Abstract
A 10-year-old male domestic shorthair cat died during anesthesia. Grossly, the lungs had multiple nodules corresponding to inflamed airways (bronchitis and bronchiolitis). Microscopically, cuffs and nodular aggregates of lymphocytes and plasma cells surrounded airways. Peribronchiolar fibrosis was also common. Globule leukocytes infiltrated the respiratory epithelium of noninflamed or mildly inflamed bronchi. Argyrophilic and filamentous organisms, consistent with cilia-associated respiratory bacillus-like organisms (CLO), were intermixed with cilia of respiratory epithelium. Ultrastructurally, CLO were longer and thinner than cilia and had a distinct trilaminar membrane, central electron-lucent areas, and no specialized external structures. Silver stained lung sections from 18 additional feline airways revealed similar bacilli in 2/9 normal lungs, 1/7 lungs with bronchitis and bronchiolitis, and 1/2 lungs with pneumonia. The significance of CLO in the pulmonary lesions was not determined.
A 10-year-old sexually intact male domestic shorthair cat was presented to the referring veterinarian for a routine dental examination. It developed cardiovascular collapse and died during anesthesia. The necropsy by the referring veterinarian revealed enlargement of both cardiac ventricles and white to tan firm nodules around intrapulmonary airways (Fig. 1). No other gross abnormalities were observed. The entire heart and samples of lung, liver, and kidney were fixed in 10% neutral buffered formalin and submitted to the Veterinary Medical Diagnostic Laboratory at the University of Missouri for microscopic evaluation.
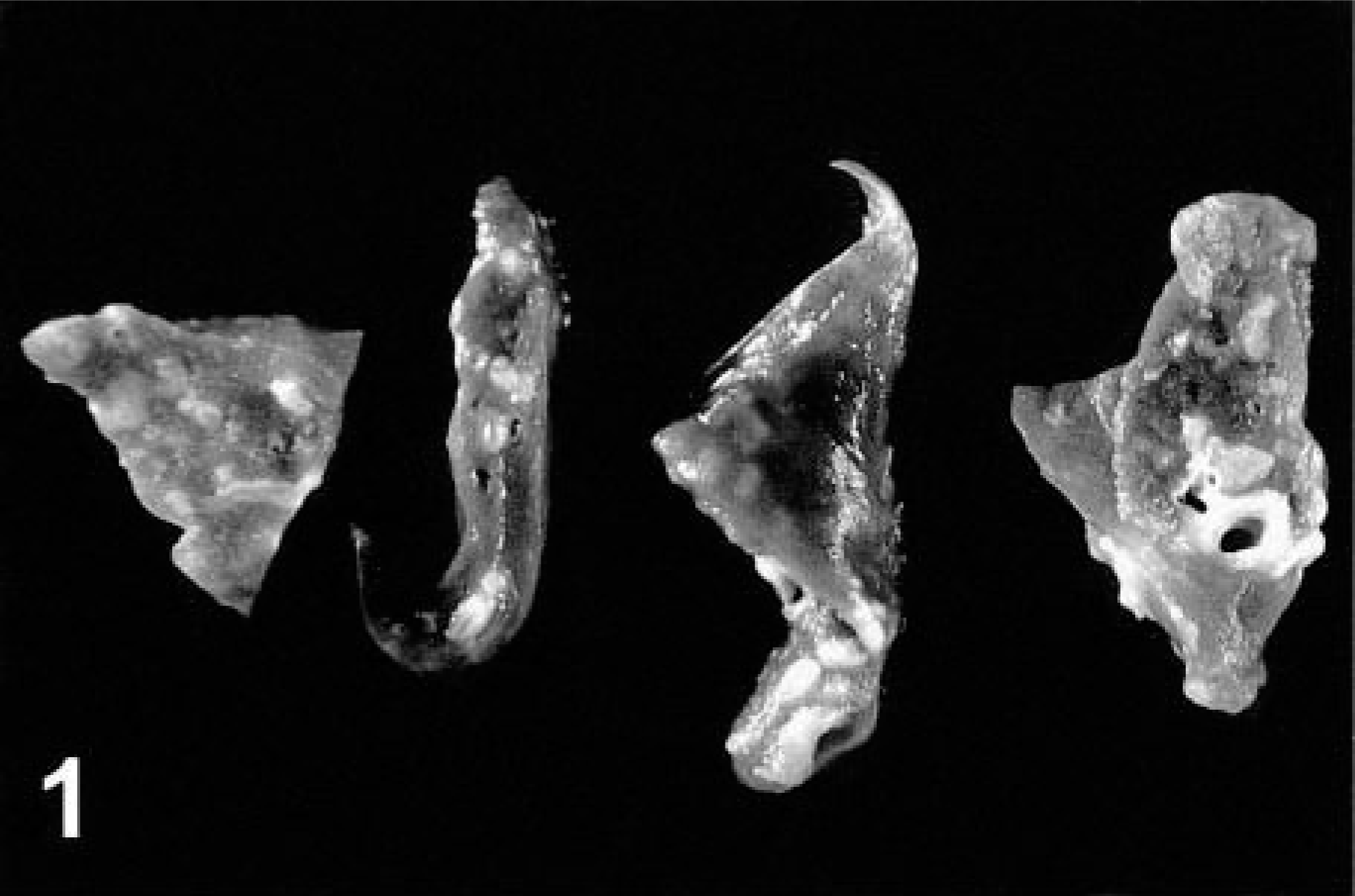
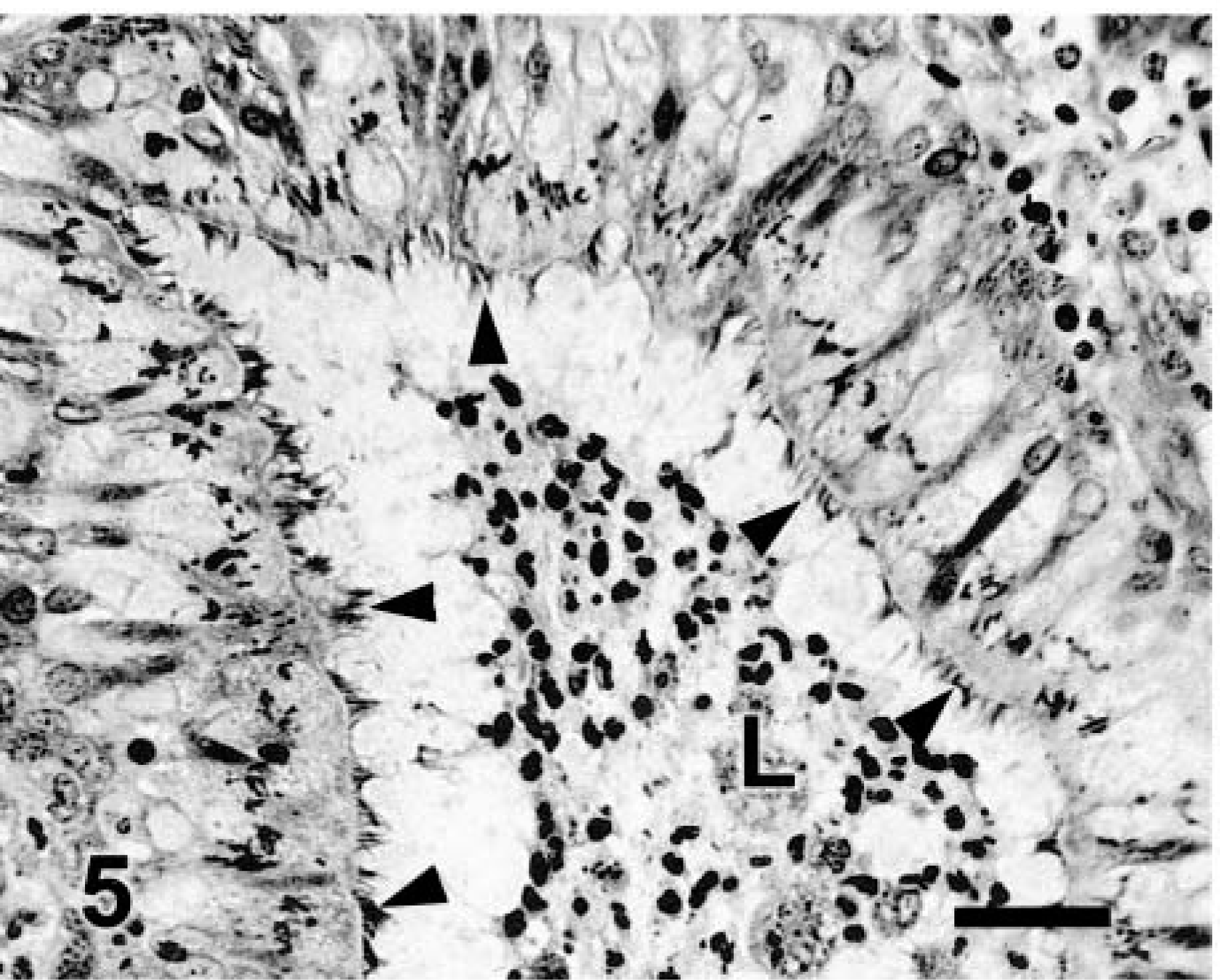
Lung; cat. Bronchitis and bronchiolitis. The surface of the lung has multiple nodules corresponding to inflamed and fibrotic airways.
