Abstract
Trypanosoma cruzi-like flagellates were incidentally noted in blood smears of a routinely monitored rhesus monkey experimentally infected with the simian immunodeficiency virus (SIV). Immunodeficiency in the course of the SIV infection reactivated a chronic infection of Chagas' disease that had been unnoticed when the macaque was imported to Europe. The animal developed no specific clinical symptoms of American trypanosomiasis, but histologically a chagasic myocarditis was detected. Analysis of the small subunit rRNA gene of the trypanosome identified the protozoan as T. cruzi.
Introduction
Trypanosoma cruzi, a protozoan parasite, is the causative agent of Chagas' disease or American trypanosomiasis.3 Initial infection with T. cruzi usually depends on intermediate arthropod vectors (Reduviidae, assassin bugs), particularly Triatoma spp., which occur naturally in Central and South America. Kissing bugs transmit the infectious stages of the protozoa through their feces, although vertical transmission of T. cruzi has been shown to occur.1,15 Various mammals, including human beings, are the natural hosts of Triatoma spp. as well as T. cruzi.23 The only nonhuman primates known to become naturally infected with T. cruzi are New World monkeys from South America and Panama.13,17,24,28 Old World monkeys, including some macaques (Macaca silenus, Macaca mulatta, or Macaca nigra) or lemurs (Lemur catta), are also susceptible when translocated into the geographic range of reduviids or when experimentally infected.4,19,20
The infection of nonhuman primates may remain subclinical for years or may infrequently produce a variety of clinical effects such as anorexia, dyspnea, fever, leukocytosis, lymphadenopathy, or myocarditis.2,12,27 In human beings, reactivation of Chagas' disease in immunodeficient patients infected with the human immunodeficiency virus (HIV) has been reported since the early 1990s.18 The clinical manifestations of reactivated Chagas' disease are severe central nervous system (CNS) alterations and cardiomyopathy.21,22 Trypomastigotes of T. cruzi, observed by direct microscopic examination of blood smears, characterize the acute phase of infection and confirm Chagas' disease reactivation.22 This article documents a case of Chagas' disease reactivation in a rhesus monkey (M. mulatta) experimentally infected with the simian immunodeficiency virus (SIV).
Clinical Course
Animal
A male rhesus monkey, M. mulatta, was born in January 1994 at a farm of a primate breeder in Yemassee, South Carolina, USA, and had been bred, reared, and socially housed in a large free-ranging colony. This colony originates from breeding animals imported from a primate breeder in Cayo Santiago, Puerto Rico, in the late 1980s. At the age of 2, the monkey arrived at the German Primate Center at Göttingen, Germany, and was quarantined for 6 weeks in an individual cage and tested for different viral (Herpes B, SIV, simian retrovirus, simian T-cell leukemia virus) and bacterial infections, including tuberculosis. All results were negative. The monkey harbored some parasites (Capillaria spp., Giardia spp., and Balantidium spp.) and was treated with antiparasitic drugs. After release from quarantine, it was caged individually with free access to tap water according to the Guide for the Care and Use of Laboratory Animals, published by the National Institute of Health. The diet consisted of primate pellets (Ssniff, Special Diet Ltd., Soest, Germany) and fresh fruits and vegetables.
Experimental infection with SIV
Two years after its arrival, the monkey was intravenously inoculated with an SIV derivate, nefSHIV-K6, in an SIV infection study.9 In the course of the experimental SIV infection, the animal was monitored for blood parameters, for neopterin levels in the urine, and for the palpation size of the peripheral lymph nodes and the spleen. The cell-associated viral load was determined by limiting dilution cocultivation with peripheral blood mononuclear cells (PBMCs), and the peripheral blood lymphocytes subsets were characterized by flow cytometry.
