Abstract
Three cases of extramedullary hematopoiesis (EMH) in the mandibular lymph nodes of rhesus monkeys, experimentally infected intravenously with a chimeric simian human immunodeficiency virus, are described. On histopathologic evaluation, multiple sections of mandibular lymph node from all animals showed evidence of EMH, which included erythroid, myeloid, or megakaryocytic precursor cells (or all) within the medullary sinuses. Immunohistochemistry was used for positive identification of multiple cell types. Evidence of EMH was not observed in numerous sections of axillary, inguinal, cervical, hilar, or mesenteric lymph nodes or in any other tissues examined. To our knowledge, this is the first report on EMH within the lymph nodes of rhesus monkeys without an obvious underlying disease process or stringent blood-sampling schedule warranting the need for increased hematopoiesis outside the confines of the bone marrow.
Extramedullary hematopoiesis (EMH) is the production of myeloid, erythroid, and megakaryocytic precursor cells at ectopic sites. Although the medullary cavity provides the optimal microenvironment for hematopoietic cells to grow and differentiate in postnatal life, other organs or sites can provide the necessary milieu under certain circumstances. Although this process most often occurs in the spleen and liver, the sites of hematopoiesis during embryogenesis, 2,13 EMH may also occur elsewhere when the necessary requirements for homing, proliferation, and differentiation of circulating stems cells are provided. Other ectopic sites reported in humans and animals include the adrenal gland, 7 kidney, 29 brain, 4 lung, 10 and lymph nodes, 16 as well as numerous other sites. EMH in the setting of normal marrow function is most unusual. This process is usually associated with stringent experimental blood sampling schedules, a neoplastic disease, or hematologic disorders that are accompanied by peripheral cytopenias and inefficient hematopoiesis. These disorders include chronic myeloproliferative disorders, leukemias, 14 thalassemias, hemoglobinopathies, and hemolytic anemias. 27
We describe three cases of EMH in the mandibular lymph nodes of rhesus monkeys infected with simian human immunodeficiency virus (SHIV). The unique feature of these three cases is that none of the animals demonstrated any underlying disease process necessitating the need for hematopoiesis outside the marrow cavity.
Twenty-nine adult rhesus monkeys were used in an experimental SHIV vaccine and peptide study. A 23-year-old intact male rhesus monkey (monkey No. 1) and a 10-year-old intact female rhesus monkey (monkey No. 2) were experimentally infected intravenously with 100 mouse infectious dose (50%) (MID50) of SHIV89.6P, a chimeric virus composed of SIVmac 239 expressing HIV-1 env and the associated auxiliary HIV-1 genes tat, vpu, and rev. Both animals were submitted for necropsy after completing a 28-week and 34-week protocol, respectively. A 14-year-old intact female rhesus monkey (monkey No. 3) was experimentally infected intravenously with 104 tissue culture infectious dose (50%) of SHIVku2, a chimeric virus with the envelope of HIV-1 IIIB and the core of SIVmac239. After 110 weeks, monkey No. 3 was infected intravenously with 100 MID50 SHIV89.6P. Monkey No. 3 was submitted for necropsy after completion of a total of 180 weeks on protocol. Each animal was euthanatized intravenously with pentobarbital before necropsy.
All animals had been cared for and used humanely according to the American Association for Laboratory Animal Science “Policy on the Humane Care and Use of Laboratory Animals” and a protocol approved by the Institutional Animal Care and Use Committee of The University of Texas M. D. Anderson Cancer Center.
Multiple complete blood counts (CBC) with differential counts were performed on each animal during their respective protocols. The only significant abnormalities observed on CBC included mild to marked leukopenias, mostly associated with lymphopenia, and occasionally monocytopenia. Most red blood cell (RBC) indices remained within normal limits, with a few minor exceptions (Tables 1, 2, 3–4). Monkey No. 1 RBC counts were slightly below normal range on all samples. However, reference blood values for animals of this age (23 years) were not available, thus values were compared with those of animals in the 5- to 10-year range. This may explain the supposed decrease. Monkey No. 2 had two hematocrits slightly below normal and a slightly lower hemoglobin value at the time of euthanasia, as well as slightly decreased mean corpuscular volume and mean corpuscular hemoglobin concentration, but these minor changes were not clinically significant. Monkey No. 3 had a few hematocrits that were less than one point below the normal range and an occasional slightly decreased mean corpuscular hemoglobin. Again, none of these changes were considered clinically significant.
