Abstract
Protozoa were present in routine sections of the gastric fundus of 15 cynomolgus monkeys (Macaca fascicularis) that were being studied in three toxicity studies with novel immunosuppressive agents. Upon detailed light microscopic and ultrastructural evaluation, all stages of parasite development (trophozoites, schizonts, gamonts, and oocysts) were seen and they structurally resembled Cryptosporidium muris, which normally is found in stomachs of rodents. Cryptosporidia were primarily present in the upper one third of fundic glands that were often concurrently colonized by a Helicobacter heilmannii-like organism; however, no clear correlation was found between bacterial burden and the number of protozoa. The primarily mononuclear cellular infiltrate appeared to coincide with the presence of protozoa only in a few animals. Changes in mucous epithelial cells mainly occurred in animals that were part of a 39-week study. Mucous epithelial cells in affected glands contained an increased amount of mucus composed of predominantly acid mucosubstances compared to the normally present neutral mucosubstances. C. muris-like protozoa are newly recognized etiologies for opportunistic infections in the stomach of immunocompromized nonhuman primates. This is the first report of C. muris-like parasite in stomachs of monkeys.
Cryptosporidium spp. are protozoan parasites of humans and animals.7 Unlike other coccidian parasites, cryptosporidia are found only in the microvillus border of epithelial cells.7 Until recently, C. muris and C. parvum were the two main species considered to infect domestic animals and humans.5,7 C. muris was the first species of the genus described by Tyzzer in 1910.15 This species primarily infects the stomach of mice and rats and is considered to be of little or no clinical significance. Tyzzer also described the second species of the genus Cryptosporidium, C. parvum.16 Unlike C. muris, C. parvum can cause gastroenteritis in animals and humans, but it is usually a parasite of the small intestine.7 Although intestinal and extraintestinal cryptosporidiosis associated with C. parvum–like organisms have been reported in several species of monkeys,7 this is the first report of cryptosporidiosis in stomach of monkeys.
Materials and Methods
Tissue samples
Samples of stomach from 15 male and female young adult cynomolgus monkeys (Macaca fascicularis) were evaluated histologically in detail (Table 1). The monkeys originated from Covance Research Products, Alice, TX. The animals were purpose-bred in Vietnam and were housed in indoor cages with access to elevated outdoor runs until approximately 1.5–2 years of age, when they were transferred to Texas and were housed indoors. They were maintained on Harlan Tekland 8775 monkey diet (Madison, WI). The animals were treated with developmental immunosuppresive compounds for 4–39 weeks as part of a routine preclinical safety assessment. The in-life phase of these studies took place at the same site in 1999 and 2000.
Summary of light microscopic fundic changes in 15 Macaca fascicularis harboring gastric Cryptosporidium sp.

Semiquantitative assessment: + = minimal; ++ = mild; +++ = moderate; ++++ = severe.
With neutrophils in glands and lamina propria.
Limited to a few glands.
Light and electron microscopy
Representative tissue samples were obtained from the stomach including cardia, fundus, and pyloric antrum, routinely processed, embedded in paraffin, and stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), PAS with Alcian blue pH 2.5 (PAS/AB), and Genta stain.8 In addition, the following tissues were routinely processed and stained with HE: adrenal gland, aorta, bone marrow, brain, cecum, cervix, colon, duodenum, epididymis, esophagus, eye, femur, gall bladder, heart, ileum, jejunum, kidney, lacrimal gland, liver, lung, lymph nodes (bronchial, mandibular, and mesenteric), mammary gland, ovary, pancreas, parathyroid, pituitary, prostate, rectum, salivary gland, sciatic nerve, seminal vesicle, skeletal muscle, skin, spinal cord, spleen, sternum, testis, thymus, thyroid, tongue, trachea, urinary bladder, uterus, and vagina.
For electron microscopy, formalin-fixed stomach samples of monkey No. 7, adjacent to sites sampled for light microscopy, were transferred to modified Karnovsky's fixative and postfixed in 0.1 M sodium cacodylate-buffered 1% osmium tetroxide. For transmission electron microscopy (TEM), samples were stained en bloc in 2% uranyl acetate in 10% ethanol. Samples were embedded in Embed (Epon) 812, and ultrathin sections were double stained with uranyl acetate and lead citrate and examined with a Zeiss EM-902 electron microscope. For scanning electron microscopy (SEM) after postfixation, samples were critical point dried with a Tosimus SamDri-790 critical point drying unit. Dried samples were fractured, mounted onto specimen holders, and sputter coated with gold/palladium, then examined with a JEOL JSM 6301FXV field emission scanning electron microscope.
Results
The light microscopic changes of the 15 stomach sections that contained protozoa are summarized in Table 1. The stomach sections of other treated animals and all controls of these studies appeared to be negative for protozoa. Moreover, no other animals were diagnosed with gastric protozoasis among those that were placed in studies with nonimmunosuppressive agents. The overall incidence of infection, including control animals, was approximately 13% in the three studies; approximately 20% of treated animals were diagnosed with parasitism. Cryptosporidia were present only in the fundus of stomachs and were primarily in the upper one third of the glands; however, the gastric luminal surface was seldom covered by protozoa. In some animals, only a few glands contained protozoa, whereas several animals had an essentially diffuse involvement of the fundic section examined. Concurrently, all stomachs were colonized by a Helicobacter heilmannii–like organism, but no clear correlation was found between bacterial burden and the number of protozoa. The lamina propria of the majority of stomachs was infiltrated by primarily mononuclear cells that occasionally were admixed with eosinophils. Only in a few animals was this cellular infiltrate associated with the presence of protozoa. Epithelial changes occurred primarily in the longer-term study and were characterized by the presence of increased amounts of mucus that had decreased staining characteristics on HE. Mucous epithelial cells of monkey Nos. 11 and 12 from a 4-week study had slightly increased mucus contents in those glands where protozoa were present. In animals from the 39-week study, a sharp demarcation often was found between normal mucosa and glands containing protozoa, which had altered staining characteristics (Fig. 1). In most severely affected areas, mucous cells contained predominently acid mucosubstances, characterized by deep blue staining on PAS/AB, whereas in less severely affected regions, cells had a blue-purple color, indicating a mixture of acid and neutral mucosubstances. In contrast, in areas that were free of protozoa, surface and mucous neck cells contained predominantly neutral mucus that stained a bright red-purple with PAS/AB. On Genta-stained sections8 epithelial cells with altered mucus production stained blue, whereas normal surface epithelium was gray; bacteria were easily visualized as black tightly coiled spirals and protozoa were either red or black. No other opportunistic infections were present in tissues examined.
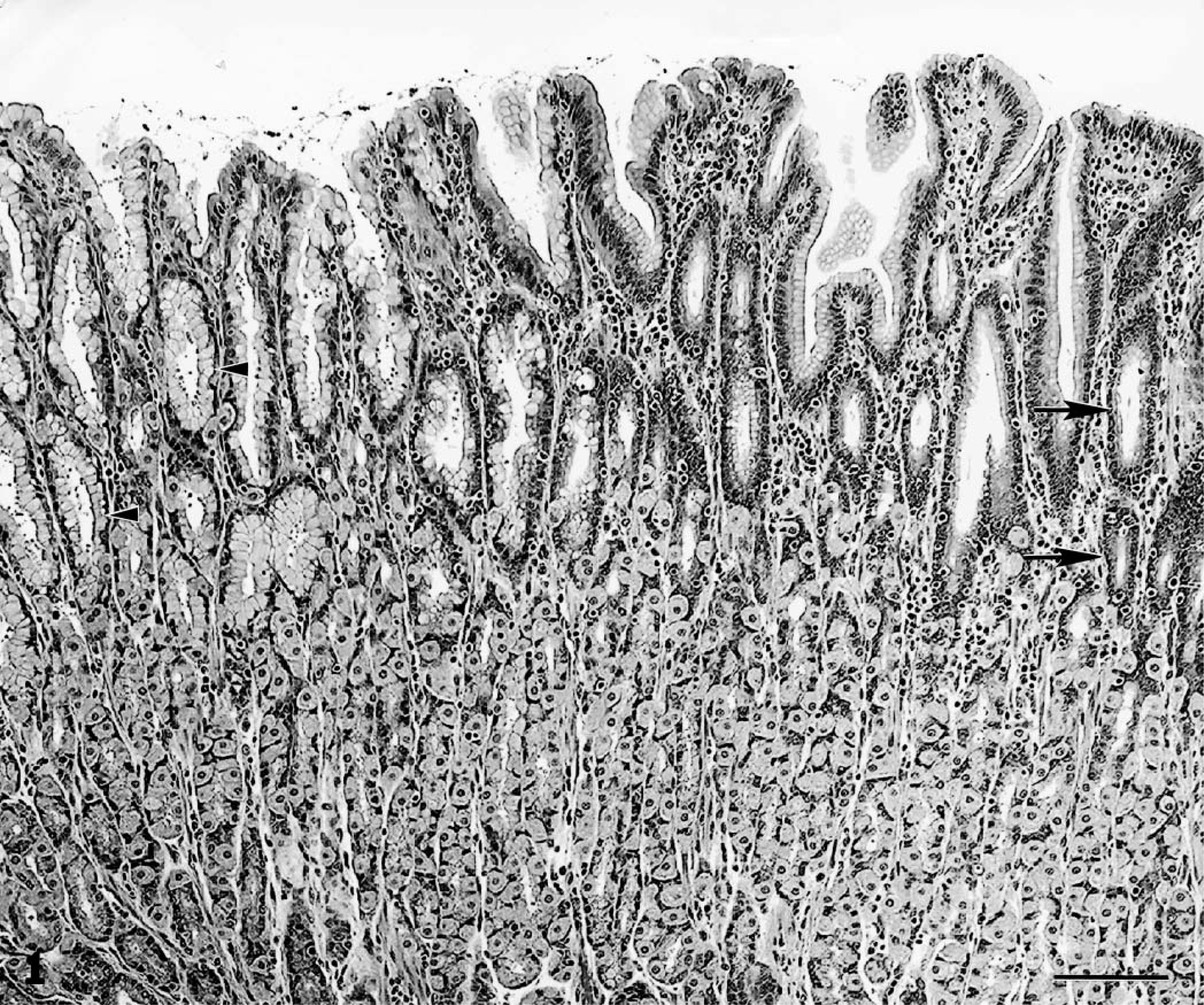
Gastric fundus; cynomolgus monkey No. 2. Glands that are parasitized (arrowheads) are lined by tall columnar mucous epithelial cells containing abundant pale-staining apical cytoplasmic mucus, whereas glands that are not parasitized (arrows) are lined by low columnar epithelial cells containing somewhat darker-staining mucus. HE. Bar = 100 μm.
