Abstract
Adhesion molecules on endothelial cells play an important role in leukocyte recruitment in several inflammatory processes. Vascular selectins mediate the initial adhesion of leukocytes to the blood vessel wall during their extravasation into inflamed tissues, and in vitro studies in dogs have shown that selectin expression can be induced by cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1 (IL-1). The objective of this study was to determine whether vascular selectins are induced by cytokines in vivo in a cutaneous model of inflammation in dogs. Skin biopsies were collected from nine dogs at various time points after an intradermal injection of TNF-α (10 ng/site) or phosphate-buffered saline containing 0.1% bovine serum albumin, and immunohistochemistry was performed using anti-P-selectin (MD3) and anti-E-selectin (CL37) monoclonal antibodies. In all animals, TNF-α induced an inflammatory reaction that was maximal at 12 hours and then decreased by 24 and 48 hours. Control skin displayed no expression of E- and P-selectin, whereas TNF-α induced the expression of P-selectin and E-selectin on dermal vessels that was highest at 12 hours and 3 hours, respectively (P < 0.05). Numerous platelet aggregates recognized by the anti-P-selectin antibody were present in the lumina of vessels and in perivascular tissues. These results demonstrate that TNF-α can induce the expression of P- and E-selectin in vivo in dog skin and suggest that these selectins are involved in leukocyte recruitment in canine dermatitis.
The accumulation of inflammatory cells into injured tissues results from a tightly controlled cascade of interactions between endothelium and leukocytes. The first adhesive event involves the margination and rolling of leukocytes along endothelial cells. 14,28 Leukocyte slowing is required for subsequent firmer adhesion and for their selective exit into the tissues. The selectins are adhesion molecules that mediate rolling, and they are present in endothelial cells and platelets and on the surface of some leukocytes. 5,11,14,28,37,48,52 They are transmembrane glycoproteins with a terminal lectin domain that is the binding site for their specific sialylated carbohydrate ligands. 27,52 P-selectin is stored in the Weibel-Palade bodies of endothelial cells and in the alpha granules of platelets, and a number of proinflammatory signals can induce its rapid translocation to the cell surface. 23,30,31 Inflammatory mediators such as tumor necrosis factor alpha (TNF-α) and bacterial lipopolysaccharide (LPS) also can induce the synthesis of P-selectin in vitro and in vivo, providing a means of sustaining leukocyte recruitment for longer periods. 1,4,13,16,20,41,55,58 In contrast, E-selectin is not found in unstimulated endothelial cells, but its expression can be induced by several inflammatory mediators. 27,48 The third member of the family of selectins, L-selectin, is constitutively expressed by granulocytes, monocytes, and most lymphocytes, and its expression is downregulated upon stimulation. 14
In humans, increased expression of endothelial adhesion molecules has been demonstrated in various skin diseases. 2,3,9,18,19,26,51,53,59 Increased expression of E-selectin, P-selectin, intercellular adhesion molecule 1, or vascular cell adhesion molecule 1 has been reported in patients with psoriasis, allergic contact dermatitis, atopic dermatitis, and chronic or dermographic urticaria. 2,3,9,18,19,26,51,53,59 Involvement of selectins in skin inflammation has been studied experimentally using different models, notably animals genetically deficient in these molecules. 8,24,25,44,46,56 For example, P-selectin–deficient mice exhibit a significantly lower baseline level of leukocyte rolling in skin postcapillary venules than do wild-type animals. 56 In models of cutaneous contact hypersensitivity, accumulation of CD4+ T lymphocytes, monocytes, and neutrophils is reduced significantly in P-selectin–deficient mice and in rats treated with anti–P-selectin antibodies. 44,46,50 Moreover, induction of an acute dermal inflammation by injection of zymosan in E- and P-selectin–deficient mice has shown that both selectins are required for neutrophil accumulation and that there is considerable overlap in their functions. 25 Both E- and P-selectin are critical in acute and chronic skin inflammation. 6 In contrast, L-selectin does not appear to play a significant role in leukocyte recruitment in the skin. 6,37
In dogs, blood vessels expressing P-selectin are increased in cases of allergic dermatitis, autoimmune dermatitis, pyogranulomatous dermatitis, dermatophytosis, and panniculitis as compared with normal skin, and there is a positive correlation between P-selectin expression and the accumulation of leukocytes in the dermis. 7 These findings suggest that P-selectin on dermal vessels participates in the local recruitment of inflammatory cells in skin diseases in dogs. Stimulation of canine endothelial cells in vitro by proinflammatory mediators such as LPS and TNF-α increases both P-selectin mRNA levels and protein synthesis. 13 However, in vivo induction of P-selectin synthesis and the role of TNF-α in dermatitis have not been investigated in dogs. Therefore, the objective of the present study was to characterize the cellular response and the expression of vascular selectins following TNF-α injection in canine skin.