RBC indices, body weights, and blood sample volumes for monkey No. 1 at euthanasia and for the preceding 6 months.∗
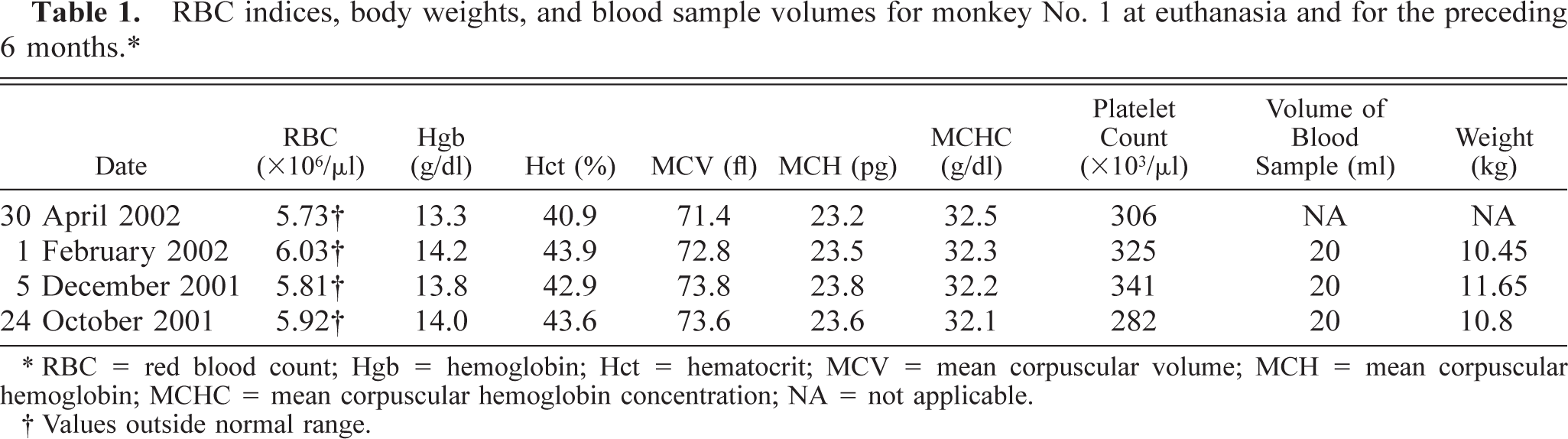
∗ RBC = red blood count; Hgb = hemoglobin; Hct = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; NA = not applicable.
† Values outside normal range.
RBC indices, body weights, and blood sample volumes for monkey No. 2 at euthanasia and for the preceding 6 months.∗
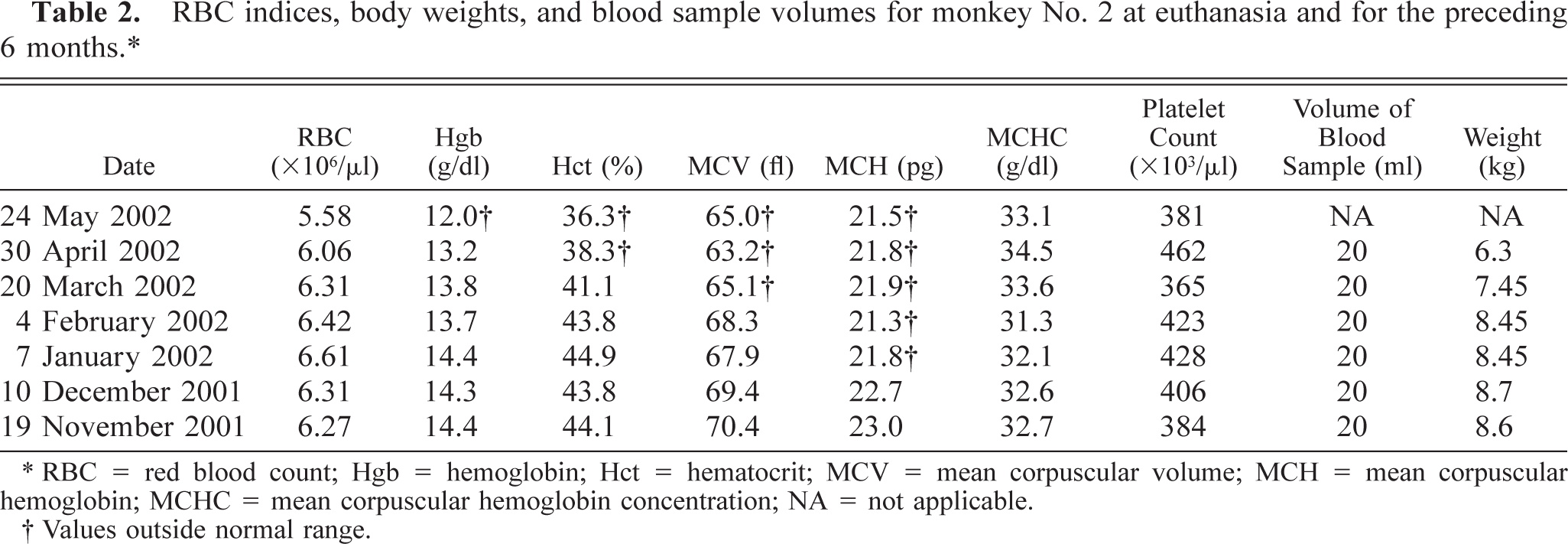
∗ RBC = red blood count; Hgb = hemoglobin; Hct = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; NA = not applicable.
