in buffered formalin (4%) at room temperature for 24 hours. The biopsies were then embedded in paraffin wax and a section of 4 μm was cut from each tissue block and stained with hematoxylin and eosin (HE). To evaluate the type and the degree of inflammation, skin biopsies from all 30 above-mentioned dogs were scored blinded.
In investigating the skin samples, the following parameters were considered: 1) the distribution pattern of the infiltrate (perivascular to interstitial or nodular periadnexal), 2) the type of the infiltrate (macrophages, lymphocytes, plasma cells, eosinophils, neutrophils, mast cells), 3) the inflammation score using a semiquantitative grading system (score 0, 1, 2, 3) to assess the degree (none, mild, moderate, severe) of cellular infiltration in the dermis, and 4) the changes in the epidermis (increased epidermal thickness, spongiosis, and cellular infiltration).
DNA preparation and real-time fluorescent PCR–based relative quantification of genomic DNA from Leishmania sp
Genomic DNA was extracted from formalin-fixed and paraffin-embedded tissue according to Müller et al. 25
Quantitative RT-PCR was carried out on a LightCycler™ Instrument (Roche Diagnostics, Rotkreuz, Switzerland) by using SYBR™ Green I as a double-strand DNA–specific fluorescent dye and continuous fluorescence monitoring as previously described. 38 Amplification of genomic Leishmania DNA providing an amplification product from a repetitive nucleotide sequence of the parasite with an approximate size of 260 base pair (bp) was performed by using previously described forward (5′-CGGCTTCGCACCATGCGGTG-3′) and reverse (5′-ACATCCCTGCCCACATACGC-3′) primers. 27 The PCR for amplification of a 162-bp canine α-actin sequence included forward primer α-ac1 (5′-GAGACCACCTACAACAGCATCATG-3′) and reverse primer α-ac2 (5′-CACCTTGATCTTCATGGTGCTGGG-3′). 25 Quantitative PCR tests were performed in duplicates and were done with 4 μl of 1 : 10 diluted DNA using the Quanti Tect™ SYBR Green PCR Kit (Qiagen, Basle, Switzerland) in a 10 μl standard reaction containing a 0.5 μM concentration of forward and reverse primers (Invitrogen, Basle, Switzerland). PCR was started by initiating the “Hot-Start” Taq DNA polymerase reaction at 95°C (15 minutes). Subsequent DNA amplification was done in 50 cycles including denaturation (94°C, 15 seconds), annealing (55°C, 30 seconds), and extension (72°C, 30 seconds) (temperature transition rates in all cycle steps were 20 C/second). Fluorescence was measured at 72°C after each annealing phase in the “single” mode with the channel setting F1. Fluorescence signals from the amplification products were quantitatively assessed by applying the standard software (version 3.5.3) of the LightCycler™ Instrument.
Quantification of PCR products was performed during the log phase of the reaction and was achieved by using the secondary derivative maximum mode for plotting of the fluorescence signals versus the cycle numbers. External standard reactions for the Leishmania-PCR were run in duplicate reactions with samples containing DNA equivalents from 1,000, 100, 10, and one parasite(s). As external standards for the canine α-actin–PCR, serial 10-fold dilutions (4 μl aliquots) of previously generated amplification products from the different target sequences were included in the quantitative PCR analyses. The standard curve from the α-actin–PCR was also run in duplicates and contained 4 log units within a linear range that essentially covered the maximal and minimal concentrations of canine DNA within the different samples. Linearity among the standard reactions was reflected by the correlation coefficient (r) which was extremely high (r = 0.99) for both PCR assays applied.
Lack of PCR inhibitory effects and overall comparability of the different standard and sample reactions were evidenced by the quasi-identity of the slopes from the amplification plots (monitoring amplification rates).
Control experiments for identification of PCR products included a DNA melting point analysis (not shown). 31 The DNA melting profile assay was run after the final PCR cycle by gradually increasing the temperature to 95°C at a transition rate of 0.1 C/second with continuous acquisition (determination of the melting profile by measuring loss of fluorescence). Data from the DNA melting profile assay were processed by using the standard software (version 3.5.3). In all, PCR tests performed, identical melting temperatures of amplicons from samples and respective standards indicated identical and specific amplification reactions without unwanted primer-dimer formation (not shown). This overall identity and specificity of reactions was confirmed by subsequent agarose gel electrophoresis (2% gels), 32 which monitored the PCR products as single DNA bands of expected sizes (not shown).
To compensate for variations in total DNA input, mean values from duplicate determinations from the Leishmania-and α-actin–PCR were taken for the calculation of the relative amounts of parasites (Leishmania-DNA level/canine host tissue DNA level).
RNA isolation and real-time RT-PCR for canine cytokines
Canine total RNA was extracted from paraffin-embedded skin biopsies by using a “User-developed protocol: isolation of total RNA from paraffin-embedded tissue using the Rneasy kit” supplied by Qiagen with slight modifications. The biopsy samples were cut out from the paraffin block and thoroughly homogenized before proteinase K treatment. Homogenization was performed in 700 μl of buffer RTL with a rotor stator (Polytron PT 10–35, Kinematica AG, Littau, Switzerland). Incubation with proteinase K was extended up to 2 hours at 55°C before centrifugation. After elution, the concentration of purified total RNA was determined by measuring the optical density at 260 nm. Two micrograms of each RNA sample was reverse transcribed into complementary DNA (cDNA) using random primers (Promega, Catalys AG; Wallisellen, Switzerland) in a final reaction volume of 50 μl. Real-time RT-PCR was performed to quantify the production of mRNA of canine IL-4, IL-13, IFN-γ, TNF-α, and 18S, respectively.
