Abstract
The degree to which the selectin inhibitor TBC1269 reduces neutrophil infiltration in specific microscopic locations of the lung during acute pneumonia of neonates was determined. Neonatal calves were inoculated intrabronchially with Mannheimia (Pasteurella) haemolytica or saline, and lung tissue was collected at 2 and 6 hours postinoculation (PI). One 6-hour group inoculated with M. haemolytica received TBC1269 intravenously before and after inoculation with M. haemolytica. Infiltrates of neutrophils were significantly higher in the alveolar lumen and septae but lower in the bronchial lumen and epithelium at 6 hours PI than at 2 hours PI. Significantly fewer neutrophils (P > 0.05) were present in the alveolar lumen and septae, and the bronchiolar lumen and lamina propria in the lungs of TBC1269-treated calves compared with untreated calves at 6 hours PI. TBC1269 did not alter the infiltration into bronchi and blood vessels or the expression of the selectin-independent adhesion molecule, ICAM-1. This work suggests that during acute pneumonia of neonates 1) neutrophil infiltrates progressively increase in the alveolar lumens and septae but decrease in the bronchial lumen and epithelium with time, 2) TBC1269 reduces neutrophil infiltration into specific regions of alveoli and bronchioles rather than uniformly throughout the lung, and 3) selectin inhibition does not affect the location and intensity of ICAM-1 expression.
Neutrophil infiltration is a vital component of the innate immune system. The protective value of neutrophils infiltrating the respiratory tract is underscored by the fact that children and young animals with impaired adhesion molecule expression (leukocyte adhesion deficiency [LAD]) often suffer from severe and recurrent infections.14,20 On the other hand, a certain degree of lung damage that occurs during acute bacterial pneumonia of neonates, in infants on ventilators, and in conditions such as acute respiratory distress syndrome can be attributed to neutrophil products such as elastase, acid hydrolases, and oxygen-free radicals released during infiltration.35,36 The lungs of children with cystic fibrosis, for example, are often colonized by Pseudomonas or Burkholderia sp., which incite recurrent infiltrates of neutrophils that impair gaseous exchange through their accumulation in alveoli and their damage to the alveolar wall.6,25 Temporary regulation or modulation of neutrophil infiltration may have a beneficial value in protecting the lung against damage during bacterial pneumonia, especially when it is concurrent with traditional therapies such as antibiotics, oxygen, nebulization, and chest percussion.
Initial neutrophil adherence in acute inflammation involves a rolling phase mediated by interactions of selectin molecules (E-, P-, and L-selectin) expressed by neutrophils and endothelial cells. Ligands for the selectins include CD34, PSGL-1, GlyCAM-1, and sialyl Lewisx (sLex).11,13 After activation of the neutrophils, L-selectin is proteolytically cleaved from the neutrophil membrane, and β2 integrins are expressed on the neutrophil membrane. Firm adherence between the neutrophils and the endothelial cells is mediated by certain β2 integrins (CD11a, b, c/CD18 [LFA-1, Mac-1, and p150,95, respectively]) expressed by neutrophils and the immunoglobulin family of adhesion molecules, such as intercellular adhesion molecule-1 (CD54; ICAM-1) expressed on endothelial cells.23 In the lung the role of selectins has not been fully established, and selectin-mediated rolling may be limited to certain regions.
Neutrophil sequestration in the lung involves a variety of adhesion molecules,8 but the activity of individual families of adhesion molecules within specific compartments (bronchi, bronchioles, alveoli, blood vessels) and microscopic locations (lumen, epithelium, lamina propria, adventitia) of the lung has not been fully characterized.18,36 In a neonatal calf model of acute Gram-negative bacterial (Mannheimia [Pasteurella] haemolytica) pneumonia, β2 integrin–ICAM-1 adherence mediates neutrophil infiltration into the bronchi but is not required for infiltration into the bronchioles and alveoli.3,4 In the alveoli there are special anatomic and physiologic features that affect infiltration, such as the thin basement membrane and overlying epithelium, the small amount of perivascular connective tissue, and the low hydrostatic pressure in alveolar capillaries. Some studies suggest that β2 integrins, α4 and α5 integrins, selectins, and ICAM-13,4,8 contribute to adherence in the alveoli. Inhibition of excessive neutrophil infiltration at the level of the alveolus would have potential therapeutic benefits because the alveolar wall is essential for gaseous exchange, and many bacterial pathogens induce strong neutrophilic infiltrates that accumulate in the alveolar lumen and impair air exchange.
In vitro studies of a synthetic selectin inhibitor, TBC1269 (1,6-bis[3-(3-carboxymethylphenyl)-4-(2-α-
The aims of this study were to determine the in vivo effects of parenteral administration of a synthetic analog of sialyl Lewisx (TBC1269) on the intensity and the microanatomic location of neutrophil infiltration in a newborn calf model with acute pulmonary infection caused by M. haemolytica. Neutrophil infiltration in bronchi, bronchioles, alveoli, and pulmonary blood vessels was assessed using a computerized image analysis system. The extent and cellular localization of ICAM-1 messenger ribonucleic acid (mRNA) expression was also assessed to identify the possible sites at which non–selectin-mediated infiltration may occur.
Methods
Experimental design
Colostrum-deprived Holstein calves (1–3 days old) were inoculated intrabronchially with pyrogen-free saline or M. haemolytica, using a fiber-optic bronchoscope as previously described.7 The calves appeared healthy, were afebrile, and had normal hematologic parameters. Groups of calves were organized as follows: group 1 (n = 3) received pyrogen-free saline and were necropsied at 2 hours PI; group 2 (n = 4) received M. haemolytica and were necropsied at 2 hours PI; group 3 (n = 3) received pyrogen-free saline and were necropsied at 6 hours PI; group 4 (n = 3) received M. haemolytica and were necropsied at 6 hours PI; group 5 (n = 4) received TBC1269 intravenously (25 mg/kg) 30 minutes before and 2 hours after inoculation with M. haemolytica, and were necropsied at 6 hours PI. TBC1269 was given at this time point (6 hours PI) because 1) previous studies3 have shown that neutrophil infiltration is generally greater at 6 hours than at 2 hours PI, which provides a more stringent assessment of TBC1269 activity against neutrophil infiltration; 2) alveolar infiltration is highest at 6 hours PI, and the alveolar capillaries are especially important for gaseous exchange at the site where selectin inhibition would potentially be the greatest; and 3) this time point is prior to the development of parenchymal necrosis and florid passage of neutrophils. A 24-hour group was not included because one M. haemolytica–inoculated calf did not survive up to this time point.
