Abstract
In a retrospective study, 51 cases of gastritis (14%) were identified from among 341 necropsies performed on simian immunodeficiency virus (SlV)-infected rhesus macaques (Macaca mulatta) at the New England Primate Research Center from 1993 to 2001. Protozoa were seen in the stomach of 13 monkeys (25%) with gastritis. Two histopathologic manifestations of gastritis were observed: seven cases of lymphoplasmacytic gastritis with trichomonad trophozoites within lumens of gastric glands and four cases of necrosuppurative gastritis containing intralesional periodic acid-Schiff-positive protozoa; two cases of gastritis had morphologic features of both types of gastritis. In instances of necrosuppurative and combined lymphoplasmacytic and necrosuppurative gastritis, protozoa were 4-35 μm in diameter and round to tear-shaped. Because of the unusual morphology of the protozoa in these latter cases, transmission electron microscopy and polymerase chain reaction (PCR) were used to further identify these organisms. The protozoa were definitively identified as Tritrichomonas in all cases on the basis of ultrastructural characteristics (flagella and undulating membranes) and amplification of a 347-bp product of the 5.8S ribosomal RNA gene of Tritrichomonas foetus, Tritrichomonas suis and Tritrichomonas mobilensis by PCR using DNA extracted from stomach tissue. On the basis of these observations, we conclude that Tritrichomonas can be a significant cofactor in the development of necrosup-purative gastritis in SIV-infected rhesus macaques.
Keywords
Trichomonads are anaerobic flagellated protozoa that are commensal organisms in many species of mammals and birds and, with some exceptions, are considered nonpathogenic. By light microscopy, trichomonad trophozoites are round to pear-shaped, approximately 5–20 µm in length and 3–7 µm in width, and are lightly eosinophilic in hematoxylin and eosin (HE)–stained sections. 13 In monkeys, trichomonads may be present in the lumen or within crypts of the gastrointestinal tract but rarely elicit an inflammatory response or other pathologic changes. 3 In tree shrews (Tupaia belangeri, Scandentia) trichomonads were found in the large intestine with no inflammatory response. 6 In 75% of squirrel monkeys (Saimiri sciureus and Saimiri boliviensis), the natural host of Tritrichomonas mobilensis, trichomonads were observed in the lamina propria of the intestine with no associated inflammatory response. 7 , 25 Reports of trichomonad-induced lesions in nonhuman primates are rare and include a granuloma in the pelvic cavity of a rhesus monkey, suppurative to pyogranulomatous gastritis in a simian immunodeficiency virus (SIV)–infected rhesus monkey, and necrosis and ulceration of the colon in a Titi monkey (Callicebus moloch). 4 , 8 , 19 The ability of several species of trichomonads (Tritrichomonas foetus, T. mobilensis, Trichomonas vaginalis, and Trichomonas gallinae) to invade tissues or migrate to other sites in their hosts suggests that they are not strictly intraluminal or surface-dwelling endoparasites.
Several studies have been performed to evaluate the virulence of trichomonads. Coculture of trichomonads with RK-13 cells caused cytopathic effects including cytoplasmic vacuolation and disruption of the cell monolayer. 22 Babal and colleagues have shown that supernatants from trichomonad cultures contain a sialic acid–specific lectin that serves as a potent hemagglutinin. 1 , 2 Others have shown that the proteolytic activity necessary for tissue invasion by T. mobilensis is primarily attributable to cysteine proteinases. 5 , 18
Intralesional protozoal organisms were observed in several recent cases of gastritis in SIV-infected rhesus macaques at the New England Primate Research Center (NEPRC). To further investigate the role of protozoal organisms in the pathogenesis of gastritis in SIV-infected macaques, a retrospective histopathologic study was performed using necropsy tissues collected between 1993 and 2001. To our knowledge, this is the first definitive description of gastritis associated with protozoal organisms of the genus Tritrichomonas in SIV-infected rhesus macaques.
Materials and Methods
Tissue specimens
A retrospective study of 341 rhesus macaques (Macaca mulatta) infected with SIVmac251, SIVmac239, or SIV-mac239 mutant viruses and necropsied at the NEPRC between 1993 and 2001 was performed to identify cases of protozoal gastritis. Of these, 13 cases of protozoal gastritis were identified for further investigation (Table 1). In addition, three control groups consisting of four monkeys each were evaluated in this study. The first control group was composed of four SIV-negative monkeys (rhesus macaque Nos. 14–17) with no histopathologic evidence of gastric lesions, and the second group consisted of four SIV-negative macaques (rhesus macaque Nos. 18–21) with gastritis. The third control group included four SIV-infected monkeys (rhesus macaque Nos. 22–25) with no microscopic evidence of gastritis.
Histopathological classification of gastritis, gastric SIV burden and concurrent diseases observed.∗
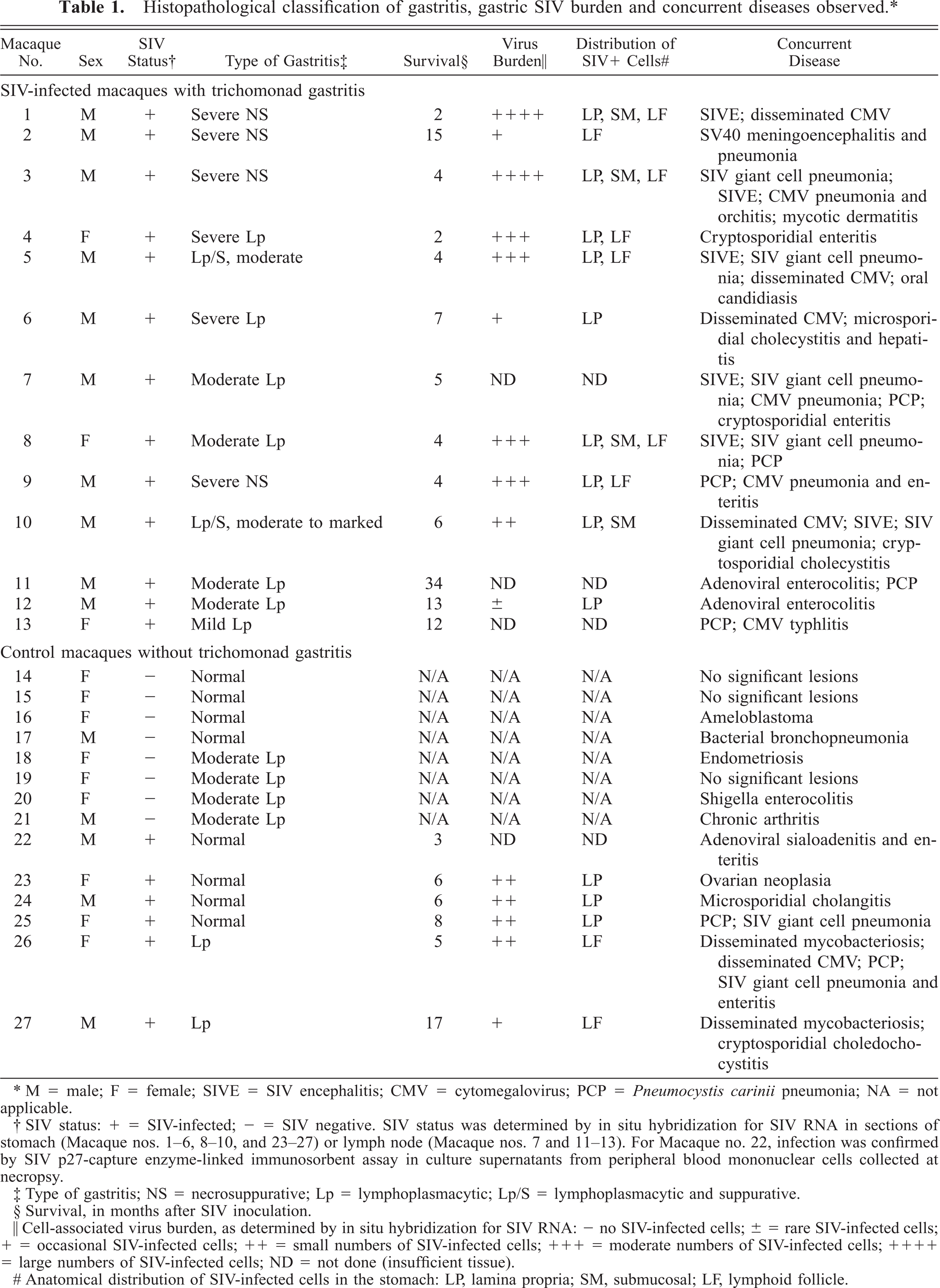
∗M = male; F = female; SIVE = SIV encephalitis; CMV = cytomegalovirus; PCP = Pneumocystis carinii pneumonia; NA = not applicable.
† SIV status: + = SIV-infected; − = SIV negative. SIV status was determined by in situ hybridization for SIV RNA in sections of stomach (Macaque nos. 1–6, 8–10, and 23–27) or lymph node (Macaque nos. 7 and 11–13). For Macaque no. 22, infection was confirmed by SIV p27-capture enzyme-linked immunosorbent assay in culture supernatants from peripheral blood mononuclear cells collected at necropsy.
‡ Type of gastritis; NS = necrosuppurative; Lp = lymphoplasmacytic; Lp/S = lymphoplasmacytic and suppurative.
§ Survival, in months after SIV inoculation.