Therefore, primers and fluorescein amadite-labeled probes were designed in-house from published sequences using the PrimerExpress software from Applied Biosystems (Rotkreuz, Switzerland). The sequences of the primers and probes are shown in Table 1. Real-time RT-PCR was performed using the TaqMan Universal PCR Master Mix (Invitrogen) at a concentration of primers and probes of 1,000 nM and 200 nM, respectively, in a final reaction volume of 25 μl in an ABI PRISM 7700 Sequence Detector (Applied Biosystems) according to the manufacturer's protocol. PCR amplification was performed for 40 cycles. Control experiments using RNA samples without reverse transcription were performed to demonstrate absence of contaminating genomic DNA. PCR amplification of the constitutively expressed 18S gene was used as a measure of input RNA. The relative units defining the relative amount of mRNA production for each sample were calculated using a cDNA pool from healthy dogs.
Sequences of primers and probes for quantification of mRNA expression by real time RT-PCR.∗
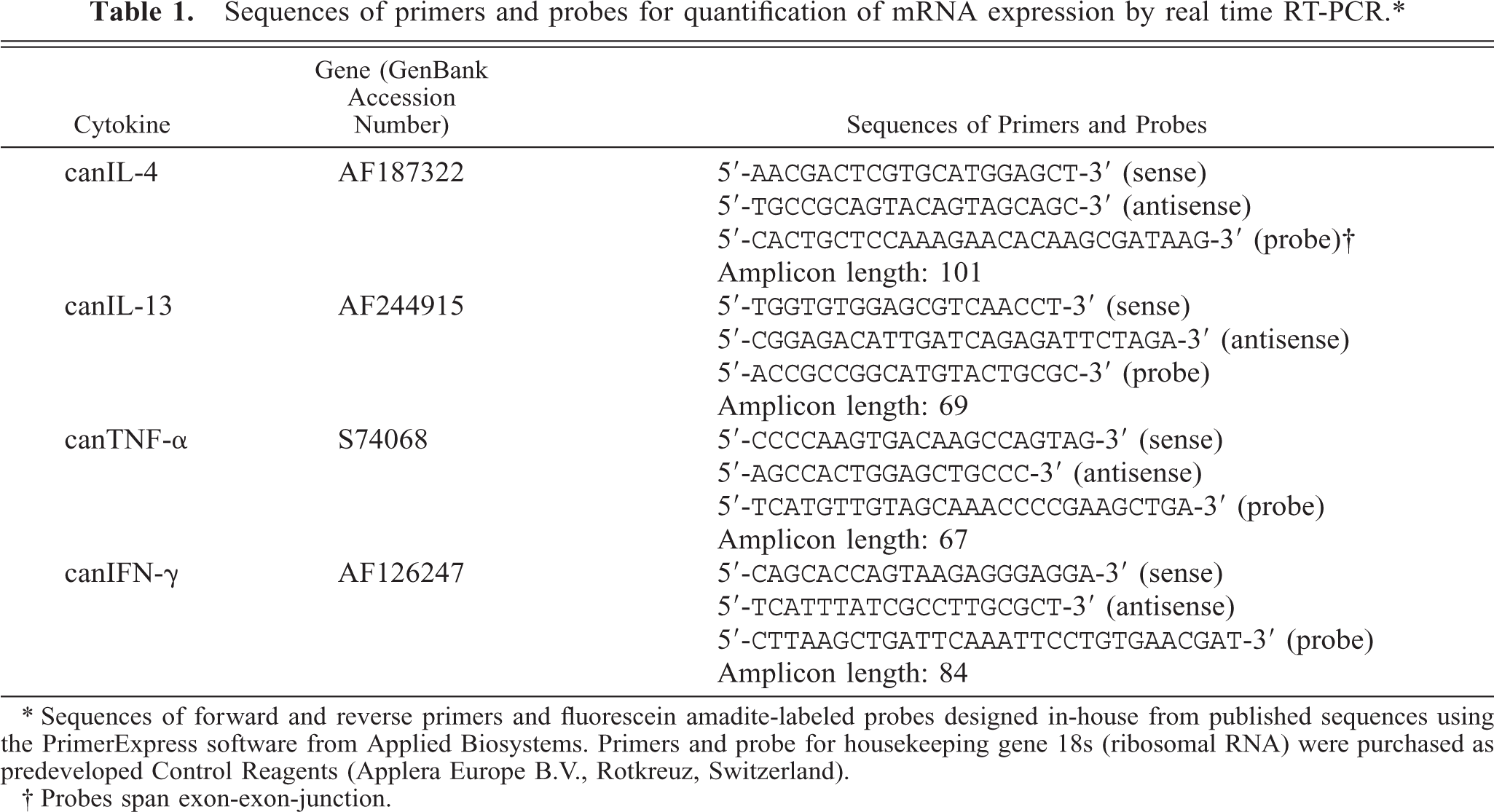
∗ Sequences of forward and reverse primers and fluorescein amadite-labeled probes designed in-house from published sequences using the PrimerExpress software from Applied Biosystems. Primers and probe for housekeeping gene 18s (ribosomal RNA) were purchased as predeveloped Control Reagents (Applera Europe B.V., Rotkreuz, Switzerland).
† Probes span exon-exon-junction.
