Abstract
Serial sections of brain and palatine tonsil were examined by immunohistochemical staining (IHC) using monoclonal antibody F89/160.1.5 for detecting protease-resistant prion protein (PrPres) in 35 hunterkilled mule deer (Odocoileus hemionus) with chronic wasting disease. Serial sections of brain were stained with hematoxylin and eosin and examined for spongiform encephalopathy (SE). Clinical signs of disease were not observed in any of these deer. On the basis of the location and abundance of IHC and the location and severity of SE, deer were placed into four categories. Category 1 (n = 8) was characterized by IHC in the palatine tonsil with no evidence of IHC or SE in the brain. Category 2 (n = 13) was characterized by IHC in the palatine tonsil and IHC with or without SE in the dorsal motor nucleus of the vagus nerve (DMNV). Category 3 (n = 2) was characterized by IHC in the palatine tonsil, IHC with SE in the myelencephalon, and IHC without SE in the hypothalamus. Category 4 (n = 12) was characterized by IHC in the palatine tonsil and IHC with SE throughout the brain. Category 1 may represent early lymphoid tissue localization of PrPres. The DMNV appears to be the most consistent single neuroanatomic site of detectable PrPres. Categories 2–4 may represent a progression of spread of PrPres and SE throughout the brain. IHC in tonsil and brain and SE in brain were not detected in 208 control deer.
Keywords
Introduction
Chronic wasting disease (CWD), a transmissible spongiform encephalopathy (SE), has been described in captive cervids.20–22 A similar SE has been described in free-ranging mule deer (Odocoileus hemionus), white-tailed deer (Odocoileus virginianus), and Rocky Mountain elk (Cervus elaphus nelsoni) from north-central Colorado.18 The neuroanatomic distribution of histologic lesions and the immunohistochemical staining (IHC) pattern of a protease-resistant prion protein (PrPres) in brain and lymphoid tissue of free-ranging mule deer in terminal stages of disease have been compared with those of captive mule deer with CWD. That report indicated that naturally occurring SE in free-ranging deer is morphologically indistinguishable from CWD in captive deer, at least in the terminal stages.19 Therefore, the free-ranging deer used in this study were considered to have CWD and not another SE.
The primary objective of this investigation was to delineate the extent and distribution of PrPres in palatine tonsil and brain and the severity and distribution of SE in free-ranging mule deer (O. hemionus) with CWD. This study would give insight into several questions: 1) Does PrPres accumulate in the palatine tonsil without detection in the brain? 2) Can PrPres be detected in a single neuroanatomic region or throughout the brain without the detection of SE and vice versa? 3) Are there different patterns of IHC and SE in the brain of free-ranging mule deer that are positive for CWD.
Materials and Methods
For the last 5 years, surveys of harvested deer and elk have been conducted in Colorado to determine the prevalence and geographic distribution of CWD.13 A total of 956 deer heads were collected during hunting seasons in Larimer County, Colorado, from August through December 1997. Brain and palatine tonsil were removed from heads and preserved in 10% neutral buffered formalin. Sections of tonsil and brain, including the obex, hypothalamus, and cerebellum, from each animal were embedded in a single paraffin block. Serial sections were cut at 5 μm and mounted on positively charged glass slides. One slide was stained with hematoxylin and eosin (HE)9 and the other with monoclonal antibody F89/160.1.5 (MAb).14 This monoclonal antibody reacts with a conserved epitope in the PrPres, an abnormal isoform of a native cellular sialoglycoprotein of mule deer, Rocky Mountain elk, domestic sheep, and cattle.14 The MAb is made to a short amino acid segment of the normal prion protein. It is not specific for detection of the altered prion protein isoform. Specificity for PrPres is conferred by the hydrated autoclaving of the sections in buffer before immunostaining, which destroys the protease-sensitive normal prion protein without affecting the resistant protein. This monoclonal antibody is specific for a peptide sequence within the prion protein that is conserved in mule deer, Rocky Mountain elk, domestic sheep, and cattle, and after removal of the protease-sensitive normal prion protein, detects only PrPres.14 PrPres has been associated with CWD of captive and free-ranging deer and elk.6,7,14,16,18,19
Before immunostaining, tissue sections were immersed in 88% formic acid (Mallinckrodt, Paris, KY) for 30 minutes, rinsed in tap water, autoclaved for 20 minutes at 121 C (liquid setting) in Tris buffer, and then cooled for 30 minutes. Sections were immunostained with MAb for 30 minutes at 37 C, and this was followed by addition of a biotinylated anti-mouse secondary antibody, an alkaline phosphatase–streptavidin conjugate, a substrate chromogen (fast red), and a hematoxylin counterstain with bluing (Ventana Medical Systems, Tucson, AZ). The technique used for the IHC was similar to previously described methods.2,10–12,15,16,18,19
Of these 956 animals, 48 deer were considered positive for CWD because they had IHC with or without SE in the dorsal motor nucleus of the vagus nerve (DMNV). None of the hunters who had killed these positive deer suspected that the animals had CWD. Ten additional deer were found to have IHC in the germinal centers of the palatine tonsil without IHC or histologic lesions of SE in the brain. Animals from these two groups were selected for further examination and included the 10 deer with IHC only in the follicles of the palatine tonsil and 25 deer with IHC in palatine tonsil and brain.
To further characterize the neural lesions in these 35 deer, 13–16 sections were made from the remaining brain, embedded in paraffin, and serial sections cut from each block. One slide was stained with HE and the other with MAb as previously described. Typical sections of brain included one to three levels of rhinencephalon with adjacent cortex and basal nuclei, hypothalamus, thalamus (including epithalamus and subthalamus), hippocampus, amygdala, mesencephalon, pons, cerebellum, obex, and spinal cord (C-1). The number of specific sites and axonal tracts evaluated for IHC and SE from each of these deer included 24 sites in the telencephalon, 43 in the diencephalon (16 in the hypothalamus and 27 in the thalamus), 26 in the mesencephalon, 25 in the metencephalon, 15 in the myelencephalon, and 4 in the cervical spinal cord (Fig. 1, category 1). Neuroanatomic locations and nomenclature used in this report were primarily taken from Singer.17 Texts by Yoshikawa24 and Jenkins8 were also consulted.
