Abstract
A 9-year-old, spayed female, Airedale Terrier was euthanatized and necropsied after a progressive clinical course that included Horner's syndrome of the left eye and unilateral atrophy of the masticatory muscles. Although gross lesions were limited, a polyradiculoneuritis and ganglionitis that was most severe in the trigeminal nerves and ganglia were confirmed histologically. The inflammatory infiltrate consisted predominantly of macrophages and B and T lymphocytes that were phenotypically confirmed by immunostaining. Horner's syndrome was the result of damage to postganglionic sympathetic fibers that were incorporated in segments of the inflamed trigeminal nerve and its ophthalmic branch. Histologically, the character and distribution of the inflammation was similar to previously described syndromes of suspected immune-mediated etiology in humans and animals.
Inflammatory diseases that are restricted to peripheral nerves, nerve roots, and specific ganglia are infrequently recognized in domestic animals. For that reason and because many cases resolve without histologic evaluation, the classification, pathogenesis, and etiologic interrelationships of these entities are not well defined. The best-characterized syndrome among this ill-defined group is acute idiopathic polyradiculoneuritis of dogs, so-called coonhound paralysis. 9,10 This syndrome generally presents as an acute ascending, symmetrical paralysis leading to tetraparesis. The disease was first recognized in dogs associated with a history of a raccoon bite or scratch. Subsequently, the syndrome was reported in dogs without history of an inciting stimulus. 17 Histologic lesions of coonhound paralysis are concentrated in the ventral roots of spinal nerves and peripheral nerves and consist of a variable mononuclear inflammatory cell infiltrate accompanied by axonal degeneration and axonal reaction in lower motor neurons. 10
Other infrequently occurring and poorly described syndromes of dogs are grouped under the term chronic polyradiculoneuritis. These diseases are characterized by slowly progressive, relapsing, motor or sensory deficits secondary to inflammation of spinal roots, and cranial and peripheral nerves. 20 Syndromes that may be classified as chronic polyradiculoneuritis include chronic relapsing polyradiculoneuritis, 7 hypertrophic neuropathy, 8 polyradiculoneuritis, 12 and chronic polyneuritis. 1
The etiology and pathogenesis of these syndromes are largely undetermined. Coonhound paralysis has many clinical and pathologic features similar to the Guillan-Barre syndrome (GBS) in humans. 6,10 Considerable evidence exists that GBS has an immunologic basis, possibly secondary to postinfectious etiologies. 13,22 Thus, presumption and circumstantial evidence suggest that coonhound paralysis has a similar, immune-mediated basis. The causative factors associated with chronic polyradiculoneuritis syndromes, although more obscure, are likewise speculated to be immunologically mediated.
Horner's syndrome is a well-recognized entity caused by deficiency in sympathetic innervation to the eye. 16,21 Sympathetic innervation to the eye is a three-neuron pathway. The cerebral or upper motor neuron pathway begins in hypothalamic neurons, passes through the midbrain and the caudal brainstem, and descends in the lateral tectotegumentospinal tract of the cervical spinal cord and synapses on lower motor neurons in thoracic segments 1, 2, and 3. Axons arising from lower motor neurons exit the spinal cord through ventral roots, join the thoracic sympathetic trunk, pass through both the cervicothoracic and middle cervical ganglia without synapse, and ascend the neck in the vagosympathetic trunk. They synapse in the cranial cervical ganglion where postganglionic fibers originate, course forward through the middle ear, then join the trigeminal nerve whereby they reach the eye through its ophthalmic branch. 4,16
Horner's syndrome can result from damage to any element in the pathway between its origin centrally and its termination in end organs of the eye and orbit. Thus, intracranial or intraspinal lesions may result in upper motor neuron deficit. Damage to spinal neurons at T 1, 2, or 3 or their axons, which extend through the vagosympathetic trunk to the cranial cervical ganglion, induce preganglionic deficits. Damage to the cranial cervical ganglion or fibers composed of axons of those ganglion cells induce postganglionic deficit. Horner's syndrome is relatively infrequently associated with postganglionic damage. 15,16 Although Horner's syndrome associated with trigeminal neuritis has been reported occasionally, 11 we have not encountered a single report that histopathologically documents trigeminal neuritis in association with Horner's syndrome in the dog.
We report here a case of polyradiculoganglioneuritis in which neural involvement is largely focused in the trigeminal nerves. Clinical expression included unilateral atrophy and later bilateral paralysis of muscles of mastication and Horner's syndrome. Histopathologic evidence is presented that strongly supports the premise that sympathetic innervation to the left eye was dysfunctional because of the proximity of ocular sympathetic fibers to the severely inflamed left trigeminal nerve.
A 9-year-old, spayed female Airedale Terrier was initially presented for examination and treatment of unilateral (left) conjunctivitis. Antibacterial medication instilled in the conjunctival sac resulted in transient improvement. At reexamination 4 days later, anisocoria, meiosis, enophthalmus, protrusion of the third eyelid, and ptosis of the left eyelid were recognized. Three weeks later, anisocoria and enophthalmus were more severe and recognizable atrophy of the left masseter and temporal muscles had occurred. Additionally, the dog was “polydipsic”; however, a urine specific gravity was elevated (1.055), suggesting that the dog was only attempting to drink, rather than successfully drinking, water. Two days later the dog was unable to close her mouth; the owner requested euthanasia.