† Values outside normal range.
RBC indices, body weights, and blood sample volumes for monkey No. 3 at euthanasia and for the preceding 7 months.∗
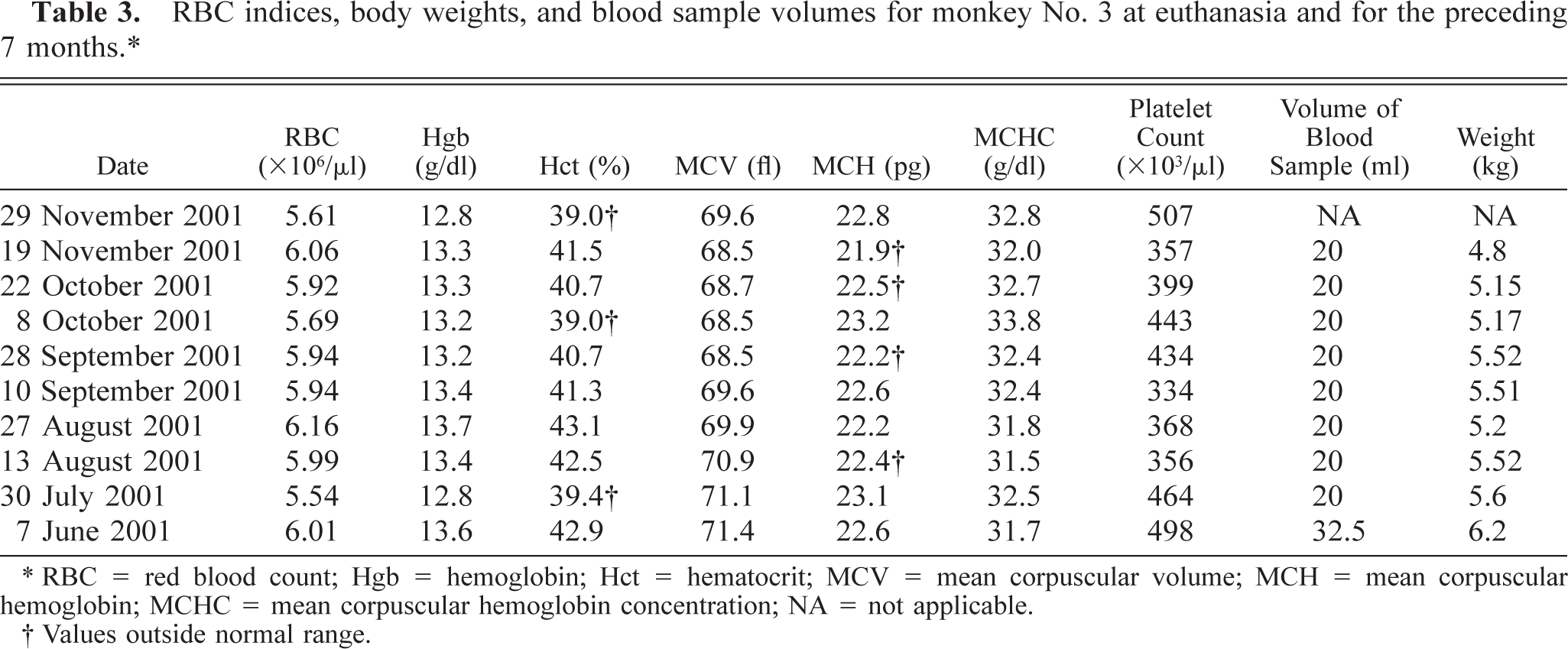
∗ RBC = red blood count; Hgb = hemoglobin; Hct = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; NA = not applicable.