Bacterial inocula and tissue collection
M. haemolytica or pyrogen-free saline (5 ml each) were deposited into the first bifurcation of the right tracheal bronchus of the cranial part of the cranial right lung lobe. Bacteria were grown as previously described7 and the inoculum routinely contained 1.3 × 108 to 2.5 × 108 CFU/ml. The animals were sedated with xylazine hydrochloride (0.1 mg/kg, IV) for inoculations. At the appropriate time points, calves were euthanatized by intravenous pentobarbital. Lung tissue was collected from three sites within the area of fiberoptic deposition of the inoculum in each animal. Site 1 was from a 1.0-cm circular area at the exact site of deposition, site 2 was from a 1.0-cm circular area just dorsal to the site of deposition, and site 3 was from a 1.0-cm circular area just ventral to the site of deposition. Five different areas of each site were collected and used for morphometry and in situ hybridization. Lung tissues were immersion-fixed in 10% neutral-buffered formalin for 18–24 hours, processed routinely, embedded in paraffin, and sectioned at 3 µ. Sections were stained with hematoxylin and eosin (HE) and used for morphometric analysis, or replicates unstained were used for in situ hybridization. Lungs from all animals were collected under the same conditions, but they were not perfused because this would wash away the neutrophils within the pulmonary airways and the vasculature.10,30 Lung tissue was also collected from noninoculated area lobes, and these sections lacked infiltrates of inflammatory cells.
Morphometric analysis of digital images
From at least five slides from each of the three sites, the numbers of neutrophils per square micrometer were measured in histologic sections of the various lung compartments by a computerized image analysis system as previously described.3,4,29,30 Tracings were made on images that included outlines of the airway lumens, epithelium, and lamina propria of the bronchi and bronchioles; the lumens and septae of the alveoli; and the tunica intima, media, and adventitia of the blood vessels. Neutrophils were marked by dots (Fig. 1A, B). Tracings were made from at least 12 bronchi, bronchioles, alveoli, and blood vessels from each of the three sites sampled.
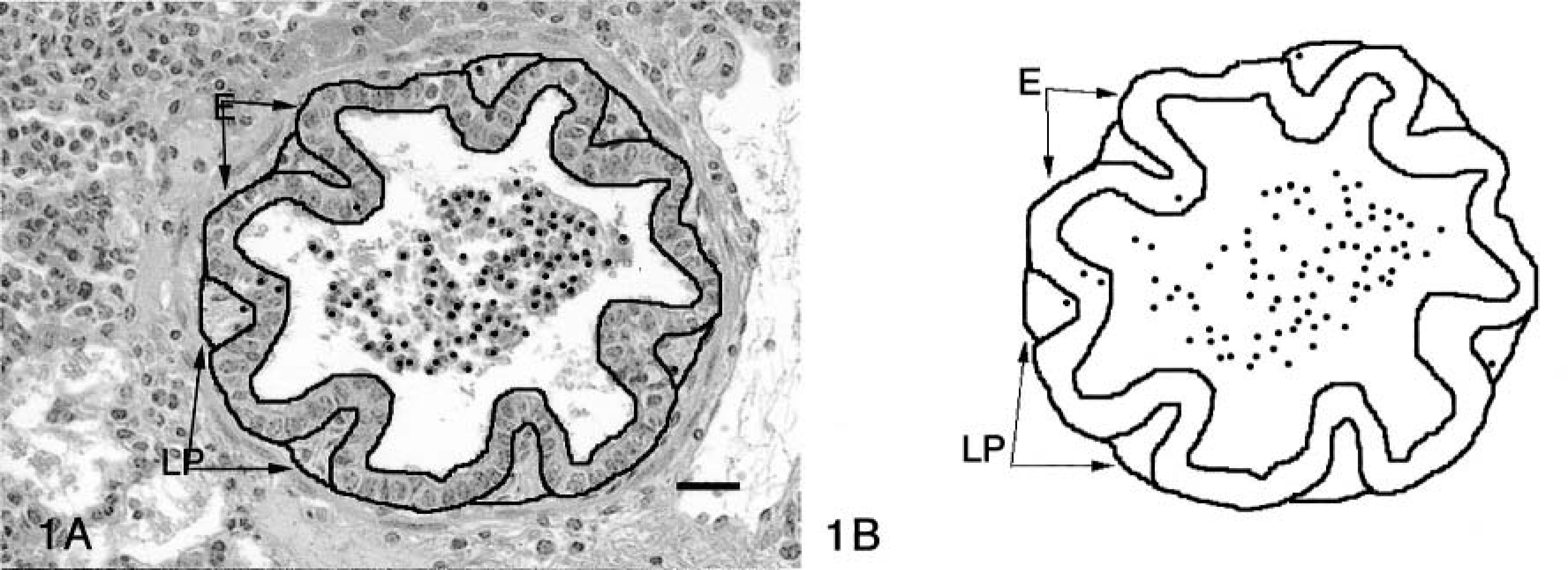
Bovine lung; M. haemolytica–inoculated calf at 6 hours PI. Computer images of bronchiolar section captured electronically for morphometric analysis.
Riboprobe preparation
Riboprobe preparation was done according to a procedure previously reported by Radi et al.29 A deoxyribonucleic acid (DNA) fragment from the complementary DNA of the bovine ICAM-1 gene was cloned and sequenced. After the in vitro transcription labeling, the RNA probe was purified, and the probe concentration was determined by dot blot.
In situ hybridization protocol and scoring system
In situ hybridization procedures were similar to those previously described by Radi et al.,29 with few modifications. A digoxigenin-labeled ICAM-1 riboprobe was used. Hybridization specificity was confirmed by incubating the sections using 1) a hybridization solution with no added ICAM-1 probe, 2) a sense digoxigenin-labeled probe, or 3) ribonuclease digestion; these sections lacked any detectable hybridization. A semiquantitative scoring system was used. Staining scores ranged from 0 to 3: 0 = no staining, 1 = minimally detectable staining in < 30% of the epithelial cells (airways) or endothelial cells (vessels), 2 = clear moderate staining intensity in > 30–60% of the cells, and 3 = intense staining in > 60% of the cells, and L = the special staining designation for intense staining of leukocytes (neutrophils and macrophages).