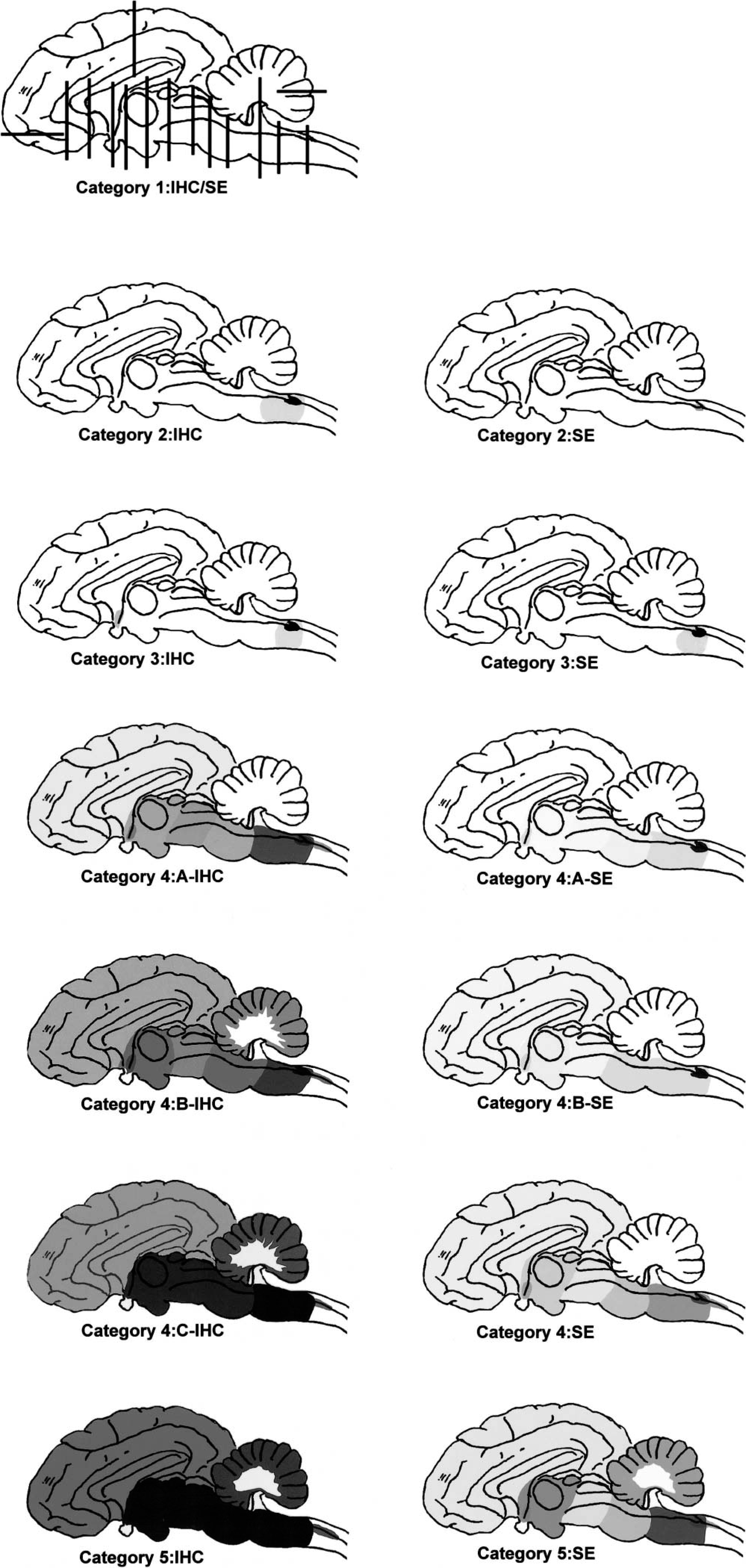
These line drawings of the brain demonstrate the location and abundance of IHC of PrPres and SE in the categories described in this article including the terminal stage (category 5). The percentages of neuroanatomic sites with positive PrPres IHC and SE are represented by gray shading. The different shades of gray are severity indicators, with darker gray indicating more intense change. For a detailed description of these categories, see the text. Category 1: This line drawing shows the location and number of sections cut from the brain of each animal. It also demonstrates a category 1 animal that is free of PrPres or SE in the brain. Category 2: This line drawing demonstrates that PrPres and SE are found only in the DMNV. Category 3: This line drawing demonstrates that PrPres and SE are found in the DMNV and adjacent nuclei and in the hypothalamic n. Category 4-A to 4-C: These three line drawings demonstrate what appears to be a progressive spread and increase in abundance of IHC and SE throughout the brain. Category 5: This line drawing demonstrates the abundance of IHC and SE throughout the brain in a terminal case of CWD.18,19.
Positive IHC has been interpreted to be a scrapie-associated prion protein or an antigenically similar PrPres that has been found in lymphoid tissues and brain of captive and free-ranging deer with CWD.6,7,14,16,18,19
The palatine tonsil was considered positive if any of the follicles exhibited positive IHC. But palatine tonsils that were positive for this IHC technique were characterized by having 50% or more of the follicles filled with positive-staining material. A negative palatine tonsil had no staining in lymphoid follicles. A typical section of tonsil contained approximately 100–200 follicles.
The relative abundance of IHC in brain was subjectively graded on a scale of 0–3 on the basis of examination with light microscopy at 20×: 0, no staining; 1, mild; 2, moderate; and 3, heavy. The intensity of the stain was bright red whether the scoring was 1, 2, or 3.
Criteria used for histologic diagnosis of CWD included intraneuronal vacuolation, microcavitation of gray matter, neuronal degeneration and loss with mild Astrocytosis, or the presence of PrPres detected by IHC in brain (or all).14,18,19,20–22 The severity of SE was graded on a scale of 0–3: 0, no lesions; 1, mild; 2, moderate; and 3, severe. The primary histologic lesions used to judge the severity of SE were neuronal cytoplasmic vacuolation and vacuolation of the neuropil. Astrocytosis was extremely difficult to evaluate using HE and was not used in evaluation of the severity of lesions. For each neuroanatomic area examined, the prevalence and severity of the SE were recorded.
Negative controls for this study included brain and tonsil from 208 deer collected from areas within Colorado where CWD had not been found. In all these deer, sections of obex, hypothalamus, thalamus, cerebellum, and tonsil were stained with HE and MAb. All these deer were examined without knowledge that they had originated from areas outside the known CWD endemic area.