∥ Cell-associated virus burden, as determined by in situ hybridization for SIV RNA: - no SIV-infected cells; ± = rare SIV-infected cells; + = occasional SIV-infected cells; ++ = small numbers of SIV-infected cells; + + + = moderate numbers of SIV-infected cells; + + + + = large numbers of SIV-infected cells; ND = not done (insufficient tissue).
# Anatomical distribution of SIV-infected cells in the stomach: LP, lamina propria; SM, submucosal; LF, lymphoid follicle.
Histologic analysis
Light microscopy and special stains
Complete necropsies were performed, and all organs were fixed in 10% neutral buffered formalin and embedded in paraffin. Five-micron-thick sections were stained with HE and special stains, including periodic acid–Schiff (PAS), modified Steiner's silver, and Ziehl–Neelsen acid fast.
Immunohistochemistry for cytomegalovirus
Sections of stomach from all cases were examined by IHC for cytomegalovirus (CMV). Paraffin-embedded tissue was sectioned at 5 µm, deparaffinized, rehydrated, and treated with 3% hydrogen peroxide. Antigen retrieval was achieved by microwaving in citrate buffer for 20 minutes, followed by blocking with universal blocking reagents (Dako, Carpinteria, CA) for 10 minutes. Tissues were incubated for 30 minutes with the primary polyclonal antibody (antirhesus CMV IE1, kindly provided by Dr. Peter Barry, University of California at Davis). After washing, sections were incubated with biotinylated goat anti-rabbit IgG for 30 minutes followed by incubation with peroxidase-conjugated streptavidin complex and diaminobenzidine (Vector Laboratories, Burlingame, CA). Sections were counterstained with Mayer's hematoxylin.
In situ hybridization
In situ hybridization for SIV RNA was performed as previously described, 21 using paraffin sections of stomach and a digoxigenin (DIG)-labeled antisense riboprobe (Lofstrand Labs, Gaithersburg, MD) that spans the entire genome of the SIVsmmPGm5.3 molecular clone of SIV. 20 Briefly, deparaffinized and rehydrated tissue sections were hydrolyzed in HCl (Sigma Chemical Co., St. Louis, MO), digested in proteinase K (Roche Diagnostics Corp., Indianapolis, IN), acetylated in acetic anhydride (Sigma), and hybridized overnight with riboprobe at 50 C. Bound probe was detected by IHC, using alkaline phosphatase–conjugated sheep anti-DIG F(ab) fragments (Roche). Tissue sections were developed in the dark for 2 hours at room temperature in the chromogen nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate (NBT/BCIP; Roche) and counterstained with nuclear fast red (Vector Laboratories). Negative controls included matching tissue sections that had been hybridized with DIG-labeled SIV sense riboprobe and tissue sections from SIV-negative macaques hybridized with antisense riboprobe, whereas tissues from two additional SIV-positive monkeys (rhesus macaque Nos. 26 and 27, Table 1) with lymphoplasmacytic gastritis but no intralesional trichomonads were used as positive controls.
Transmission electron microscopy
Archived formalin-fixed tissues from 12 of the 13 cases of protozoal gastritis were examined by transmission electron microscopy (TEM). The sections were washed in 0.1 M phosphate buffer, postfixed in 1% osmium tetroxide, dehydrated in graded ethanols and propylene oxide, and embedded in eponate 12 epoxy resin (Ted Pella, Inc., Redding, CA). Thin sections were prepared on a Sorvall MT-2 ultra-microtome and stained with uranyl acetate and Sato's lead stain. 23 The grids were examined with a Jeol 1010 Electron Microscope (Jeol, Tokyo, Japan).
DNA extraction and polymerase chain reaction
Genomic DNA was extracted from paraffin-embedded sections of stomach from the 13 monkeys with protozoal gastritis, the 12 control monkeys, and a mouse naturally infected with Tritrichomonas muris. Paraffin sections were incubated in lysis buffer (10 mmol/liter tris–HCl, containing 100 mmol/liter KCl, 2.5 mmol/liter MgCl2, and 0.45% Tween 20) for 10 minutes at 95 C. Proteinase K (2 mg/ml) was then added to the lysis buffer, and protease digestion was performed at 65 C for 2 hours. After protease treatment, lysates were heated at 95 C for 10 minutes to denature the proteinase K and then centrifuged for 5 minutes at 6,000 rpm. The aqueous layer was collected from beneath the paraffin layer, and DNA was precipitated by two extractions with phenol/chloroform/isoamyl alcohol, washed twice with 70% ethanol, and quantitated with a spectrophotometer. To confirm the presence of trichomonads and to identify the genus, a 347-bp fragment of the 5.8S ribosomal RNA (rRNA) gene was amplified as previously described. 12 Polymerase chain reaction (PCR) was performed in 50-µl reaction volumes containing 1× PCR reaction buffer, 200 µmol/liter deoxynucleoside triphosphate, 2 U of Taq DNA polymerase (Roche), 1 µmol/liter of each primer, and 100 ng of genomic DNA. Forty cycles of amplification were performed, using primers TFR3 (5′-CGGGTCTTCCTATAT-GAGAGAGAACC-3′) and TFR4 (5′-CCTGCCGTTGGAT-CAGTTTCGTTAA-3′), which are specific for species of Tritrichomonas (e.g., T. foetus, Tritrichomonas suis, and T. mobilensis). Thermal cycling conditions were denaturation at 94 C for 30 seconds, annealing at 67 C for 30 seconds, extending at 72 C for 90 seconds, and a final extension step of 15 minutes at 72 C. Amplification products were observed on an ethidium bromide–stained 2% agarose gel.
Statistical analysis
The relationship between gastric virus burden and survival time was evaluated by simple least-squares linear regression. Significant differences were assumed for probability values of P ≤ 0.05.
Results
Gross and microscopic examination
The gastric mucosa from SIV-infected rhesus macaques with trichomonad gastritis was thickened with multiple irregular erosions and hemorrhages (Fig. 1). Two distinct histopathologic presentations of trichomonad gastritis were observed in affected macaques: necrosuppurative gastritis and lymphoplasmacytic gastritis.
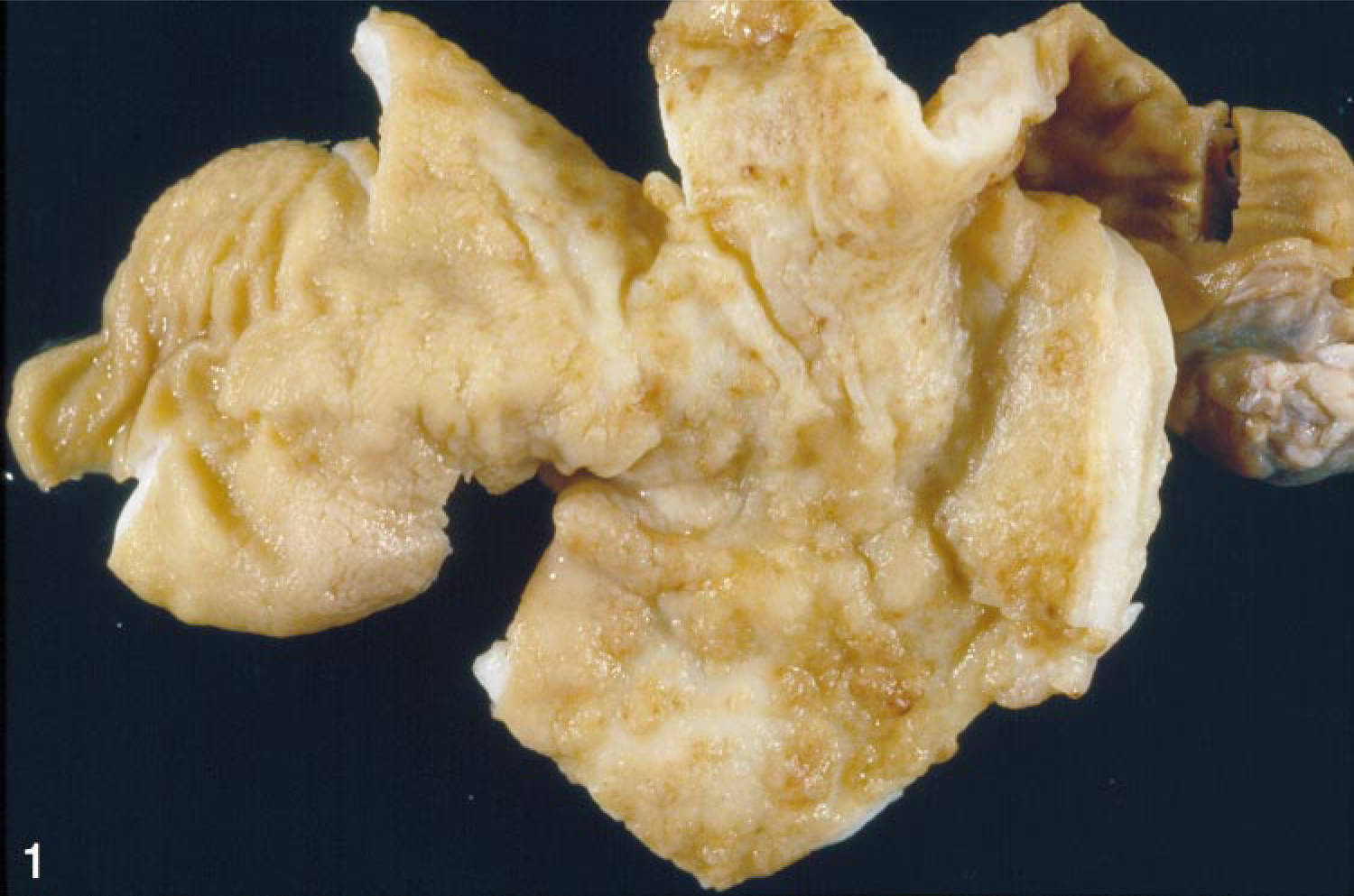
Stomach; SIV-infected rhesus macaque No. 1. The gastric mucosa is roughened and edematous with numerous multifocal to coalescing petechial and ecchymotic hemorrhages.