Clinical findings
Four weeks postinfection (wpi) a mild thrombocytopenia was reported. Eight wpi a moderate lymphadenopathy and splenomegaly could be palpated. From 20 wpi the animal developed a severe thrombocytopenia, and from 24 wpi this was accompanied by a severe lymphadenopathy and splenomegaly. From 46 wpi the monkey showed recurrent loss of appetite accompanied by slight weight loss. From 60 wpi an abundant number of T. cruzi–like flagellates were observed in blood smears and in coculture with PBMCs. At this time point the absolute CD4+ T-cell count had declined to 158/mm3. The CD4+ to CD8+ ratio was 0.43. Physical examination of the animal at the time of diagnosis revealed an apparently healthy animal, although it was noted that the monkey still showed loss of appetite. When sacrificed at 70 wpi the content of neopterin had increased to eight times the normal values, the peripheral blood lymphocytes showed a high cell-associated viral load, and the ratio of CD4+ to CD8+ cells was 0.23. Necropsy was carried out immediately after euthanasia.
Pathologic findings
Histopathologic examination revealed three main diagnostic findings. First, typical signs of an SIV-associated lymphadenopathy were observed. These alterations were characterized by an irregular hyperplasia of the lymphatic tissues, with severe proliferation of the lymphocytes.
Further striking evidence were alterations in the digestive system, likewise connected to a progressive immunodeficiency. Histologically, the stomach was characterized by a moderate superficial chronic gastritis (gastritis score 2), with diffuse hyperemia and focal occurrence of large gastrointestinal spiral-shaped bacteria, probably Helicobacter spp. The intestine was marked by a moderate chronic enteritis, with infiltration of eosinophilic granulocytes and the occurrence of adhesive bacteria and Balantidium spp. in the lumen. These pronounced, inflammatory alterations, responsible for the clinical course, can be judged as primary and secondary SIV-induced alterations.
The third main diagnosis was a nonpurulent interstitial myocarditis, related to an infection with T. cruzi. Histologically, the myocardium was multifocally infiltrated by lymphohistiocytic inflammatory cells, which were predominantly found in the perivascular interstice. The lesions were accompanied by moderate vacuolar degeneration of myocardial muscle cells. Giemsa-stained sections revealed pseudocysts with intracellular amastigote-like organisms (Fig. 1).
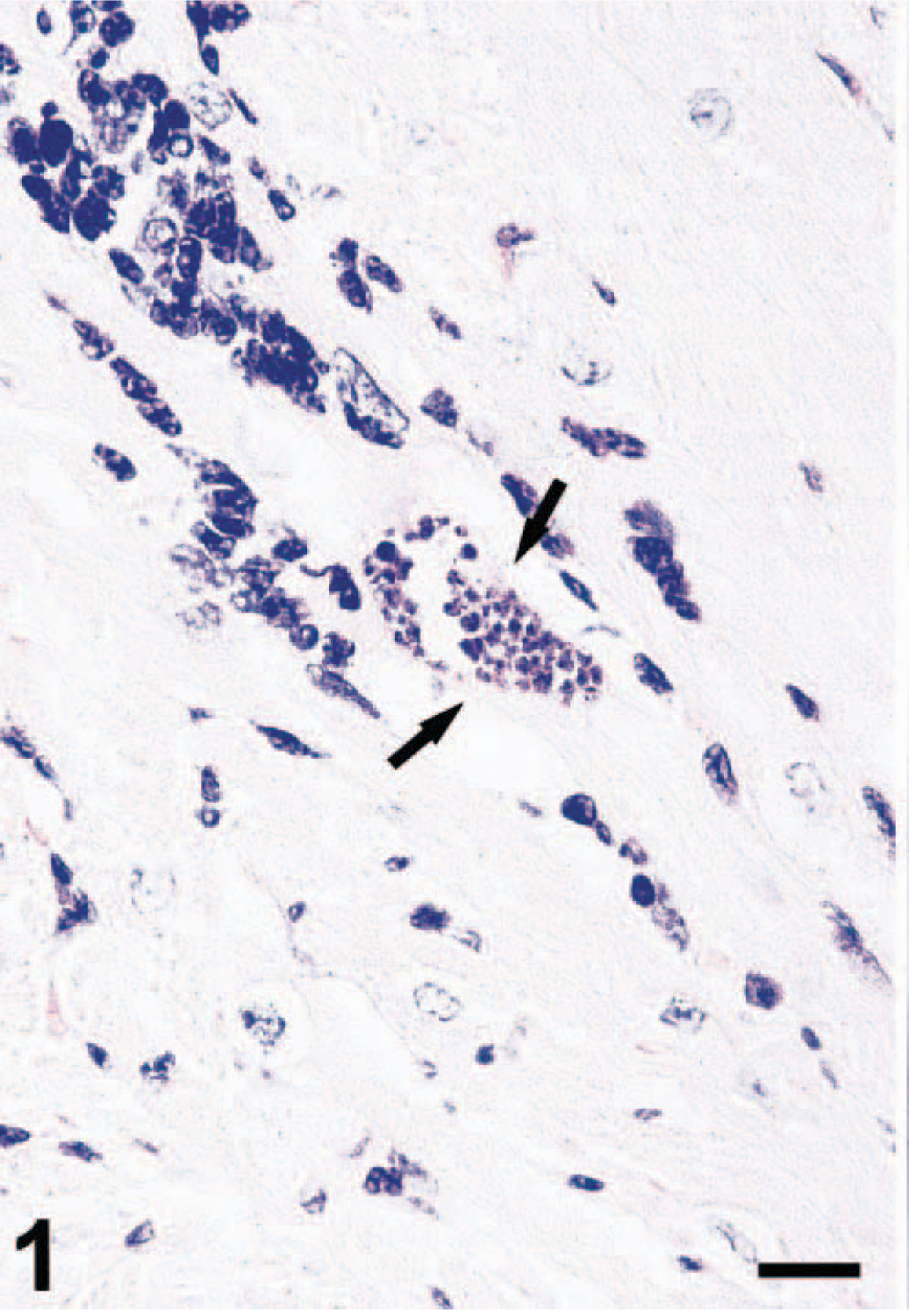
Myocardium; M. mulatta. Nonpurulent interstitial myocarditis with perivascular infiltration of lymphohistiocytic inflammatory cells. Pseudocyst with amastigote forms of T. cruzi (arrows). Giemsa. Bar = 17 μm.