† Values outside normal range.
Normal RBC indices for rhesus macaques based on age and sex. 6 ∗

∗ RBC = red blood count; Hgb = hemoglobin; Hct = hematocrit; MCV = mean corpuscular volume; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration.
At necropsy, monkey No. 1 was in good nutritional condition except for multiple bony proliferations throughout the vertebral column, colonic lymphadenopathy, and unilateral testicular atrophy. Monkey Nos. 2 and 3 were moderately anorectic with no lesions observed grossly. A number of tissues including the spleen, tonsil, thymic remnants, femoral bone marrow, hilar, mandibular, cervical, axillary, inguinal, and mesenteric lymph nodes, as well as numerous gut-associated and mucosal-associated lymphoid tissues, were collected and placed in 10% formalin. Tissues were then processed, paraffin embedded, cut at 5 µm, and stained with hematoxylin and eosin. Microscopic evaluation of six sections of mandibular lymph node from each animal revealed similar lesions.
In monkey No. 1, one section contained many erythroid and granulocytic precursor cells and a few megakaryocytes within the medullary sinuses. Other findings included a moderate sinus histiocytosis with many erythrophagocytic and hemosiderin-laden macrophages and many adipocyte infiltrates. The cortex had good lymphoid cellularity, with normal cellularity to moderate hypocellularity of the germinal centers. Hyaline material was observed in the hypocellular centers.
Monkey No. 2 had few megakaryocytes and even fewer granulocytic precursor cells in the medullary sinuses of three sections. Erythroid elements were not observed. A marked sinus histiocytosis with excessive numbers of erythrophagocytic macrophages and numerous hemosiderin-laden macrophages was observed. The cortex had good cellularity and showed moderate depletion of germinal centers that contained varying amounts of hyaline material and karyorrhectic debris.
Monkey No. 3 had many megakaryocytes, erythroid, and granulocytic precursor cells within the medullary sinuses of all six sections. Mild to marked sinus histiocytosis with many erythrophagocytic macrophages and multiple hemosiderin-laden macrophages was observed. The cortex had good cellularity, and mild to moderate depletion of germinal centers was observed.
Hematopoietic elements were not observed in any of the other lymph nodes or in the spleen, liver, kidney, or adrenal glands. The femoral bone marrow from each animal had normal cellularity. A diagnosis of EMH of the mandibular lymph node was made for each animal.
The identity of the hematopoietic cells was verified immunohistochemically. Factor VII–related antigen, macrophage HAM56, CD3, CD79a, and neutrophil elastase (DAKOCytomation, Carpinteria, CA) were used to identify megakaryocytes, macrophages/monocytes, T lymphocytes, B lymphocytes, and neutrophils, respectively. The LSAB2 system or Envision + systems (DAKOCytomation) using diaminobenzidine as the chromogen and Mayer's hematoxylin as the counterstain were used. Negative controls consisted of replacing the primary antibody with mouse or rabbit serum.