Results
Thirty-five of the 58 hunter-killed deer with positive IHC of the palatine tonsil had sufficient tissue available for a detailed study of the distribution and abundance of PrPres and the distribution and severity of SE in the brain. From the evaluation of serial sections of brain, these animals could be placed into four categories.
Category 1
Eight deer ranging from 1.5 to 2.5 years of age were placed in this group. The hallmark of this group was positive IHC in the follicles of the palatine tonsil, with no detectable PrPres staining or SE in the brain (Fig. 1, category 1). IHC in the palatine tonsil was characterized by abundant, bright-red chromogen deposition filling the germinal centers (Fig. 2). Histologic lesions of SE and IHC were not found in any of the 133 sites examined in the brain. Two of the 10 deer that only had IHC in the tonsil during the initial survey of the 958 animals were found upon further sectioning of the obex to have several neurons surrounded by intense IHC in the lateral region of the middle third of the DMNV; therefore, they were placed in category 2.
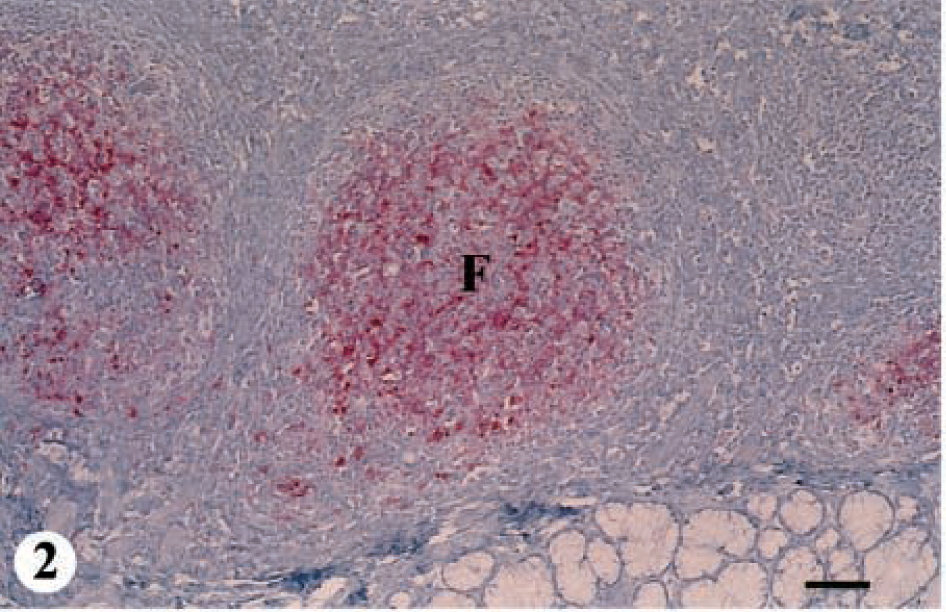
Palatine tonsil; mule deer. Photomicrograph of a lymphoid follicle in a deer with CWD. Note the abundant bright-red chromogen deposition in the lymphoid follicle (F). IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 240 μm.
Category 2
Thirteen deer ranging in age from 1.5 to 5+ years were placed in this group. The hallmark of this category was IHC in germinal centers of the palatine tonsil and positive IHC with or without SE in the DMNV (Fig. 1, category 2: IHC and SE; Table 1). Positive IHC in the DMNV was characterized by coarse granular, bright-red chromogen deposition on the periphery of neurons and in the neuropil (Figs. 3, 4). Positive IHC was also found in the solitarius nuclei (n) and reticular n in one deer. IHC also was found in the radix of the DMNV and the tract of the solitarius n. Of these 13 animals, 8 did not have any histologic lesions of SE in the brain, and the remaining 5 had SE only in the DMNV. Histologic lesions of SE and IHC were not found in any of the other neuroanatomic locations of the brain.
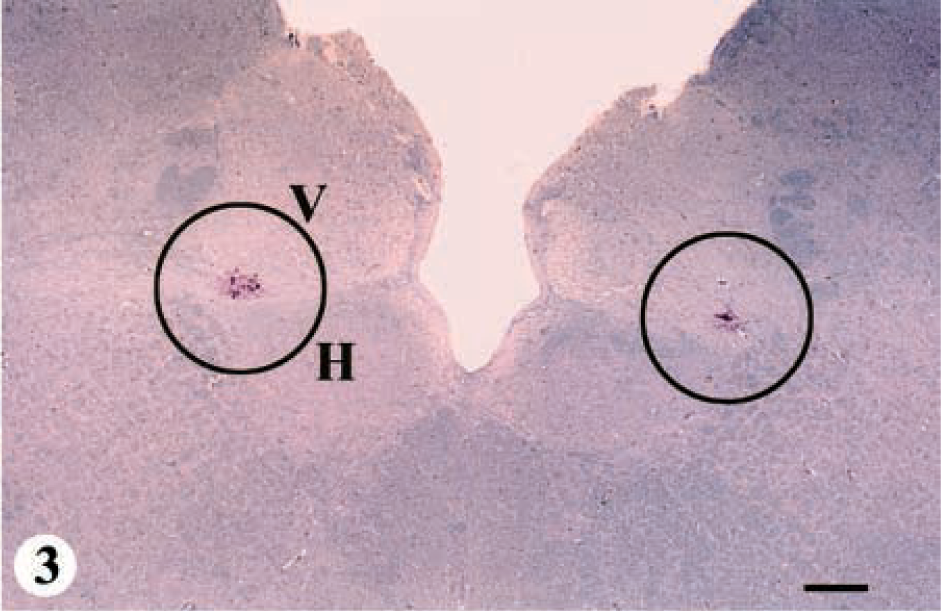
Obex, myelencephalon, brain: mule deer with CWD (category 2). Note the small clusters of bright-red chromogen (circles) in the lateral aspects of the DMNV (V). Staining is not present in the hypoglossal n (H). IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 667 μm.