Statistical evaluation
The levels in individual cytokine expression among the three groups of dogs were compared in a nonparametric Kruskal-Wallis one-way analysis of variance on Ranks test. The Bonferroni correction was used to correct for multiple comparisons. Within the Leishmania-infected group, the cytokine expression between the two levels of parasite burden and between different inflammation scores was evaluated using the same statistical test. The overall P value for statistical significance was set to 0.05. Statistical analysis was performed with the software package NCSS 2001 (www.ncss.com).
Results
Histopathologic evaluation
The histopathologic evaluation of the skin biopsies revealed that in the Leishmania-infected dogs, the predominant inflammatory cells were macrophages. They were usually more abundant in the superficial dermis and characterized by a moderate amount of finely vacuolated and lightly eosinophilic cytoplasm (epithelioid cells). Within the cytoplasm of macrophages, variable numbers of amastigote forms of Leishmania were identified as round to oval organisms with a round nucleus and a kinetoplast surrounded by a clear halo. They were 2–4 μm in diameter. In biopsies with only few organisms, the definitive diagnosis was on the basis of positive PCR results, positive IHC, and the pattern of inflammation. The other inflammatory cells, which were present in the biopsies of the Leishmania-infected dogs, were lymphocytes and plasma cells. As depicted in Table 2, four dogs had a higher number of lymphocytes compared with plasma cells. In the other six dogs, the number of plasma cells was equal (n = 1) or higher (n = 5) than the number of lymphocytes. In the 10 healthy control dogs, when present, the inflammatory response was characterized by a minimal to mild perivascular infiltration with rare mast cells and lymphocytes. In the fleabite hypersensitive group, the superficial dermis and the periadnexal area were infiltrated with a high number of mast cells, fewer eosinophils and some lymphocytes. Epidermal lesions consisted of subcorneal pustules or serocellular crusts. In these dogs, a moderate number of neutrophils was present in the dermis.
Summary of skin lesions with regard to the parasite burden, the severity, the pattern and the predominant cell type in Leishmania-infected dogs.
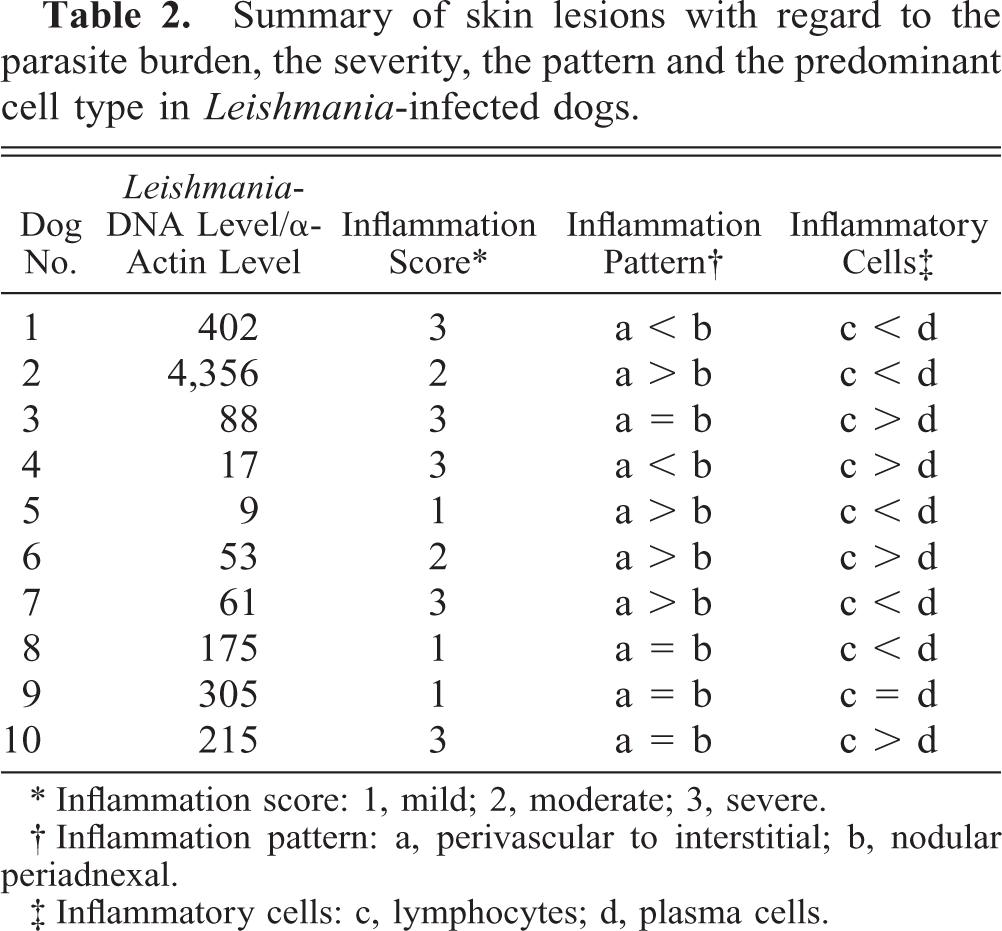
∗ Inflammation score: 1, mild; 2, moderate; 3, severe.
† Inflammation pattern: a, perivascular to interstitial; b, nodular periadnexal.
‡ Inflammatory cells: c, lymphocytes; d, plasma cells.
In the Leishmania-infected dogs, two different patterns of inflammation could be distinguished. The first pattern was characterized by a perivascular to interstitial infiltrate, which was either superficial or deep (pattern a). This pattern is shown in Fig. 1. In the second pattern, which is shown in Fig. 2, a nodular infiltrate was centered around the adnexal structures (pattern b). In four cases, pattern a was predominating, and in two cases, the periadnexal reaction (pattern b) was prevailing. In the remaining four cases, the two inflammation patterns were present in the same biopsy.