Materials and Methods
Skin inflammatory model
Healthy adult Beagle dogs (n = 9) were used. Skin on one side of the thorax was shaved. Sterile recombinant human TNF-α (R&D Systems, Minneapolis, MN; 10 ng/site) reconstituted in phosphate-buffered saline (PBS) containing 0.1% bovine serum albumin (PBS/BSA; n = 6 dogs) or PBS/BSA alone (n = 3 dogs) was administered intradermally using a 27-ga needle. Punch biopsies (6 mm) were taken under local anesthesia (subcutaneous injection of 0.7 ml of 2% lidocaine with epinephrine; Austin, Division of Vetoquinol Canada, Joliette, PQ, Canada) 0, 3, 6, 12, 24, and 48 hours following injection of TNF-α or PBS/BSA. Biopsies were bisected; one half was snap-frozen in OCT (Sakura Finetek USA, Torrance, CA) using isopentane/liquid nitrogen, and the other half was fixed in 10% formalin and embedded in paraffin. Hematoxylin–eosin–saffron (HES)-stained sections were prepared for histologic examination. The HES stain is routinely used at the Faculty of Veterinary Medicine in St. Hyacinthe. The saffron gives a yellow-orange color to collagen.
Antibodies
The anti-canine P-selectin monoclonal antibody (MD3) was produced in mice that had been immunized with thrombin-stimulated dog platelets. 10 MD3 recognizes a nonblocking epitope of P-selectin in paraffin-embedded tissues and was used as tissue culture supernatant (1:10 dilution in PBS). A monoclonal antibody directed against E-selectin (CL37) was generated by immunization of mice with human umbilical vein endothelial cells stimulated with recombinant human interleukin-1β (IL-1β). 35 CL37 recognizes human 35 and canine (Dr. C. W. Smith, personal communication) endothelial E-selectin in frozen tissue sections. PBS was used as a negative control.
Immunohistochemistry
Immunohistochemical staining was performed using the Vectastain ABC kit (Vector Laboratories, Burlingame, CA) as previously described. 7,10,13 Paraffin-embedded 3-µm sections were deparaffined through a graded alcohol series and used for the P-selectin staining with antibody MD3. Frozen sections were air-dried at room temperature for 2 hours, fixed for 30 seconds in acetone, rinsed in PBS for 15 minutes, and used for the E-selectin staining with antibody CL37. Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in methanol for 30 minutes. After rinsing in PBS for 15 minutes, sections were incubated with diluted normal horse serum for 20 minutes at room temperature. Primary antibody diluted in PBS (MD3, 1:10 dilution; CL37, 1 µg/ml) was applied, and sections were incubated overnight at 4 C. Control sections were incubated with PBS. After rinsing in PBS for 10 minutes, a biotinylated horse anti-mouse antibody (1:222 dilution; Vector Laboratories) was applied, and sections were incubated for 45 minutes at room temperature. Sections were washed in PBS for 10 minutes and incubated with the avidin DH-biotinylated horseradish peroxidase H reagents for 45 minutes at room temperature. After washing with PBS for 10 minutes, the reaction was revealed using diaminobenzidine tetrahydrochloride as the peroxidase substrate. Sections were counterstained with Gill's hematoxylin stain and mounted.
Microscopic evaluation
HES-stained tissue sections were evaluated using a light microscope to quantify the inflammatory reaction in the dermis and the panniculus. Inflammatory cells (including neutrophils, macrophages, lymphocytes, plasma cells, and eosinophils) were counted in nine representative fields in the dermis and nine representative fields in the panniculus. Immunostained tissue sections were examined to determine the number of vessels immunoreactive for MD3 and CL37. The total number of positively stained blood vessels was evaluated in each section.
Statistical analysis
The effect of time on the number of infiltrating leukocytes and on the number of E- and P-selectin–positive vessels was analyzed using a one-way analysis of variance followed by multiple comparisons of means with the Tukey–Kramer method; statistical significance was set at P < 0.05. All values are given as mean ± SEM.
Results
TNF-induced inflammatory reaction
Histologic examination revealed that injection of TNF-α (10 ng/site) induced an acute inflammatory reaction in all dogs (n = 6) (Figs. 1–6). Although normal skin (from dogs injected with PBS/BSA alone) displayed no or rare inflammatory cells (Fig. 1), neutrophils were seen adherent to and extravasating from dilated dermal and subcutaneous venules by 3 hours after injection of TNF-α (Fig. 2). Thereafter, there was a progressive increase in the number of extravascular neutrophils at 6 and 12 hours, accompanied in some areas by a perivascular exudation of fibrin (Figs. 3, 4), and the number of leukocytes peaked at 12 hours (P < 0.05; Figs. 4, 7). At 24 and 48 hours, the leukocyte infiltration had decreased (Fig. 7), and some macrophages were observed among the population of neutrophils. Few eosinophils were observed in one animal.