Parasitologic findings
Isolation of the protozoa was performed by hemoculture on blood agar. For further propagation, the T. cruzi–like flagellates were cultivated with RPMI-medium containing 10% fetal calf serum. Under direct microscopic examination T. cruzi–like epimastigotes were observed (Fig. 2).
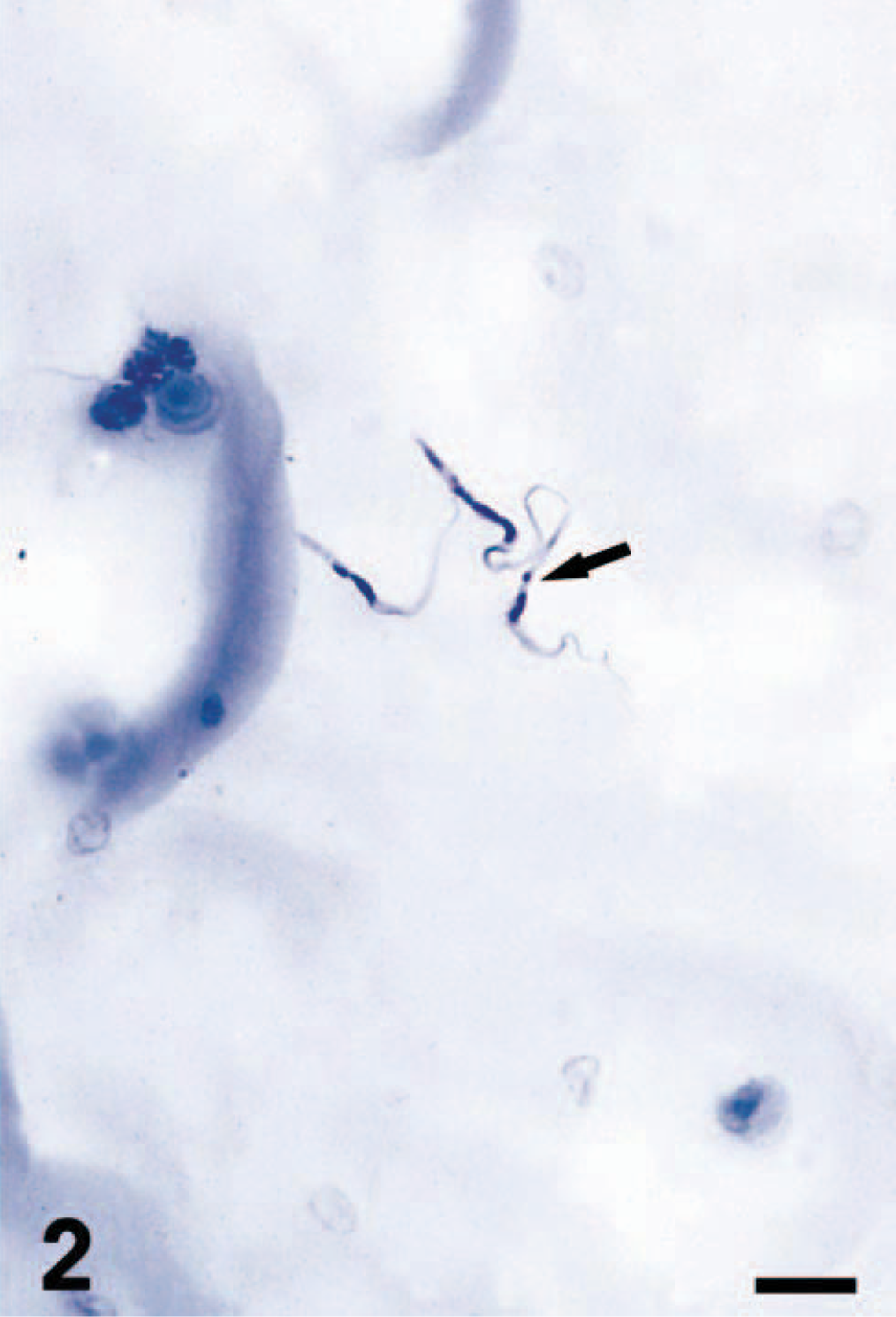
Hemoculture. Epimastigotes of T. cruzi (arrow). Giemsa. Bar = 11 μm.
For specific identification of the trypanosome, a small subunit (ssu) rRNA gene analysis was performed as described previously.25 A polymerase chain reaction (PCR) was performed on a crude lysate of the cultured trypanosomes. The approximately 2.2 kb fragment containing the 18S ssu rRNA sequence was obtained in two overlapping 1.2 kb fragments by PCR using conserved primers.14 The products of eight separate PCR reactions were purified and sequenced using 12 additional internal primers. A consensus sequence was assembled using the ABI program AutoAssembler v. 2.0. A BLAST search was performed on 2,202 bases of the ssu rRNA gene. The sequence had 98% similarity to four strains of T. cruzi including the strains MT4166 and MT4167 (both isolates from triatomine bugs, Rhodnius brethesi, Brazil) and CAN III cl.1. (human origin, Brazil).