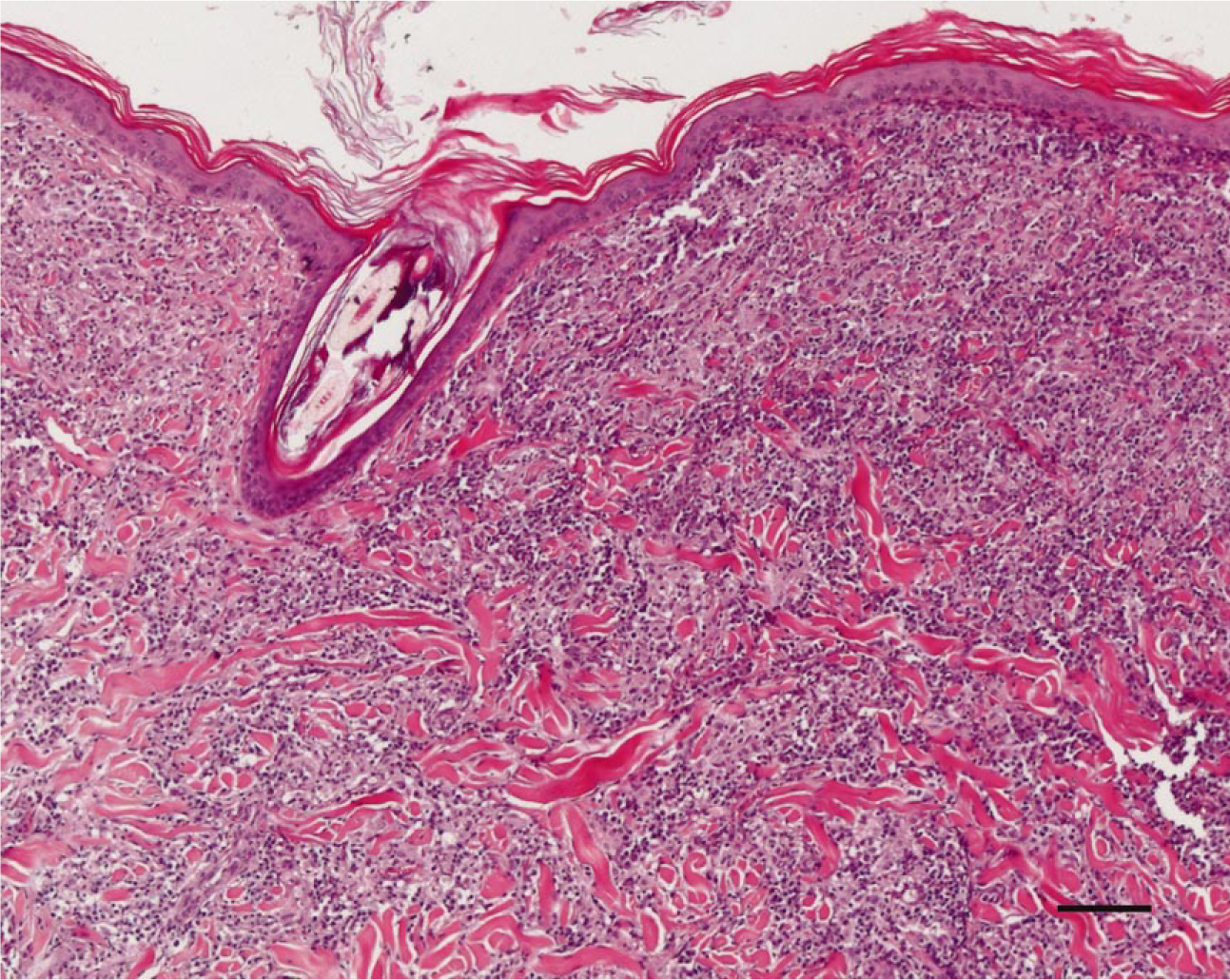
Skin; Leishmania-infected dog No. 7. Note the perivascular to interstitial infiltration in the superficial and deep dermis. HE. Bar 5 100 mm.