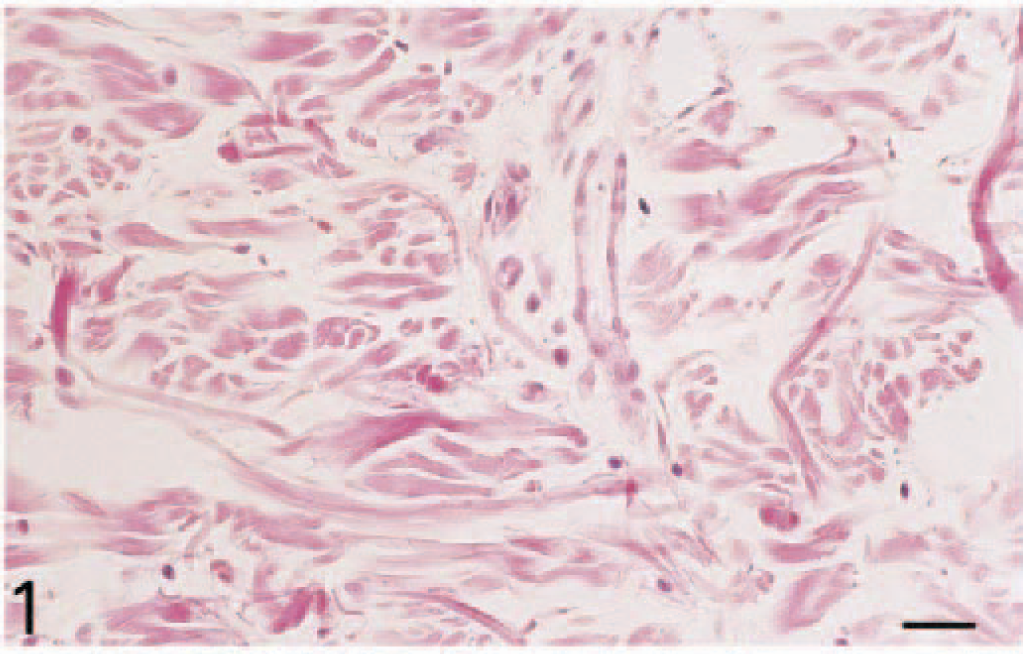
Skin; dog. Control skin (injected with PBS/BSA) shows only rare inflammatory cells. HES. Bar = 25 µm.
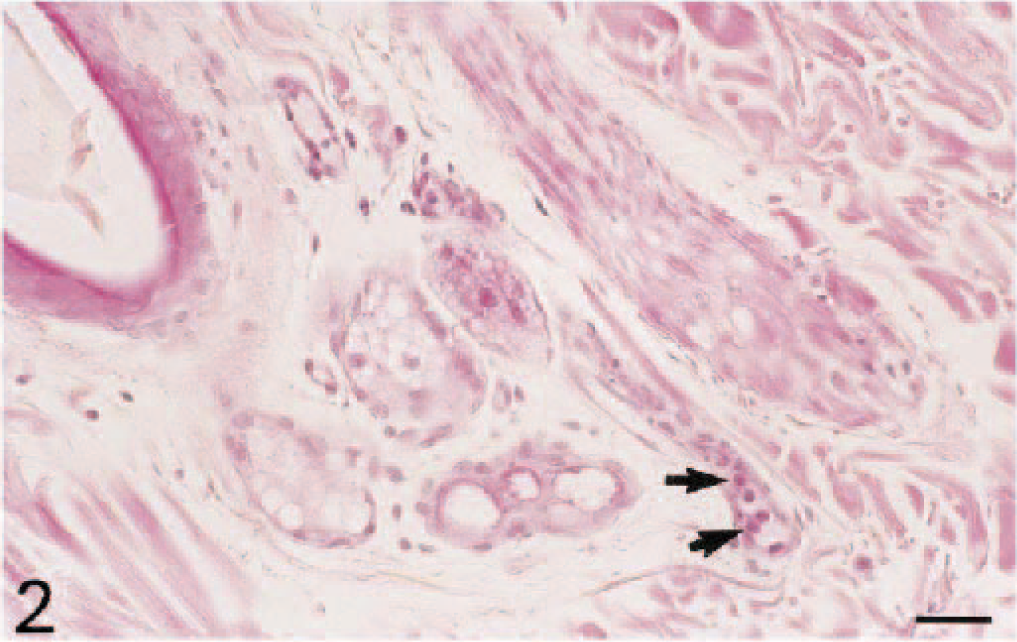
Skin; dog. Three hours after intradermal injection of TNF-α, neutrophils (arrows) are adherent to venular endothelium and are starting to extravasate. HES. Bar = 25 µm.