Microscopic changes in the lung were associated with airways. There was prominent thickening of many bronchi and large to medium-size bronchioles, with infiltration of lamina propria and submucosa by lymphocytes, plasma cells, fewer neutrophils, and rare eosinophils. Distinct lymphoid aggregates and cuffs around bronchioles were also present (Figs. 2, 3). Bronchiolectasis was common and associated with mural fibrosis. The epithelium of some airways (mainly bronchioles) had focal squamous metaplasia or hyperplasia. Cellular debris and mixed leukocytes filled the lumina of many airways. These changes were consistent in multiple sections of lung regardless of lobar location. Smooth muscle hyperplasia and hypertrophy, eosinophils in airways, and bronchial gland hyperplasia were not prominent. Many bronchi had 20–30-μm-diameter intraepithelial mononuclear cells with numerous cytoplasmic eosinophilic granules (Fig. 4). Morphologically, these cells were consistent with globule leukocytes. Numbers of globule leukocytes were greater in noninflamed or slightly inflamed airways than in severely inflamed airways. Globule leukocytes were not apparent in terminal bronchioles or in the pulmonary parenchyma. Globule leukocytes were negative for CD3 and lysozyme by immunohistochemistry.19 Monoclonal antibody to feline CD1815 produced an equivocal reaction in globule leukocytes but labeled numerous adjacent leukocytes. Warthin–Starry