In the four cases of necrosuppurative gastritis, the lesions were characterized by multifocal erosions or ulcerations (or both) of the gastric mucosa with extension into the submucosa. The inflammation within the mucosa was characterized by infiltrates of moderate to extensive numbers of neutrophils with fewer lymphocytes and macrophages, whereas the lesions within the submucosa were typically composed of multiple circumscribed abscesses (Figs. 2, 3). Small to moderate numbers of round, oval to polygonal, PAS-positive organisms, measuring 4–35 µm in diameter, were present within the necrosuppurative lesions of both the mucosa and submucosa (Figs. 4, 5).
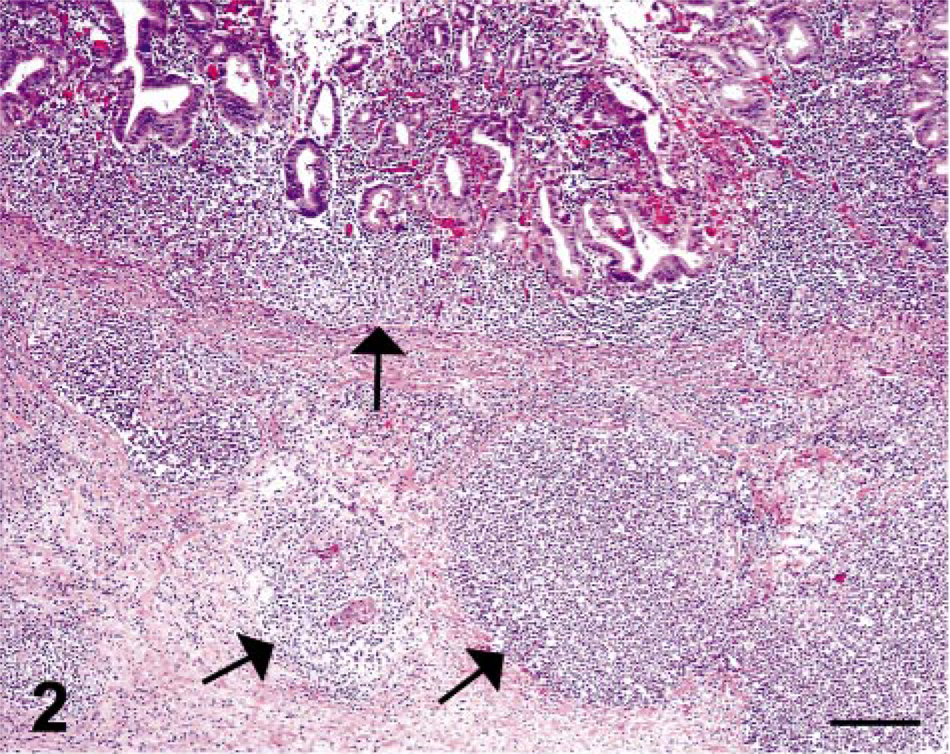
Stomach; SIV-infected rhesus macaque No. 1. Necrosuppurative foci in the gastric mucosa and submucosa (arrows) contain intralesional trichomonads. HE. Bar = 200 µm.
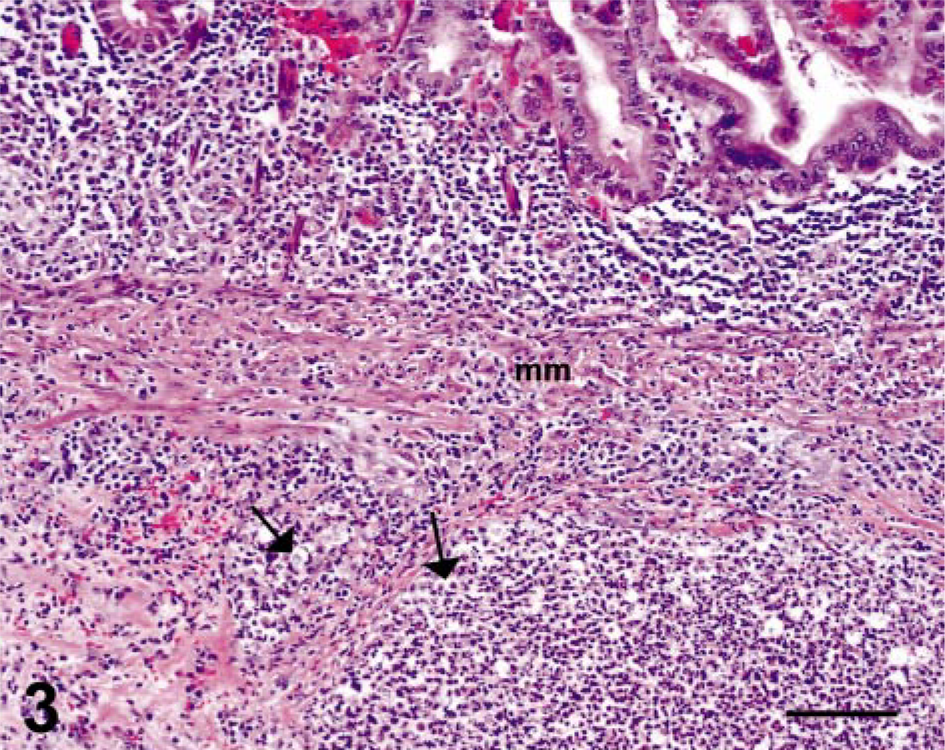
Stomach; SIV-infected rhesus macaque No. 1. Higher magnification of field displayed in Fig. 2 shows trichomonads (arrows) within a submucosal lesion (mm identifies the gastric muscularis mucosae). HE. Bar = 100 µm.
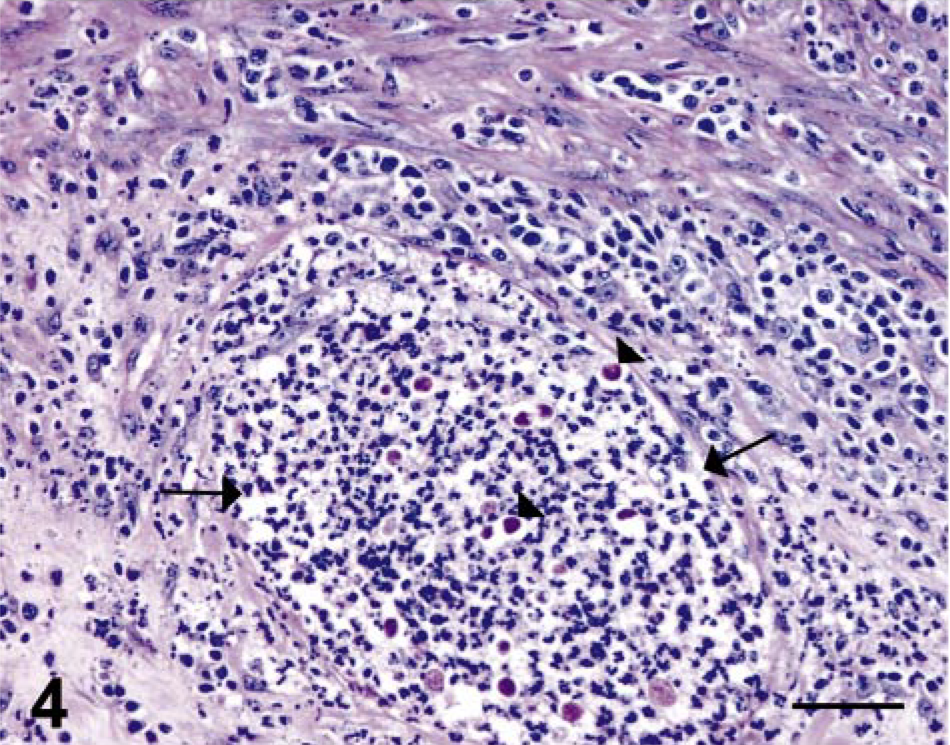
Stomach; SIV-infected rhesus macaque No. 1. PAS-positive protozoa (arrowheads) within a microabscess (delineated by arrows) in the gastric submucosa. Periodic acid–Schiff. Bar = 50 µm.