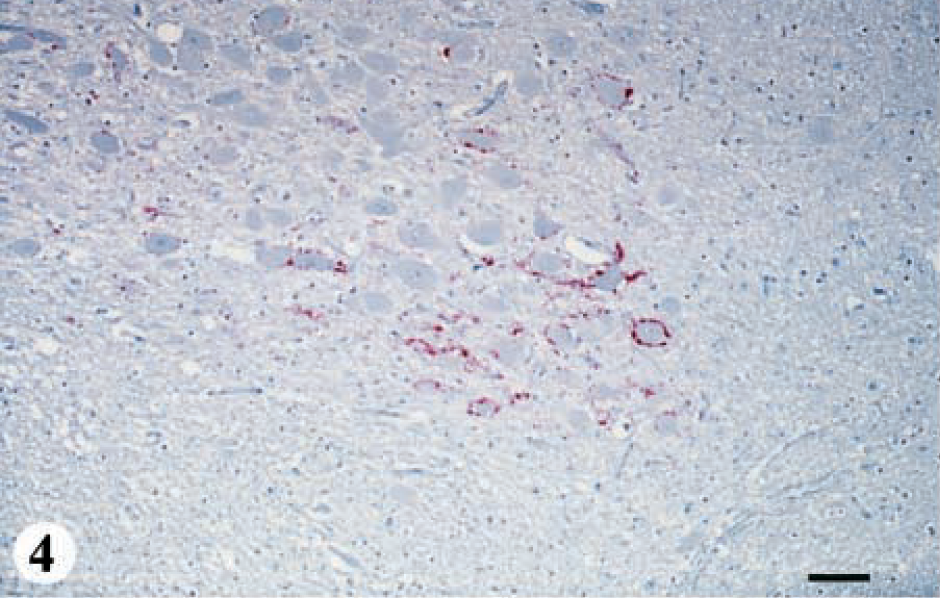
DMNV, obex, myelencephalon, brain: mule deer (not same deer as in Fig. 2) with early CWD (category 2). Note the bright-red chromogen staining around neurons. IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 133 μm.
Summary of the percentages of occurrence of IHC and SE in brain and palatine tonsil of 35 free-ranging mule deer with CWD. These mule deer have been divided into four categories according to the prevalence of positive IHC of PrPres with monoclonal antibody (F89/160.1.5) and the occurrence of SE in nine regions of the brain. Note the progressive increase of prevalence of IHC and SE in category 1 to 4-C.
Percentage of the total number of neuroanatomic sites examined in a specific region of brain (i.e., telencephalon) that had positive IHC for PrPres.
Percentage of the total number of neuroanatomic sites examined in a specific region of brain (i.e., telencephalon) that had histologic lesions of SE.
Percentage of deer in which palatine tonsil showed positive IHC for PrPres in follicles with monoclonal antibody (F89/160.1.5).
Category 3
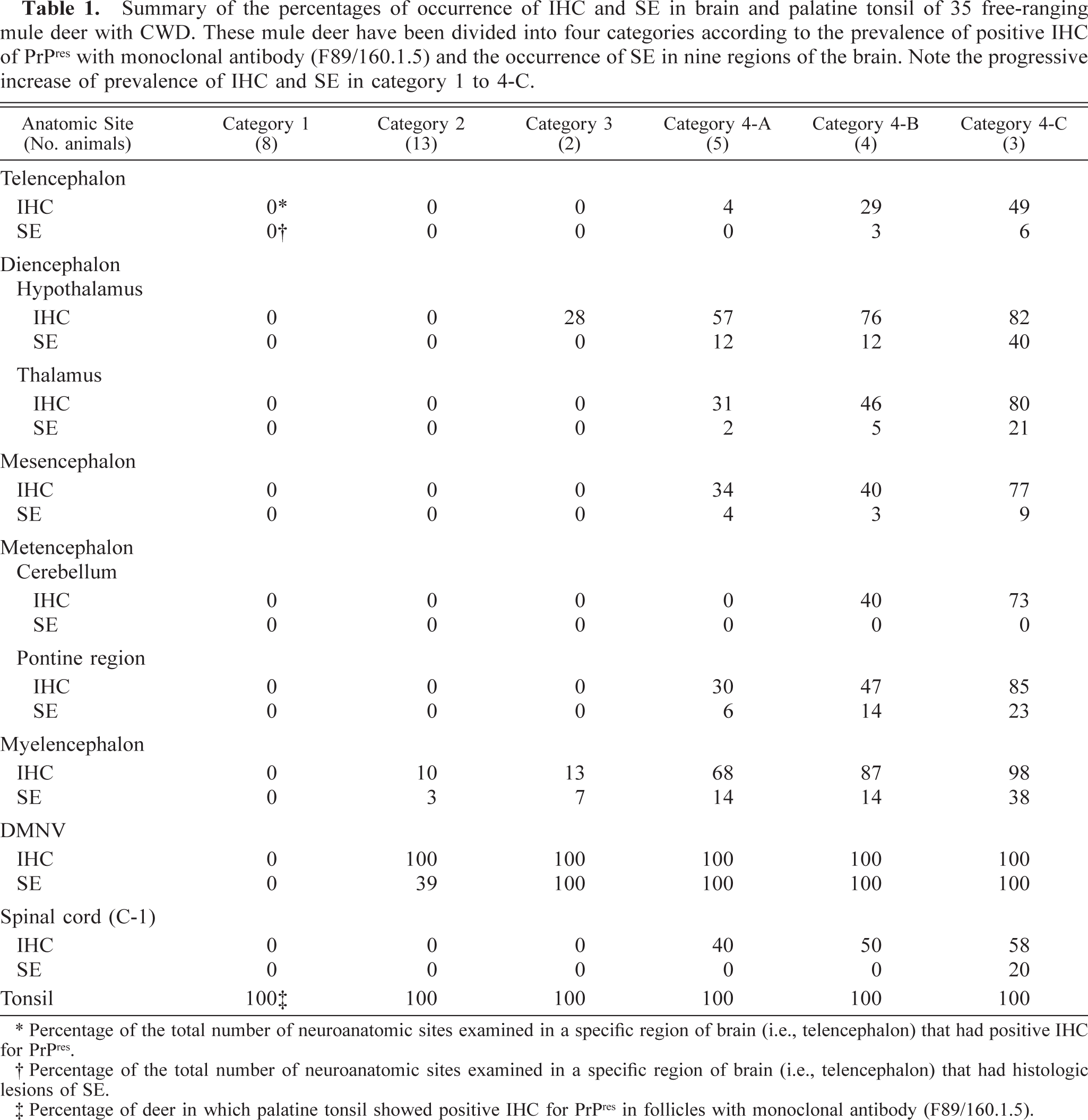
Two deer, 3.5 and 5+ years of age, were placed in category 3. The hallmark of this group was IHC filling the germinal centers of the palatine tonsil, IHC and SE in the DMNV and solitarius n, and in at least 5 hypothalamic n (anterior hypothalamic, paraventricular n of hypothalamus, area dorsalis of hypothalamus, dorsomedial n, and supraoptic n). The IHC totally filled the DMNV. Both deer had SE only in the DMNV (Table 1). SE and IHC were not found in any of the other regions of the brain (Fig. 1, category 3: IHC and SE, Fig. 5).