Cryptosporidia were located in microvillus border of glandular epithelial cells. In HE-stained sections, organisms stained faintly (Fig. 2). In 1-μm sections stained with toluidine blue, all stages of parasite development were seen, including trophozoites, schizonts, gamonts, and oocysts (Fig. 3). By TEM, organisms were located in the lumen and in epithelial cells of gastric glands (Fig. 4). Parasites were located in parasitophorous vacuoles and attached to host cells with a prominent feeder organelle (FO; Figs. 5–8). The FO consisted of membranous lamellae that appeared as whorls in cross section (Fig. 8). The FO was surrounded by filamentous processes of the host cell (Fig. 8). The parasite was separated from the host cell cytoplasm by a dense band (Fig. 6). Trophozoites were globular and contained a large nucleus but very few other organelles (Fig. 5). Merozoites budded from a large residual body (Figs. 5, 7). By TEM, most schizonts contained slender merozoites (Figs. 4, 9, 10). Mature schizonts in sections were 4.0–7.5 × 4.0–6.0 μm (n = 10). At least six merozoites were identified in mature schizonts (Figs. 6, 9). Because merozoites are sickle-shaped, it was not possible to measure them accurately. By TEM, the inner curvatures were 5.3 and 6.3 μm long and outer curvatures were 8.3 and 8.9 μm long in two longitudinally cut merozoites (Fig. 10); these merozoites were approximately 1 μm wide at their widest point (Fig. 10). In an SEM preparation of a mature schizont, six sickle-shaped merozoites are visible (Fig. 9). These merozoites had blunt ends and a pitted surface (Fig. 9).
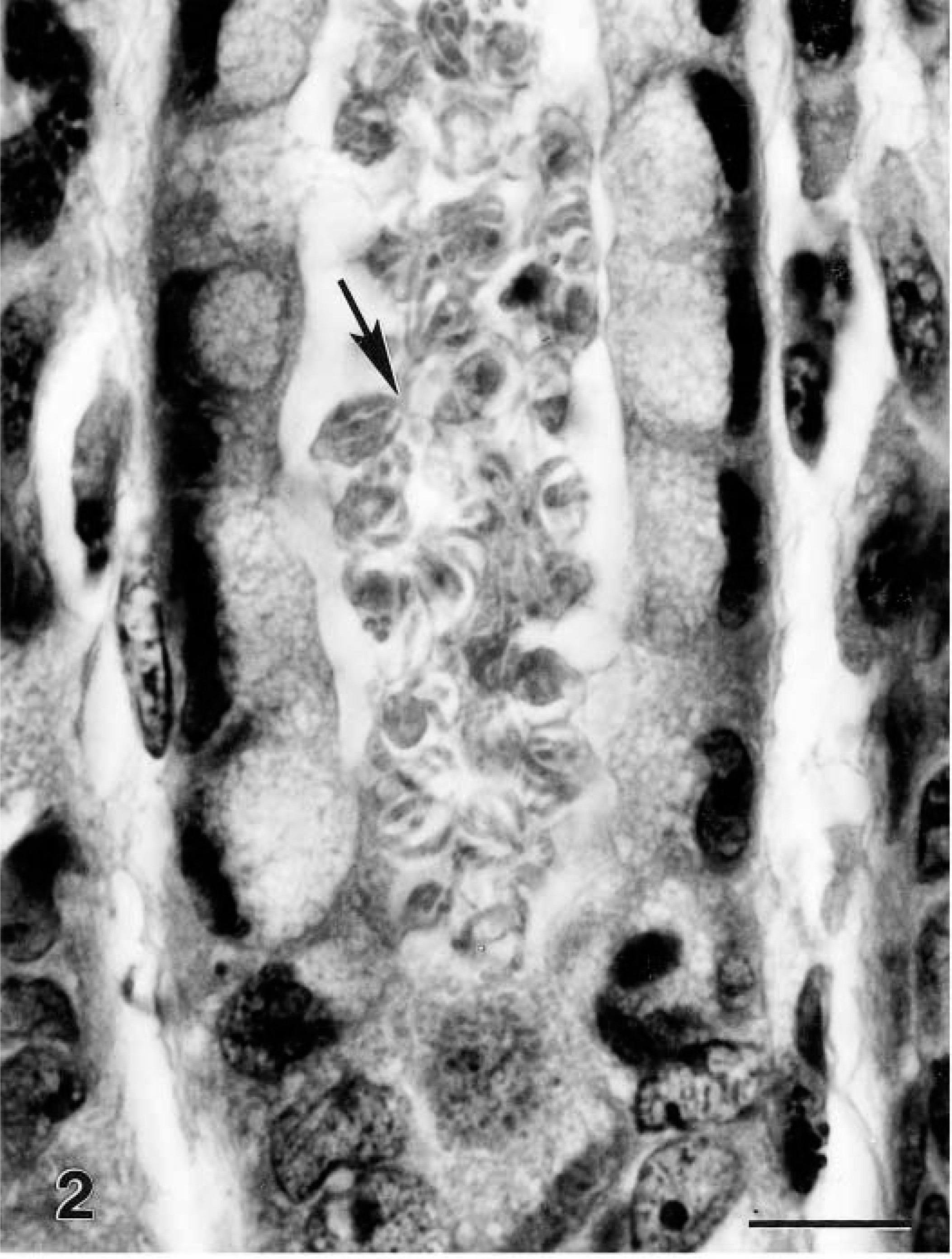
Gastric fundus; cynomolgus monkey No. 2. Gastric gland with numerous cryptosporidia (arrow) in lumen. HE. Bar = 10 μm.
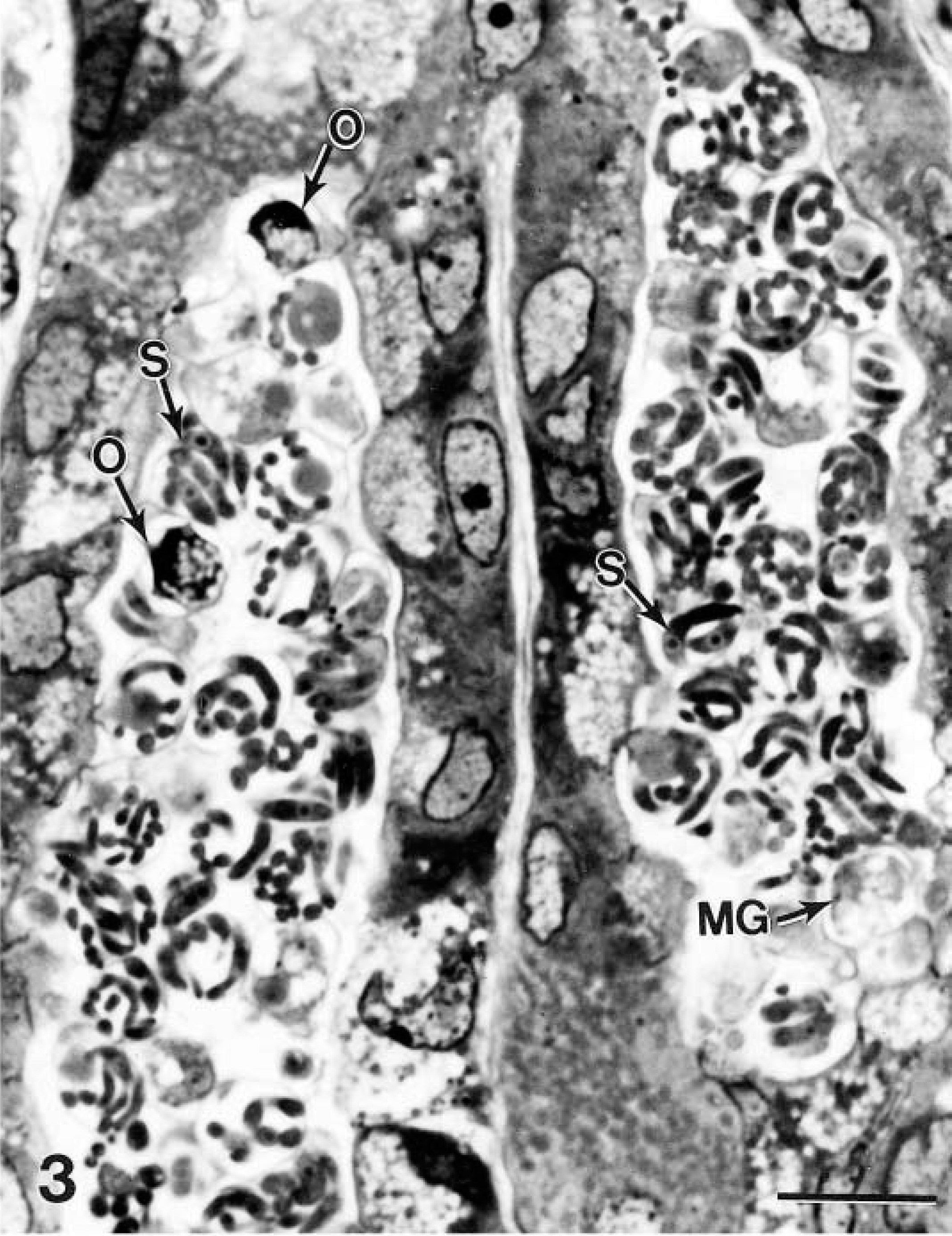
Gastric fundus; cynomolgus monkey No. 7. Two gastric glands packed with cryptosporidia in various stages of development including schizonts (S), male gamonts (MG), and oocysts (O). Epon-embedded 1-μm section, toluidine blue. Bar = 10 μm.