Statistical analysis
Neutrophil morphometric data were analyzed by the general linear models procedure of SAS.32 The statistical model used included effects of treatments, time of necropsy, pulmonary tissue type, and pulmonary tissue type location. ICAM-1 data were analyzed using a nonparametric analysis (chi-square test and Fisher's exact test). The variables tested consisted of treatments, pulmonary cell types, and time of necropsy. The significance level used was P < 0.05.
Results
Histopathology
Neutrophil infiltration, typical of M. haemolytica infection, was present in the lungs of calves from the M. haemolytica–inoculated groups at both 2 and 6 hours PI (groups 2, 4, and 5). Neutrophils were present in all three sites of the M. haemolytica–inoculated area of the right lung lobe. Only occasional minimal neutrophil infiltrates were present in the lungs of the control (saline-inoculated) groups (groups 1 and 3), and no inflammatory cells were present in the noninoculated lung lobes of any animal.
Morphometric analysis of digital images
Data from all three sites collected from the area of inoculum deposition were combined for morphometric assessment for each animal. For each animal the numbers of neutrophils per micrometer2 in each pulmonary compartment (alveolus, bronchiole, bronchus, or vessel) and each subcompartment (epithelium, lamina propria, or lumen) were determined, and the groups were compared for 1) inoculum (M. haemolytica versus saline inoculation at 2 and 6 hours), 2) time PI with M. haemolytica (2 versus 6 hours PI), and 3) TBC1269 treatment (TBC1269 treatment versus no treatment and saline controls at 6 hours PI).
M. haemolytica–inoculated versus saline-inoculated control at 2 and 6 hours. At 2 and 6 hours PI the lungs of the M. haemolytica–inoculated calves (groups 2, 4, and 5) had overall (when combining all compartments) greater infiltrates of neutrophils (P < 0.05) than the saline-inoculated control lung lobes (groups 1 and 3). At 2 hours PI, the bronchioles, bronchi, and alveoli, but not the blood vessels, of M. haemolytica–inoculated calves (group 2) had significantly higher (P < 0.05) neutrophil counts than the saline-inoculated controls (group 1). The infiltrate was highest in the bronchiolar and bronchial compartments, lower in the alveolar compartment, and lowest in the vessels (Table 1). At 6 hours PI in the M. haemolytica–inoculated groups (groups 2, 4, and 5), neutrophil infiltrates were significantly higher in all compartments when compared with the saline-inoculated controls (groups 1 and 3) (Table 2). Infiltrates were highest in the lumens of the bronchioles and alveoli.
Neutrophil counts (per μm2) (means ± SEM) within each pulmonary compartment in saline-inoculated control calves and M. haemolytica–inoculated calves at 2 hours/PI.
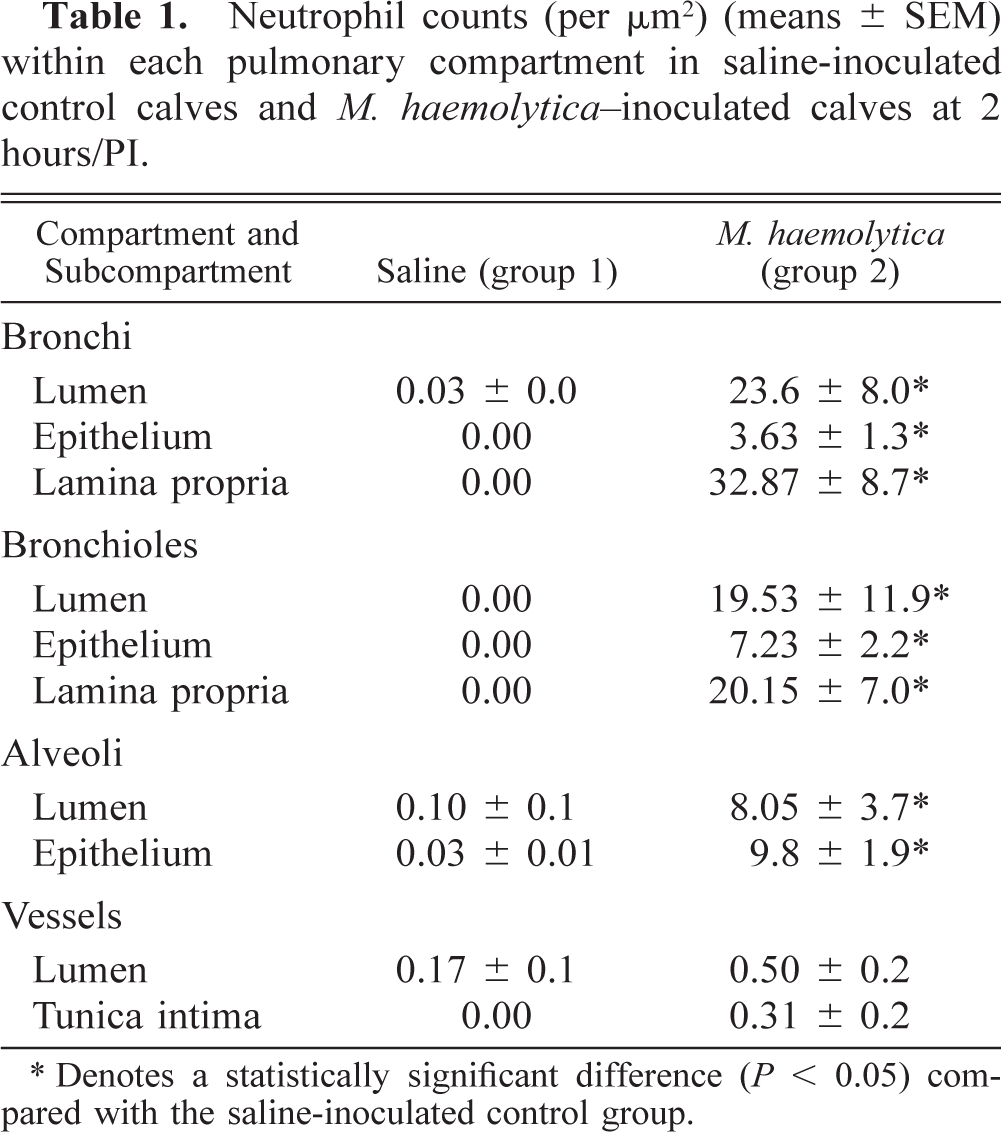
∗ Denotes a statistically significant difference (P < 0.05) compared with the saline-inoculated control group.