stain also revealed variable numbers of cilia-associated respiratory (CAR) bacillus-like organisms (CLO) located parallel to cilia in bronchi and main bronchioles (Fig. 5). Six of seven (86%) bronchi, 17/32 (53%) bronchioles, and 0/10 terminal bronchioles had bacilli consistent with CLO. Frequently, these organisms formed dense clumps (Fig. 6). Organisms were present in inflamed and noninflamed areas. Lesions of hypertrophic cardiomyopathy were evident in both ventricles. The liver and kidney were within normal limits.
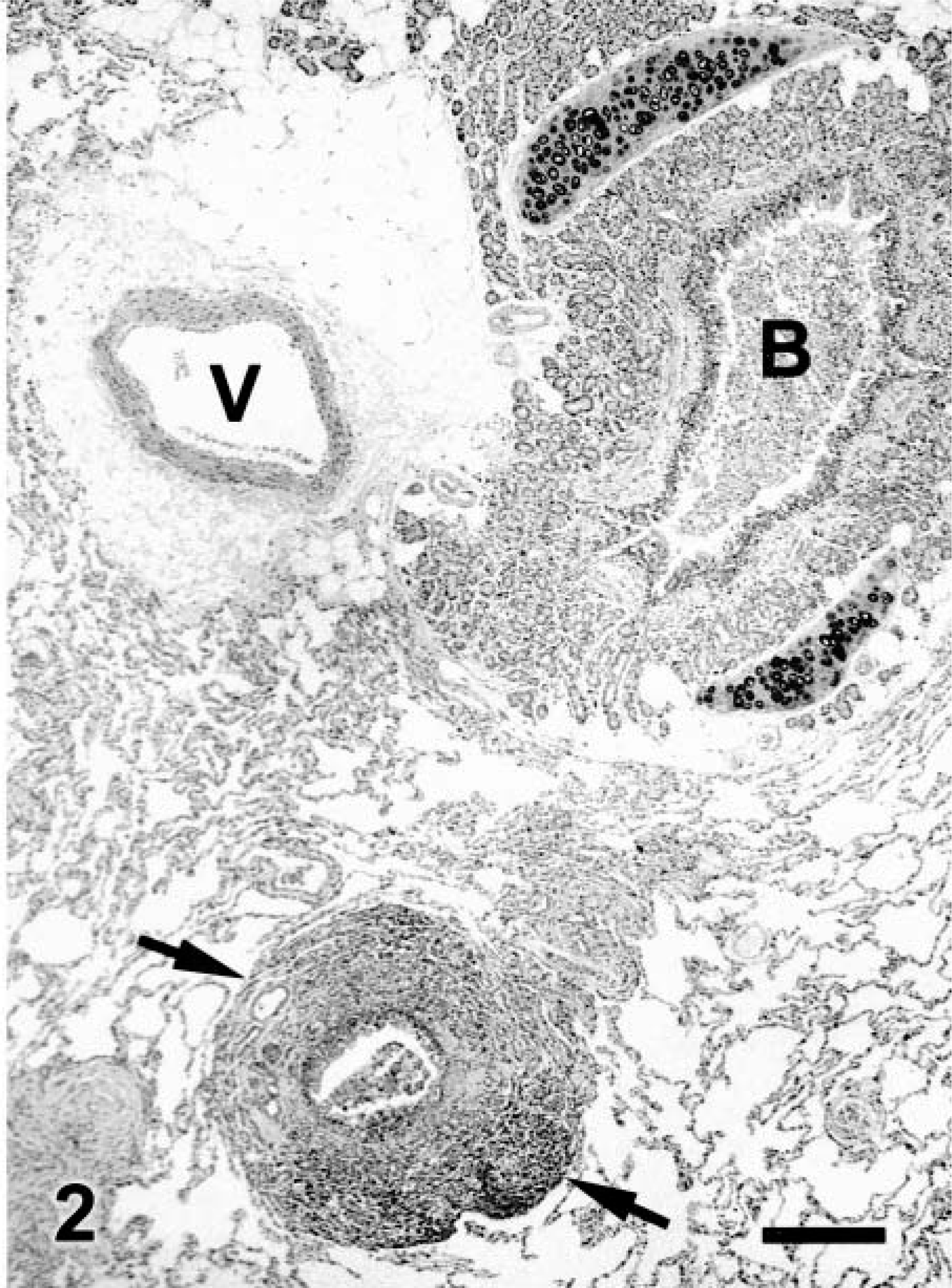
Lung; cat. A bronchus (B) has luminal inflammatory cells and cellular debris. A bronchiole (arrows) has prominent lymphoid cuffing. V = vessel. HE. Bar = 285 μm.