Obex, myelencephalon, brain: mule deer with CWD (category 3). Note the abundant bright-red chromogen filling the DMNV (V) and the lack of staining in the hypoglossal n (H). IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 667 μm.
Category 4
Category 4 (n = 12) was characterized by IHC in the germinal centers of the palatine tonsil and IHC with SE throughout the brain. These deer could be further divided into three subgroups (4-A, 4-B, and 4-C) on the basis of differences in the prevalence of IHC and SE in the telencephalon, mesencephalon, metencephalon, and spinal cord (Table 1).
Category 4-A
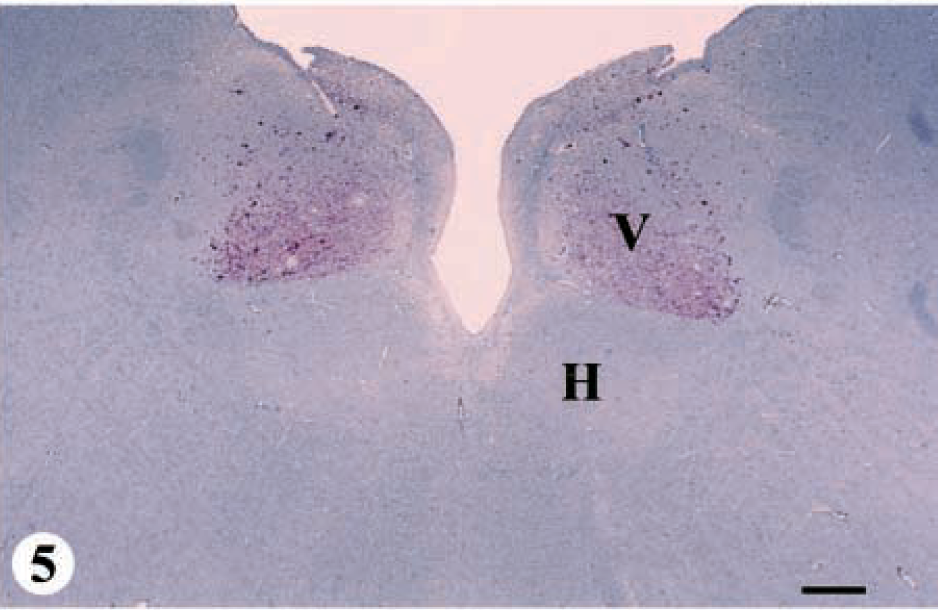
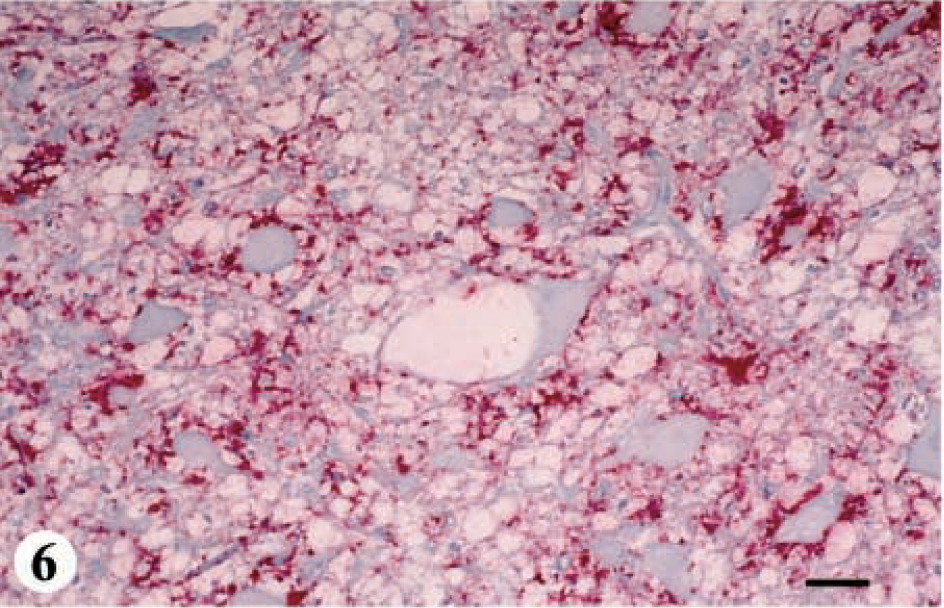
Five deer ranging in age from 2.5 to 8+ years were placed in category 4-A. IHC was mild in the telencephalon, thalamus, mesencephalon, and pontine region of the metencephalon; moderate in the hypothalamus and spinal cord; and severe in the myelencephalon (Fig. 1, category 4-A: IHC and SE, Figs. 6, 7).
DMNV, obex, myelencephalon, brain: mule deer with CWD (category 4-A). Note the abundant bright-red chromogen that appears to be on the cell surface of neurons and in the adjacent neuropil. Note the large intracytoplasmic vacuole in the neuron in the center of the field. IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 33 μm.
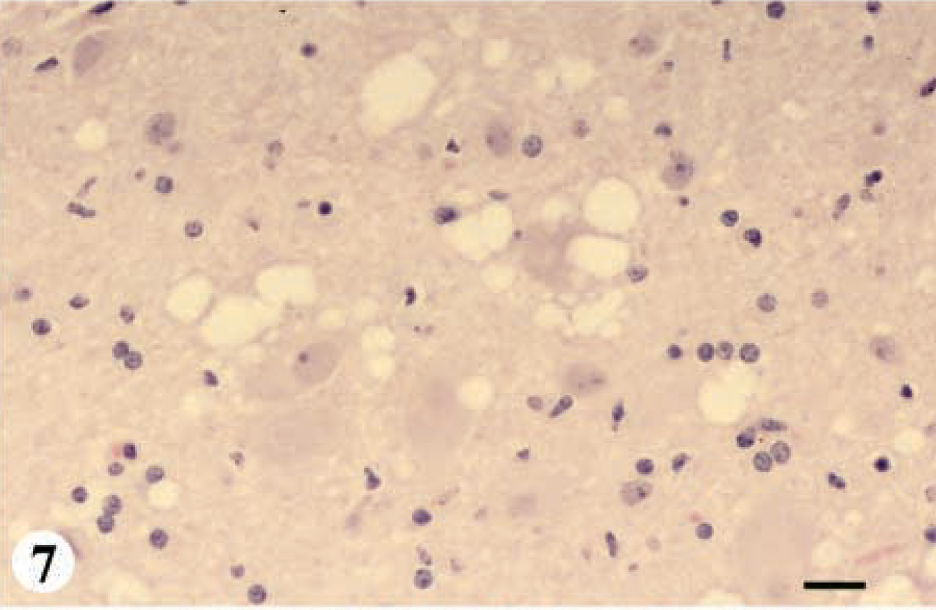
DMNV, obex, myelencephalon, brain: mule deer with CWD (category 4-A). Note the extensive degree of spongiform degeneration of the neuropil and vacuolated neurons. HE. Bar = 33 μm.