Neutrophil counts (per μm2) (means ± SEM) within each pulmonary compartment in saline-control inoculated calves (group 3), untreated (no TBC1269 treatment) M. haemolytica–inoculated calves (group 4), and TBC1269-treated M. haemolytica–inoculated calves (group 5) at 6 hours PI.

∗ Statistically significant (P < 0.05) compared with the saline-inoculated control group.
∗∗ Statistically significant (P < 0.05) compared with the M. haemolytica-inoculated group (with no TBC1269 treatment).
∗∗∗ Statistically significant (P < 0.05) compared with the M. haemolytica-inoculated group 2 hours PI.
M. haemolytica inoculated at 2 hours PI versus 6 hours PI. As expected, there was an overall increase in neutrophil infiltration in the lungs of calves at 6 hours PI (group 4) compared with that at 2 hours PI (group 2). In the specific lung compartments, infiltrates of neutrophils were significantly higher (P < 0.05) in the alveolar lumen and septae at 6 hours PI (group 4) than at 2 hours PI (group 2) in the M. haemolytica–inoculated groups (Tables 1, 2), but they were significantly lower in the bronchial lumen and epithelium at 6 hours PI than at 2 hours PI. There were no significant differences in neutrophil infiltration between saline-inoculated control animals at 2 hours PI (group 1) and at 6 hours PI (group 3).
TBC1269-treated M. haemolytica–inoculated versus M. haemoltytica–inoculated and saline-inoculated controls at 6 hours PI. TBC1269-treated M. haemolytica–inoculated calves (group 5) had significantly lower (P < 0.05) overall neutrophil infiltration in the lung lobes than the untreated M. haemolytica–inoculated calves (group 4) (Table 2). But the treated calves had significantly higher infiltrates of neutrophils (P < 0.05) than the saline-inoculated control calves (group 3).
Lung lobes from the TBC1269-treated M. haemolytica–inoculated (group 5) and saline-inoculated control calves (group 3) had significantly fewer (P < 0.05) neutrophils in the alveolar lumen and epithelium as well as in the bronchiolar lumen and lamina propria than lung lobes from the untreated M. haemolytica–inoculated calves (group 4; Table 2). There were no significant differences in neutrophil infiltration between the TBC1269-treated M. haemolytica–inoculated calves (group 5) and the untreated M. haemolytica–inoculated calves (group 4) in the bronchi and blood vessels.
In situ hybridization assessment of ICAM-1 mRNA
In situ ICAM-1 mRNA expression ranged from 0 to 3 (negative to strong) and was present in various pulmonary cell types in lung tissues from all groups (Figs. 2–4). Sections of tissues used as controls for hybridization specificity (i.e., no ICAM-1 probe or incubated with sense probe) lacked any detectable staining (Fig. 5). The intensity of ICAM-1 staining on the epithelium and endothelium of saline control lung tissue at 2 or 6 hours PI (groups 1 and 3) was not detectable to minimal (0–1 score) in all pulmonary cell types except bronchiolar cells, which ranged from minimal to moderate to strong expression (Figs. 2, 3, 6). Bronchiolar epithelium in the M. haemolytica–inoculated calves at 2 hours PI (group 2) also had moderate to strong ICAM-1 expression, and there was no significant difference between the M. haemolytica–inoculated (group 2) and saline-inoculated control calves (group 1) at 2 hours PI.
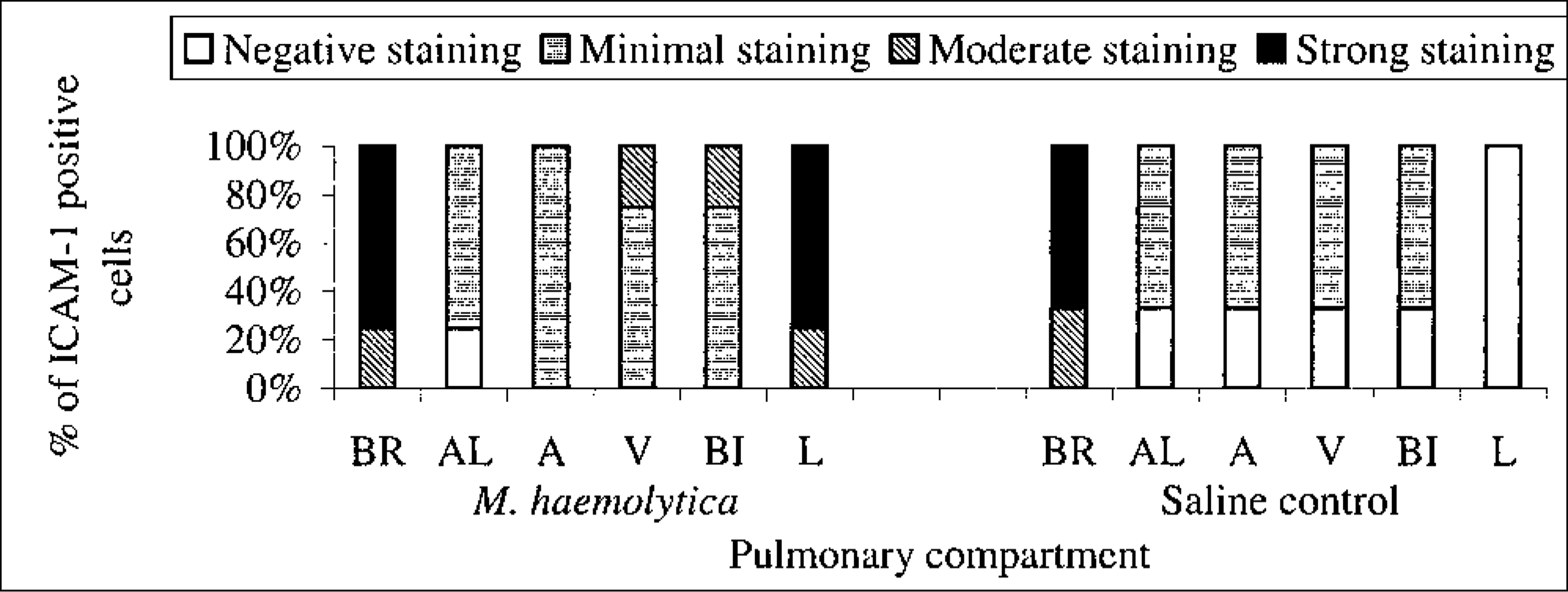
The percentage of staining intensity for ICAM-1 expression in the lungs of M. haemolytica–inoculated (first six bars at left) and saline-inoculated (last six bars at right) newborn calves at 2 hours PI. BR = bronchioles, AL = alveoli, A = arteries, V = veins, BI = bronchi, L = leukocytes.