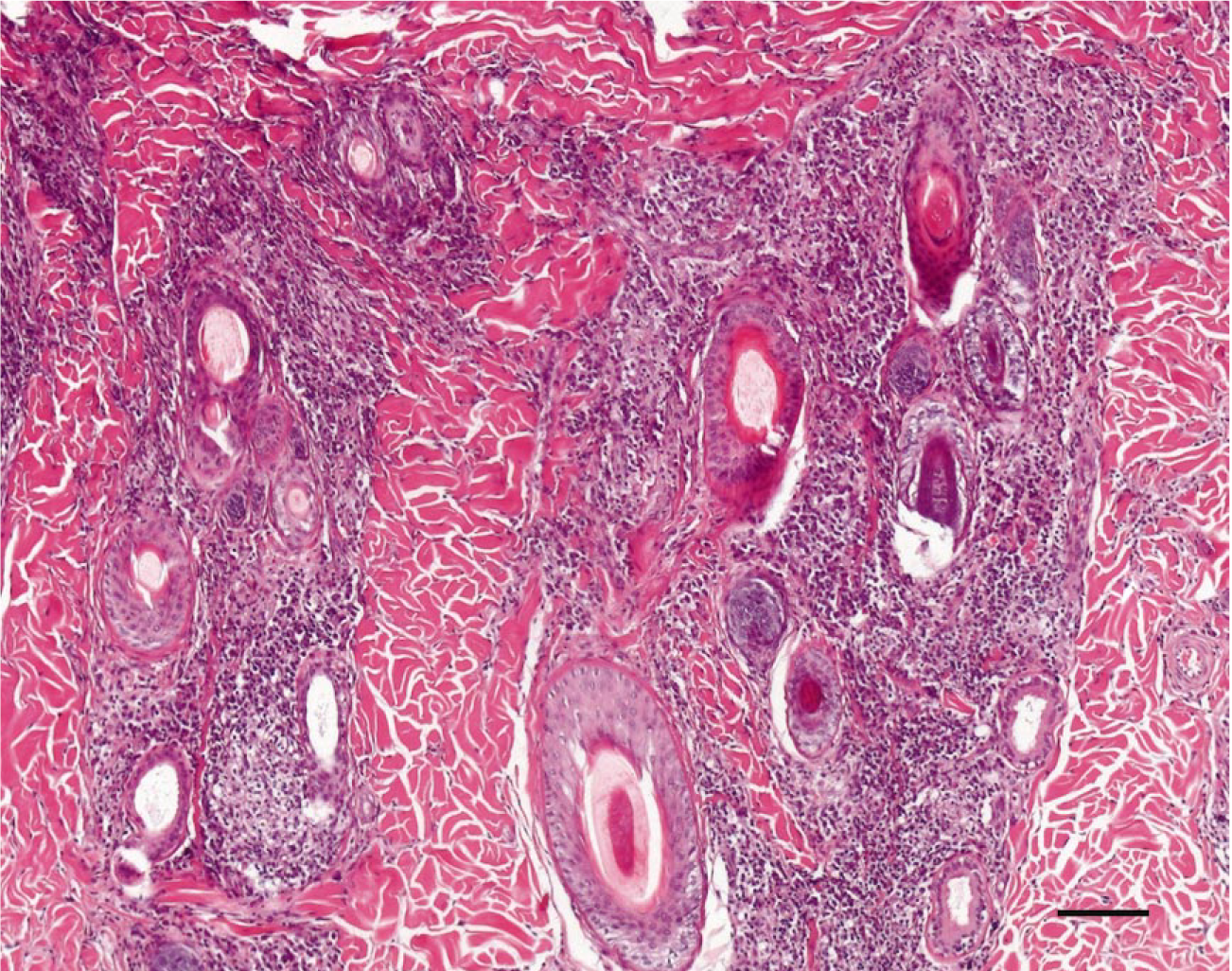
Skin; Leishmania-infected dog No. 1. Note the nodular infiltration of inflammatory cells centered around the adnexal structures. HE. Bar 5 100 mm.
Evaluating the severity of the cellular infiltrate in the Leishmania-infected group depicted that three dogs had a mild inflammatory response (score 1), two dogs had a moderate infiltrate (score 2), and five dogs had a severe infiltrate (score 3). Epidermal lesions were characterized by variable degrees of orthokeratotic hyperkeratosis and epidermal hyperplasia.
In the healthy control dogs, three animals had a mild perivascular inflammatory infiltrate (score 1) and seven dogs had no infiltrate (score 0). In the fleabite hypersensitivity group, the inflammation score varied from 1 (n = 3), to 2 (n = 4), to 3 (n = 3).
The results of the histopathologic evaluation of biopsies from the Leishmania-infected group are summarized in Table 2.
Quantitative PCR for detection of genomic DNA from Leishmania sp
For the first time, we showed that quantification of Leishmania by PCR from paraffin-embedded tissue sections is possible (Table 2). To achieve this, the amount of parasite DNA was calculated relative to the amount of α-actin, as a measure for canine host tissue input (Leishmania-DNA level/canine host tissue DNA level). The obtained values were then related to a standard curve. In the 10 biopsies from the Leishmania-infected dogs, the amount of parasites varied from 9 to 4,356. In the two control groups, no Leishmania DNA was detected. The results for the Leishmania-infected dogs are shown in Table 2.
Real-time RT-PCR for canine cytokines
Cytokine mRNA production in the Leishmania-infected dogs, in the fleabite hypersensitive group, and in healthy animals varied considerably between individual dogs. In the Leishmania group, relative units for IL-4 ranged from 0.3 to 415. The relative units for IL-13 varied between 0.31 and 6.1. The lowest value for TNF-α was 2.3, whereas the highest value was 50. IFN-γ ranged from 544 up to 24,406. In the fleabite hypersensitive group, relative units for IL-4 ranged from 3.8 to 117. The relative units for IL-13 varied between 12 and 515. The lowest value for TNF-α was 0.38, whereas the highest was 11. IFN-γ ranged from 124 to 929. The relative units of IL-13, TNF-α, and IFN-γ in the healthy group ranged between 0 and 1 in all dogs. The relative units of IL-4 ranged between 0.34 and 9, although six out of 10 values were close to one. Although the cytokine mRNA production varied considerably between individual dogs, it became evident that Leishmania-infected dogs had a statistically significant higher production of IL-4, TNF-α, and IFN-γ mRNA when compared with the fleabite hypersensitive dogs (P < 0.05) (Fig. 3A, C, D). On the other hand, fleabite hypersensitive dogs expressed higher levels of IL-13 as did the Leishmania-infected dogs (P < 0.05) (Fig. 3B). In both the Leishmania-infected and the fleabite hypersensitive group, the expression of each cytokine was statistically significantly higher than in the biopsies from the healthy dogs (P < 0.05).