In the telencephalon, 24 different sites were examined, and IHC was found in three sites: the anterior commissure, column of the fornix, and the entopeduncular n. SE was not observed in the telencephalon.
Seventeen nuclear sites were examined in the hypothalamic region of the diencephalon, and 15 had IHC. These nuclei included the six reported in category 3 and 9 others (ventromedial n, perifornicalis n, lateral hypothalamic n, posterior hypothalamic n, supra-chiasmaticus n, the area tegmentalis, supra and premammilaris n, mammillary body). All these nuclei had SE except in the ventromedial n and area tegmentalis. Of the three white-matter tracts examined, IHC was only found in the column of the fornix.
Within the thalamus, 26 sites were examined. IHC was found in 15 of the 24 nuclear sites examined. These areas included the medial central n of thalamus, habenular n, ventral medial n, ventral lateral n, medial dorsal n, paraventricular n, central medial n, ventral posterior medial n, ventral posterior lateral n, lateral posterior n, nucleus posterior, lateral geniculate n, and medial geniculate n. SE was found in only one area, the central medial n. Three of the four axonal tracts had IHC: the stria medullaris thalami, decussation of the ventral tegmental tract, and the crus cerebri.
Of the 26 sites examined in the mesencephalon, 11 of 17 nuclear sites and 6 of 9 axonal tracts had IHC. The nuclear sites included the superior colliculus, central gray substance, interpeduncular n, pretectalis n, red n, substantia nigra, profundus, oculomotor n, dorsal n of raphae, trochlear n, and reticular formation. SE was found in the central gray substance, oculomotor n, dorsal n of the raphae, and the reticular formation. Axonal tracts found with IHC included the medial longitudinal fasciculus, dorsal tegmental decussation, spinothalamic tracts, corticospinal and bulbar tracts, decussation of the ventral tegmental tract, and the dorsal longitudinal fasciculus.
Twenty sites were examined in the pontine region of the metencephalon, of which eight nuclear areas (tegmental n, dorsal n of raphae of the tegmental n, reticular formation, trigeminal n, n centralis, peduncularis, vestibular n, and trochlearis n) had IHC. SE was found in only one nuclear area, the dorsal n of the raphae of the tegmental n. The cerebellum was free of IHC and SE.
Thirteen sites were examined in the myelencephalon, 10 nuclear areas and 3 axonal tracts. IHC was found in the all 10 sites: the solitarius n, DMNV, hypoglossal n, raphae, n of the spinal tract of the trigeminal nerve, n ambiguus, reticular n, olivary n, cuneatus n, and gracilis n. SE was found in 2 of the 10 nuclear areas: the DMNV and solitarius n. IHC was found in the three axonal tracts examined: the tracts of the solitarius n, radix of DMNV, and the pyramis.
IHC was found in the dorsal and intermediate horns of the spinal cord at the level of C-1 but not in the ventral horn. SE was not observed in these sections of the upper spinal cord.
Category 4-B
Four deer ranging in age from 2.5 to 6+ years were placed in category 4-B. This group was characterized by mild IHC in the telencephalon; moderate in the thalamus, mesencephalon, cerebellum and pons region of the metencephalon, and spinal cord; and severe in the hypothalamus and myelencephalon (Fig. 1, category 4-B: IHC and SE. SE was not present in the cerebellum or spinal cord but was present and more advanced in all other regions of brain as compared with category 4-A.
In the telencephalon, eight nuclear areas had IHC: the olfactory tubercle, olfactory cortex, septal n, corpus striatum, caudate n, putamen, globus pallidus, and gray matter of the neocortex (frontal and parietal lobes). SE was found only in the olfactory tubercle and olfactory cortex. In addition to the white-matter tracts with IHC in category 4-A, IHC also was found in subcortical white matter and the internal capsule.
Within the pontine region of the metencephalon, in addition to the nuclear areas with IHC in category 4-A, five additional nuclei were found to be positive: the pontine n, superior olivary n, facial n, lemnisci n, and the n medial profundus. SE was found in five additional nuclear areas: the tegmental n, reticular formation, n centralis superior, superior olivary n, and facial n.
In category 4-B, IHC was found in the molecular layer, Purkinje cell layer, granular cell layer, and the white lamina but not in the roof nuclei. SE was not found in the cerebellum.
The distribution and abundance of IHC and SE in the diencephalon (hypothalamus, subthalamus, epithalamus, and thalamus), mesencephalon, myelencephalon, and spinal cord for category 4-B were essentially the same as those for category 4-A.
Category 4-C
Three deer ranging in age from 2.5 to 3.5 years were placed in category 4-C. IHC was moderate in the telencephalon and spinal cord and severe in the hypothalamus, thalamus, mesencephalon, cerebellum and pons region of the metencephalon, and the myelencephalon (Fig. 1, category 4-C: IHC and SE, Figs. 8, 9). SE was present in all regions of brain except in the cerebellum.