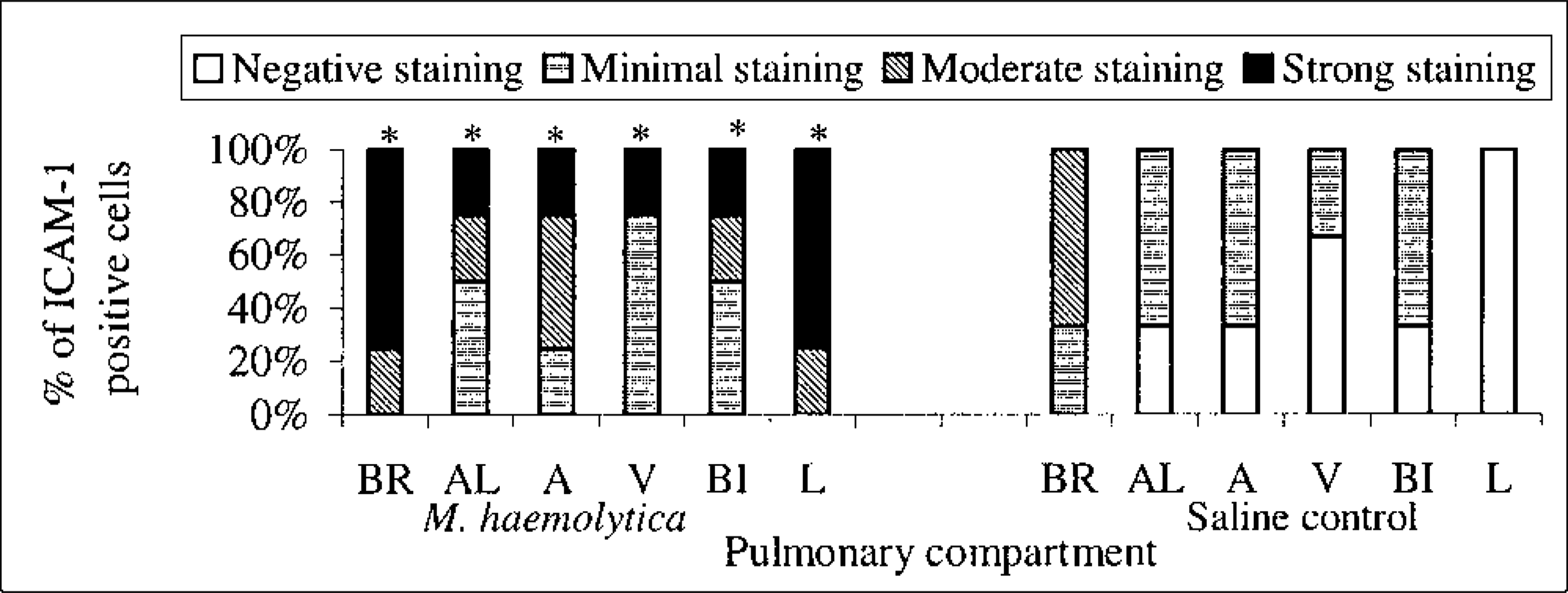
The percentage of staining intensity for ICAM-1 expression in the lungs of M. haemolytica–inoculated (first six bars at left) and saline-inoculated (last six bars at right) newborn calves at 6 hours PI. BR = bronchioles, AL = alveoli, A = arteries, V = veins, BI = bronchi, L = leukocytes. “∗” Denotes significant level (P < 0.05) compared with the saline-inoculated group.
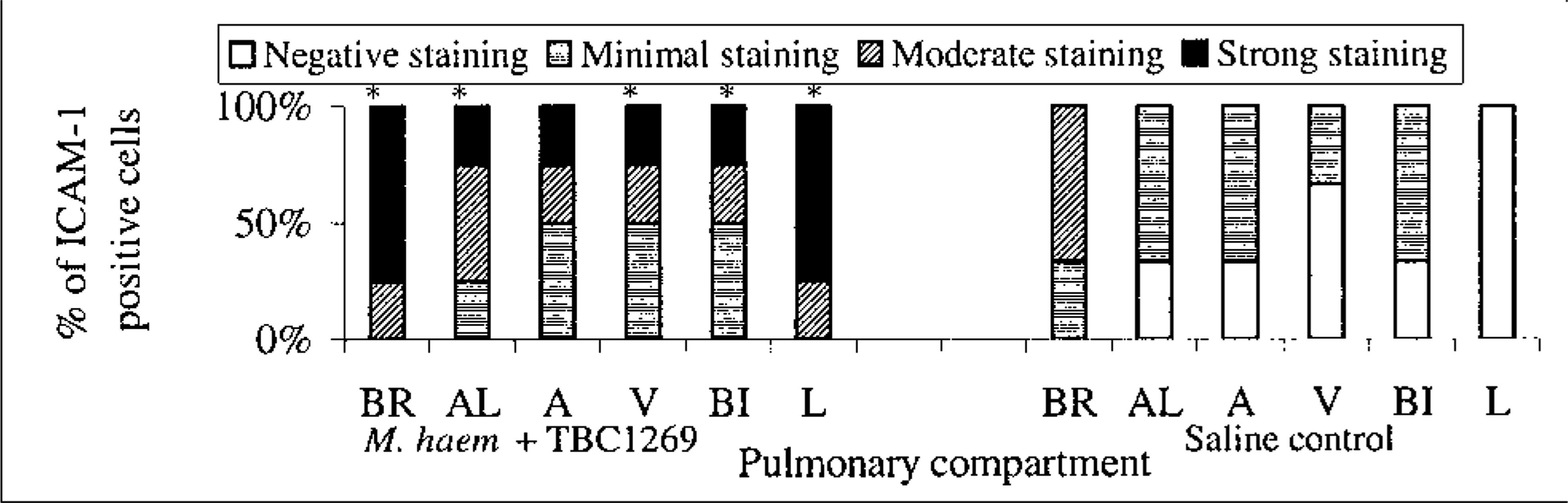
The percentage of staining intensity for ICAM-1 expression in the lungs of treated (M. haemolytica-inoculated & TBC1269-treated) (first six bars at left) and saline-inoculated (last six bars at right) newborn calves at 6 hours PI. BR = bronchioles, AL = alveoli, A = arteries, V = veins, BI = bronchi, L = leukocytes. “∗” Denotes significant level (P < 0.05) compared with the saline-inoculated group.