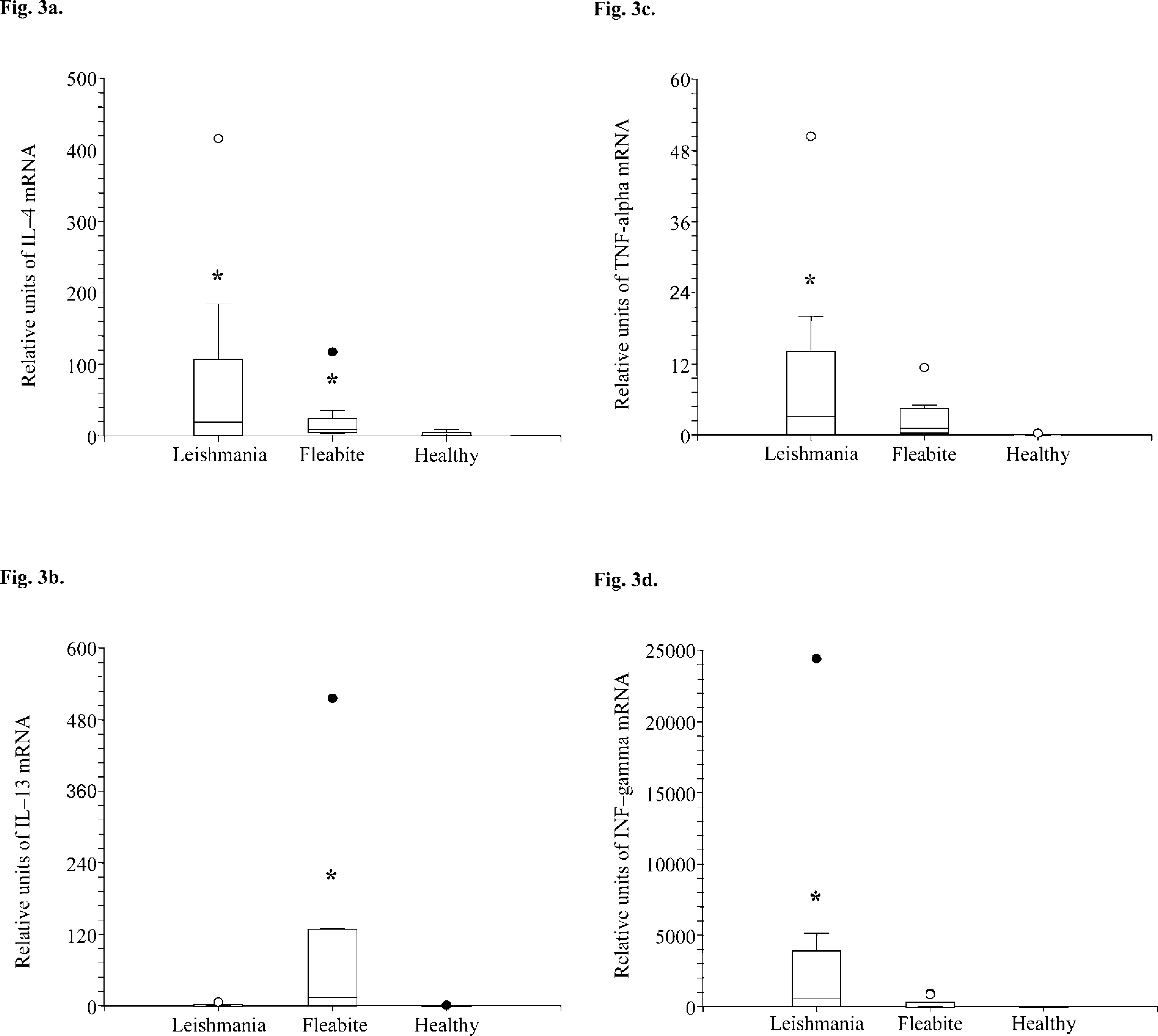
Production of IL-4
To compare the mRNA production for the different cytokines and the number of Leishmania, we dichotomized the number of organisms into two groups (group I < 100 Leishmania and group II > 100 Leishmania). A statistically significant higher expression of IL-4 could be shown in group II (Fig. 4), whereas no difference was noted between the two groups for the expression of TNF-α and IFN-γ (data not shown). For IL-13, a negative association was seen; however, the difference was not statistically significant.
