Abstract
Cytomegalovirus (CMV)-associated gastrointestinal masses have been reported in human acquired immune deficiency syndrome patients. This is the first report on CMV-associated gastrointestinal masses in simian immunodeficiency virus (SIV)-infected macaques. Two SIV-infected macaques presented at necropsy with multiple nodular or umbilicated masses within the gastrointestinal tract. In one animal, the masses were located throughout the gastrointestinal tract, whereas in the other, the masses were restricted to the proximal small intestine. Grossly, the masses were indistinguishable from those caused by neoplastic conditions such as lymphoma and, histologically, were composed of hyperplastic glandular tissue, dense neutrophilic infiltrates within the lamina propria, and multifocal proprial hemorrhage. Frequent cytomegalic cells with basophilic intranuclear inclusions were found in affected regions. Immunohistochemistry for CMV demonstrated frequent immunopositive cells within affected areas. Furthermore, immunohistochemistry for the proliferation marker Ki-67 demonstrated increased proliferation in hyperplastic glands and crypts. CMV should be considered a cause of discrete mass lesions in the gastrointestinal tract of SIV-infected macaques.
Cytomegaloviruses (CMV) are members of the family Herpesviridae, subfamily Betaherpesvirinae. 4 CMV are host-specific viruses that infect humans, nonhuman primates, and a number of other species. 4 Human cytomegalovirus (HCMV) infects between 40 and 100% of individuals, depending on the population sampled. 2,5 In immunocompetent hosts, HCMV is usually a latent, persistent infection but can be reactivated in immunocompromised states. 5 HCMV is the most common opportunistic infection in patients with acquired immune deficiency syndrome (AIDS), affecting up to 40% of AIDS patients, and infection of the alimentary tract is the most frequent extraocular manifestation of HCMV in AIDS patients. 2 Although the esophagus and colon are the most commonly affected sites, all portions of the alimentary tract are susceptible to infection. 2,5 HCMV infection of the gastrointestinal tract usually causes erosive to ulcerative lesions that can progress to transmural necrosis and perforation. 5 CMV-associated focal mass lesions in the gastrointestinal tract of AIDS patients are rare, but they are being reported with increasing frequency. 11,13 Mass lesions in the gastrointestinal tracts of AIDS patients are almost invariably associated with Kaposi's sarcoma and non-Hodgkin's lymphoma. 3 The gross appearance of CMV-associated mass lesions can result in misdiagnosis of neoplasia in these cases.
Simian immunodeficiency virus (SIV)–infected rhesus macaques (Macaca mulatta) are well established as an excellent model of AIDS, whereas SIV-infected pig-tailed macaques (Macaca nemestrina) have also become a frequently used model of AIDS in recent years. 9,12 Simian AIDS shares extensive similarities with its counterpart in humans, including the spectrum of opportunistic infections encountered. 9 CMV infection is the most common opportunistic viral infection in both HIV-infected humans and SIV-infected macaques. 2,8 In both humans and macaques, the tissues most commonly involved in CMV infection include the brain, lung, lymph node, liver, spleen, intestine, testicle, nerves, and arteries. 1,2 CMV-associated lesions are characterized by a predominantly neutrophilic infiltrate with variable numbers of cytomegalic and inclusion-bearing cells. 1
Analogous to the disease in human AIDS patients, CMV infection of the alimentary tract in SIV-infected rhesus macaques is common. 1 CMV gastrointestinal lesions in SIV-infected macaques typically consist of dense sheets of neutrophils with focal erosions and ulceration. 1,9,10 Mass lesions in the alimentary tract of SIV-infected macaques are most often due to lymphoma and far less frequently due to mycobacterial infection. 9,10 CMV-associated masses of the gastrointestinal tract of SIV-infected macaques have not previously been documented. In this study, we present two cases of CMV-related gastrointestinal masses in two species of macaques that were infected with SIV.