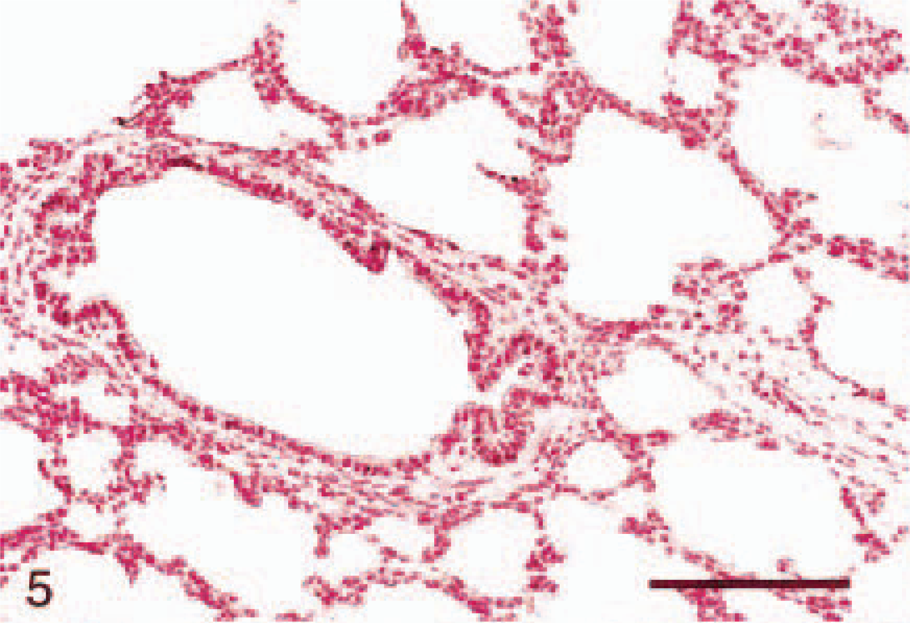
In situ hybridization on lung tissue; Holstein calf. Six hours PI, nuclear fast red counterstain. Saline-inoculated calf. No detectable staining (score = 0) of ICAM-1 mRNA expression in the pulmonary epithelium of alveoli and a bronchiole. Bar = 100 μm.
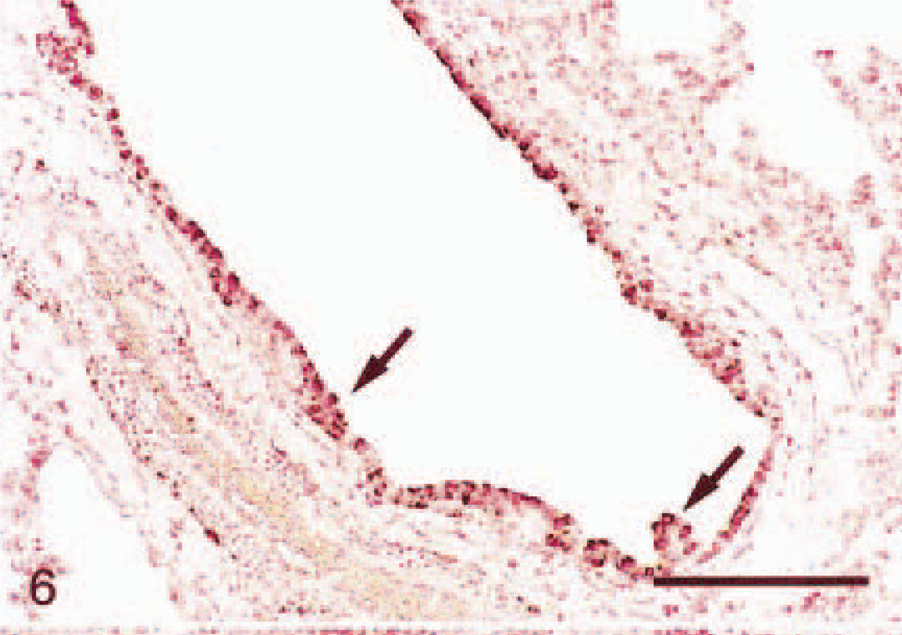
In situ hybridization on lung tissue; Holstein calf. Six hours PI, nuclear fast red counterstain. Saline-inoculated calf. Intense staining (score = 3) ICAM-1 mRNA expression in the pulmonary bronchiolar epithelium (arrows). Bar = 150μm.
At 6 hours PI the expression in lung tissues from both TBC1269-treated M. haemolytica–inoculated (group 5) and untreated M. haemolytica–inoculated animals (group 4) was significantly more intense (P < 0.05) in 1) the bronchiolar and bronchial epithelium, 2) the endothelium of arteries and veins, and 3) the alveolar epithelium, when compared with the saline-inoculated control group (group 3). The highest staining intensity was in the bronchiolar epithelium (Fig. 7). The only significant difference in ICAM-1 expression that was related to the time PI was in the endothelium of the pulmonary arteries, in which staining at 6 hours PI (groups 4 and 5) was greater than at 2 hours PI (group 2) in M. haemolytica–inoculated animals. In TBC1269-treated M. haemoltyica-inoculated (group 5) and untreated M. haemolytica–inoculated (group 4) calves, most of the neutrophils and alveolar macrophages that were present in airways, alveolar septa, or interstitium had intense staining for ICAM-1 (Fig. 8). There were no statistically significant differences in ICAM-1 mRNA expression between TB1269-treated M. haemolytica–inoculated (group 5) and untreated M. haemolytica–inoculated animals (group 4) (Fig. 9).