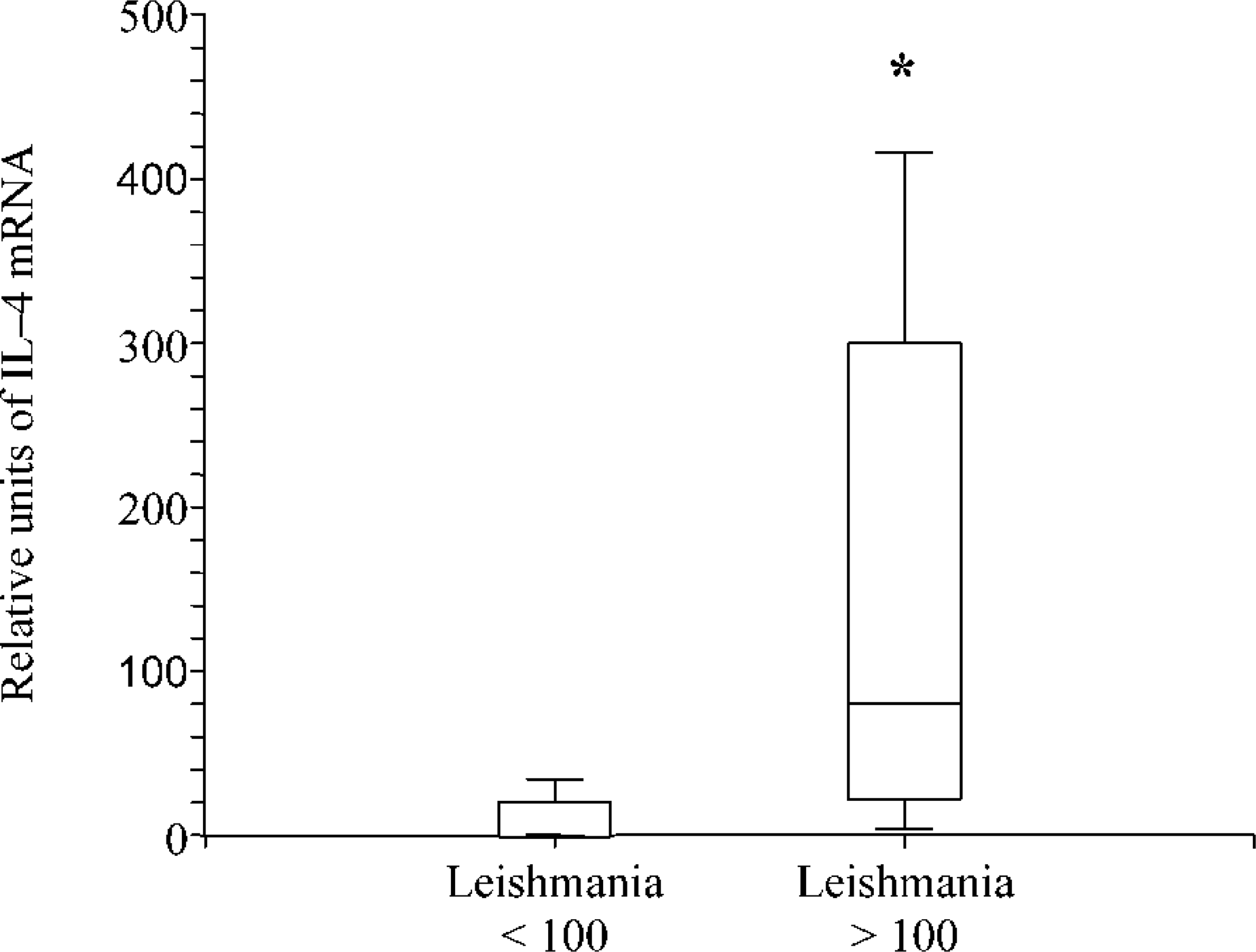
Production of IL-4 mRNA in skin biopsies from Leishmania-infected dogs with different parasite burden. Note that IL-4 mRNA expression in dogs with a low (<100) parasite burden is significantly lower than in dogs with a high (>100) parasite burden (P∗<0.05). Values in the box plots reflect the 25th, 50th (median), and 75th percentiles. The whiskers represent approximately 90 to 95% of the values.
By comparing the histologic pattern of inflammation and the cytokine expression a clear trend was seen in that the expression of IL-4, TNF-α, and IFN-γ was higher in biopsies with a periadnexal nodular pattern and in biopsies where the severity of the periadnexal infiltrate was equal to, or higher than, the perivascular to interstitial infiltrate (mean Rank for cytokine production comparing periadnexal versus perivascular pattern: IL-4, 6.08 versus 4.63; TNF-α, 5.67 versus 5.27; IFN-γ, 5.83 versus 5.00). The expression of IL-13 was higher in the group where a perivascular to interstitial infiltration predominated (4.83 versus 6.5). These results were statistically not significant.
Although not significant, the expression of IL-4, IL-13, and TNF-α was increased in the biopsies where plasma cells prevailed on lymphocytes, whereas the expression of IFN-γ was higher when lymphocytes were predominating (data not shown).
To assess whether there was any correlation between the severity of the inflammation and the expression of cytokines, we compared the inflammation score and the expression of individual cytokines. Because of the small number of dogs, inflammation scores 0 and 1 were analyzed together as group A and inflammation scores 2 and 3 were analyzed as group B. No statistically significant correlation could be detected between the two groups for any of the analyzed cytokines (IL-4, IL-13, TNF-α, and IFN-γ). However, there was an evident trend indicating that cytokine expression in group A biopsies was higher than that in group B biopsies (data not shown).
Discussion
In this study, we demonstrate by real-time RT-PCR the local cytokine response in paraffin-embedded skin biopsies from Leishmania-infected dogs. Up to now, a number of studies evaluated cytokine expression in experimental and naturally occurring leishmaniasis by analyzing the immune response at the level of the PBMCs or serum, but to our knowledge, no study investigated the local immune response in the skin.
The histopathologic evaluation of the skin biopsies revealed that in all Leishmania-infected dogs the predominant inflammatory cells were macrophages. Lymphocytes and plasma cells were the second most frequent cell type. The prevailing number between lymphocytes and plasma cells varied between individuals. In the biopsies from the Leishmania-infected dogs, two different patterns of inflammation could be distinguished. The first pattern was characterized by a perivascular to interstitial infiltrate. In the second pattern, a nodular infiltrate was centered around the adnexal structures. These findings are in accordance with the literature, where granulomatous perifolliculitis, superficial and deep perivascular dermatitis, and interstitial dermatitis are reported to be most common. 16 The presence of more than one pattern as we have found in some of our biopsies has been reported in the same article. The severity of the cellular infiltrate in the Leishmania-infected group varied from a mild inflammatory infiltrate to a severe one.