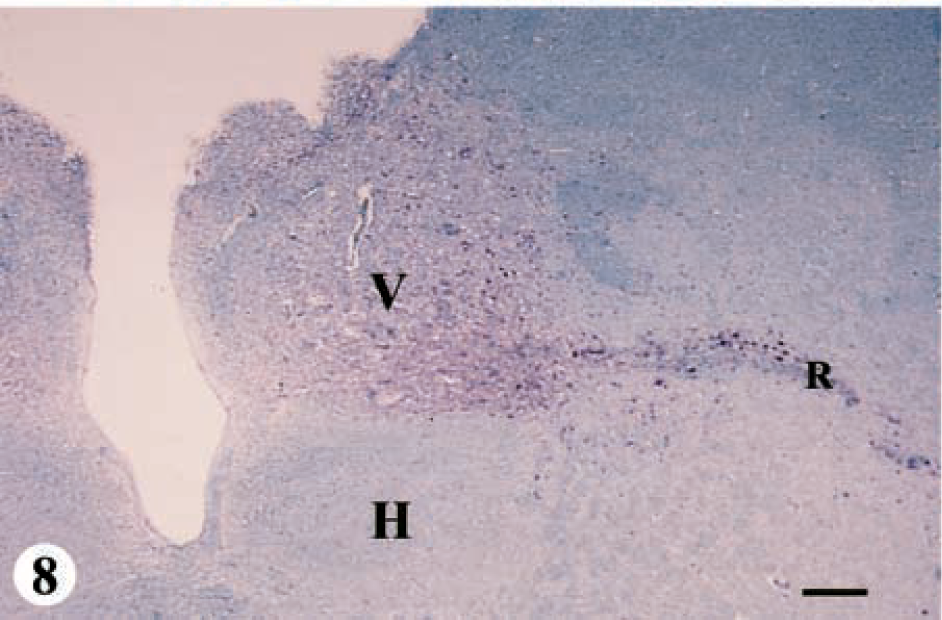
Obex, myelencephalon, brain: mule deer with CWD (category 4-C). Note the abundant bright-red chromogen filling the entire DMNV (V) and the radix of the vagal nucleus (R). A minimal amount of immunopositive material is found on the periphery of the hypoglossal nucleus (H). IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 333 μm.
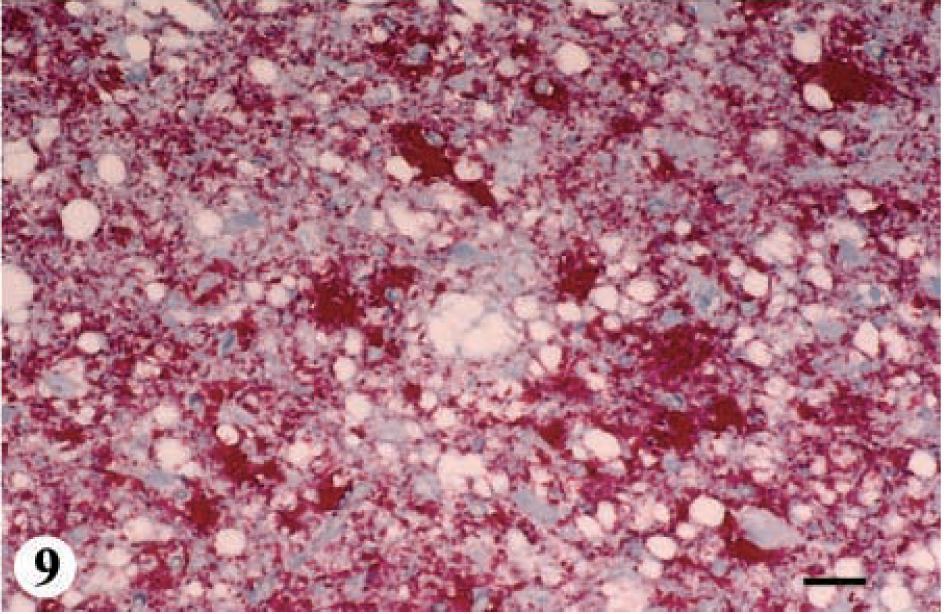
DMNV, obex, myelencephalon, brain: mule deer with CWD (category 4-C). Note the abundance of bright-red chromogen that appears to be on the cell surface of neurons and in the adjacent neuropil. IHC: Streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 67 μm.
In the telencephalon, five additional nuclear areas had IHC: the claustrum, n of the anterior commissure, amygdala, hippocampus, and cortex of the pyriform lobe. SE was found only in the pyriform lobe. In addition to white-matter tracts with IHC as in category 4-B, IHC was found in the anterior commissure, olfactory stria, optic chiasm, and fimbra of the hippocampus.
IHC was found in six additional nuclei in the thalamus as compared with category 4-B: the anterior dorsal n, anterior medial n, anterior ventral n, reticular n, zona incerta of the subthalamus, and lateral anterior n. SE was found in an additional 10 nuclei and included the anterior medial n, anterior ventral n, endopenduncular n, pulvinar, ventral medial n, ventral lateral n, paraventricular n, ventral posterior lateral n, zona incerta of subthalamus, and lateral anterior n. IHC was found in the corpus callosum in one deer.
Within the mesencephalon, two additional areas with IHC were found: the subcommissural organ and the pineal body. SE was found in three additional nuclear areas: the red n, substantia nigra, and trigeminal n of mesencephalon. IHC was found in the optic tracts in the mesencephalon and in the medial lemniscus and spinothalamic tracts.
Within the pontine region of the metencephalon, in addition to the nuclear areas with IHC in category 4-B, five additional nuclei were found to be positive: the pontine n, superior olivary n, facial n, lemnisci n, and the n medial profundus. SE was found in five additional nuclear areas: the tegmental n, reticular formation, n centralis superior, superior olivary n, and facial n.
An additional finding was IHC within the roof nuclei of the cerebellum, but SE was not observed in these nuclei.
The IHC found in the gray matter of the spinal cord in category 4-C was similar to that of category 4-B. SE was present in the dorsal, intermediate, and ventral horns.
The distribution and abundance of IHC and SE in the hypothalamus and myelencephalon in category 4-C were similar to those in category 4-B. IHC was not found in the choroid plexus in any of the deer.
Control deer
Histologic lesions of SE or IHC were not detected in brain or palatine tonsil from the 208 negative control deer collected outside the endemic region for CWD in Colorado. Of the 956 animals examined from northeastern Colorado in which CWD was endemic, 908 did not have any histologic lesions or IHC suggestive of CWD. These animals were not considered to be negative controls because they originated within the endemic area for CWD, but they did aid in ruling out problems of nonspecific IHC staining and neurologic lesions.
Discussion
Even though this investigation did not examine deer sequentially killed after inoculation with known CWD-infected material, the identification of PrPres in the palatine tonsil and the four different patterns of distribution of PrPres and SE in the brain may give several important insights into the pathogenesis of this disease.