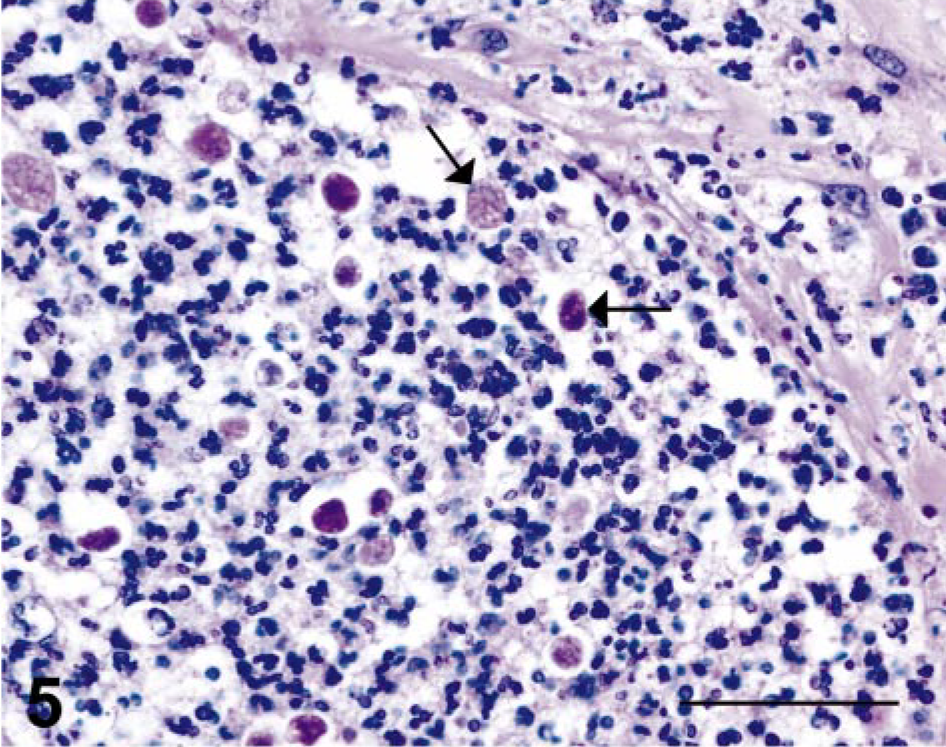
Stomach; SIV-infected rhesus macaque No. 1. PAS-positive protozoa (arrows) of variable sizes and shapes. Periodic acid–Schiff. Bar = 50 µm.
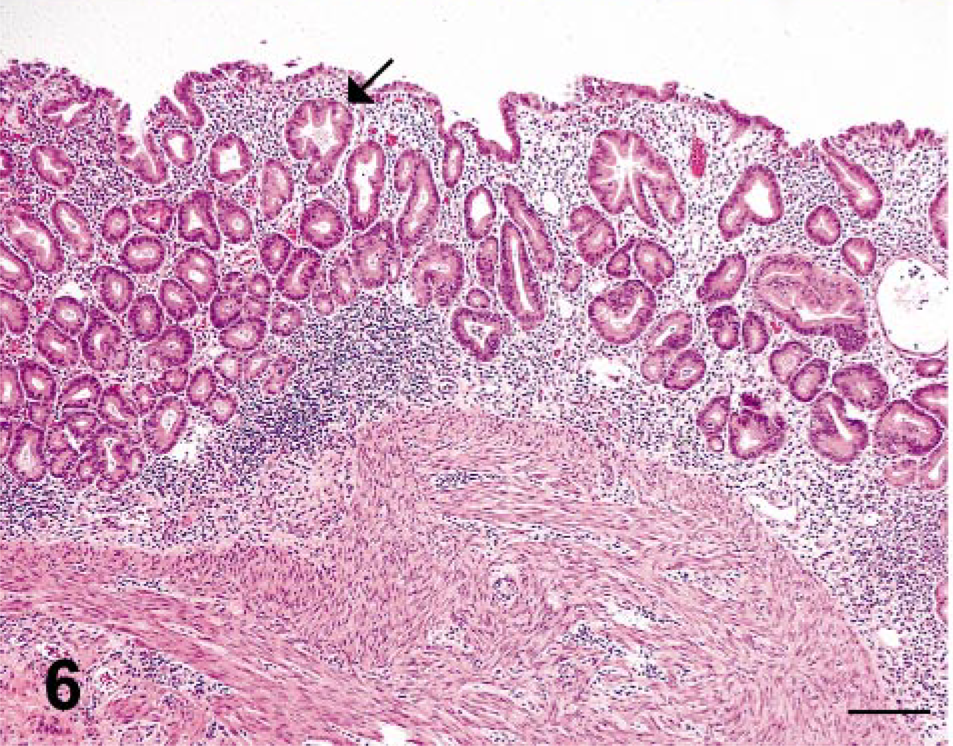
Stomach; SIV-infected rhesus macaque No. 6. Lymphoplasmacytic gastritis is confined to the lamina propria, and often includes dilated gastric glands which contain intraluminal trichomonads (arrow). HE. Bar = 200 µm.
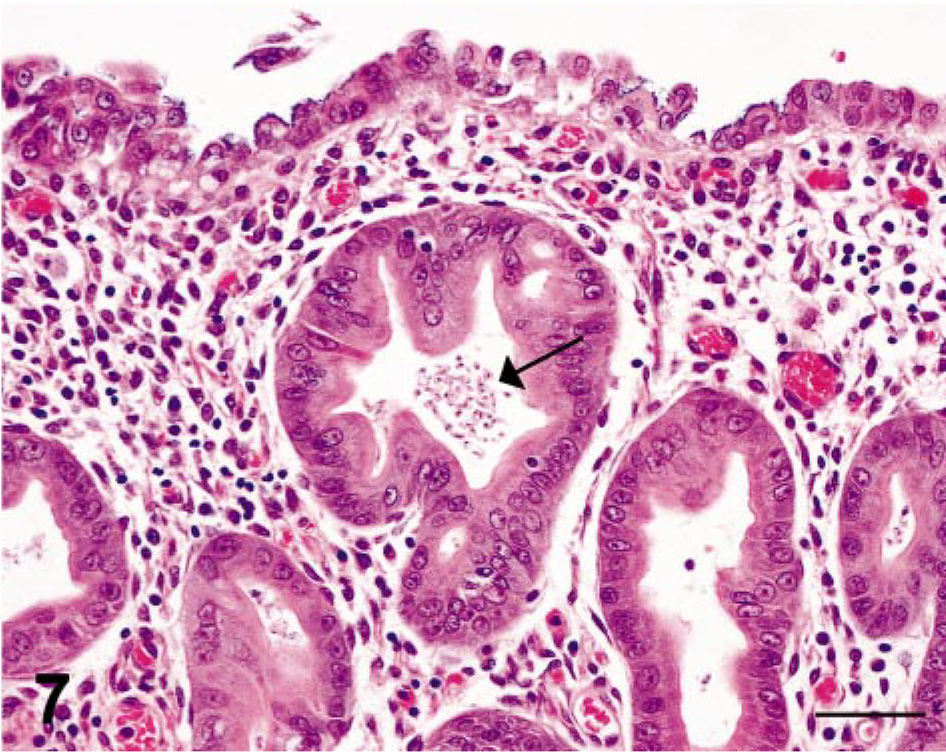
Stomach; SIV-infected rhesus macaque No. 6. Higher magnification of field shown in Fig. 6; arrow identifies an accumulation of tear-shaped trichomonads in the lumen of an ectatic gastric gland. HE. Bar = 50 µm.
Protozoal organisms were also located within the lumen of some gastric glands, occasionally scattered in the lamina propria, and rarely within the cytoplasm of glandular epithelial cells at the periphery of the necrosuppurative lesions. Multifocal necrotizing vasculitis with hemorrhage, fibrin, and edema was seen within the submucosa of two cases of necrosuppurative gastritis.
In the seven cases of lymphoplasmacytic gastritis, the lesions were confined to the mucosa and characterized by a diffuse, mild to moderate infiltrate of lymphocytes, plasma cells, and macrophages with scattered small aggregates of neutrophils within the lamina propria. Small to moderate numbers of 3–5 µm, tear-shaped trichomonads were observed in multiple lumina of dilated gastric glands (Figs. 6, 7). For two cases of predominantly lymphoplasmacytic protozoal gastritis (rhesus macaque Nos. 5 and 10), the histopathologic features included scattered crypt abscesses and ruptured gastric glands accompanied by neutrophilic infiltrates within the gastric lamina propria. In all 13 cases, the number of gastric trichomonads was proportional to the severity of gastritis.
Special stains and IHC
Additional techniques were used to determine whether other infectious agents were associated with the gastritis. All experimental and control cases were negative for acid-fast bacilli. Modified Steiner's silver stain revealed mild to moderate colonization of the gastric mucosa with Helicobacter spp. in 9 of 13 (70%) cases of protozoal gastritis compared with 4 of 12 (33%) control cases. CMV was localized to the lamina propria and submucosa of two monkeys (rhesus macaque Nos. 1 and 9) with severe necrosuppurative gastritis and in one macaque (rhesus macaque No. 6) with severe lymphoplasmacytic gastritis (3/13 or 23%).
Electron microscopy
TEM was performed on stomach tissue collected from 12 of the 13 macaques with histopathologic evidence of protozoal gastritis (there was insufficient archived formalin-fixed stomach tissue from rhesus macaque No. 13 to permit ultrastructural examination). In each of the 12 cases of protozoal gastritis examined, TEM revealed intralesional unicellular organisms with ultrastructural features typical of trichomonads (Fig. 8). Trichomonads had prominent costa with pelta, anterior flagella, recurrent flagellum, a well-defined undulating membrane, and a single nucleus (Fig. 9). Many of the intralesional trichomonads in the severe necrosuppurative lesions were round to oval and 10–35 µm in diameter with invagination of the flagella, which is characteristic of the intermediate form of trichomonads (Fig. 10).