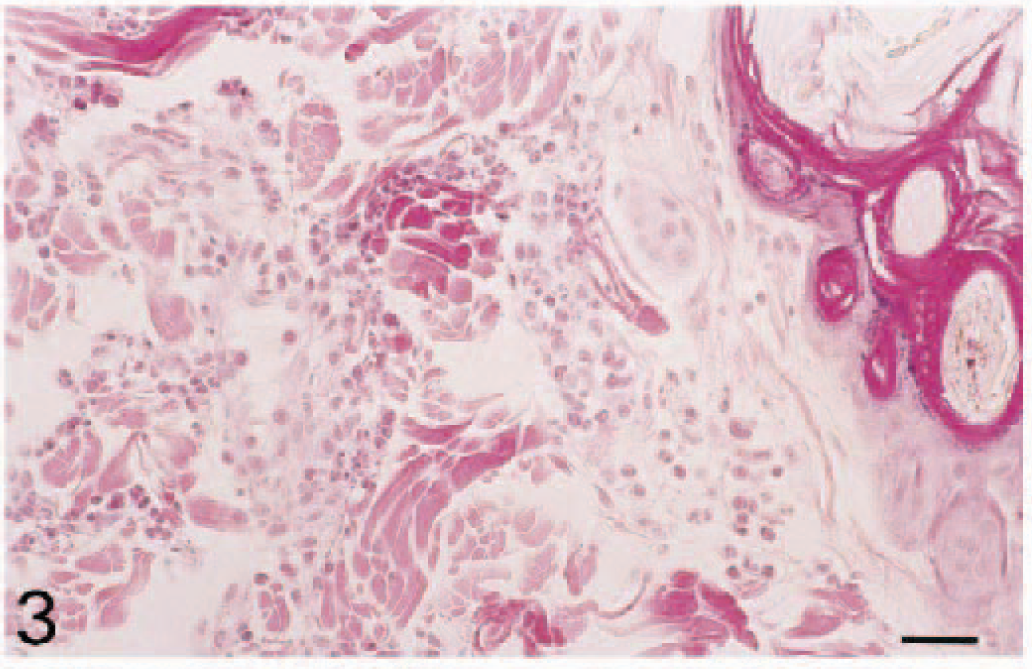
Skin; dog. Six hours after intradermal injection of TNF-α, more neutrophils are present within blood vessels and in interstitial tissues. HES. Bar = 25 µm.
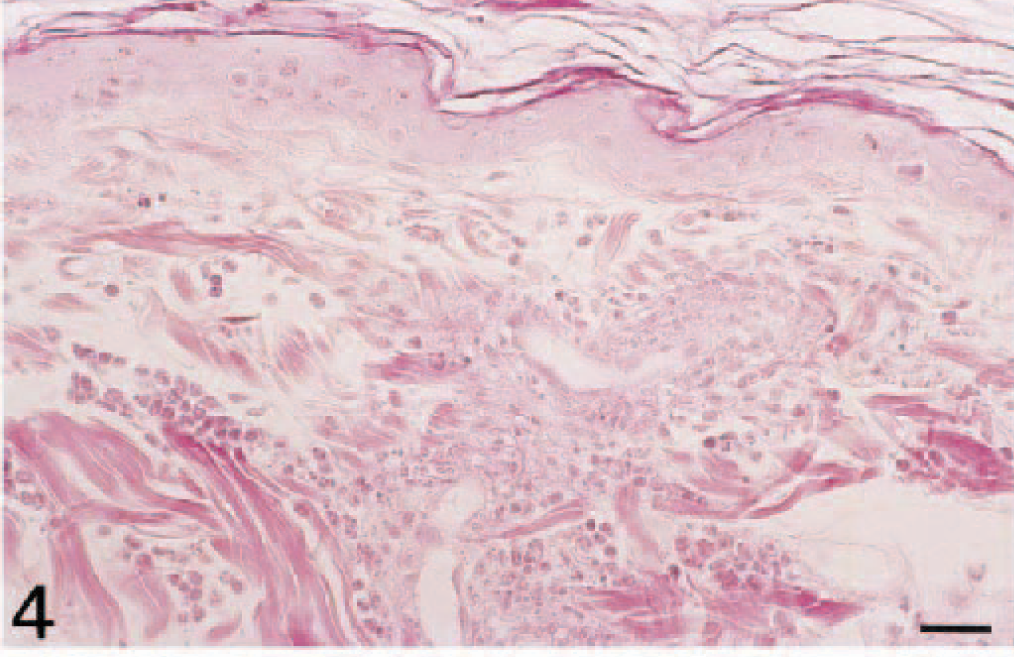
Skin; dog. Twelve hours after intradermal injection of TNF-α, the leukocyte infiltration is composed mostly of neutrophils and is accompanied by perivascular exudation of fibrin. HES. Bar = 25 µm.