Macaque No. 1 was an adult female rhesus macaque (M. mulatta) that presented 1 year post-SIVmac316 inoculation for neurologic signs, pyrexia, and diarrhea. Abdominal palpation and ultrasound revealed irregularly thickened small and large intestines and a fluid-filled stomach. The animal was treated with intravenous cefazolin for 6 days. The animal became moribund and was euthanatized 7 days after the initial presentation. On postmortem examination, multiple, 0.5-to 1.5-cm, raised, hemorrhagic, umbilicated mucosal masses were found in the gastric fundus and in the small and large intestines, with the ileum, cecum, and proximal colonic mucosa being the most severely affected (Figs. 1, 2).
Small intestine; M. mulatta, macaque No. 1. Multiple, raised, reddened, umbilicated mucosal masses. Inset: Cross sectional view demonstrates polypoid masses in the gastrointestinal mucosa of macaque no. 1.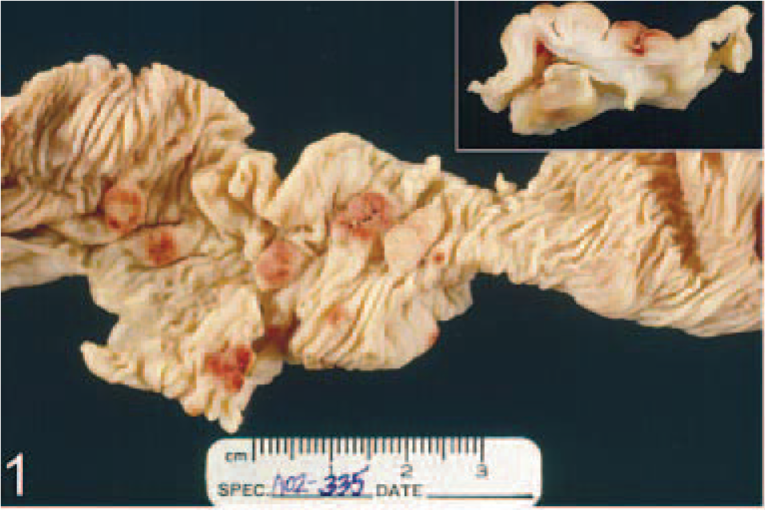
Small intestine; M. nemestrina, macaque No. 2. Multiple, smooth, tan, raised mucosal masses.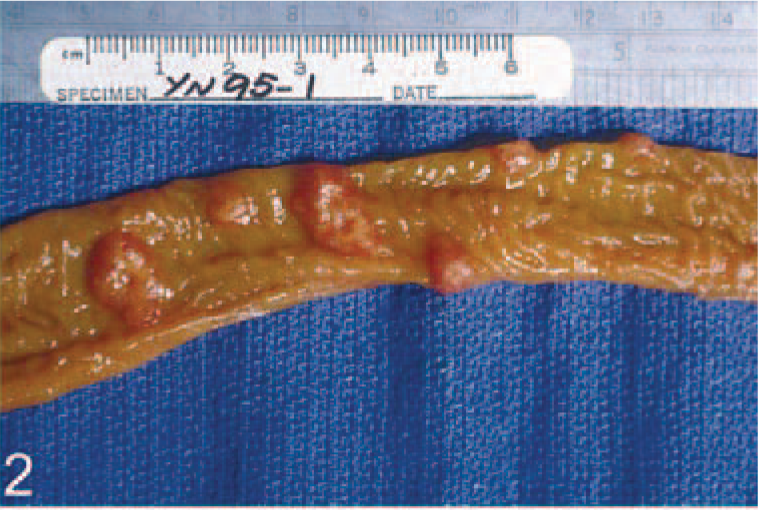
Histopathologic examination of the gastrointestinal masses revealed marked infiltration and expansion of the lamina propria by neutrophils and fewer macrophages, lymphocytes, and plasma cells (Fig. 3), with multifocal extension of inflammation into the submucosa and muscularis externa. Within the areas of inflammation, frequent enlarged cells (cytomegalic cells) contained large, basophilic to amphophilic intranuclear inclusions surrounded by clear halos (Fig. 3) and frequent macrophage-like cells that had abundant cytoplasm filled with variably sized eosinophilic inclusions (Fig. 3). Occasional intranuclear inclusions were seen in proprial stromal cells, macrophages, smooth muscle cells, and in endothelial cells. Moderate to marked hemorrhage and edema were associated with necrotizing vasculitis in the superficial mucosa and submucosa. Gastric glands and intestinal crypts in affected areas were tortuous and lined by hyperplastic epithelium, with frequent mitotic figures extending the length of the glands. Additional histopathologic findings include suppurative meningoencephalitis, myelitis, and neuritis of the cauda equina with cytomegalic cells with intranuclear inclusions, and lymphoproliferative disease characterized by marked splenic lymphoid hyperplasia and lymphoid nodules in multiple organs, including kidney, lung, and liver.