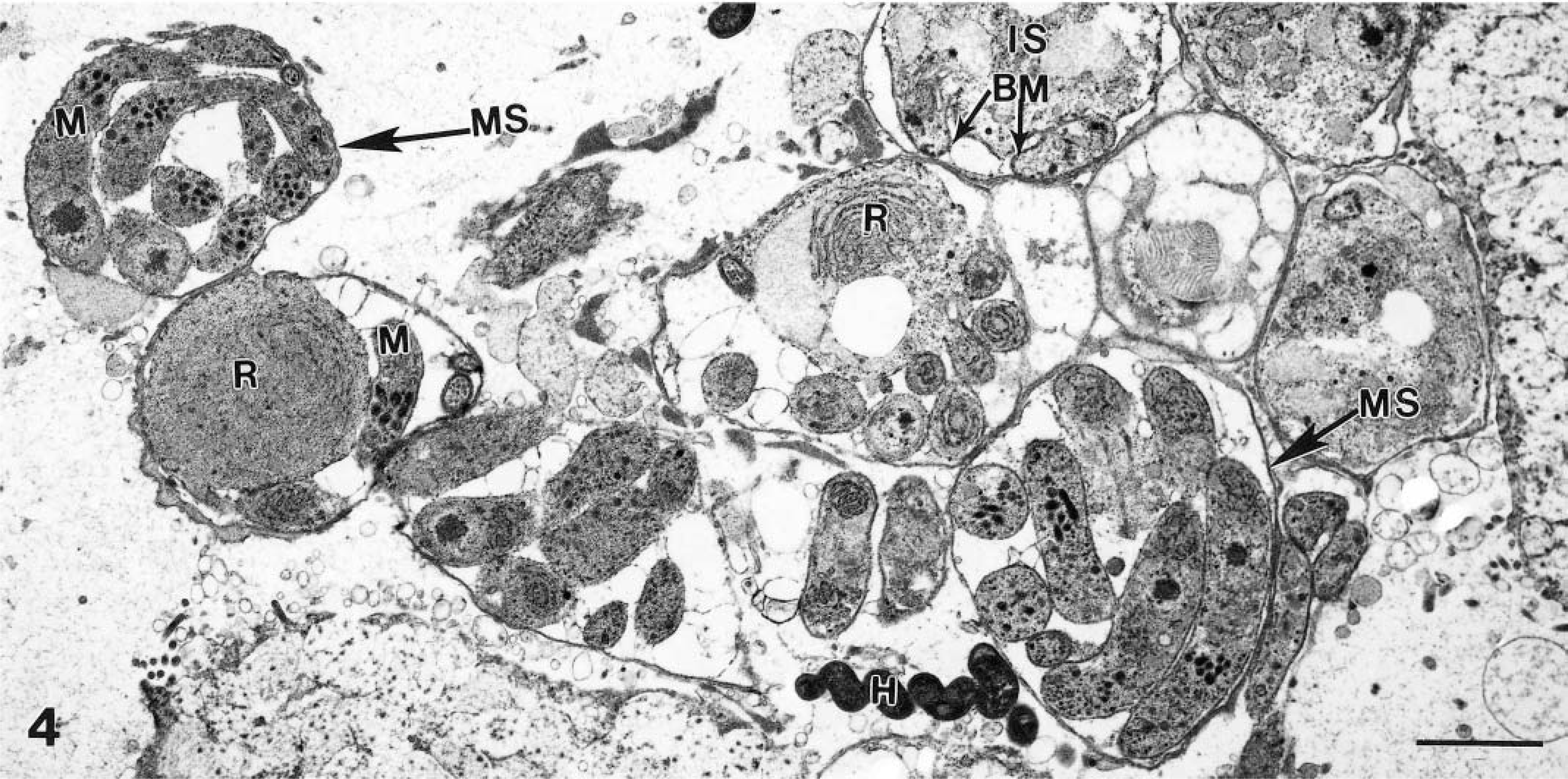
Transmission electron micrograph of several schizonts in various stages of development. Immature schizonts (IS) have a large residual body (R). Mature schizonts (MS) contain sickle-shaped merozoites (M). Budding merozoite is indicated (BM). Also note a Helicobacter heilmannii–like (H) organism. Bar = 2 μm.
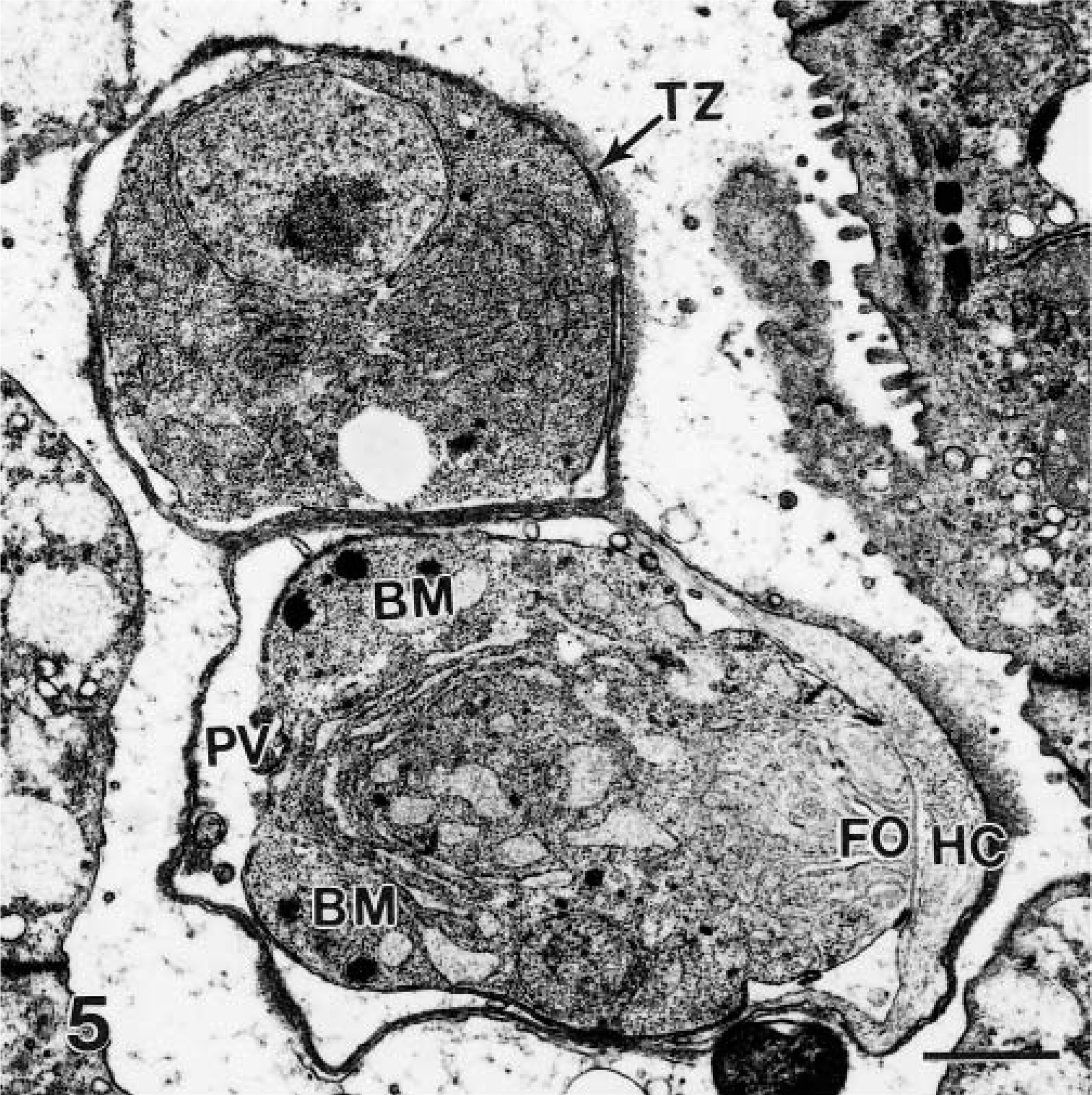
Transmission electron micrograph of a trophozoite (TZ) and an immature schizont in parasitophorous vacuoles (PV). Note the large nucleus, prominent reticulum, and absence of other organelles in the trophozoite. The schizont is immature and has two budding merozoites (BM). Note feeder organelle (FO) attached to host cell (HC). Bar = 1 μm.