Discussion
T. cruzi infection is widespread in Latin America among humans and mammals, including nonhuman primates. In the USA, the flagellate is occasionally found in the southeastern part of the country, where infections of translocated Old World primates with indigenous strains of T. cruzi have been reported previously.4,7,8 The rhesus monkey could have become infected in a number of different ways. If the monkey colony was already chronically infected, a nonvectorial mode of transmission could have been possible: the blood forms could either be transmitted vertically from mother to offspring or horizontally through mucous membranes (biting wounds or copulation) or by wound-wound contact.5 If the monkey colony was Trypanosoma-free when it was established, the colony could have become infected through the bites of infected triatomine bugs, but transmission through fecal contamination of oral mucosa or lips can occur when the animals eat infected bugs.8
In the southern USA there are only a few native Triatoma spp. and Rhodnius spp. that transmit Chagas' disease. However, the identified T. cruzi had the greatest homology (98%) to Brazilian T. cruzi isolates detected in R. brethesi, a kissing bug indigenous in Brazil and Venezuela.10
There are three main subgroups of T. cruzi: zymodemes Z1, Z2, and Z3.16,26 This T. cruzi isolate had greatest similarity to three trypanosomes in the Z3 subgroup. This subgroup has a distribution restricted to the Amazon basin and is associated with terrestrial animals such as armadillos. Z1 is the only subgroup found in North American mammals. If the monkey had picked up a trypanosome infection that naturally occurred in the area, we would expect the trypanosome to be in the Z1 subgroup. There are several possible explanations for the homology of this T. cruzi to trypanosomes in the Z3 subgroup.
Because the ancestors of the monkeys in the colony were bought from Puerto Rico, the colony may originally have become infected in South America. Alternatively the colony could have become infected from a triatomine bug imported from a Z3 region. Triatomine bugs could have been imported in building materials imported from Puerto Rico. A search for South American triatomine species such as R. brethesi on the monkey island could be informative. A less likely explanation is that the Z3 subgroup could have a wider geographical range than that previously thought.
Once infected with T. cruzi, the monkeys do not necessarily develop signs of Chagas' disease such as anorexia, fever, lymphadenopathy, or even sudden death from severe diffuse myocarditis.6 The majority of T. cruzi infections appear to have no detectable adverse effects and infected monkeys often stay essentially asymptomatic.20 However, the clinical findings are not necessarily symptomatic either for SIV infection or for Chagas' disease. M. mulatta infected only with SIV show symptoms similar to alterations described for Chagas' disease: anorexia, loss of weight, lymphadenopathy.11 In this animal, the course of SIV infection suggests that the clinical findings were connected with SIV infection: at 8 wpi a lymphadenopathy and splenomegaly could be palpated, but it was not until 60 wpi, at a stage of severe immunosuppression, that the flagellates were detected for the first time. However, left ventricular dysfunction, a typical sign for chronic chagasic myocarditis, could not be auscultated. At 70 wpi and 10 weeks after detection of the trypanosomes, the animal was sacrificed for its poor general condition. The histologic alterations were evidence of a nonpurulent interstitial myocarditis, which is typical for Chagas' disease. The observation of a pseudocyst with amastigotes in the interstitium of the muscular tissue confirmed the diagnosis. In HIV-infected patients, reactivation of T. cruzi infection not only leads to degeneration of the myocardium but also commonly affects the CNS with expansive lesions or meningoencephalitis. In this case, the CNS of the rhesus monkey was not involved, but encephalitis in association with T. cruzi has been reported in a Celebes black macaque.19
The rhesus macaque was already infected with T. cruzi on its arrival at the German Primate Center. The flagellate is usually only detectable in blood smears during the acute phase or during reactivation of the infection. Because there are no routine tests for T. cruzi, it is possible for a chronically infected animal to be given a false health certificate and be imported from an area where T. cruzi is endemic into a T. cruzi–free region. Because there are no cures available for chronically infected animals, such naturally occurring infections in imported animals still present a risk to humans. Even in regions without vectorial insects there are potential sources of accidental transmission to animal handlers and uninfected laboratory animals. A bite from an animal infected with T. cruzi or contamination with a wound containing epimastigote blood forms during the acute phase of infection could result in a human becoming infected.
This work shows that occult infection with T. cruzi can reactivate and lead to histologic or even symptomatic changes associated with Chagas' disease and could distort the results of otherwise well-designed studies. This should always be a consideration when working with monkeys from colonies derived from T. cruzi–endemic areas.
Footnotes
Acknowledgements
We wish to acknowledge the excellent technical assistance from E. Nicksch, K. Kaiser-Jarry, H. Gilhaus, and H. Zuri and generous advice and support from Dr. W. Gibson and Dr. S. Graefe.