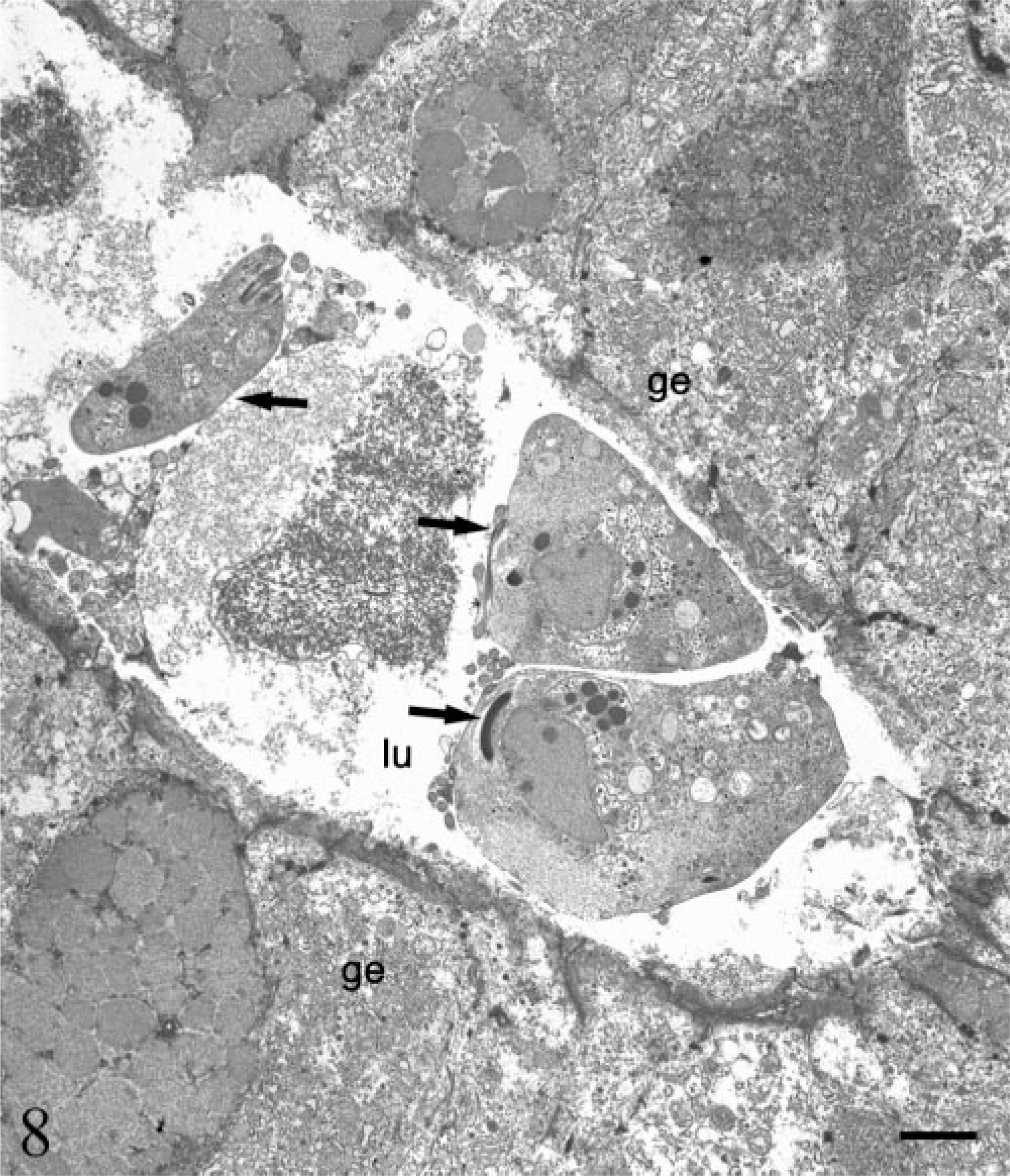
Electron micrograph. Stomach; SIV-infected rhesus macaque No. 2. Three trichomonads (arrows) are present in the lumen (lu) of a gastric gland, ge = gastric epithelial cell. Bar = 2 µm.
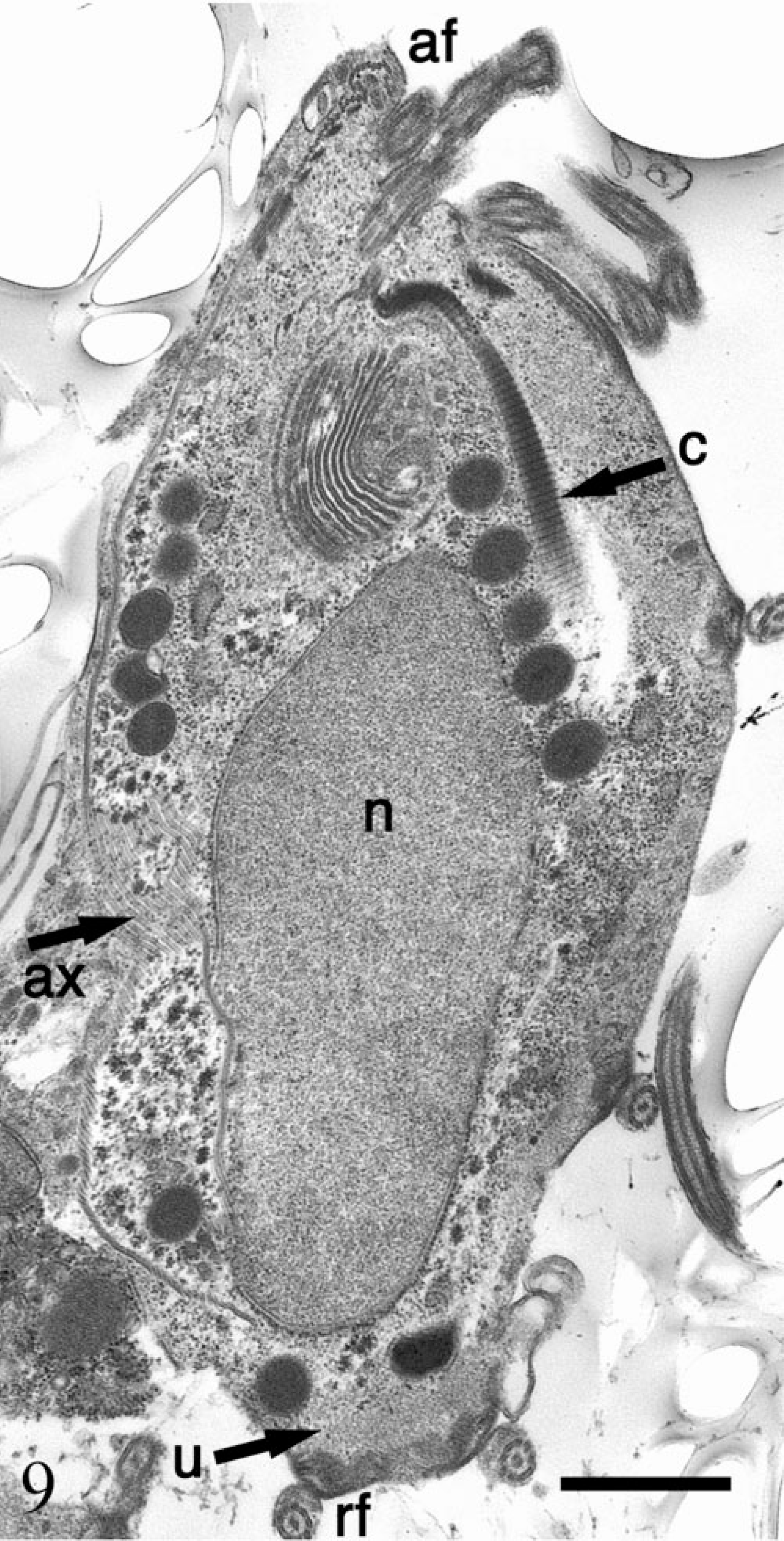
Electron micrograph. Stomach; SIV-infected rhesus macaque No. 4. Trichomonad trophozoites with anterior flagella (af), axostyle (ax), prominent costa (c), nucleus (n), recurrent flagellum (rf), and undulating membrane (u). Bar = 1 µm.