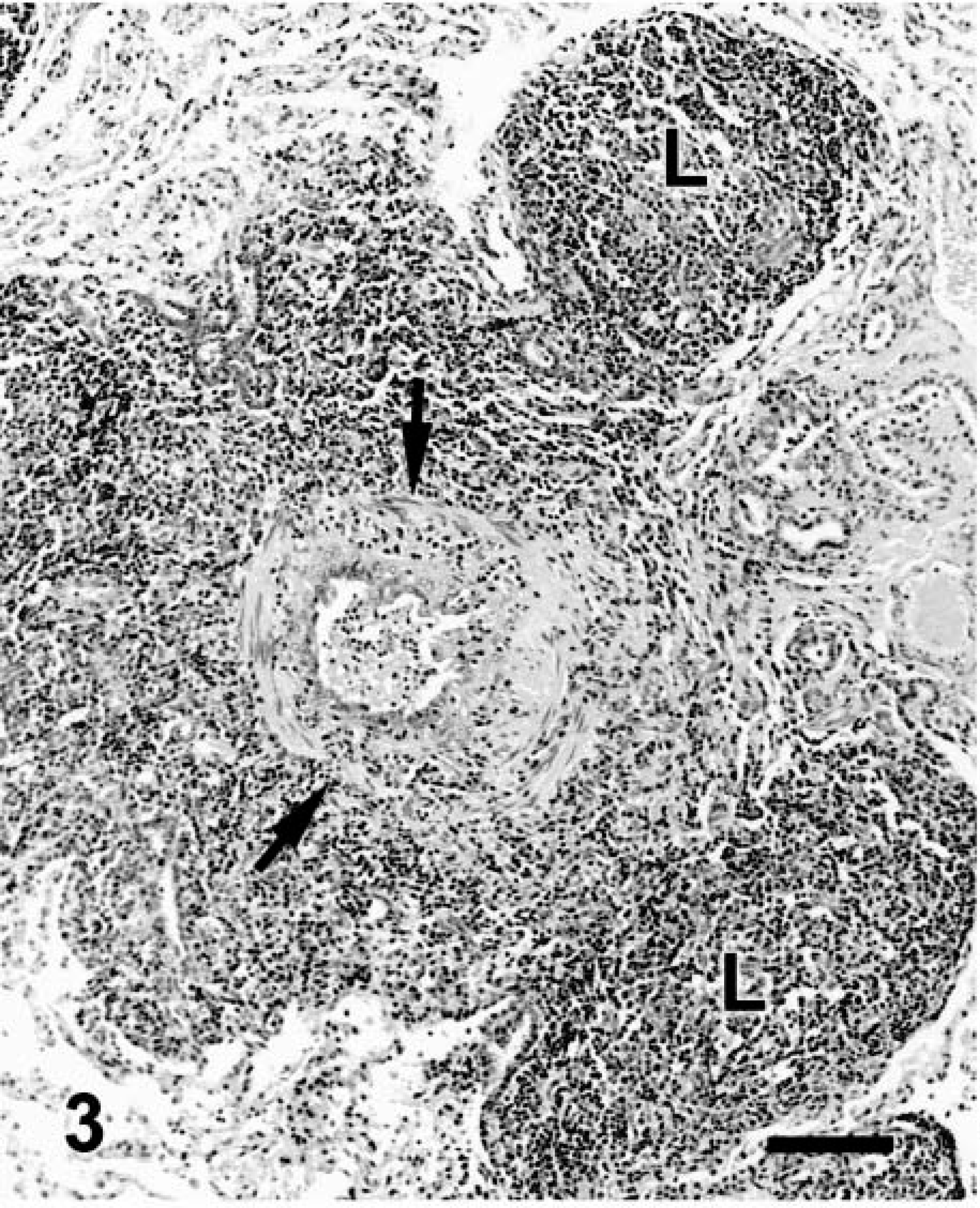
Lung; cat. Bronchiolitis. The wall of the bronchiole is delimited by arrows. Note mural fibrosis, prominent lymphoid cuff, and hyperplasia of BALT (L). HE. Bar = 114 μm.
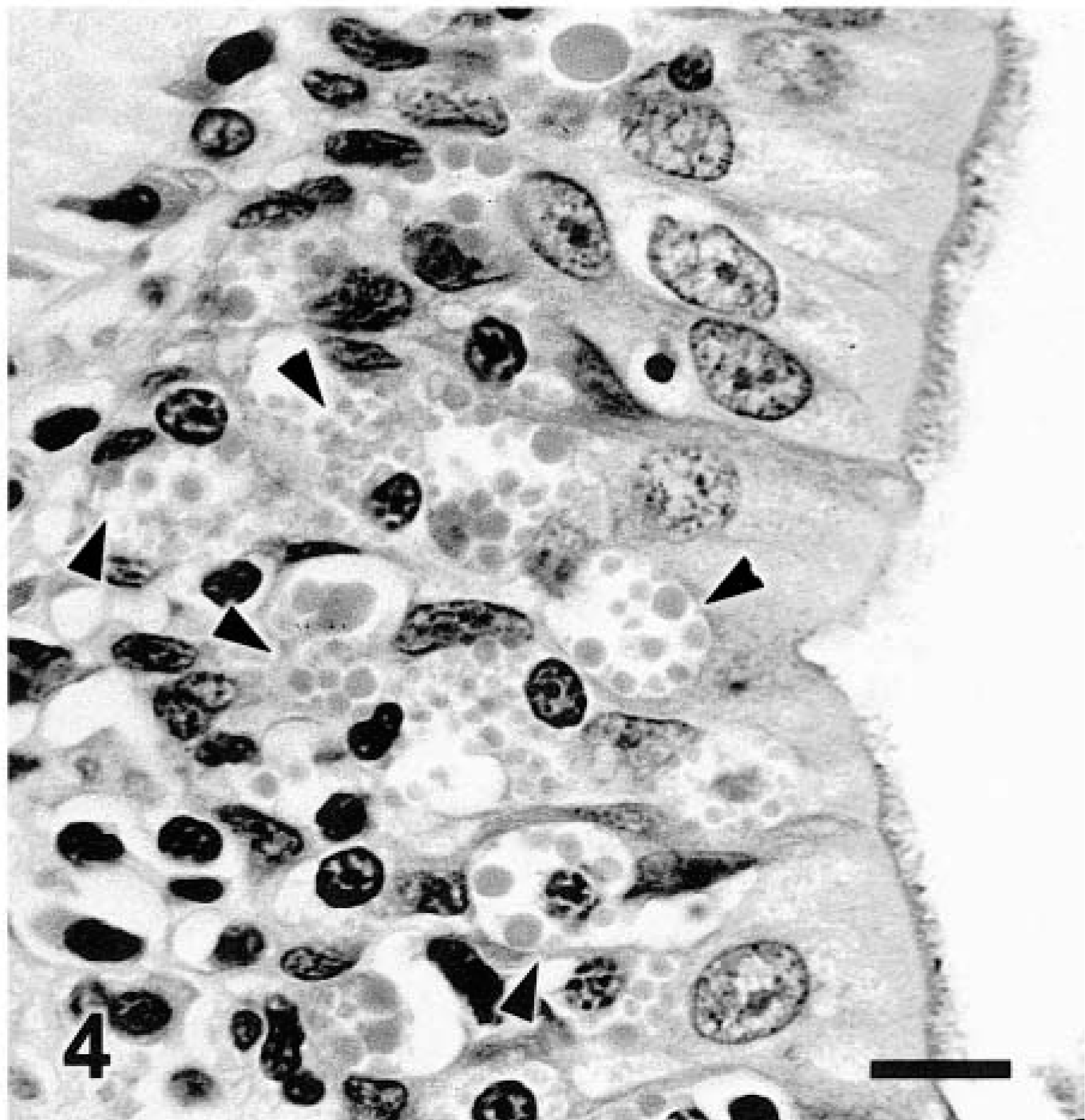
Bronchus; cat. Note numerous intraepithelial globule leukocytes (arrowheads). HE. Bar = 11 μm.
Lung; cat. Bronchitis. Numerous argyrophilic bacilli (arrowheads) are interspersed with cilia of the respiratory epithelium. L = lumen with inflammatory exudate. Warthin–Starry. Bar = 29 μm.
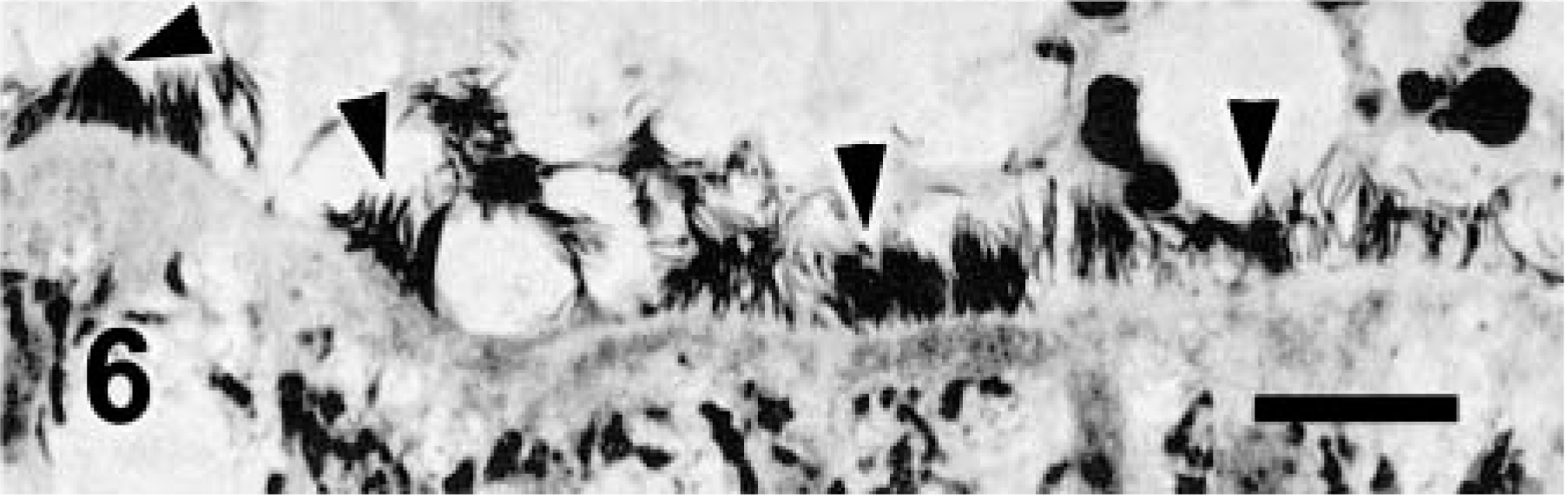
Lung; cat. Detail of respiratory epithelium with clusters of bacilli (arrowheads). Warthin–Starry. Bar = 11 μm.