EMH, the ectopic production of hematopoietic cells, is a compensatory mechanism that results when stressed bone marrow is unable to keep up with the body's requirements because of either increased demand or ineffective erythropoiesis. EMH may also be secondary to pathologic fractures that result in extrusion of hematopoietic marrow into the surrounding soft tissues. 24 EMH can occur in virtually any organ but most commonly occurs in areas of fetal hemoglobin production, such as the spleen, liver, and lymph nodes, in humans. 15 The liver and spleen are common sites of EMH in animals. In certain species, EMH commonly occurs without underlying disease; for example, EMH can be found in the mouse spleen throughout life, and EMH occurs naturally in the spleen, liver, kidneys, mesenteric lymph nodes, adrenal glands, and choroid plexuses of Common marmosets (Callithrix jacchus). 20,29
EMH is generally assumed to be a compensatory bone marrow hyperplastic phenomenon, which begins with seeding of circulating hematopoietic progenitor cells at various sites. 30 The possibility of pluripotential stem cells existing in various organs and differentiating into hematopoietic precursor cells with the proper stimuli has been postulated as well. 1 The most common sites of EMH in the adult humans are the liver and spleen, supporting the theory that the pathogenesis of EMH is a reversion of these organs to their fetal hematopoietic function after stimulation by certain cytokines and growth factors. This reversion is believed to arise from pluripotential stem cells in the mesenchymal tissue of these organs. These cells can be present in numerous other organs as well. 21 The inductive microenvironment for the formation of blood elements from these pluripotential cells is composed of macrophages, fibroblasts, endothelial cells, appropriate growth factors, and cytokines. 4
Cytokines, interleukin (IL)-3 and IL-12, and granulocyte colony-stimulating factor have all been reported to induce EMH experimentally in nonhuman primates 19,26 and in humans. 22 IL-6, IL-12, and granulocyte-macrophage colony-stimulating factor are secreted by macrophages and enhance erythropoiesis. 25 All three rhesus monkeys studied in this report had sinus histiocytosis admixed with EMH. One or more cytokines may have been secreted by macrophages, which may have induced or stimulated the pluripotential cells of the lymph node to produce hematopoietic cells, but what stimulated macrophages to secrete these cytokines is not known. SHIV may itself have a mechanism that stimulates the secretion of macrophage-synthesized cytokines. A previous study found that IL-6 levels were elevated in lymph nodes from SIV-infected cynomolgus monkeys, compared with control monkeys. 9 In contrast, IL-6 levels measured in lymph nodes from HIV-infected humans were no different than those from normal reactive lymph nodes of noninfected humans. 12
The stimulus for cytokine secretion in these animals was most likely not the experimental blood-sampling schedule in the months preceding euthanasia because the volumes taken were far lower than those that would be likely to incite hematopoiesis. The recommended interval for recovery from a blood sample consisting of 7.5% of circulating blood volume is 1 week. 11 The percentage of blood taken from the animals in this report neither approached nor exceeded this recommended level. Blood sample size and sampling frequency never exceeded levels that other authors consider to be maximal safe levels for blood sampling in nonhuman primates. 3,18
Variations in local cytokine or growth factor concentrations could account for the variable EMH phenotypes observed in the three animals. The preponderance of erythropoiesis observed in monkey Nos. 1 and 3 did not reflect the number of macrophages observed in the medullary region. Monkey No. 2 did not have erythropoiesis but appeared to have more sinus macrophages than the other two animals.
EMH has been described previously in nonhuman primates and has been associated with anemia in both old-world 27 and new-world 8,28 primates. EMH has also been induced experimentally in nonhuman primates. 5,19,26 The immunodeficiency-inducing retroviruses are, to some degree, associated with anemia. This anemia may be severe enough to cause extramedullary hematopoietic splenomegaly, a lesion that is sometimes diagnosed in HIV-infected humans with no other underlying disease process. 17,23 The three monkeys in this study did not have anemia or bone marrow alterations that would have resulted in EMH. Even if they had anemia, the spleen and liver would most commonly have been the first sites of EMH. Lymph nodes usually are not a site for EMH unless a severe chronic disease altering the bone marrow is present. EMH had not occurred in either spleen or liver of the three monkeys, and it was interesting that the mandibular lymph node was the only lymph node of the six types evaluated to be affected. The reason why EMH would affect only the mandibular lymph nodes is unknown. A predilection of EMH for lymph nodes had not been described in the literature, and these lymph nodes are not in close proximity to other hematopoietic structures. Some hypotheses about the occurrence of EMH in the mandibular lymph nodes could be that hematopoietic precursor cells remain from the fetal development stage, that pluripotential stem cells are stimulated in these lymph nodes, or that hematopoietic elements are released into the circulation and home in on these lymph nodes. Although all of these hypotheses are plausible, the reason that EMH occurred in the mandibular lymph nodes and not in the other six lymph node locations evaluated cannot be fully explained.
EMH in the mandibular lymph nodes of rhesus monkeys infected with SHIV may be an epiphenomenon of little clinical significance, or it may be a particularly uncommon manifestation of a previously unrecognized aspect of SHIV infection. Continued evaluation of multiple lymph nodes, including the mandibular lymph nodes, may offer more insight into the sites of predilection for EMH in rhesus monkeys.
Footnotes
Acknowledgements
We thank Mr. Gerald Costello, Mrs. Bharti P. Nehete, Ms. Talitha S. Keeney, Mrs. DeeAnn Brandenberger, and Mr. Howard Bulack for their technical support of this project. This work was supported in part by funds from the National Institute of Allergy and Infectious Diseases AI 42694.