Small intestine, mucosa, lamina propria; macaque No. 1. Cytomegalic cell with intranuclear inclusion and infiltration of the lamina propria by neutrophils. HE. Bar = 30 µm.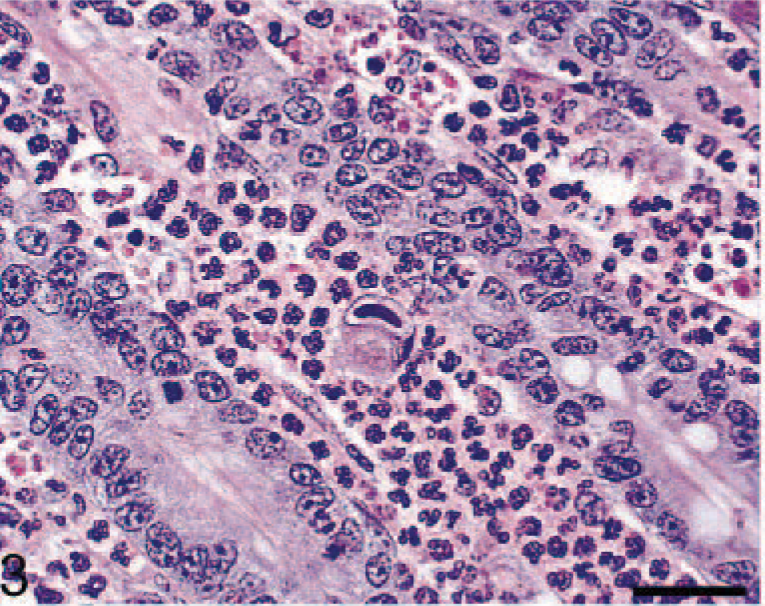
Macaque No. 2 was an adult male pig-tailed macaque (M. nemestrina) that was euthanatized 11 weeks post-SIVpgm/mln inoculation at the Yerkes National Primate Research Center due to progressive neurologic signs, weight loss, anorexia, and dyspnea. Macroscopic findings included 25–30 slightly raised, mostly pale tan nodules, which ranged from 0.3 to 1.0 cm in width, in the upper half of the small intestine (Fig. 2). Smaller nodules were pale tan, whereas larger nodules were more reddened and finely umbilicated. Histologically, the masses were composed of dense neutrophilic infiltrates within the lamina propria and crypt hyperplasia, similar to macaque No. 1 (Fig. 4). Lymphatic vessels in the villar tips were often greatly distended (Fig. 5). Numerous cytomegalic cells with typical CMV intra-nuclear inclusions were found within the areas of inflammation.