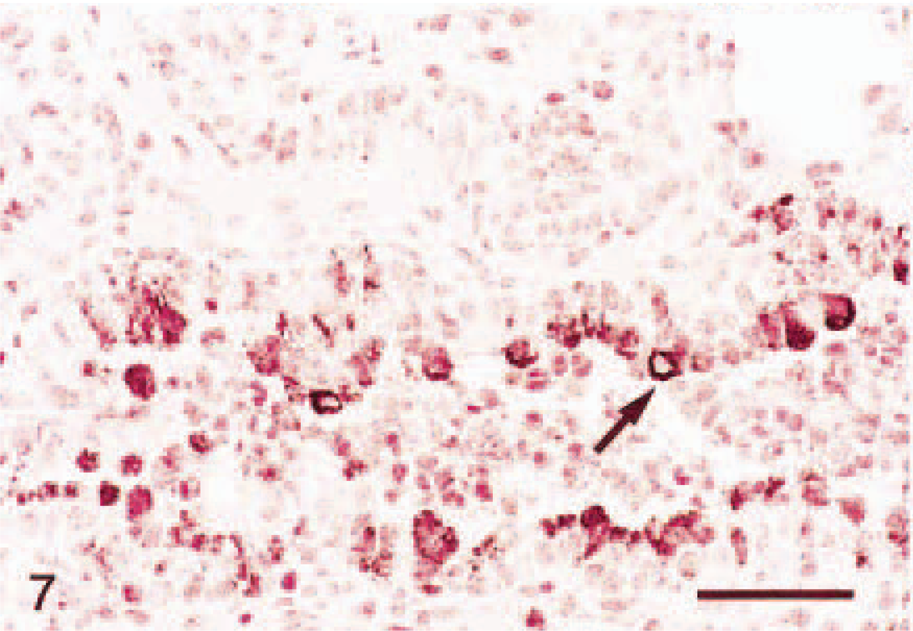
In situ hybridization on lung tissue; Holstein calf. Six hours PI, nuclear fast red counterstain. M. haemolytica–inoculated calf. Intense staining (score = 3) ICAM-1 mRNA expression in the bronchiolar epithelium (arrows). Bar = 40μm.
In situ hybridization on lung tissue; Holstein calf. Six hours PI, nuclear fast red counterstain. M. haemolytica–inoculated calf. Strong staining (score = 3) ICAM-1 mRNA expression of leukocytes (arrows) in the alveoli. Hybridization signals are seen as black grains. Bar = 100 μm.
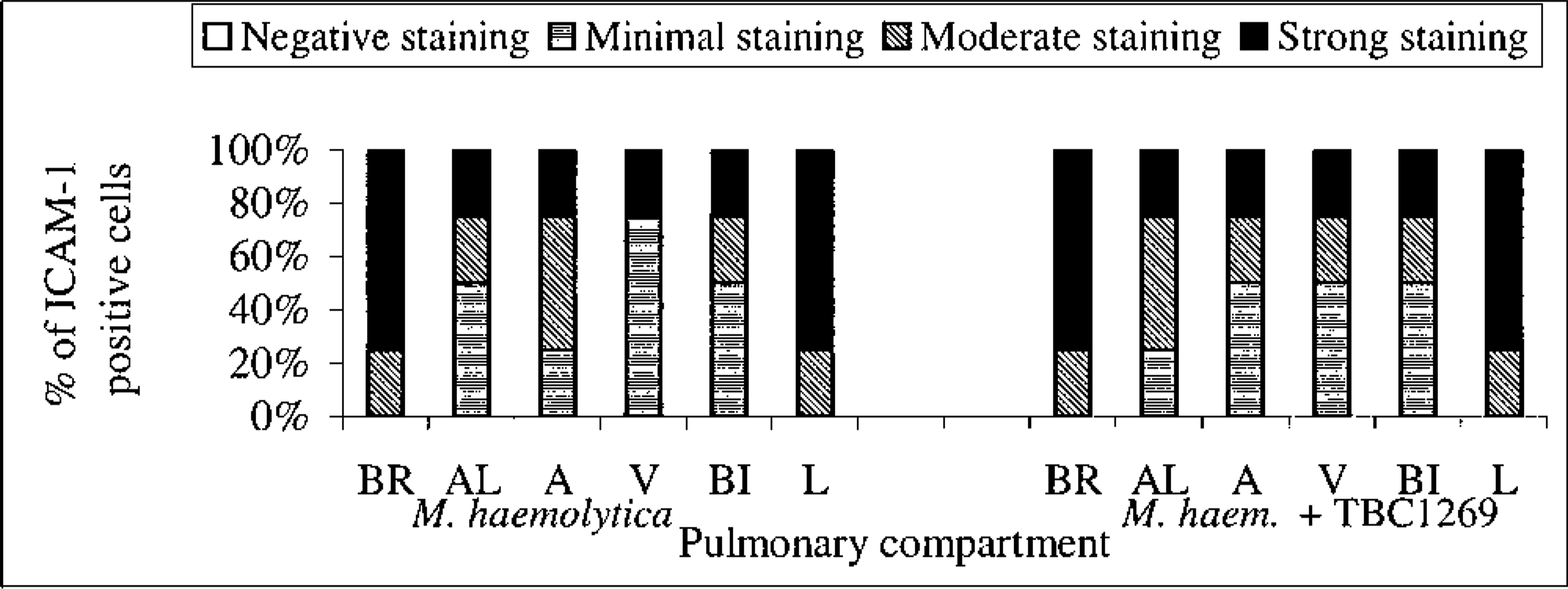
The percentage of staining intensity for ICAM-1 expression in the lungs of untreated (M. haemolytica-inoculated) (first six bars at left) and treated (M. haemolytica-inoculated and TBC1269 treated) (last six bars at right) newborn calves at 6 hours PI. BR = bronchioles, AL = alveoli, A = arteries, V = veins, BI = bronchi, L = leukocytes.
Discussion
The increased neutrophil infiltration in various pulmonary compartments 2 or 6 hours after M. haemolytica inoculation is consistent with other reports.2–4,33 The highest degree of infiltration was in the bronchiolar compartment, whereas the lowest was in the vascular compartment.