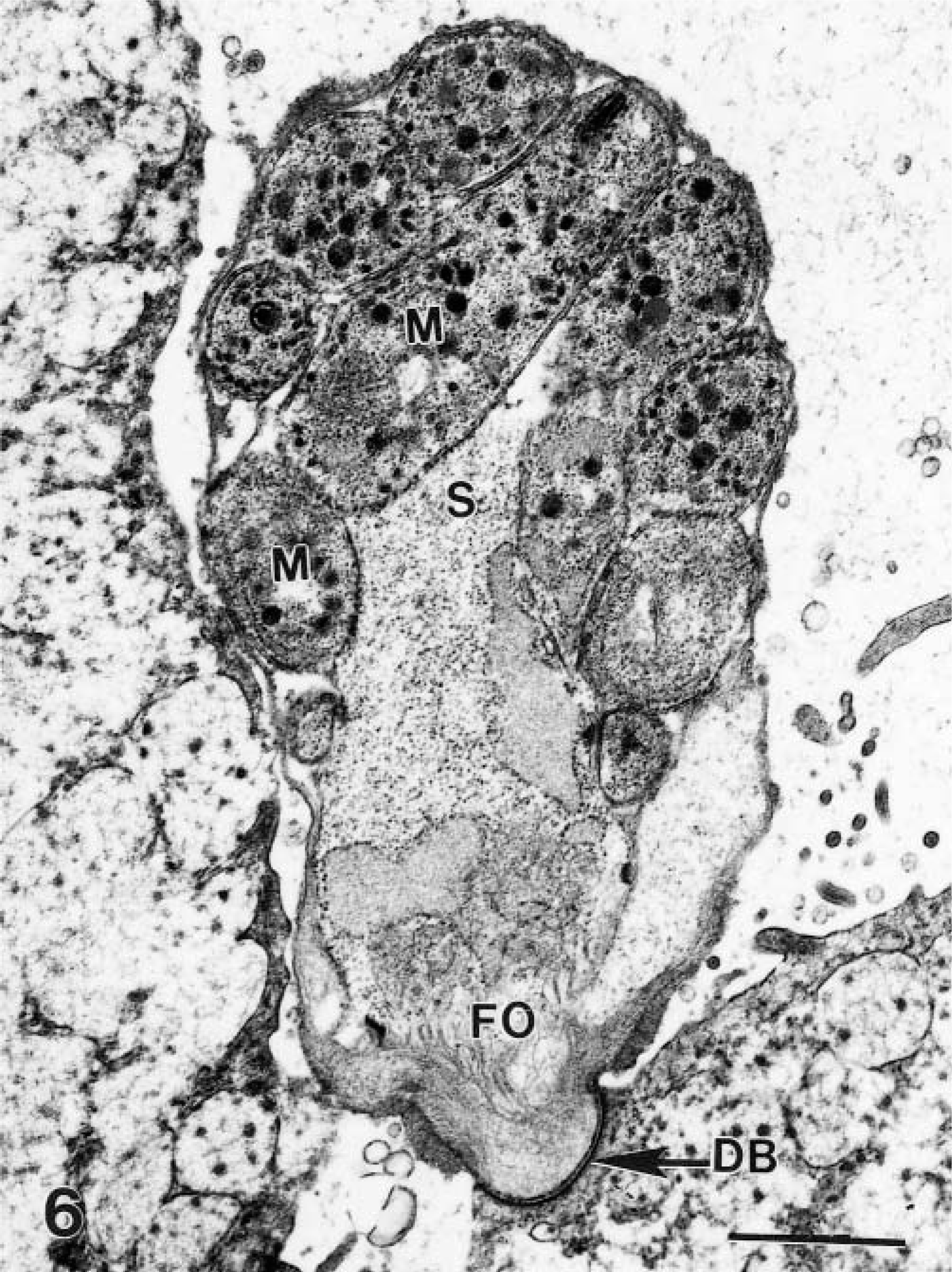
Transmission electron micrograph of a schizont (S) with mature merozoites (M), and a feeder organelle (FO) of the parasite separated from the host cell by a dense band (DB). Bar = 1 μm.
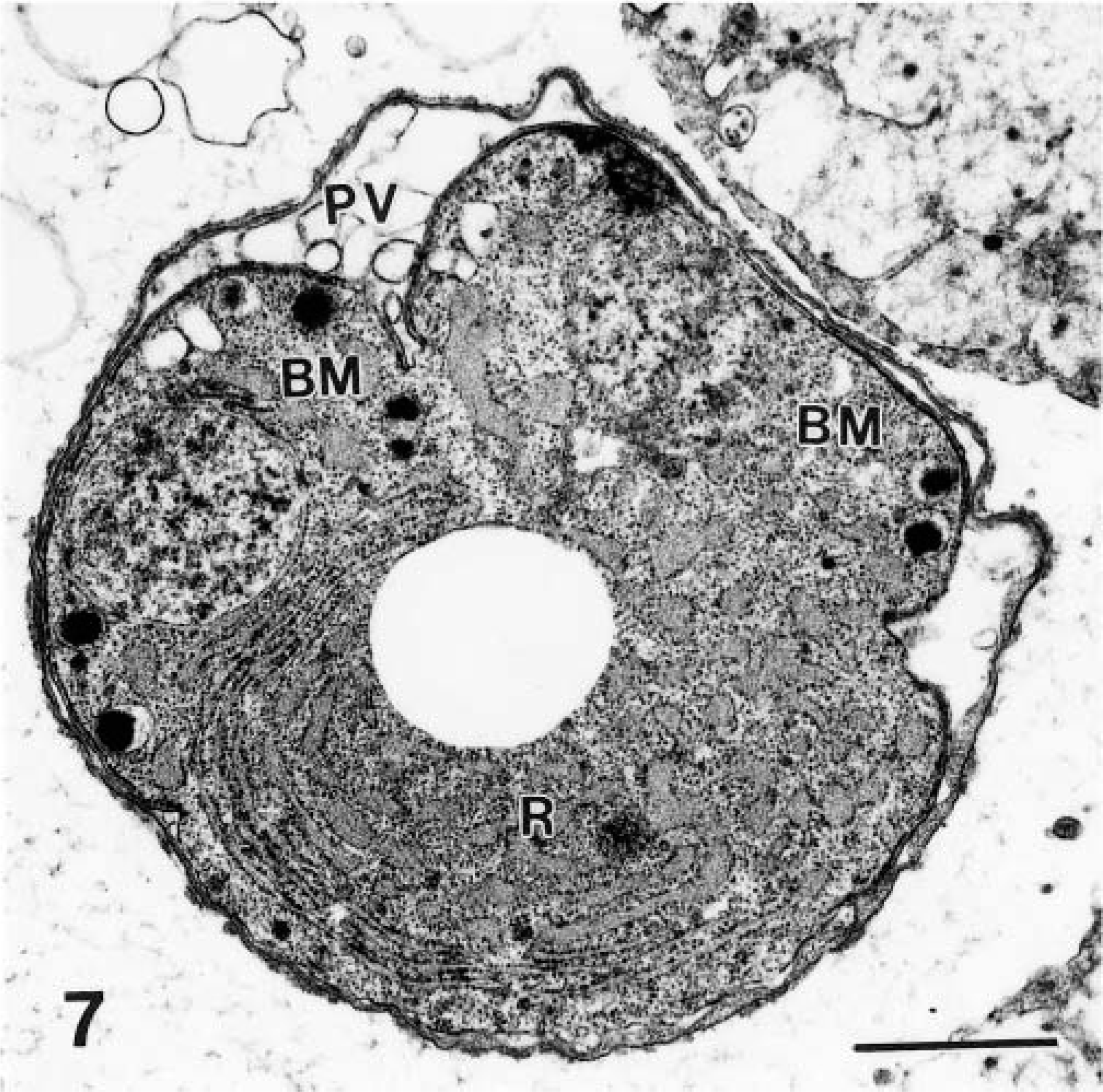
Transmission electron micrograph of an immature schizont with merozoites budding (BM) from a residual body (R). The parasitophorous vacuole (PV) is indicated. Bar = 1 μm.