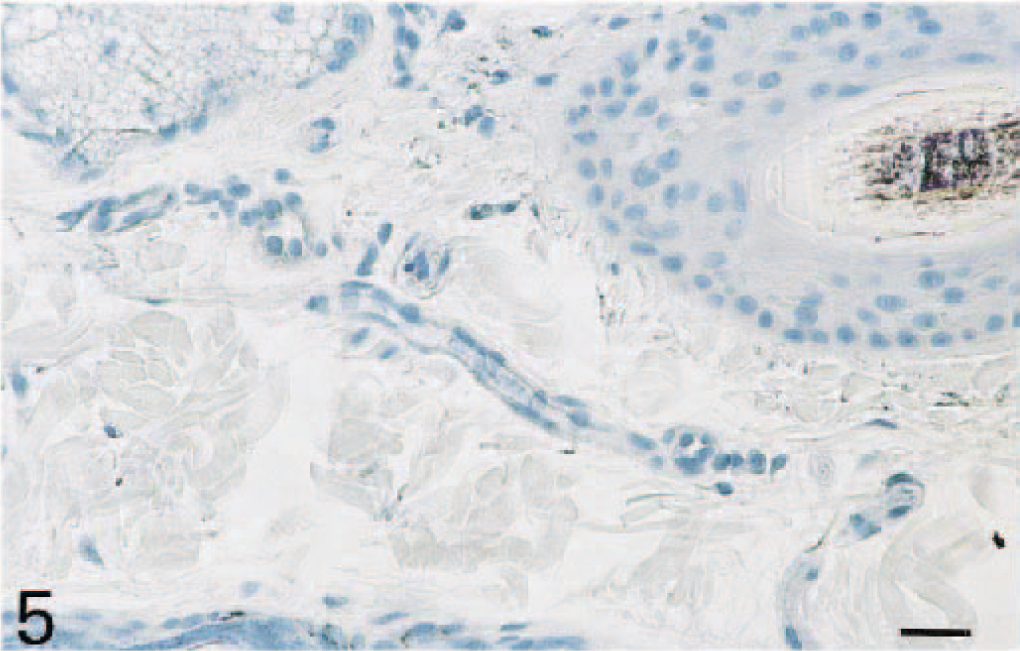
Skin; dog. Immunohistochemistry was performed on formalin-fixed tissues using the anti-canine P-selectin monoclonal antibody MD3. No P-selectin expression is present in normal skin. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 25 µm.
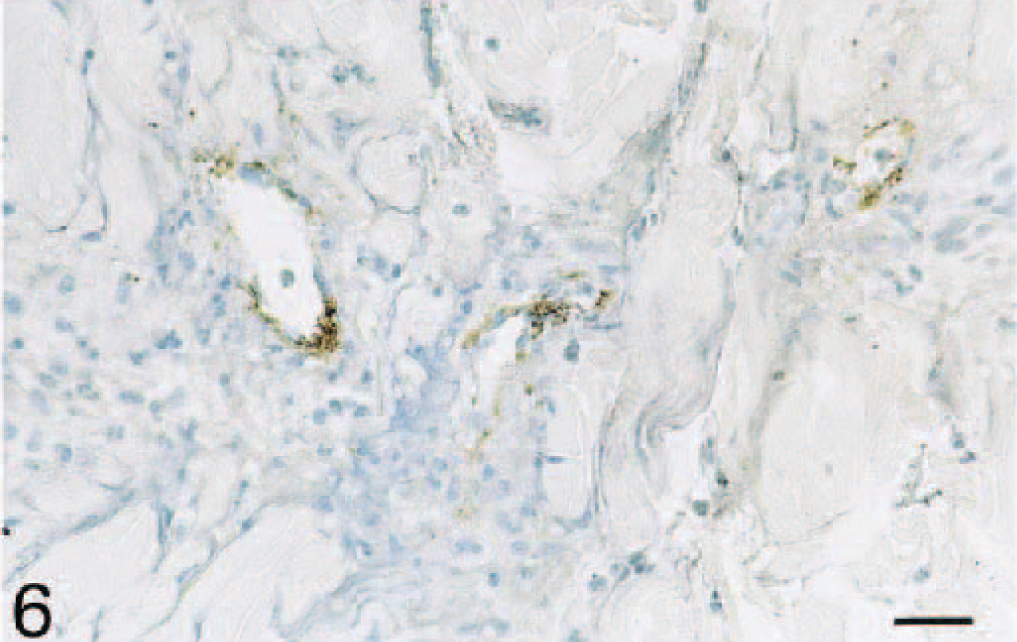
Skin; dog. Immunohistochemistry was performed on formalin-fixed tissues using the anti-canine P-selectin monoclonal antibody MD3. Many blood vessels are immunoreactive for P-selectin at 12 hours following TNF-α injection. Avidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 25 µ.