Small intestine, mucosa; macaque No. 2. Hyperplasia of crypt epithelium, infiltration of the lamina propria by neutrophils, macrophages, lymphocytes, and plasma cells. Scattered macrophages with pyknotic nuclei containing numerous eosinophilic cytoplasmic inclusions. HE. Bar = 65 µm.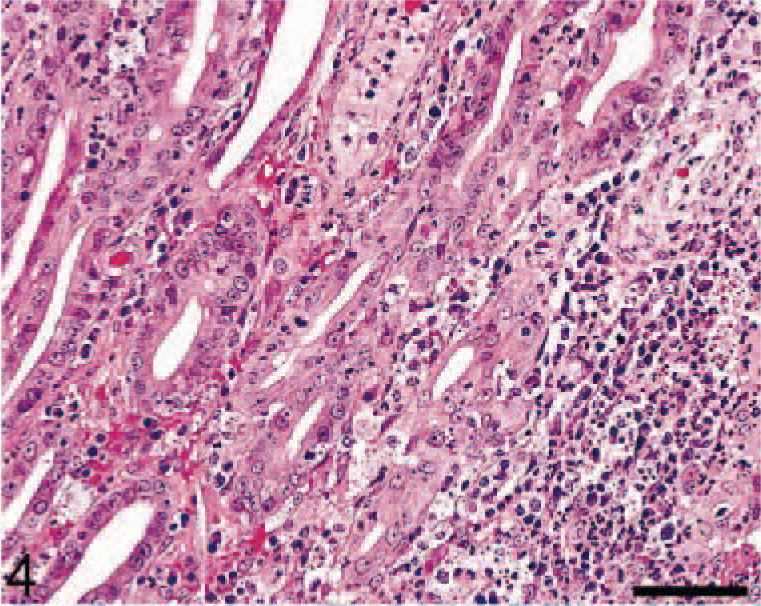
Small intestine; macaque No. 2. Polypoid mucosal thickening with crypt hyperplasia, edema, and hemorrhage. HE. Bar = 500 µm.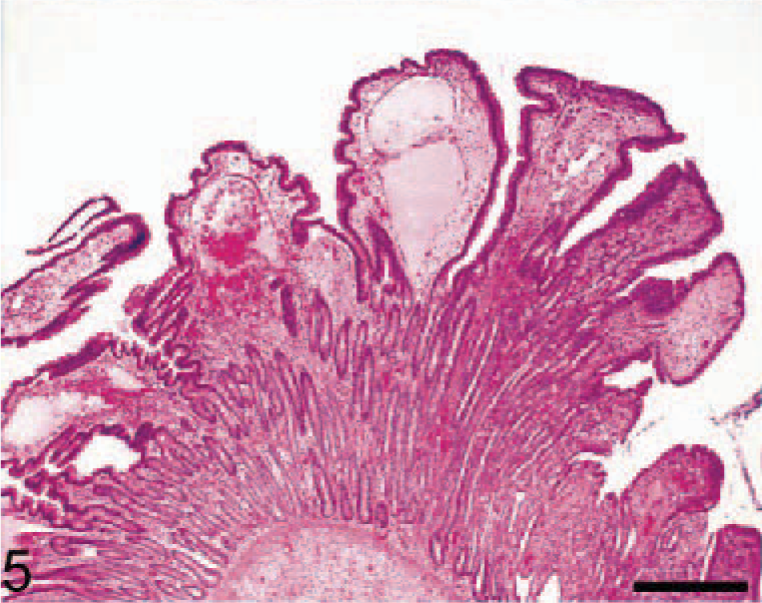
Immunohistochemistry for Ki-67, a proliferation marker, performed using an anti-human Ki-67 monoclonal antibody (MIB-1 clone, DAKO, Carpenteria, CA) demonstrated an increased number of Ki-67–positive mononuclear cells within the lamina propria (Figs. 6, 7) and submucosa of affected regions of the gastrointestinal tracts of both animals. Ki-67–positive epithelial cells could be identified within hyperplastic gastric glands and intestinal crypts throughout the length of the glands and crypts (Figs. 6, 7). Immunopositivity for Ki-67 was restricted to the basal third of normal gastric glands and intestinal crypts.