IHC has significantly improved the histopathologic diagnosis of cutaneous leishmaniasis, but the quantification of the parasites is still a problem especially if numerous amastigotes are within one macrophage. We were able to demonstrate for the first time that quantification of Leishmania by PCR from paraffin-embedded tissue sections is possible. The quantitative PCR carried out with the LightCycler allowed an accurate quantification of the parasites present in the biopsies. The possibility to use paraffin-embedded material strongly enhances the applicability of this diagnostic technique and is of great value for both retrospective studies and ongoing trials investigating the local immune response or the efficacy of treatment.
Leishmania-infected dogs had a statistically significant higher expression of IL-4, TNF-α, and IFN-γ mRNA when compared with the fleabite hypersensitive dogs. Furthermore, a higher expression of IL-4 could be shown in biopsies from dogs with a high Leishmania burden, whereas no difference was noted with regard to the expression of TNF-α and IFN-γ. In the literature, the role of IL-4 in the pathogenesis of Leishmania infections is discussed controversially. In a study investigating L. major infections in Balb/c mice, high levels of IL-4 in the draining lymph nodes correlated with the lesion size, thus indicating the disease severity. 10 Other authors have associated a TH2 response with high production of IL-4 to an enhanced susceptibility of some strains of mice to Leishmania amazonensis. 2 In contrast, Kropf et al. have shown that IL-4-deficient BALB/c mice infected with L. major developed progressive lesions. 17 In humans, IL-4 and IL-13 are involved in the regulation of B-cell development, immunoglobulin-E synthesis, and the allergic response. 8, 23 The finding indicating that more IL-4 is expressed in the skin of Leishmania-infected dogs than in fleabite hypersensitive dogs was surprising because allergic reactions are known to be linked to a TH2 response. On the other hand, as expected, fleabite hypersensitive dogs expressed higher levels of IL-13 than Leishmania-infected dogs. This constellation might be partially because of the fact that, after allergen stimulation, PBMCs secret more IL-13 than IL-4. An additional reason for this could be that allergen-stimulated IL-4 expression only occurs in a transient manner.
The results of this study demonstrate that IL-4 is expressed in the skin of all dogs with clinical disease and that a high expression of TH2-type cytokine IL-4 is associated with a high parasite burden. In contrast, no correlation between IFN-γ or TNF-α expression and parasite burden could be established. These find-ings indicate that susceptibility of dogs to cutaneous leishmaniasis is linked to a TH2-biased immune response. However, the expression of IFN-γ and TNF-α in all biopsies from the Leishmania-infected dogs but not in the control groups confirms the participation of a TH1 response to the presence of the parasite. The development of a TH1-type immune response is critical for controlling many intracellular pathogens. In particular, it has been shown that a TH1-type response is associated with resistance to Leishmania. 29, 30 A further study evidenced that the cytokines IFN-γ and IL-2 activate human and rodent macrophages to destroy the parasite. 20 In addition, Pinelli et al. reported that the treatment of a L. infantum–infected canine macrophage cell line with supernatants from a Leishmania-specific canine T cell line, containing IFN-γ, TNF-α, and IL-2 resulted in NO production and enhanced anti-leishmanial activity of the parasitized cells. 28
The prevalence of a TH1- or a TH2-type response depends on the ability of Langerhans cells and keratinocytes, expressing class II major histocompatibility complex molecules, to present the parasite antigen and to prime an effective T cell response. 12 The vertebrate host defense strongly depends on T lymphocyte activity, which supports macrophage-mediated killing of intracellular amastigotes. 34
A TH1-TH2 dichotomy was observed in humans who had recovered from different forms of leishmaniasis. Regression of symptoms were associated with either IFN-γ (cutaneous leishmaniasis) or combined INF-γ/IL-4 (visceral leishmaniasis) production. 20 In some patients with mucosal leishmaniasis, it has been demonstrated that the decreased ability to produce IL-10 and TGF-β resulted in a noneffective response although high levels of proinflammatory cytokines such as IFN-γ and TNF-α were present. 1
When we compared the pattern of inflammation and the cytokine expression, a clear trend was evident that the expression of IL-4, TNF-α, and IFN-γ was higher in biopsies with a periadnexal nodular pattern, and in biopsies where the severity of the periadnexal infiltrate was equal to the perivascular to interstitial infiltrate. However, the distribution of the lesions was not related with parasite burden. This finding contradicts the results of another study, where parasite burden, quantified by histologic means, was higher in biopsies with a nodular periadnexal infiltrate. 9 In this study, we were not able to associate the parasite burden with the inflammatory cell type. However, in another study, the number of lymphocytes was inversely associated to the number of Leishmania. 9 Although not significant, the expression of IL-4, IL-13, and TNF-α was increased in the biopsies where plasma cells prevailed on lymphocytes, whereas the expression of IFN-γ was higher when lymphocytes were predominating.
In conclusion, the clinical presentation and evolution of leishmaniasis is a consequence of complex interactions between the parasite and the host immune response. Our data demonstrated that the severe form of naturally occurring cutaneous leishmaniasis in dogs is characterized by a TH2-biased local immune response. Future studies investigating the expression of other cytokines (such as IL-2, IL-10, and IL-12) related to Leishmania infections in both symptomatic and asymptomatic dogs will further improve our knowledge on those immunologic processes that determine the outcome of the parasite infection.
Footnotes
Acknowledgements
This study was supported in part by a PhD grant from the University of Perugia, Italy. We thank Dr. Michael Stoffel for his help in taking the photographs, Prof. Maja Suter and Prof. Luca Mechelli for their support, and the Novartis team in St. Aubin.