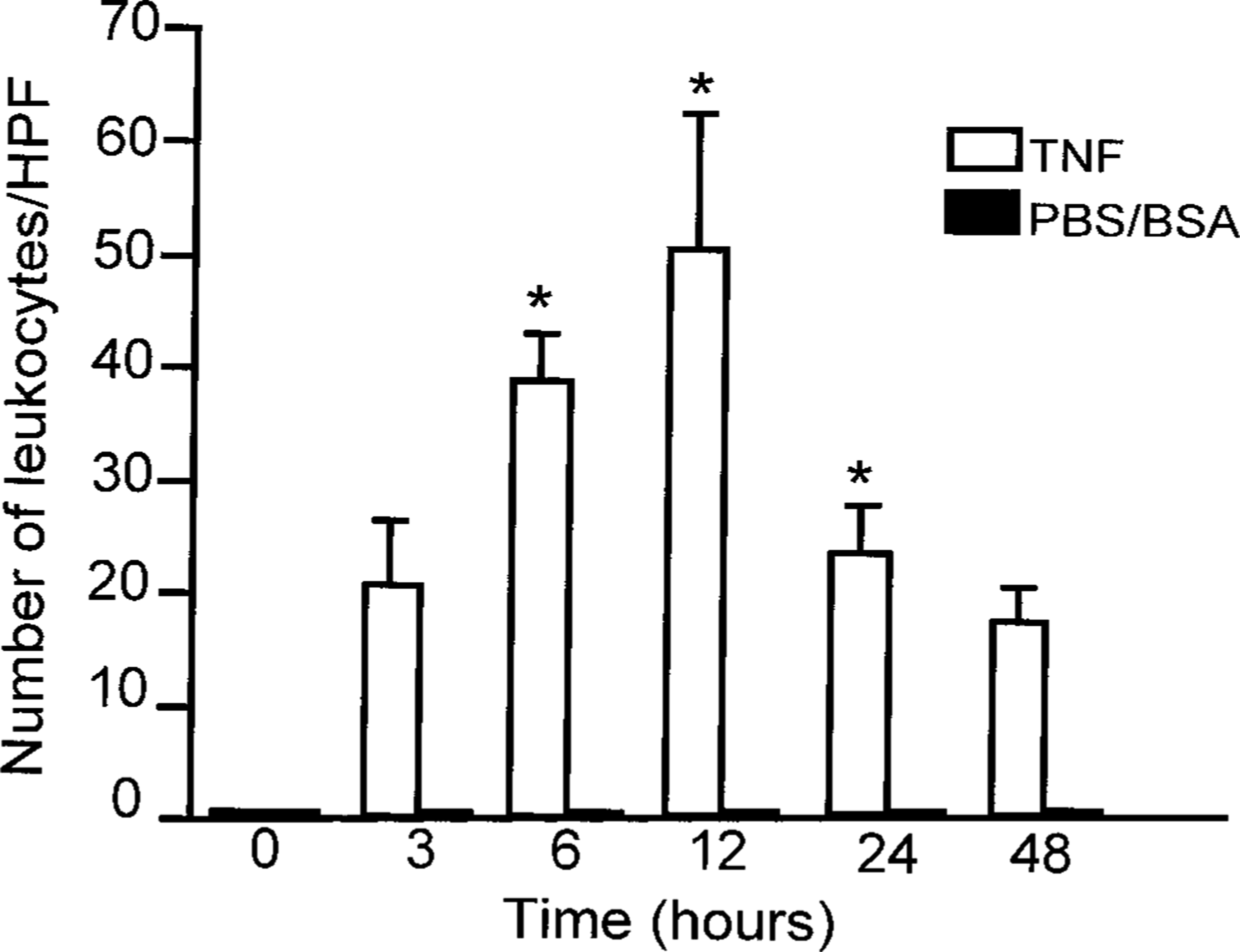
Leukocyte infiltration in TNF-α-induced dermatitis in dogs. The number of leukocytes (neutrophils, macrophages, plasma cells, lymphocytes, and eosinophils) infiltrating the lesions per high-power field (HPF) was determined. Results are presented as mean ± SEM. ∗Significantly different from control skin (PBS/BSA), P < 0.05.
Expression of P- and E-selectin in TNF-α–induced dermatitis
No significant P-selectin expression was detected in endothelial cells from blood vessels in the normal skin (Figs. 5, 8). However, following TNF-α injection, P-selectin–expressing blood vessels were observed in the dermis at all time points, with a maximal number of positive blood vessels observed at 12 hours after TNF-α administration (P < 0.05; Figs. 6, 8). In three of the six dogs, several positive vessels were present at 12 hours (20 ± 6), whereas the other three dogs showed very few vessels (1 ± 1). P-selectin–positive blood vessels were often associated with the presence of leukocytes in the area surrounding the vessels, but leukocyte infiltration was not always associated with P-selectin–reactive vessels in the area. In addition to endothelial cells, P-selectin–expressing platelets were present both within the lumina of blood vessels and in the perivascular dermis at all time points after TNF-α injection (data not shown).
As with P-selectin, E-selectin was not found in blood vessels in the normal skin (data not shown). However, TNF-α injection induced E-selectin expression on dermal vessels at 3, 6, and 12 hours (P < 0.05; Fig. 8), with a subsequent decrease to negligible levels by 24 hours.