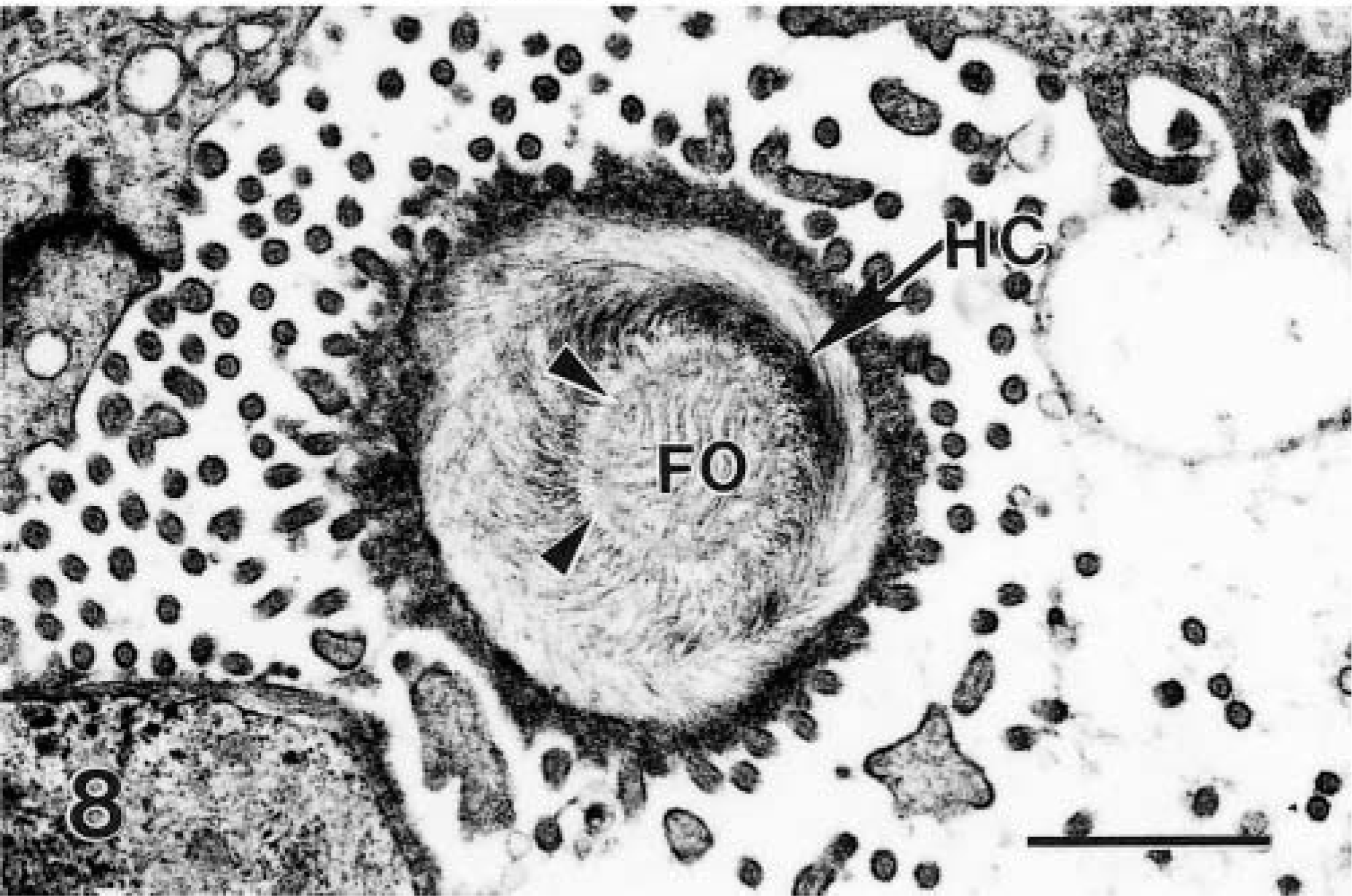
Transmission electron micrograph of transverse section of attachment of Cryptosporidium to the host cell (HC). The central area is the feeder organelle (FO), surrounded by a rim (arrowheads) of filamentous processes of the host cell. Bar = 1 μm.
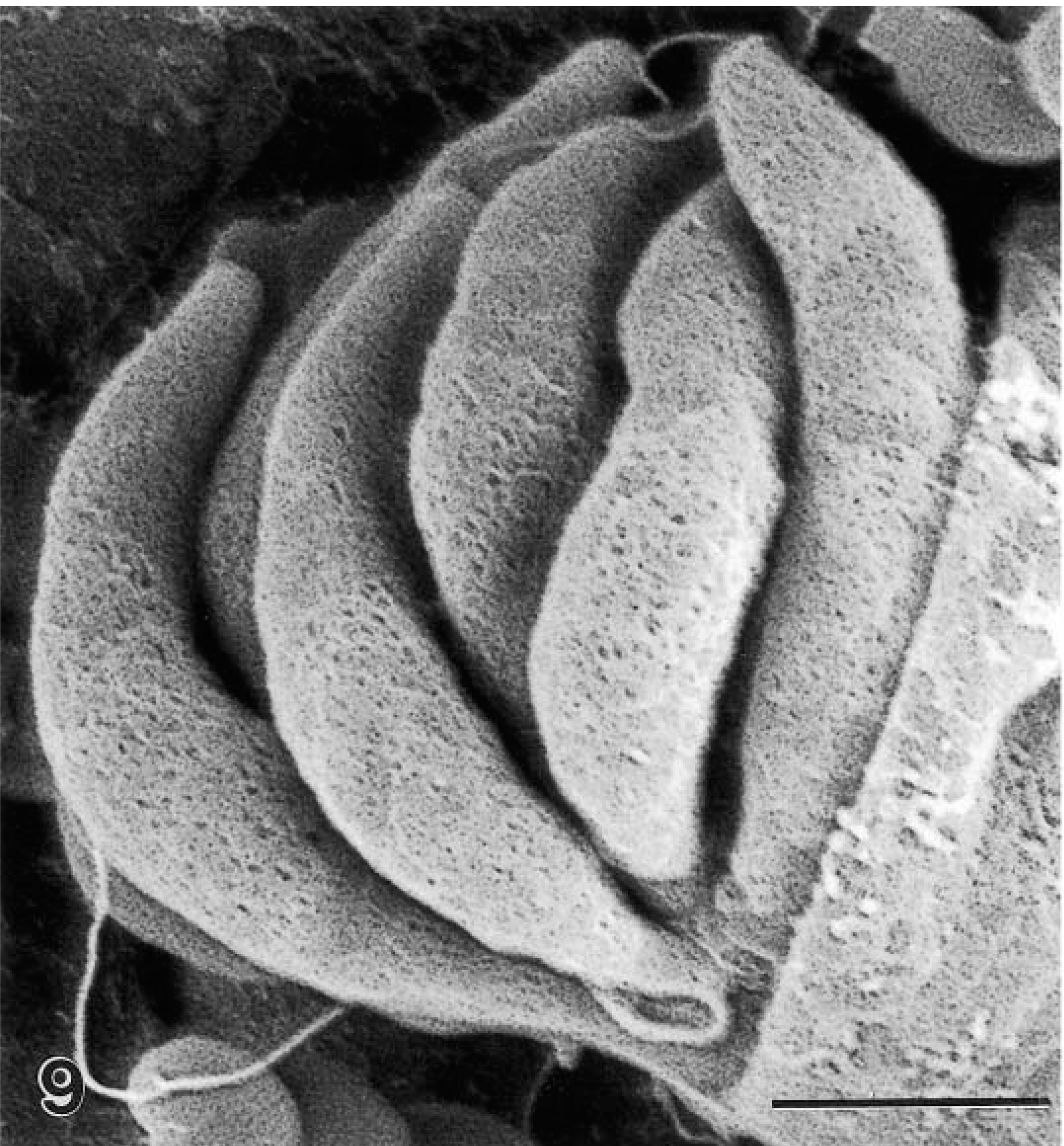
Scanning electron micrograph of a mature schizont exposed to show six sickle-shaped merozoites. The merozoite surfaces are pitted. Bar = 1 μm.