Selected airways were removed from the paraffin block and processed for electron microscopy. Samples were postfixed in 2% glutaraldehyde containing ruthenium red and then in osmium tetroxide, dehydrated in a graded ethanol series, and embedded in epoxy resin. Thin sections were stained with uranyl acetate and lead citrate and examined with an electron microscope at 100 kV. The number of bacilli was always lower than the number of cilia. Bacilli were interspersed and parallel to cilia. Direct contact with the cytoplasmic membrane of ciliated epithelial cells was not apparent. Bacilli had a length of at least 4 μm and a maximum diameter of 120–175 nm (Fig. 7) and were consistently thinner than adjacent cilia, which had a diameter of 165–200 nm. These organisms had a distinct trilaminar membrane with central electron-lucent areas (Fig. 7). No specialized external structures were identified. Polymerase chain reaction procedures used with paraffin-embedded sections of the lung yielded negative results with primers specific for Lawsonia spp., Mycoplasma spp., rat CAR bacillus, and rabbit CAR bacillus.5,8 However, a generic primer for bacteria also did not amplify any products in this cat's lung sample, indicating the possibility that prolonged fixation affected the results of this test.9
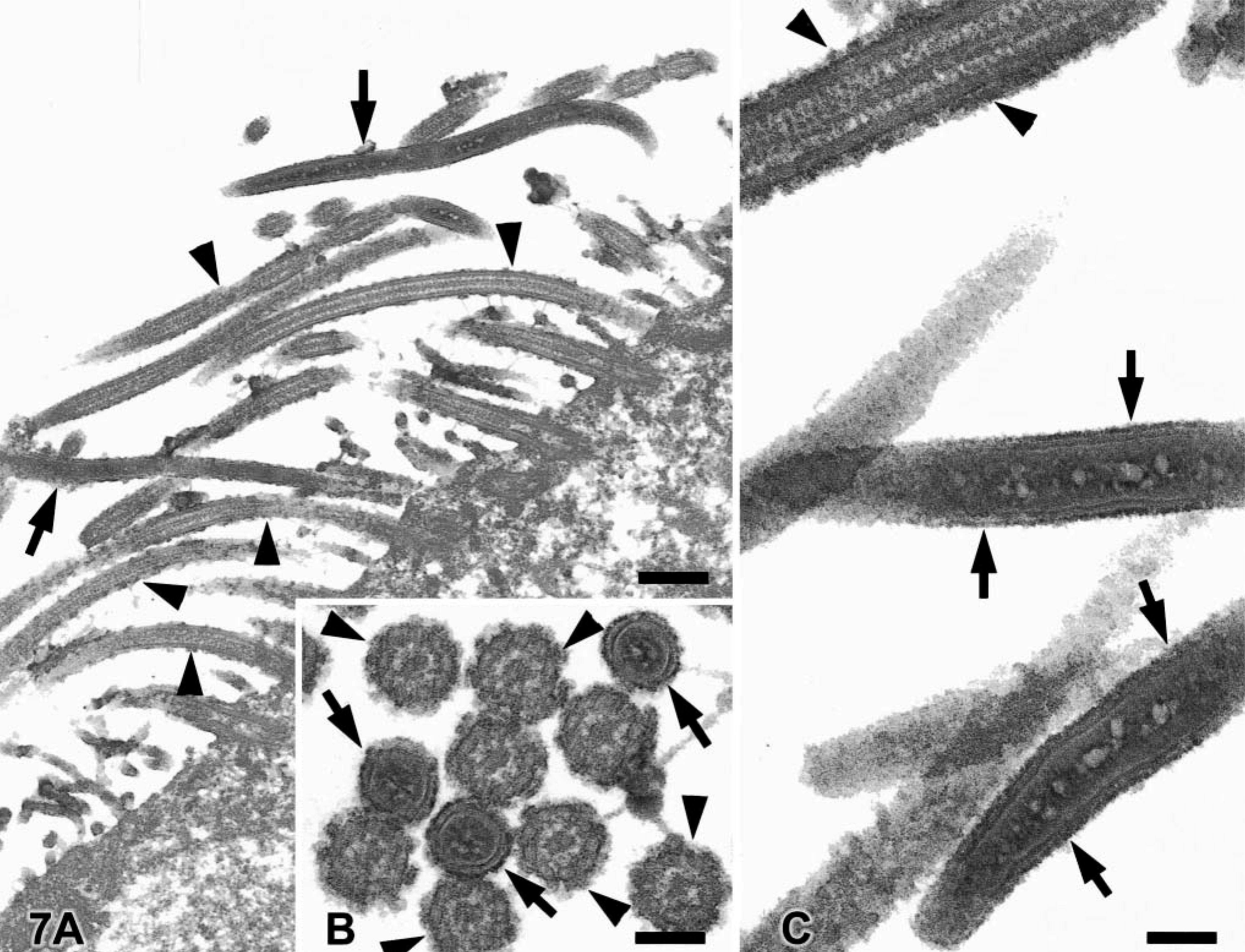
Electron micrograph. Lung; cat.