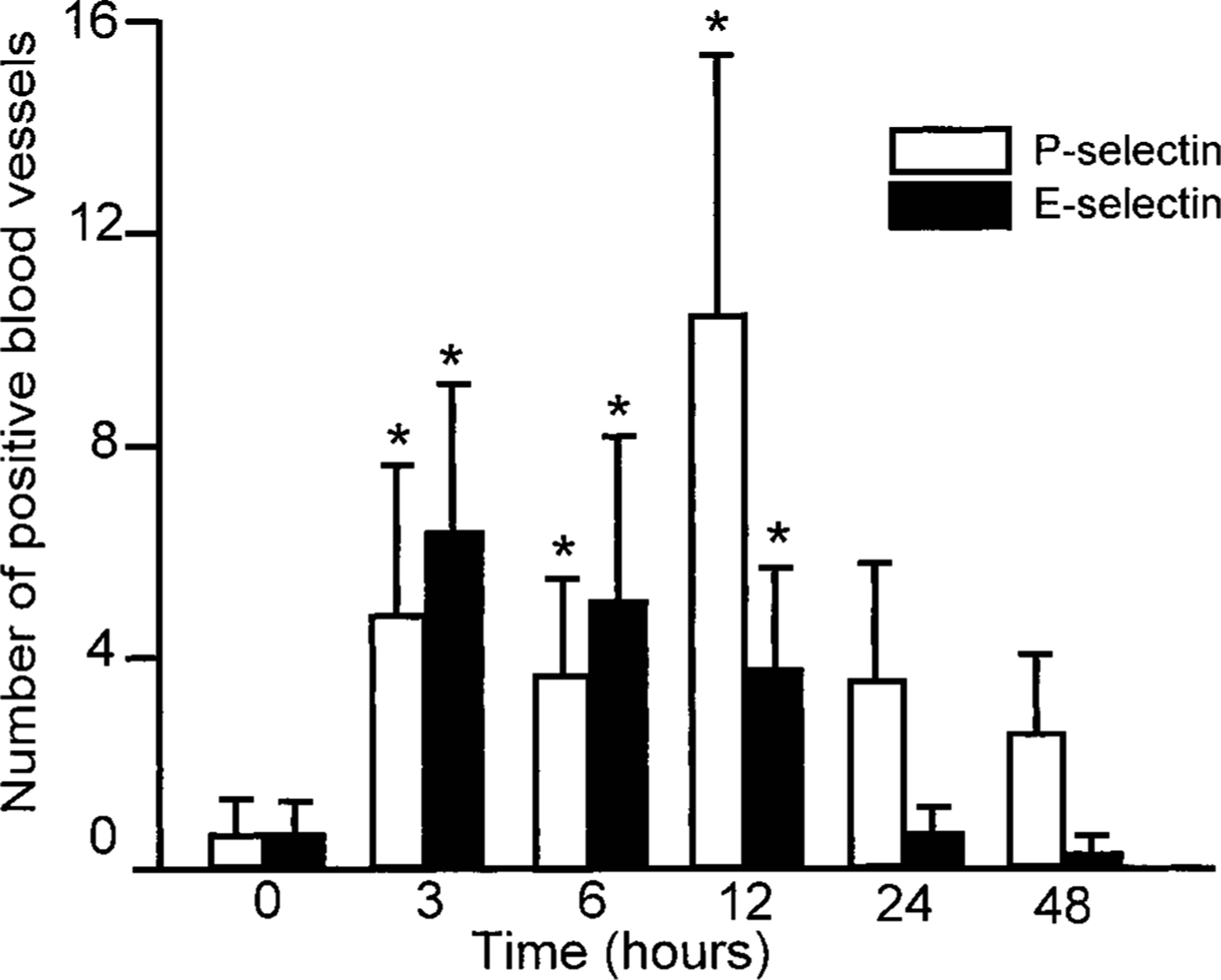
E- and P-selectin expression in TNF-α–induced dermatitis in dogs. Immunohistochemistry was performed on formalin-fixed or frozen tissues using the anti-canine P-selectin monoclonal antibody MD3 or the anti-E-selectin monoclonal antibody CL37. Blood vessels staining positively were counted and expressed as the number of E- or P-selectin–expressing blood vessels per tissue section. ∗Significantly different from 0 hours, P < 0.05.
Discussion
This study demonstrates for the first time the in vivo induction of P-selectin expression in dog tissues by an inflammatory mediator such as TNF-α. Previously, TNF-α was shown to induce P-selectin mRNA and protein in canine endothelial cells in vitro, but in vivo induction had never been reported in dogs. P-selectin is prestored in Weibel-Palade bodies of endothelial cells and is therefore readily available to be expressed on the cell surface in the very early phases of an inflammatory reaction. However, its expression is only transient; it is soon either proteolytically released into the circulation or rapidly endocytosed. 17,22,42,45 Endocytosed P-selectin can be recycled to the Weibel-Palade bodies or degraded in lysosomes. 17,22,42,45 However, increased transcription and translation of P-selectin due to the effect of proinflammatory cytokines could represent a means of sustaining P-selectin expression and the recruitment of leukocytes at later stages of inflammation.
Important species differences have recently been reported in the induction of P-selectin by inflammatory mediators such as TNF-α and LPS. Studies have shown that TNF-α, IL-1β, and LPS increase expression of P-selectin mRNA and protein in mouse endothelial cells both in vitro and in vivo and in bovine endothelial cells in vitro. 4,20,41,55 However, these same mediators do not increase P-selectin mRNA in human endothelial cells in vitro. 57 The binding sites for NF-kappa B and ATF-2, two transcription factors necessary for the activation of the murine P-selectin gene, are not present in the promotor of the human P-selectin gene. 38,39 In a recent study, intravenous administration of Escherichia coli, which markedly elevates plasma LPS and TNF-α, did not increase mRNA for P-selectin in baboons. 29 These results suggest that important species differences exist and that caution is needed when extrapolating the function of P-selectin from one species to another. The results of the present study indicate that the expression of canine P-selectin is more similar to that of murine and bovine P-selectin; both in vitro and in vivo upregulation of P-selectin have been demonstrated in these species.