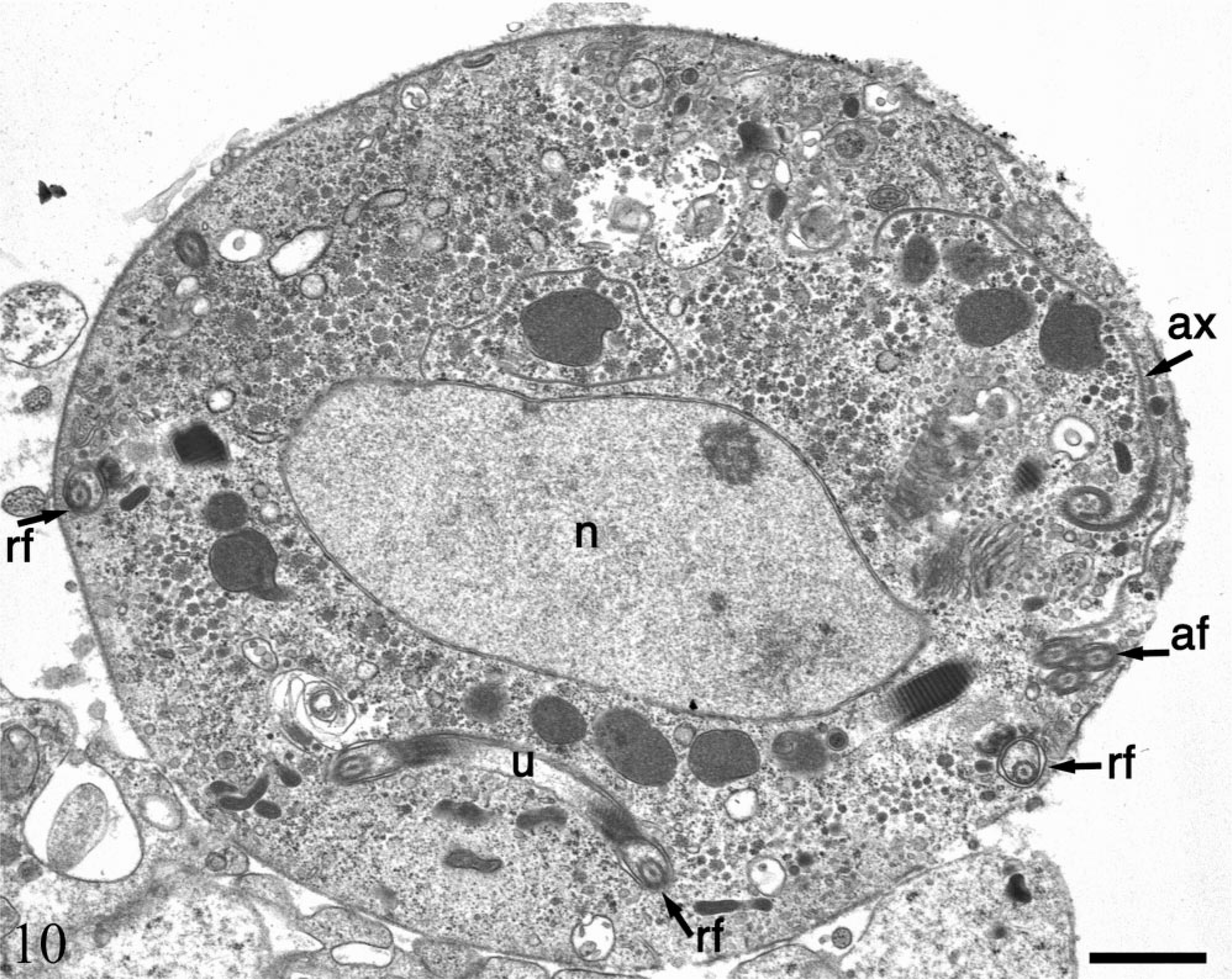
Electron micrograph. Stomach; SIV-infected rhesus macaque No. 1. Ultrastructural features consistent with an intermediate form of trichomonad include three internalized anterior flagella (af), axostyle (ax), nucleus (n), internalized recurrent flagellum (rf) and undulating membrane (u). Bar = 1 µm.
Polymerase chain reaction
A 347-bp product was amplified by PCR using genomic DNA extracted from the gastric mucosa from each of the 13 cases with protozoal gastritis (Fig. 11). In contrast, no amplification product was observed in reactions using DNA extracted from control macaque tissues or control mouse tissue. The TFR3/TFR4 primer set amplifies a product of the same size from T. foetus, T. suis, and T. mobilensis confirming the classification of the protozoa as a species of Tritrichomonas. 12
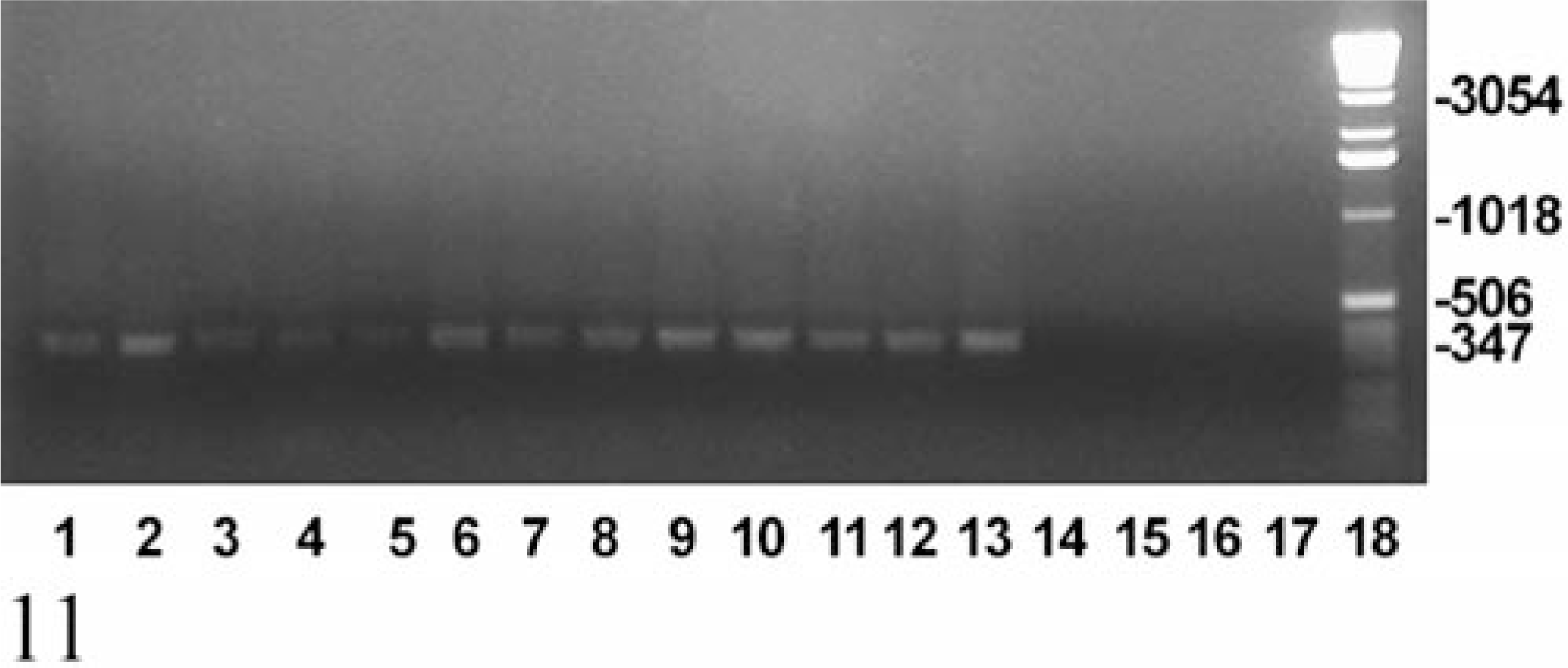
Ethidium bromide–stained agarose gel showing the results of PCR for a 347-bp fragment of the 5.8S rRNA gene of Tritrichomonas spp. Genomic DNA, extracted from stomach tissue of macaques with gastric trichomoniasis (lanes 1–13 as per Table 1), representative macaques from the first, second, and third control groups (lanes 14, 15, and 16, respectively), and a mouse naturally infected with Tritrichomonas muris (lane 17), was subjected to PCR amplification, using primers TFR3 and TFR4, which are specific for Tritrichomonas foetus, Tritrichomonas suis, and Tritrichomonas mobilensis. Lane 18 = 1-kb DNA ladder.
In situ hybridization
In situ hybridization for SIV revealed moderate to very high (+++ or ++++) gastric virus burdens in four of the six macaques with necrosuppurative gastritis (Table 1); SIV-positive cells were distributed in the mucosal lamina propria, submucosa, and gastric lymphoid follicles of these animals (Figs. 12, 13). In the cases of lymphoplasmacytic gastritis, scattered SIV-positive cells were found only in the lamina propria or lymphoid follicles (Figs. 14, 15), similar to what is typically seen in SIV-positive macaques without gastritis (Figs. 16, 17).
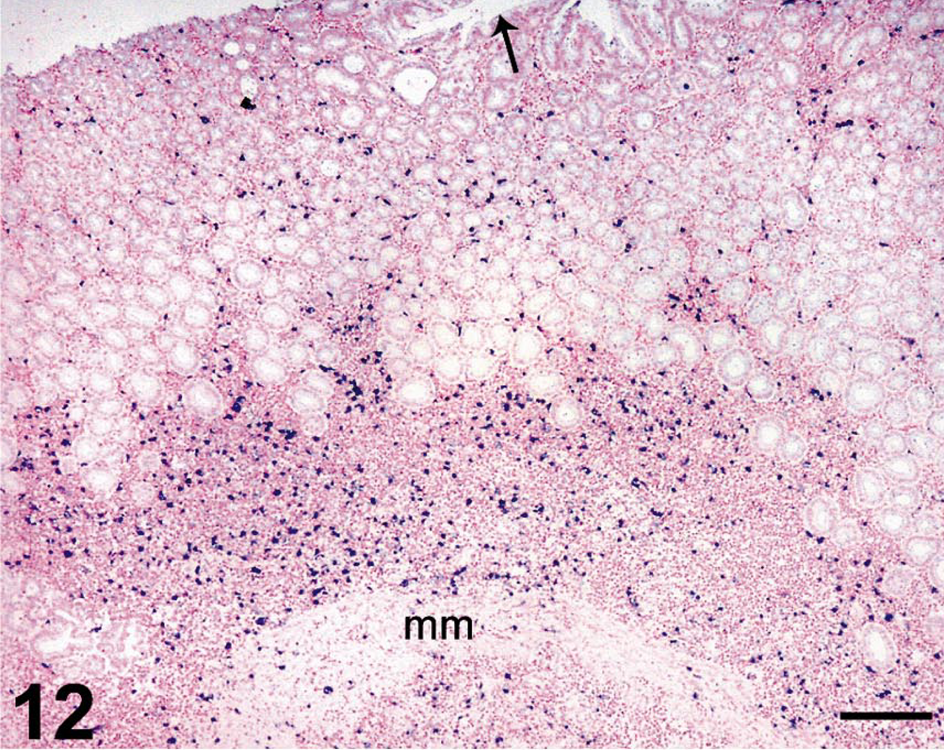
Stomach; rhesus macaque No. 3. In situ hybridization for SIV RNA, showing large numbers of SIV-infected cells (dark blue) throughout the gastric mucosa of a SIV-positive macaque with necrosuppurative gastritis and intralesional trichomonads. SIV-infected cells extend beneath the gastric muscularis mucosae (mm) and into the submucosa. Arrow identifies gastric lumen. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 200 µm.