Results from this investigation suggest that deer may have positive IHC within germinal centers of palatine tonsil in the absence of IHC or SE in the brain. This finding suggests that PrPres may be deposited in the palatine tonsil before being deposited in the brain. This is consistent with what has been found when mule-deer fawns were fed infected brain from captive deer that had CWD. In that experiment, alimentary tract–associated lymphoid tissues (retropharyngeal lymph node, tonsil, Peyer's patches, and ileocecal lymph node) were found to be positive within 42 days after oral infection.16 Hadlow reported infectivity of lymphoid tissues (tonsil, retropharyngeal and mesenteric lymph nodes, ileum, and spleen) by mouse bioassay from a 10-month-old naturally infected domestic lamb born to a ewe that later developed scrapie.5 If lymphoid infection precedes infection of the brain, then early lymphoid infection may be an integral part of the pathogenesis of CWD in deer. This is exemplified by the fact that further sectioning of the obex in two deer that were initially placed in category 1 revealed neurons surrounded by positive IHC in the lateral aspects of the middle third of the DMNV. This observation supports the suggestion that PrPres accumulates in lymphoid tissues (at least in the palatine tonsil) before it accumulates to detectable levels in the brain. The finding of positive lymphoid tissues such as the palatine tonsil may serve as an early test in nonclinical animals.
Deer with positive IHC of palatine tonsil (category 1) may be extremely early cases of CWD; alternatively, animals with lymphoid tissue involvement alone could be resistant to neural infection or even perhaps be asymptomatic shedders. But the ages of the deer with positive IHC in tonsil and not in brain were 1.5–2.5 years. If many of the deer were resistant to neural infection or were asymptomatic shedders, one would have expected a number of these animals to be older (at least 4–5 years of age).
The significance of finding three different patterns of distribution of PrPres and SE in the brain of these deer is not known, but several inferences can be made. First, the DMNV can have IHC and SE with the remaining brain being free of IHC or SE as in category 2. Also, IHC and SE can be found in the DMNV and hypothalamus with the remaining brain being free of lesions as in category 3. In several instances, IHC was found without SE, but SE was not found without IHC.
These different patterns of IHC and SE suggest at least three different explanations. The first is that the patterns demonstrate a progression of deposition of PrPres in lymphoid tissue and brain, followed by the development of SE (categories 1–4). These different patterns may demonstrate evidence of spread of PrPres and SE throughout the brain. PrPres could possibly have been deposited in the environment (ground or food) via feces and saliva from an infected animal. After ingestion and uptake, the PrPres may then have a multiplication phase in lymphoid tissue, especially in those associated with the head and the gut. At least one route of entry into the brain may be via the vagus and glossopharyngeal nerves that lead to the vagus and solitarius n of the myelencephalon. PrPres first accumulates in the lateral aspects of the middle third of the DMNV, then spreads throughout this nucleus, and then to the adjacent nuclei. This would be similar to what has been described in hamsters that were fed the scrapie agent. These authors suggest that the scrapie agent enters the brain via the vagus nerve and infects the DMNV first, then spreads to the commissural solitary tract nucleus.1 PrPres then may spread via axonal tracts to the hypothalamus, then to the thalamus, mesencephalon, pons, and finally to the telencephalon, spinal cord, and cerebellum.
These patterns of detectable PrPres followed by histologic lesions of SE seem to be progressive and spread throughout the brain with increasing severity. Positive IHC accumulates in white tracts early in the disease, perhaps indicating that PrPres travels within axons in the brain. Positive IHC is prominent in the subependyma, especially in the third and fourth ventriculi, and accumulates in Virchow-Robins spaces around neural vessels, suggesting that there may be a clearance mechanism of PrPres as described in hamsters.3
The finding of 13 deer in category 2, 2 deer in category 3, and 12 deer in category 4 may suggest that there is an initial multiplication or adaptation phase for PrPres in the DMNV. PrPres may spread to the hypothalamus and then rapidly spread throughout the brain via axonal tracts. This may explain why only two animals were found in category 3.
There is actually a category 5 that is similar to category 4-C. The lesions and pattern of IHC of category 5 have been described in free-ranging and captive deer that were exhibiting clinical signs or were found moribund or dead with CWD.19 But deer with clinical signs or in the terminal stage of disease were not observed by any of the hunters who contributed positive mule deer examined in this study; therefore, deer in category 5 were not investigated in this study. The only additional lesions described in category 5 were the presence of SE in the cerebellum and the abundance of IHC and increased severity of SE (Fig. 1, category 5: IHC and SE).19
The ages of the positive deer suggest that the animals may become infected at any age, but if they are infected early in life, the disease may progress more rapidly than in an animal infected later in life. This would also be associated with the initial or subsequent dose (or both) received by the animal.
A second explanation for these different patterns of IHC and SE would be that each pattern could be caused by a different strain of PrPres. Different patterns of SE have been documented in various breeds of sheep with scrapie, but the differences in these breeds of sheep are much more subtle than the differences in the categories described in this study.4,23 The likelihood of multiple strains of PrPres in this group of deer from a relatively small area in northeastern Colorado is unlikely.
A third possible explanation for these different patterns may be different polymorphisms of the prion protein gene and, therefore, slightly different isoforms of the normal cellular prion protein in brain. The genomic makeup of the normal cellular prion protein of these deer was not investigated.
These three possible explanations for the different patterns of IHC and SE in brain are purely speculative in nature. The cause or combination of causes is not known. But the most logical explanation is that these patterns do demonstrate progressive spread of PrPres throughout the brain. The proof of this explanation awaits experimental infections in deer with sequential examination of the brain.
Footnotes
Acknowledgements
We thank the hunters for submitting heads from harvested deer; Kate Larsen, Liz Wheeler, Suzanne Tracy, and Sam Berry for collecting deer heads from numerous locations throughout Colorado; Sam Hendrix, Danielle Hettler, Wendy Walden, Peggy Bartling, Matthew Brooks, Matthew Organ, Steve Herman, and Troy Spraker for collection of brain and lymphoid tissue from deer heads; Lee Bie, Danielle Hettler, and Matthew Organ for trimming brain sections; Maryanne Tryjan for embedding them; and Yang Sun Shin and Frank Aquino for cutting the tissues. We thank Charlie Kerlee and Jenger Smith for aid with the photography and line drawings. This project was partially funded by the Colorado Division of Wildlife, Fort Collins, Colorado, and the Colorado State University Diagnostic Laboratory, College of Veterinary Medicine, Fort Collins, Colorado.