In the present study, some P-selectin–positive blood vessels were observed 3 hours after TNF-α injection, but maximal expression was reached only at 12 hours. This time course of expression is different from the time course observed in vitro with canine endothelial cells, where induction of P-selectin mRNA by TNF-α and LPS occurred between 3 and 6 hours. 13 Previous in vitro studies have also shown P-selectin protein in endothelial cells at 3.5 hours following LPS stimulation. 13 These results suggest that there are differences in the time course of induction of P-selectin between in vitro and in vivo conditions. The reasons for the maximal P-selectin expression at 12 hours are unclear. It may be that P-selectin expression in vivo appeared early and thus was missed in the present study. However, the skin from one animal was analyzed at 1 hour postinjection, and there were no P-selectin–expressing vessels (data not shown). Also, in vitro studies were performed using endothelial cells derived from jugular veins, and there might be differences in the response of endothelium depending on the localization of the vascular bed (small dermal vessels versus jugular vein).
P-selectin expression has been documented in various skin diseases in humans and in some forms of dermatitis in dogs, including allergic dermatitis, autoimmune dermatitis, pyogranulomatous dermatitis, dermatophytosis, and panniculitis. 2,3,7,9,18,19,26,51,53,59 In humans, evidence of an important role for TNF-α in the induction of adhesion molecules, including P-selectin, in inflammatory skin diseases has recently been presented in patients with psoriasis. 49 In these patients, large amounts of TNF-α were detected in the supernatant of freeze-thawed samples, and there was strong staining for TNF-α on keratinocytes and endothelial cells and for P-selectin on endothelial cells from involved skin. 49 A rapid and transient expression of TNF-α mRNA and protein has also been shown in experimental skin sensitization in mice, suggesting that this cytokine plays a role in the initiation of cutaneous immune responses. 15 Moreover, the importance of P- and E-selectin in the early phases of leukocyte recruitment in contact hypersensitivity has been highlighted by studies using mice deficient in these adhesion molecules. 6,46 In a recent study, researchers also reported the persistence of both E- and P-selectin expression in mice in vivo in a model of immune-mediated inflammatory response of the skin. 21
In the present study, platelet aggregates were observed in the lumina of many blood vessels and in the perivascular interstitial tissues. These platelets could be readily identified by their positive reactivity with the anti–P-selectin antibody MD3. 12 The clumping of these platelets in the vessel lumen and around blood vessels likely results from the administration of TNF-α. In humans and mice, intravenous injection of TNF-α induces a thrombocytopenia that appears to be due to an increase in platelet consumption. 47 Platelet consumption is believed to be the result of platelet activation, with the surface expression of molecules such as P-selectin. 12,32,33 TNF-induced thrombocytopenia is mainly mediated by the TNF receptor I (p55), acting not on platelets directly but on other cells. 47 An important pathway of the TNF-induced thrombocytopenia involves mast cells and the generation of monoamines. 40 The presence of platelets in blood vessels and in the dermis suggests that the platelets could participate in leukocyte recruitment; in vitro studies have shown that P-selectin–expressing canine platelets can mediate the rolling and firm arrest of flowing canine neutrophils. 12
The time course of E-selectin expression observed in the present study (between 3 and 12 hours) is comparable to previously reported in vivo expression of E-selectin in rhesus monkeys and baboons. 36,43 E- and P-selectin both may be necessary for neutrophil accumulation in the skin. 34,54 For example, in models of acute and chronic dermal inflammation, mice deficient in both E- and P-selectin exhibit a significantly reduced neutrophil accumulation, whereas neutrophil accumulation in mice deficient in only E- or only P-selectin is unchanged. 25,34 Also, E-selectin expression has been documented in the lesional skin of patients with atopic dermatitis as compared with nonatopic individuals. 9 This expression was more pronounced after epicutaneous application of aeroallergen (atopy patch test). In psoriasis, as in other chronic dermatoses, persistent E-selectin expression on endothelium has also been reported. 49,51
TNF-α injected intradermally in dog skin induces an acute inflammatory reaction characterized by an increased expression of vascular selectins. E- and P-selectin have distinct patterns of expression on endothelial beds, and their precise regulation permits the initial phase of leukocyte recruitment. New antiadhesive therapies based on the inhibition of the selectin-dependent leukocyte infiltration could contribute to diminish the tissue damage in skin inflammatory processes. Further investigations will be necessary to determine the precise role of vascular selectins in dogs.
Footnotes
Acknowledgements
This study was supported by grants from the Natural Sciences and Engineering Council of Canada (M. Doré) and the Fonds du Centenaire de l'Université de Montré al (M. Doré and M. Paradis). We thank Dr. C. W. Smith (Baylor College of Medicine, Houston, TX) for kindly providing antibody CL37.