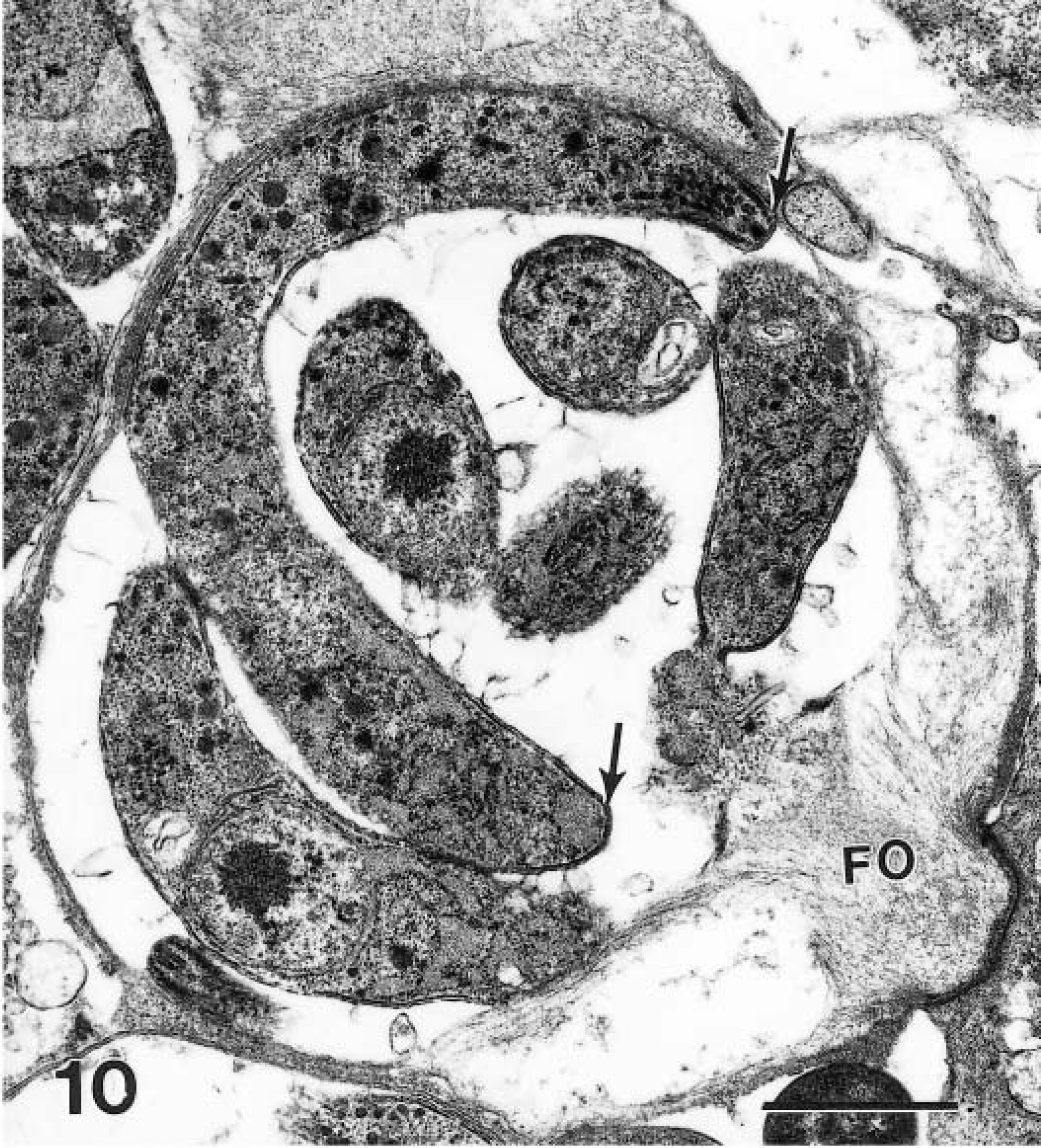
Transmission electron micrograph of a mature schizont with one longitudinally cut merozoite (arrows). This merozoite is about 8 × 1 μm. Note the feeder organelle (FO). Bar = 1 μm.
Two schizonts were found to have four short merozoites attached to a large residual body (Figs. 11, 12). By TEM, the residual body was 3.2 × 3.8 μm and contained prominent endoplasmic reticulum.
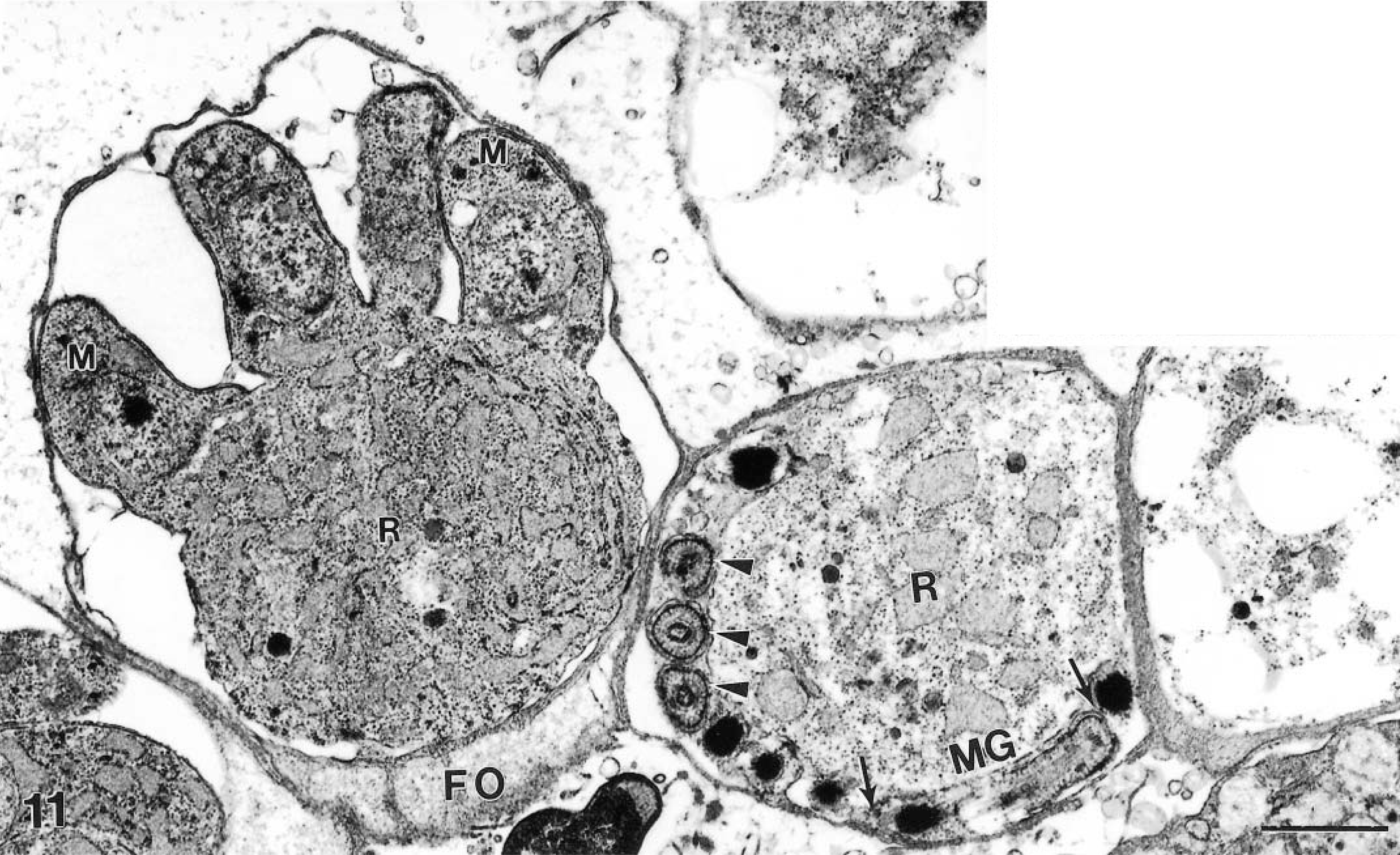
Transmission electron micrograph of an immature schizont, with four stubby merozoites (M) attached to the large residual body (R) which is rich in endoplasmic reticulum. Next to the schizont is a mature male gamont with one longitudinally cut bullet-shaped male gamete (MG, arrows). Other male gametes are cut in cross section (arrowheads). A residual body (R) and feeder organelle (FO) are indicated. Bar = 1 μm.
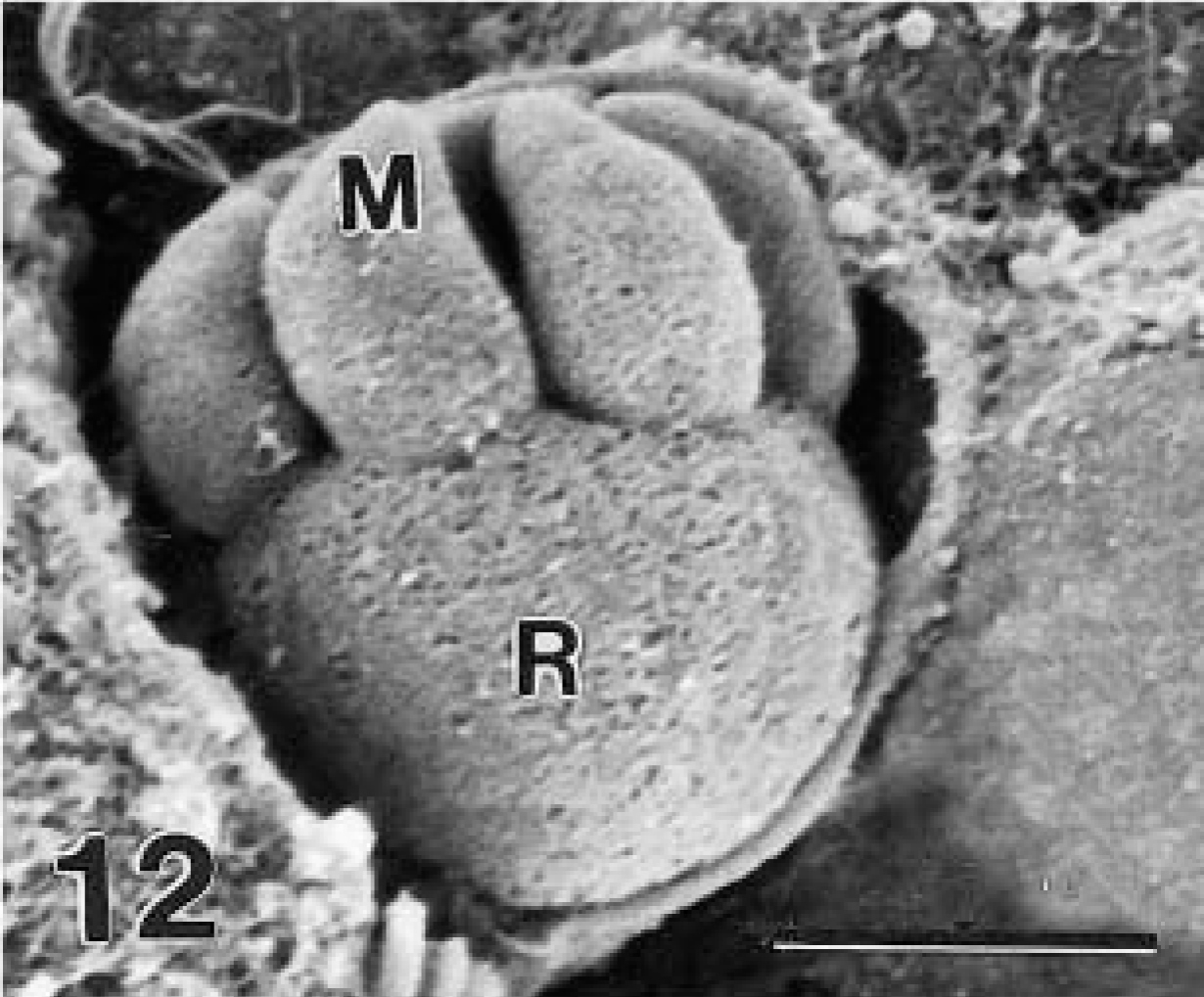
Scanning electron micrograph of an immature schizont with 4 small stubby merozoites (M) still attached to a large residual body (R). Bar = 1.6 μm.