Small intestine, mucosa; macaque No. 2. Glandular epithelial cells and scattered cells within lamina propria immunopositive for Ki-67 (nuclear, diffuse). Avidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 65 µm.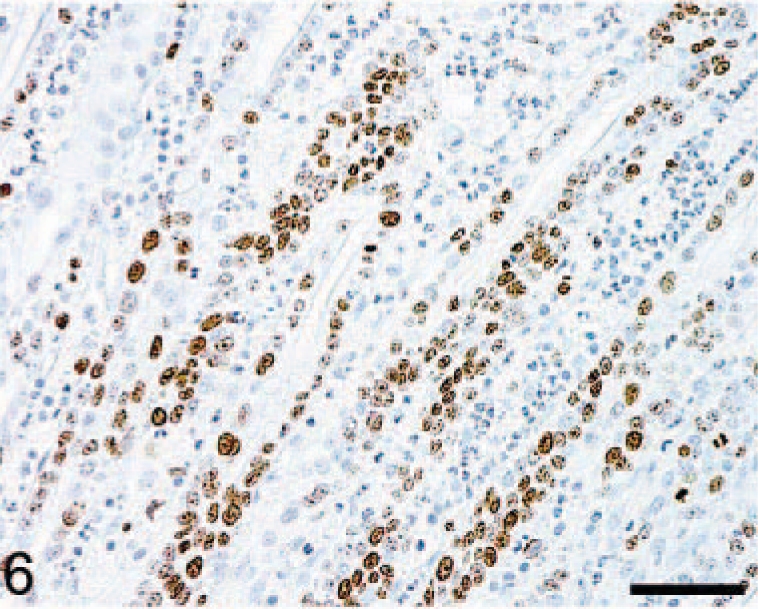
Small intestine; macaque No. 2. Polypoid mucosal thickening with hyperplastic crypt epithelium immunopositive for Ki-67. Avidin–biotin complex method, Mayer's hematoxylin counterstain. Bar = 500 µm.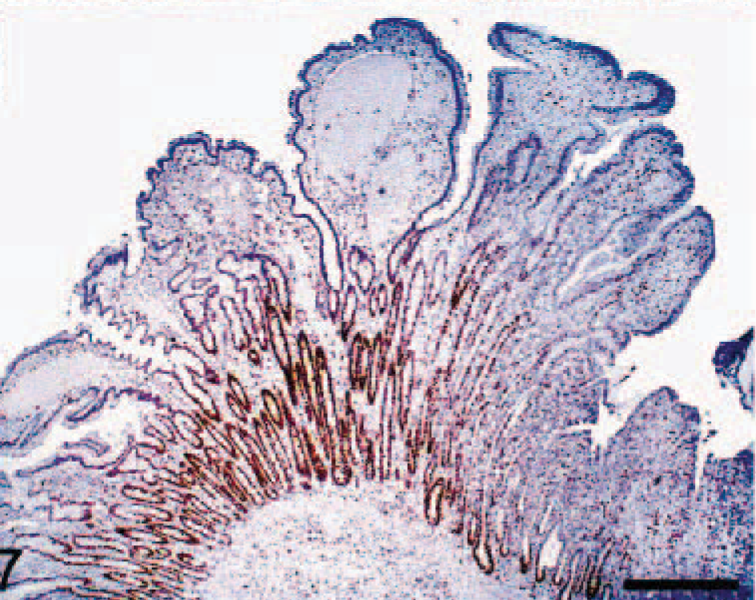
Immunohistochemistry using an anti-human CMV monoclonal antibody (clone CCH2, DAKO) was performed using a previously described avidin–biotin immunostain technique 7 and demonstrated numerous positive cells within the lamina propria (Fig. 8), submucosa, and muscularis externa of affected regions of the gastrointestinal tract. The location and morphology of the cells were consistent with endothelial cells, macrophages, smooth muscle cells, and proprial stromal cells. CMV-positive cells were not identified in unaffected regions of the gastrointestinal tract.