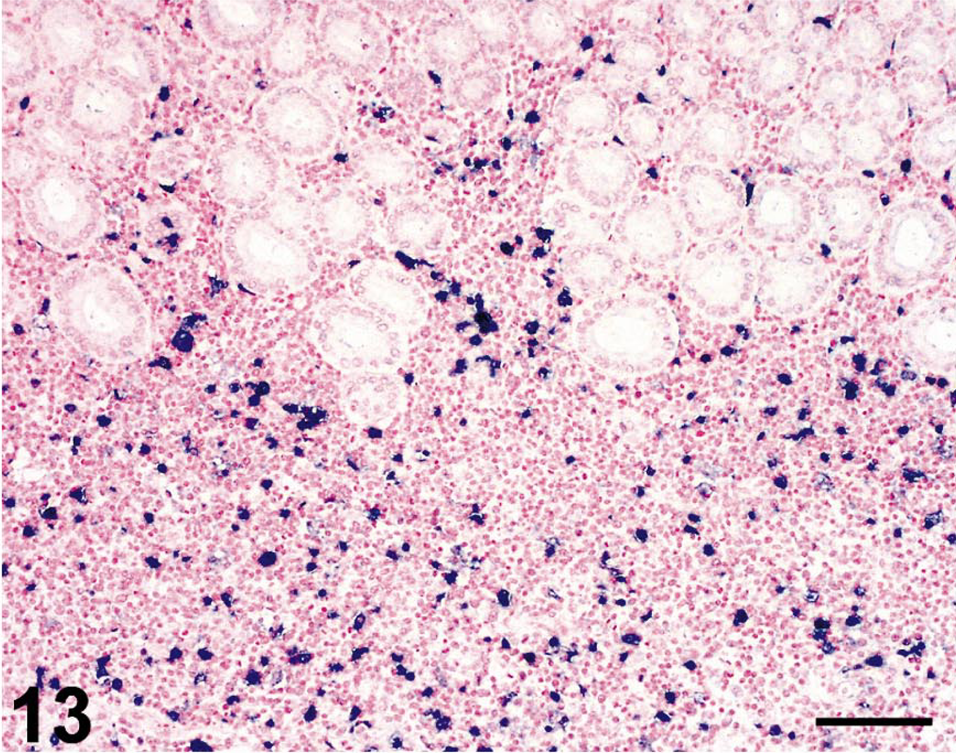
Stomach; rhesus macaque No. 3. Higher magnification of field shown in Fig. 12, revealing large numbers of SIV-infected cells (dark blue) throughout the gastric lamina propria. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 100 µm.
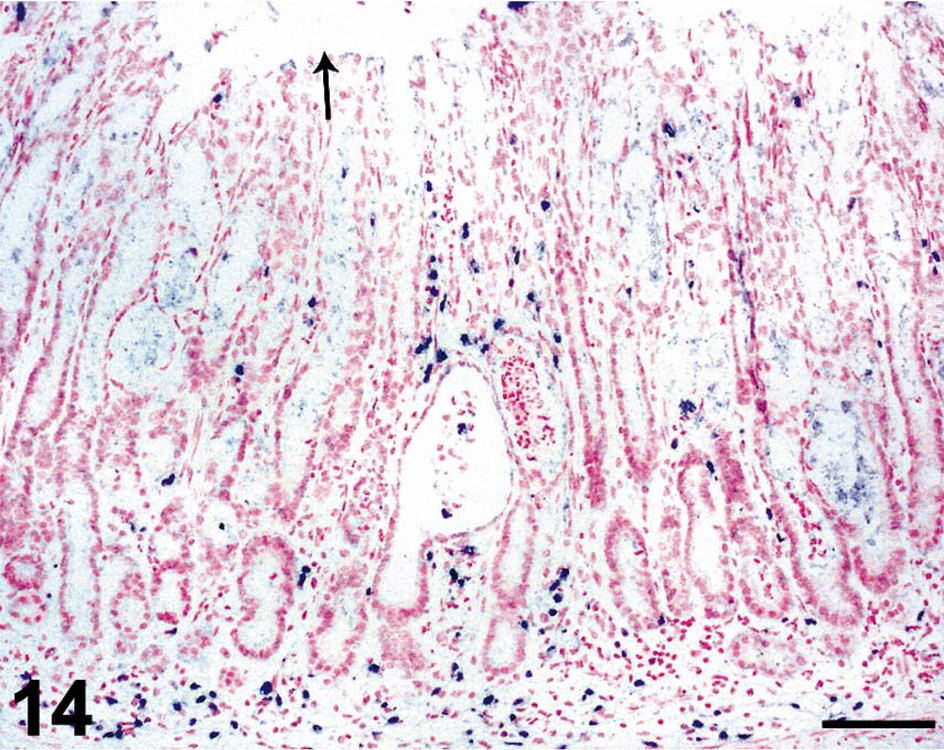
Stomach; rhesus macaque No. 4. In situ hybridization for SIV RNA, showing moderate numbers of SIV-infected cells (dark blue) throughout the gastric mucosa of a SIV-positive macaque with lymphoplasmacytic gastritis. Arrow indicates location of gastric lumen. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 100 µm.
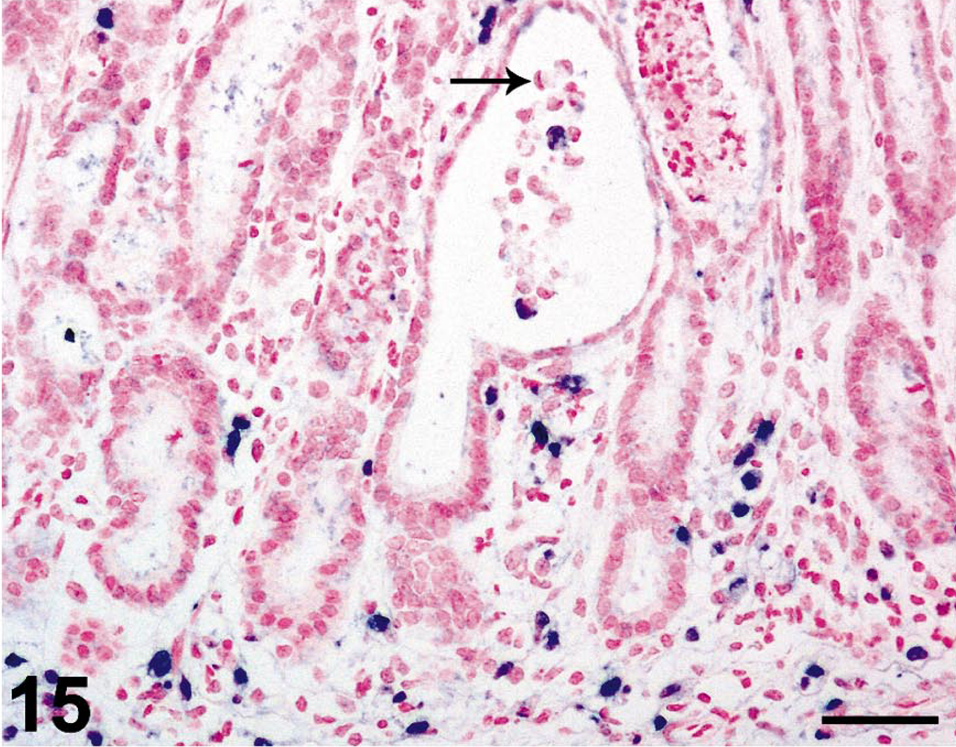
Stomach; rhesus macaque No. 4. Higher magnification of field depicted in Fig. 14, showing intralesional trichomonads (arrow) within an ectatic gastric gland. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 50 µm.