Only a few gamonts and oocysts were seen. Mature male gametes were approximately 2.0 × 0.5 μm in size and were located at the periphery of the gamont (Fig. 11). Female gamonts had a single nucleus and wall-forming bodies that were PAS-positive. Oocysts in section (n = 10) were 5–8 × 4–6 μm (Figs. 13, 14). Sporulated oocysts contained a prominent residual body (Figs. 13, 14). Sporozoites contained amylopectin granules (Fig. 14). The oocyst contained a characteristic suture in its wall (Fig. 15).
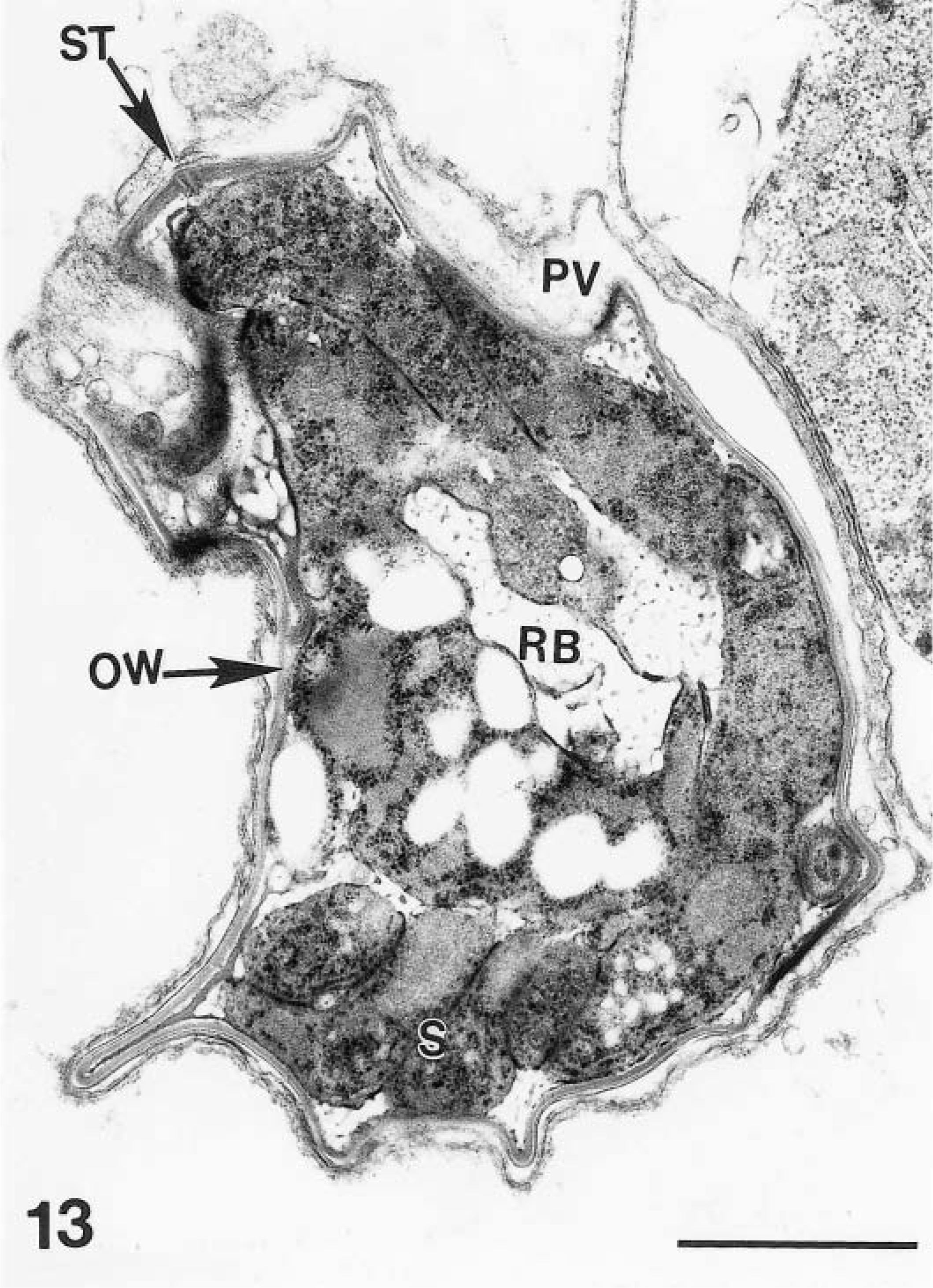
Transmission electron micrograph of a sporulating oocyst with sporozoites (S) at the periphery and an amylopectin-rich residual body (RB) in the center. The oocyst is located in a parasitophorous vacuole (PV). Note a suture (ST) in oocyst wall (OW). Bar = 1 μm.
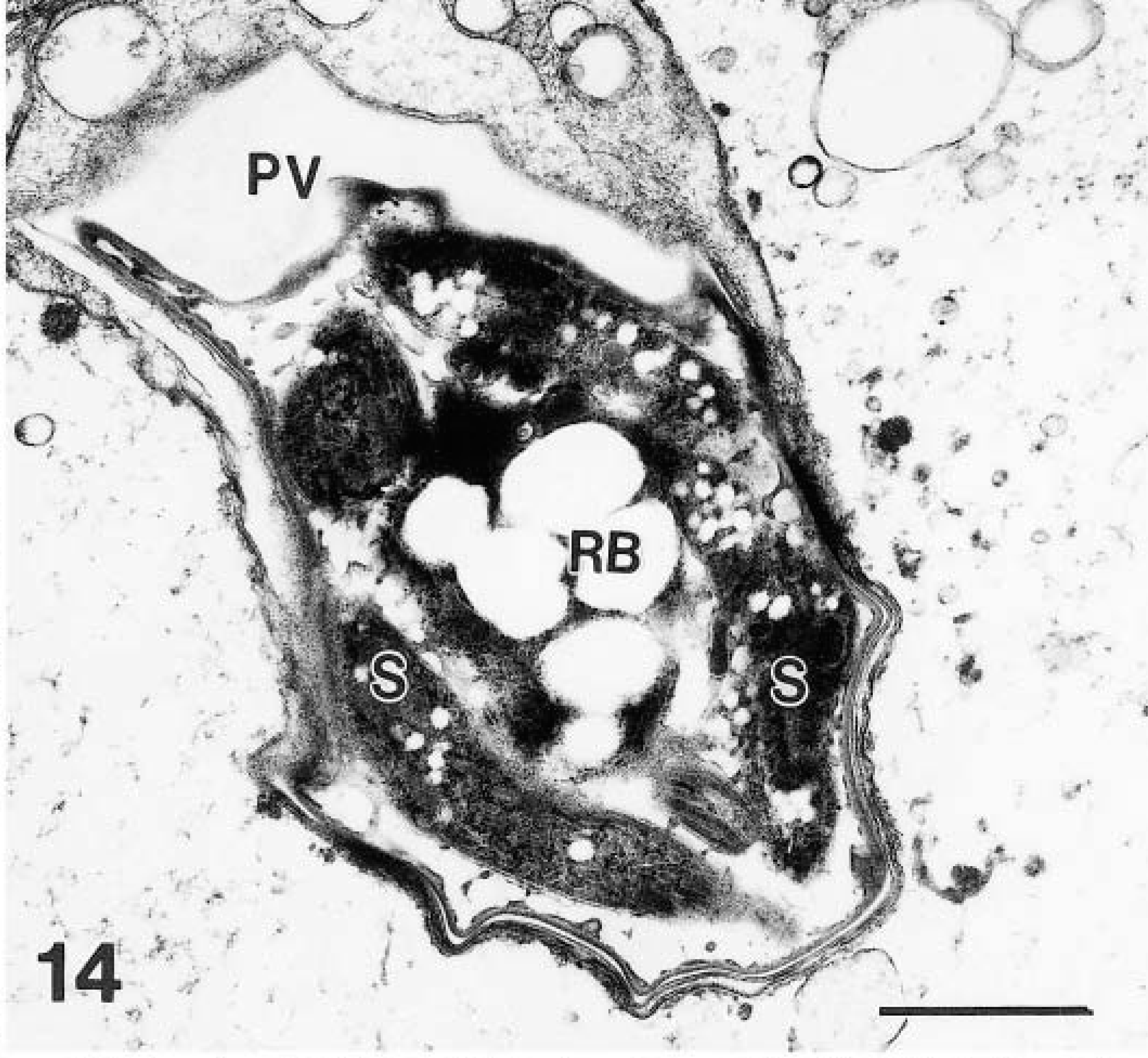
Transmission electron micrograph of a sporulated oocyst with sporozoites (S); note amylopectin granules in sporozoites and a centrally located residual body (Rb). Bar = 1 μm.