The role of selectin adhesion molecules (L-, E-, P-selectin) during acute inflammation has been studied but has not been fully established.17 L-selectin–deficient mice have reduced leukocyte infiltration into sites of inflammation, and humans who lack functional sialyl-Lewisx (LAD type II [LAD II]) have decreased impaired leukocyte egress, which results in recurrent infections of the mucosal surfaces.15,34 Inhibiting the selectin interactions with their ligands has been shown to have potential therapeutic effects by inhibiting neutrophil infiltration and subsequent tissue damage.26,31 In the present study, neonatal calves treated with the selectin inhibitor, TBC1269, had a significantly lower overall degree of neutrophil infiltration after intrabronchial M. haemolytica–induced pulmonary inflammation than untreated calves. This indicates that the selectin inhibitor, TBC1269, reduces the selectin-mediated infiltration of neutrophils into the lung. This is in agreement with other reports using TBC1269 in both an in vivo ruminant model of allergic inflammation and in in vitro studies.1,12 Manufacturing limitations make it impractical to administer most of the other inhibitors of adhesion molecules, such as MAbs and synthesized peptides, to larger animals such as calves. In addition, some inhibitors can induce hypersensitivity. TBC1269 appears to lack these limitations because sufficient quantities can be synthesized for large animals and humans, and no hypersensitivity reactions have been observed in calves in this study and in human trials.
In contrast to previous studies, this study determined the degree of neutrophil infiltration in specific compartments of the lung (bronchi, bronchioles, alveoli, and blood vessels) and in specific subcompartments (lumens, lining epithelium [or alveolar septa], lamina propria, and adventitia) to determine more precisely the regional influences of selectin-mediated infiltration. This analysis revealed that TBC1269 had the highest degree of inhibition of neutrophils entering the alveolar lumen and septae, and this was followed by the bronchiolar lumen and lamina propria. In previous studies with this model using the same inoculum (fiberoptic deposition of M. haemolytica) in the same breed (Holstein), we have determined that in the bovine lung there is a CD18 (β2 integrin)–dependent mechanism of leukocyte infiltration in the airways (bronchi and bronchioles) and a CD18-independent mechanism that is operative in the alveoli.3 Biologically, the significant reduction in neutrophil infiltration into these locations of alveoli and bronchioles with TBC1269 indicates that selectin-mediated adherence is integral to adherence in the lower portions of the lung during acute pneumonia. Therapeutically, TBC1269 may temporarily reduce neutrophil infiltration and its associated lung damage, and this would allow improved gaseous exchange and distribution of nebulized and intravenous drugs during episodes of intense neutrophil infiltration.
Although the reduction was not significant in one CD18-dependent compartment (bronchi), there was a significant reduction in neutrophil infiltration in the other CD18-dependent compartment (bronchioles) after TBC1269 treatment. The bronchiolar but not bronchial reduction in neutrophil infiltration could have several causes. First, it may be the result of age differences in the groups that were used in this study as compared with previous studies. In this study we used 1- to 3-day-old newborn calves because this is the period when neutrophil activity is especially vital for protection against pathogens. In contrast, previous studies assessed young or adult cattle.3 Second, in some species, including cattle, there is reduced L-selectin expression in neonates in comparison with adults, and this may alter neutrophil infiltration into the bronchi and bronchioles.16,24 For example, it has been found that MAbs against L-selectin did not attenuate the inflammatory process in neonatal rabbits, whereas it resulted in a significant reduction of leukocyte infiltration in adult rabbits.16 Third, the expression of P- and E-selectin has not been studied in neonatal calves, and these may also have a role in bronchiolar adherence. Ohnishi et al.27 have demonstrated that P-selectin had a vital role in lipopolysaccharide-induced lung injury, and in a study in rabbits it was demonstrated that anti–P-selectin MAb significantly reduced acute lung injury.19 Neutrophils likely had a role in lung damage in both these studies, but the studies did not specify the exact location of decreased selectin activity in the lung. Therefore, this aspect of neutrophil infiltration cannot be completely compared. Fourth, it may be that selectin receptor expression by endothelial cells is simply reduced in newborn calves only in the bronchi but not in the bronchioles. This may explain why using the TBC1269 selectin antagonist in our calf model inhibited neutrophil infiltration into the bronchioles but not into the bronchi. Fifth, TBC1269 may inhibit or alter selectin-mediated signaling and activity downstream to selectin signaling. Sixth, the deposition of bacteria, and thus the stimulus for neutrophil infiltration, may be less in bronchi than in bronchioles and alveoli. Finally, the pathway from the blood into the airway lumen appears less complicated in bronchioles and alveoli, which have limited perivascular stroma, than in bronchi.
It is well-established that ICAM-1 expression is important for tight leukocyte adhesion and subsequent extravasation to sites of infection.5,37 In the present study, ICAM-1 mRNA expression in the lungs of all saline control newborn calves was absent or minimal in the epithelium of bronchi and alveoli and in the endothelium of arteries and veins. This is consistent with our previous studies in adult cattle.29 The significant increase in ICAM-1 expression after M. haemolytica inoculation in both TBC1269-treated (group 5) and untreated calves (group 4) and the differential expression of ICAM-1 in various pulmonary cell types after M. haemolytica inoculation were similar to what was reported in adult cattle.9 The intensity of staining was significantly increased on the bronchiolar epithelium, the endothelium of arteries and veins, and the alveolar epithelium and was slightly increased on the bronchial epithelium. ICAM-1 mRNA was expressed at high levels in the locations of greatest neutrophil infiltration, i.e., the bronchioles and the alveolar epithelium. Therefore, CD18-ICAM-1–mediated neutrophil infiltration likely occurs despite selectin inhibition.
Leukocyte infiltration in the lung is complicated by the numerous adhesion molecule families8 and the dynamics of the intrathoracic vascular and air pressures. Findings in this study suggest that inhibition of selectin activity alone temporarily reduces neutrophil infiltration into specific microscopic regions (alveolar lumen and epithelium, and bronchiolar lumen and lamina propria) of the lungs of neonates during the early stages of Gram-negative bacterial pneumonia. The selectins appear to have a predominant role in mediating alveolar infiltration, whereas CD18 has an important role in mediating bronchial infiltration. Bronchiolar infiltration appears intermediate and is likely optimal with both selectin- and CD18-mediated adherence.
Footnotes
Acknowledgements
We thank Drs E-K. Lee and K. Register for providing the ICAM-1 plasmid; J. Nott, E. Snook, and M. Carter, Carter Image Analysis Facility, Iowa State University; D. Nordman for his help in the statistical analysis; and J. Fosse for preparation of the figures. This work was supported by USDA grant 97-35204-4769.