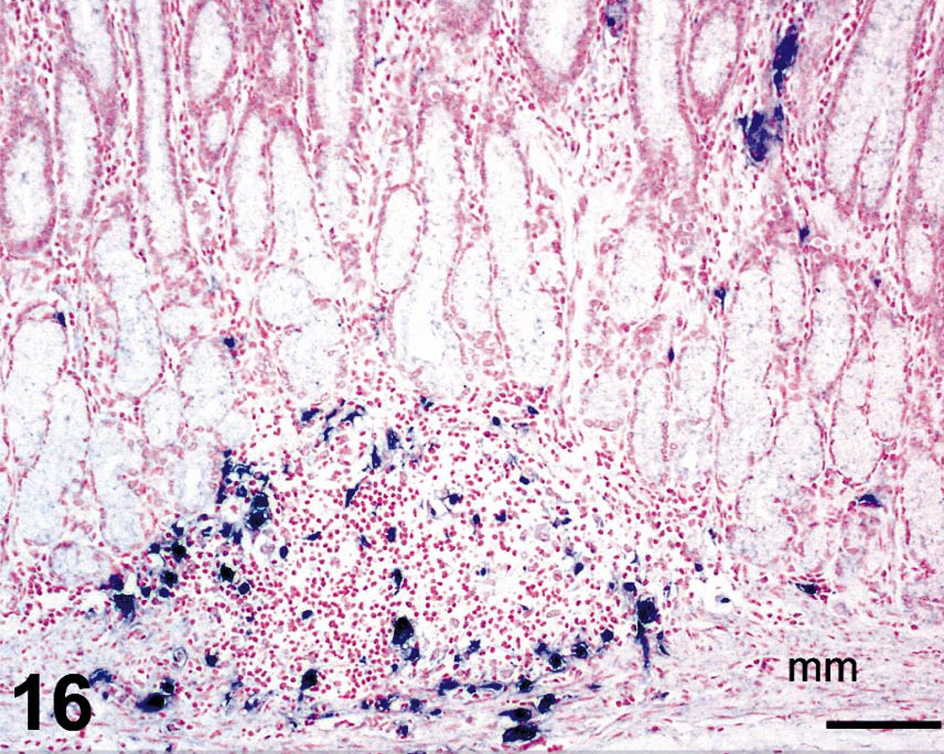
Stomach; rhesus macaque No. 26. In situ hybridization for SIV RNA on a section of stomach from a SIV-positive macaque without gastric trichomoniasis. In contrast to macaques with gastric trichomoniasis, SIV-positive cells are fewer in number in the lamina propria and are primarily located within mucosal lymphoid follicles. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 100 µm.
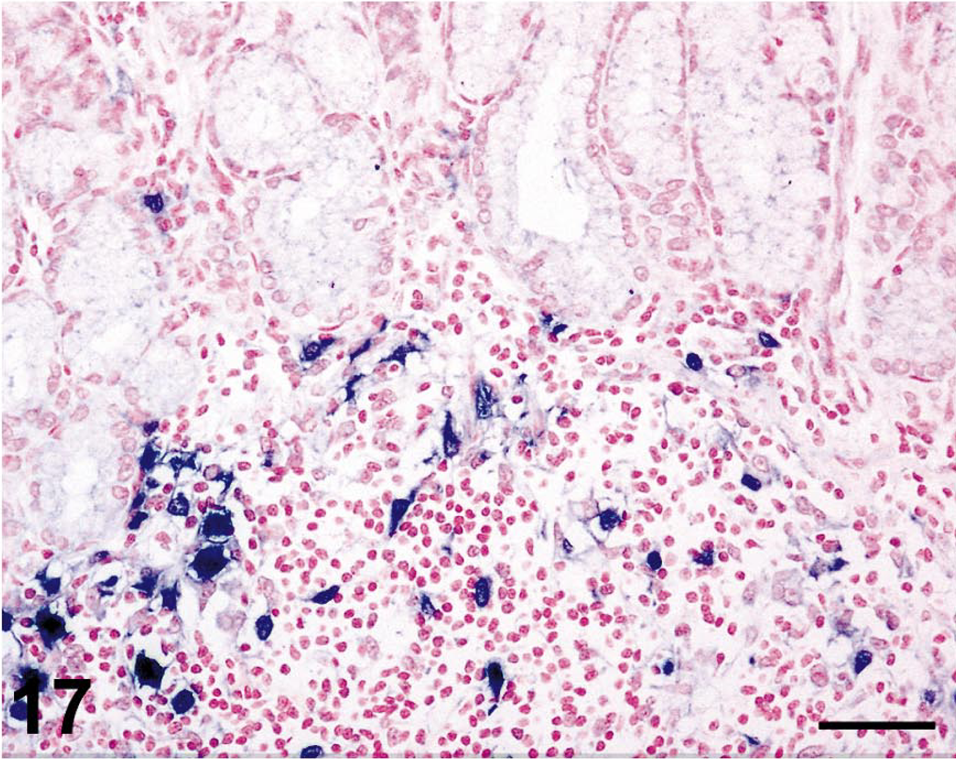
Stomach; rhesus macaque No. 26. Higher magnification of field shown in Fig. 16, showing SIV-infected cells within a mucosal lymphoid follicle. Nitroblue tetrazolium/5-bromo-4-chloro-3-indolyl-phosphate chromogen (NBT/BCIP) with nuclear fast red counterstain. Bar = 50 µm.
Gastric SIV burden was inversely correlated with survival time (P < 0.002). Five of the six macaques with necrosuppurative gastritis were rapid acquired immunodeficiency syndrome (AIDS) progressors, surviving less than 6 months after inoculation. In contrast, only three of the seven macaques with lymphoplasmacytic trichomonad gastritis were rapid progressors.
Discussion
In this study, we show that Tritrichomonas can be associated with the development of necrosuppurative gastritis in SIV-infected rhesus macaques. In the cases of lymphoplasmacytic gastritis, light microscopy revealed that protozoal organisms within the lumens of gastric glands had morphologic features that were typical of trichomonad trophozoites. In contrast, the protozoa in the four cases of necrosuppurative gastritis and the two cases with combined necrosuppurative and lymphoplasmacytic gastritis possessed atypical morphologic features that did not permit definitive classification by light microscopy. In all cases, the ultra-structural characteristics of the protozoa, including anterior and recurrent flagella, an undulating membrane, an axostyle, and a costa with distinct banding, were compatible with Trichomonas spp. 9 , 24 Confirmation of the protozoa as tritrichomonads was accomplished by PCR using primers specific for a 347-bp portion of the 5.8S rRNA of T. mobilensis. 10–12 Because T. mobilensis is the only known enteric trichomonad of lower and New World primates (tree shrews and squirrel monkeys, respectively), 6 , 25 whereas T. foetus and T. suis are pathogens of cattle and swine, respectively, we propose that the tritrichomonads identified in this study may be T. mobilensis. However, further studies are warranted to conclusively identify the species of gastrointestinal tritrichomonads in rhesus macaques.
Although trichomonads were present within the lumens of gastric glands in the cases of lymphoplasmacytic gastritis, it is unlikely that the trophozoites incited the mononuclear inflammatory cell infiltrate in the lamina propria. In comparison, the necrosuppurative gastric lesions were likely induced by the intra-lesional trichomonads. The two cases of gastritis with combined histopathologic features of lymphoplasmacytic and necrosuppurative gastritis may represent a transition between these two manifestations of gastritis. Rupture of the gastric glands may facilitate the spread of the trichomonads from the lumens of the gastric glands into the lamina propria.
The mechanism of entry of trichomonads into the gastric mucosa is unknown. SIV infection of gut-associated lymphoid tissue may alter the local gastrointestinal microenvironment, creating conditions (e.g., decreased gastric acidity and impaired immune function) that favor the colonization of gastric glands by trichomonads. The gastrointestinal tract is a primary site of SIV viral amplification and persistence during pathogenic progression to AIDS. 16 , 26 , 27 Opportunistic infections are common in the gastrointestinal tract of HIV-infected humans and SIV-infected macaques; however, there is also abundant evidence to support a primary lentiviral etiology in the pathogenesis of AIDS enteropathy, a diarrheal and wasting syndrome of humans and macaques that occurs in the absence of opportunists. 16 , 27 We speculate that factors induced by SIV infection, including hypochlorhydria or inadequate mucosal inflammatory responses, may promote colonization by protozoal organisms.
In this study, Helicobacter pylori was found in 70% of the cases with trichomonad gastritis as compared with 33% of the controls. It is unknown if H. pylori is a predisposing factor in the development of trichomonad gastritis. There is evidence that H. pylori induces local expression of certain monocyte/macrophage–derived inflammatory cytokines (e.g., interleukin 1, interleukin 6, and tumor necrosis factor-alpha), 15 many of which are also expressed during the course of SIV infection.
Many of the trichomonads in the severe necrosuppurative lesions were round to oval and 10–35 µm in diameter, and TEM examination revealed internalization of the flagella in organisms bearing this morphology. Intermediate and pseudocyst forms have been described for T. foetus and T. muris; 14 , 17 , 25 at the ultra-structural level, the intermediate form of Tritrichomonas is identified by its oval to pear shape and internalized flagella, whereas the pseudocyst form is round, with internalized flagella and a trilaminar wall. It is likely that other species of Tritrichomonas can develop similar intermediate and pseudocyst forms under suitable environmental conditions. The predominance of the intermediate rather than the trophozoite form of Tritrichomonas in the cases of necrosuppurative and combined lymphoplasmacytic/necrosuppurative gastritis would explain the atypical appearance of trichomonads reported here. Based on the findings of this study, tritrichomonads should be considered as a possible pathogen in SIV-infected macaques.
Footnotes
Acknowledgements
This work was supported by NIH grants RR00168, DK55510, and RR07000. We thank Karen Boisvert, Courtney Romsey, Michael O'Connell, and Kristen Toohey for technical support and Douglas Pauley for a critical review of the manuscript.