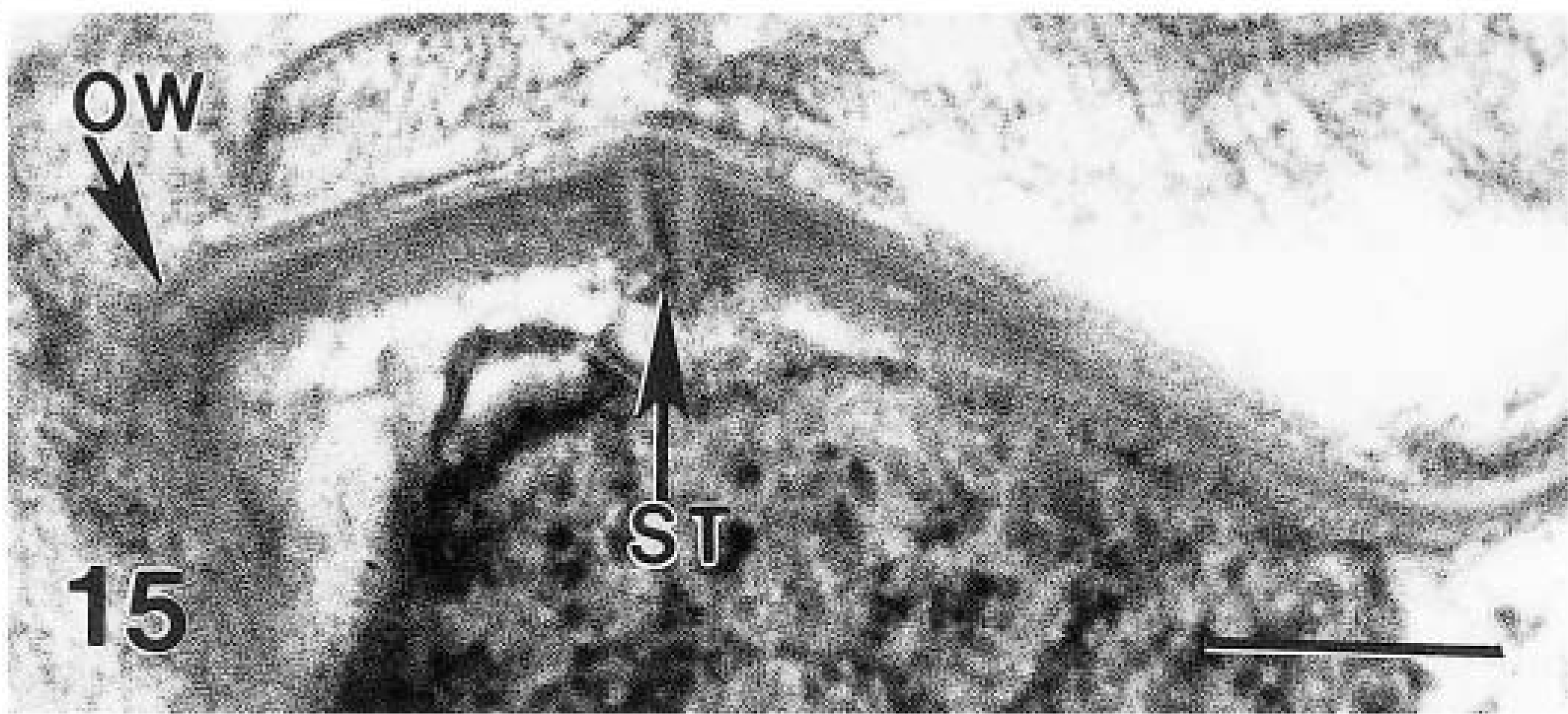
Transmission electron micrograph. Higher magnification of Fig. 13. The suture (ST) runs through the multilayered oocyst wall (OW). Bar = 1 μm.
Discussion
The species of Cryptosporidium in the present study was not identified. Cryptosporidiosis of the digestive tract in nonhuman primates has been reported in nursery-reared infants or in animals with acquired immunodeficiency syndrome (AIDS).14 These cases were caused by C. parvum and within the digestive tract, typically involved the small intestine, although large intestinal and hepatic, pancreatic, and biliary involvements also have been reported. C. parvum was present in the pyloric antrum of a patient who had AIDS and also had cryptosporidiasis in the small and large intestine.11 In the monkeys of the present report, only the fundic glands, but not the intestine, were parasitized with a Cryptosporidium sp. Moreover, in histologic sections, the oocysts in cynomolgus monkeys were 5–8 × 4–6 μm and were structrually distinct from C. parvum oocysts, which measure approximately 5.2 × 5.0 μm.5 Until recently, only C. muris was thought to parasitize gastric glands of rodents, cattle, and camels.6,7 Recently, a new species, C. andersoni, was proposed for the parasite found in the abomasum of cattle.10 C. andersoni differs from C. muris molecularly, structurally, and biologically. The fecal oocysts of C. andersoni are 6.0–8.1 × 5.0–6.5 μm and are not infective for mice, whereas oocysts of C. muris are 7.5–9.8 × 5.5–7.0 μm and are infective to mice and rats.4,10 Because fecal oocysts from monkeys were not available for molecular and structural studies, we are unable to compare C. muris with C. andersoni.5,10 Our attempts to recover good-quality DNA from paraffin sections of stomachs of monkeys were unsuccessful.
Little is known of the pathogenic potentials of cryptosporidial species in stomachs of mammals. Both C. muris and C. andersoni are considered to be of little or no clinical significance. In a recent report, two healthy girls were diagnosed to have a subclinical infection.9 Although C. muris causes no overt disease, abomasal parasitism of cattle, now considered to be due to C. andersoni, has been associated with decreased production in feedlot as well as in dairy operations.2 Parasitized cattle had increased abomasal pH and weight and had elevated pepsinogen levels. The abomasal mucosa of these cattle was thickened and granular, resembling type 2 ostertagiasis. The fundus glands of nude mice that were experimentally infected with C. muris were dilated and had mild epithelial changes.13 An inflammatory reaction in experimentally infected mice was present only in animals that received the largest inoculum of 1,000,000 oocysts. In our monkeys, the epithelial changes were less severe than in the experimentally infected mice. When mice that were naturally infected with C. muris were infected with Helicobacter felis, gastric glands were more severily parasitized by the protozoa and their stomach had more severe cellular infiltrates.12 A H. heilmannii–like bacterium was present among cryptosporidial parasites found in monkeys in the present report but its role in the pathogenesis of cryptosporodial infection is unknown. The pathogenesis of the gastric cryptosporidiosis of these monkeys is undetermined, but as with infection with C. parvum, their immunocompromized status was a factor because we did not see parasites in control, immune-competent animals or in animals that were treated with nonimmunosuppressive developmental compounds at our facility concurrently to these studies. Similar to C. muris infections in other species, these monkeys had no clinical signs.
The developmental stages of Cryptosporidium sp. observed in the stomach of monkeys could not be compared with those of C. andersoni because little is known of the structure of developmental stages of C. andersoni in infected abomasa.1,10 In naturally infected cattle, a 10% increase occurred in abomasal mucosal thickening; the gland lumens were widened and the epithelium was atrophied.1
The structure of the parasite in monkeys most closely resembled C. muris from stomachs of mice.17 Unlike other coccidian parasites, cryptosporidial organisms are attached to host cells by an FO.5,7,17 Although all species of Cryptosporidium have an FO, the FO in C. muris is distinct.17,18 The FO in C. muris consists of concentric folds or filaments surrounded by filamentous processes of the host cell.17
Two generations of schizonts have been reported for C. muris.3 The first generation has six to eight slender merozoites.3,17 The second generation contains four stubby merozoites.3 Uni et al.17 found only the first-generation schizonts. In the present study, two schizonts with four stubby merozoites were considered to be second-generation schizonts.
Footnotes
Acknowledgements
We thank the following pathologists for presenting these cases to us: D. Gunson, S. Francke, and F. Voelker. We also thank G. Argentieri, P. Grosenstein, F. Grossman, and B. Zdawczynski for their technical assistance. We thank Drs. R. Fayer and C. A. Speer for their advice.