The bacilli in the respiratory epithelium of this cat share morphologic features with the CAR bacillus. CAR bacillus is a gram-negative, argyrophilic, gliding and flexing filamentous rod found in the respiratory tract of rats, mice, rabbits, hamsters, guinea pigs, cattle, pigs, and goats.3,7,11,16,20 Alone or in conjunction with Mycoplasma, CAR bacillus can produce chronic respiratory disease in laboratory animals.20 However, the role of CAR bacillus in respiratory disease of livestock is not clear.16 The diameter of CAR bacillus in this cat was always smaller than that of adjacent cilia, usually 120–175 nm. The reported diameter of the CAR bacillus in other species is usually bigger than that in this cat and in some cases is larger than that of the cilia.4,7,10,11,14,18 Other ultrastructural characteristics of CAR bacillus reported in other species (trilaminar membrane, central electron-lucent areas) were present in this cat.4,10,14,17,18 The number of bacteria present in electron microscopy specimens was lower than expected based on light microscopy, and the bacteria were always outnumbered by cilia. The extraction procedures followed for the preparation of samples for electron microscopy might have accounted for some loss of organisms.
Cells with morphologic characteristics of globule leukocytes were present in the respiratory epithelium of noninflamed or slightly inflamed airways. Globule leukocytes are present in mucosal epithelium of many vertebrates, including humans, cattle, sheep, goats, dogs, cats, chickens, rats, and mice.1,2,12,13,21 Although the source of these cells is still controversial, there is evidence that they might originate from large granular lymphocytes.13 Although globule leukocytes have been previously reported in the gastrointestinal tract of the cat,13,21 such cells have not been reported in the feline lung.13
This is the first description of CLO and globule leukocytes in the respiratory tract of cats. The significance of CLO in the pathogenesis of bronchitis and bronchiolitis in this cat is unclear. Causes of bronchitis and bronchiolitis in cats include bacterial, viral, parasitic, and allergic diseases.6 There was no evidence of parasitic or allergic reaction in this cat. With only fixed specimens, isolation of other pathogens was not possible.
Although the CAR bacillus is considered pathogenic in some laboratory animal species, its significance as a pathogen in pigs and cattle is questionable. To determine the presence of similar bacilli in the respiratory epithelium of other cats, the lungs of 18 other cats submitted to the VMDL were examined. The criteria for selection was recent consecutive cases examined by two of us (J. A. Ramos-Vara, M. A. Miller) regardless of the presence or absence of pulmonary lesions. Two of nine normal lungs, 1/7 lungs with bronchitis, and bronchiolitis, and 1/2 lungs with pneumonia had CLO, as revealed by light microscopy. The number of bronchi and bronchioles with CLO was similar; however, in a given animal both types of airways did not necessarily have CLO. Terminal bronchioles did not have CLO. The presence of inflammation or lymphoid hyperplasia in airways was not correlated with the presence of CLO. These results indicate that CLO might be present in low numbers in feline lungs but might not necessarily be related to inflammation. Additional studies including complete microbiologic and virologic examination of the lungs and trachea of cats are needed to determine the significance of this CLO in feline respiratory disease.
Footnotes
Acknowledgements
We appreciate the technical work of C. Jensen and R. Tindall for electron microscopy and Howard Wilson and Don Connor for photography, and we thank C. Pennington for submission of this case.