Colon, mucosa, lamina propria, macaque No. 1. Multiple cells, intensely positive with anti-CMV monoclonal antibody (cytoplasmic and nuclear, diffuse, finely granular). Avidin–biotin complex method, Mayer's hematoxylin counter-stain. Bar = 30 µm.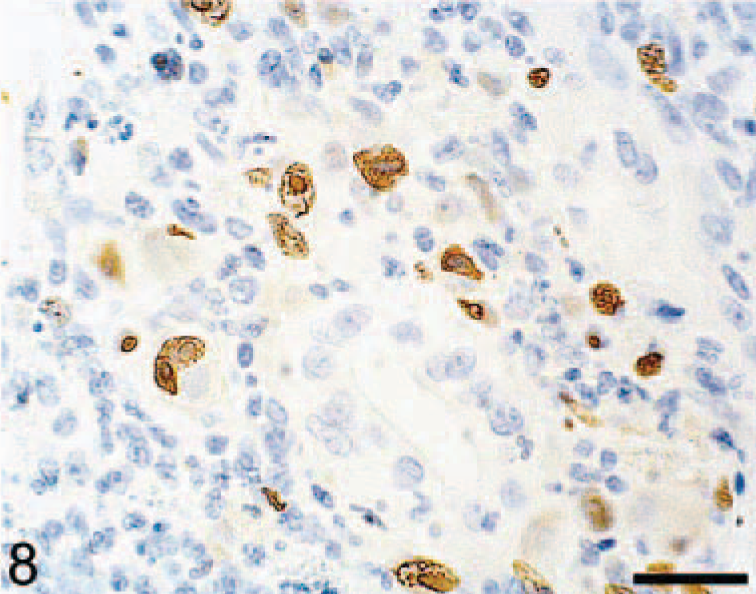
Gastrointestinal masses associated with CMV infection in humans are infrequent lesions. 11,13 The formation of mass lesions has been attributed in part to marked hemorrhage and edema secondary to infection of the vascular endothelium, which may result in vascular occlusion and vasculitis. 13 Dense accumulation of inflammatory cells within the lamina propria and submucosa, proliferation of fibroblasts, and granulation tissue also contribute to the formation of these inflammatory masses. 11,13 Although hemorrhage, edema, and inflammation are significant features of the lesions described here, glandular epithelial hyperplasia is also a prominent feature. Immunohistochemistry for the proliferation marker Ki-67 confirmed increased proliferation of both glandular epithelial cells and inflammatory cells in the lamina propria. Compared with normal mucosa in which epithelial proliferation is confined to the basal third of glands or crypts, Ki-67–positive cells extended into the surface epithelium in affected regions of the gastrointestinal tracts.
The proliferative lesion here most closely resembles that described in the localized form of Ménétrier's disease. In humans, HCMV has been associated with adult and pediatric Ménétrier's disease. 6,14 Ménétrier's disease is characterized as a proliferative gastropathy with hyperplasia of the foveolar epithelium. 6,14 Grossly, classical Ménétrier's disease is characterized by thickened rugal folds in the fundus, whereas the localized form can have occasional discrete mucosal nodules or polypoid masses located anywhere within the gastric mucosa. 14 Overexpression of growth factors, such as transforming growth factor–α, by hyper-plastic foveolar epithelium has been implicated in the pathogenesis of Ménétrier's disease. 14 The proliferative nature of the lesions described here and their resemblance to the localized form of Ménétrier's disease suggests that the overexpression of growth factors may play a role in the glandular hyperplasia seen in our cases of CMV-associated gastrointestinal masses.
Ethical Treatment of Animals
The animals described in this article were housed and treated in accordance with standards of the Association for Assessment and Accreditation of Laboratory Animal Care and Harvard Medical School's and Emory University's Animal Care and Use Committees.
Footnotes
Acknowledgements
We thank Kristen Toohey for Graphic services and Douglas Pauley for technical services. This research was funded in part by grants RR00168, DK55510 (New England Primate Research Center), and RR00165 (Yerkes National Primate Research Center).