Complete necropsy was performed within 1 hour of euthanasia. Gross lesions were limited to markedly reduced muscle mass of the left temporal and masseter muscles and severe erosion of articular cartilage of stifle joints. Other organs and tissues as well as the cavities of each tympanic bulla were normal. Histologic examination was conducted on specimens of all organ systems and segments of the nervous system that included multiple specimens of brain, and spinal cord, thoracic segments 1, 2, and 3, and selected dorsal root ganglia. Also examined were the thoracic and cervical sympathetic trunks, the cervicothoracic and cranial cervical sympathetic ganglia, the semilunar ganglion of each trigeminal nerve, each trigeminal trunk proximal and distal to the ganglion, the mandibular cluster of nerves after emergence from the oval foramen, each eye, and segments of the sciatic and radial nerves. The ophthalmic nerve was not specifically identified.
Extensive histologic lesions were present in pre- and postganglionic segments of the trigeminal nerve trunk and the ganglion (Figs. 1, 2). Lesions were characterized by an extremely dense inflammatory cell infiltrate composed of variable proportions of lymphocytes, plasma cells, macrophages, and a few scattered eosinophils. Although inflammation was not present in the trigeminal root within or at the margins of the caudal brainstem, numerous isolated demyelinated, degenerate fibers were present in the trigeminal root and nerve (Fig. 3) and chromatolysis and nuclear displacement occurred in scattered neurons of the trigeminal motor nucleus. The trigeminal lesion was more severe on the left than on the right. Denervation atrophy, evidenced by discrete clusters of angular myofibers of markedly reduced dimension, existed in the left but not the right masseter muscle.
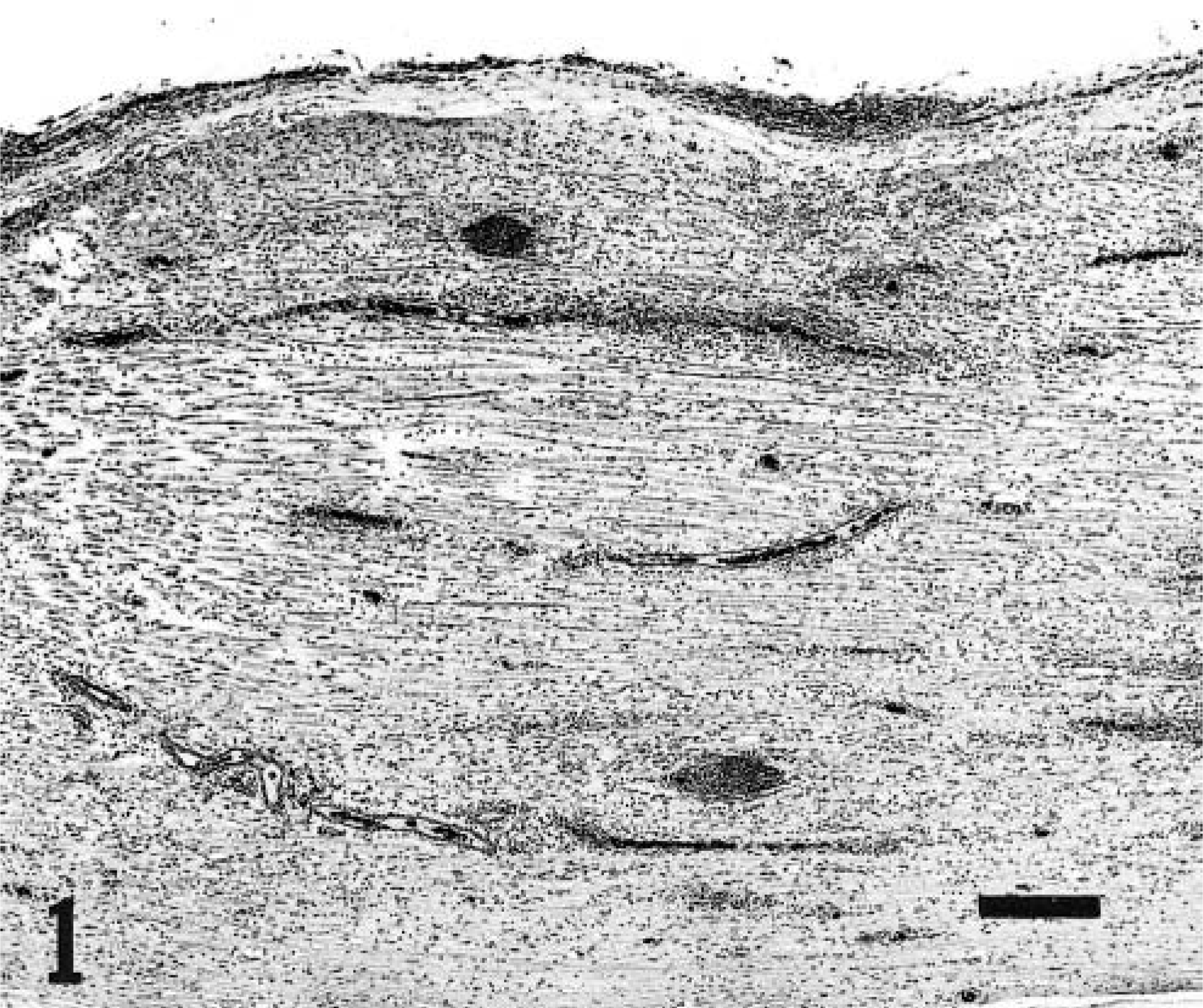
Left mandibular nerve; dog. Disseminated inflammatory cell infiltrate with intensification around blood vessels and within the epineurium. Separation of nerve fibers by edema is prominent. HE. Bar = 300 µm.
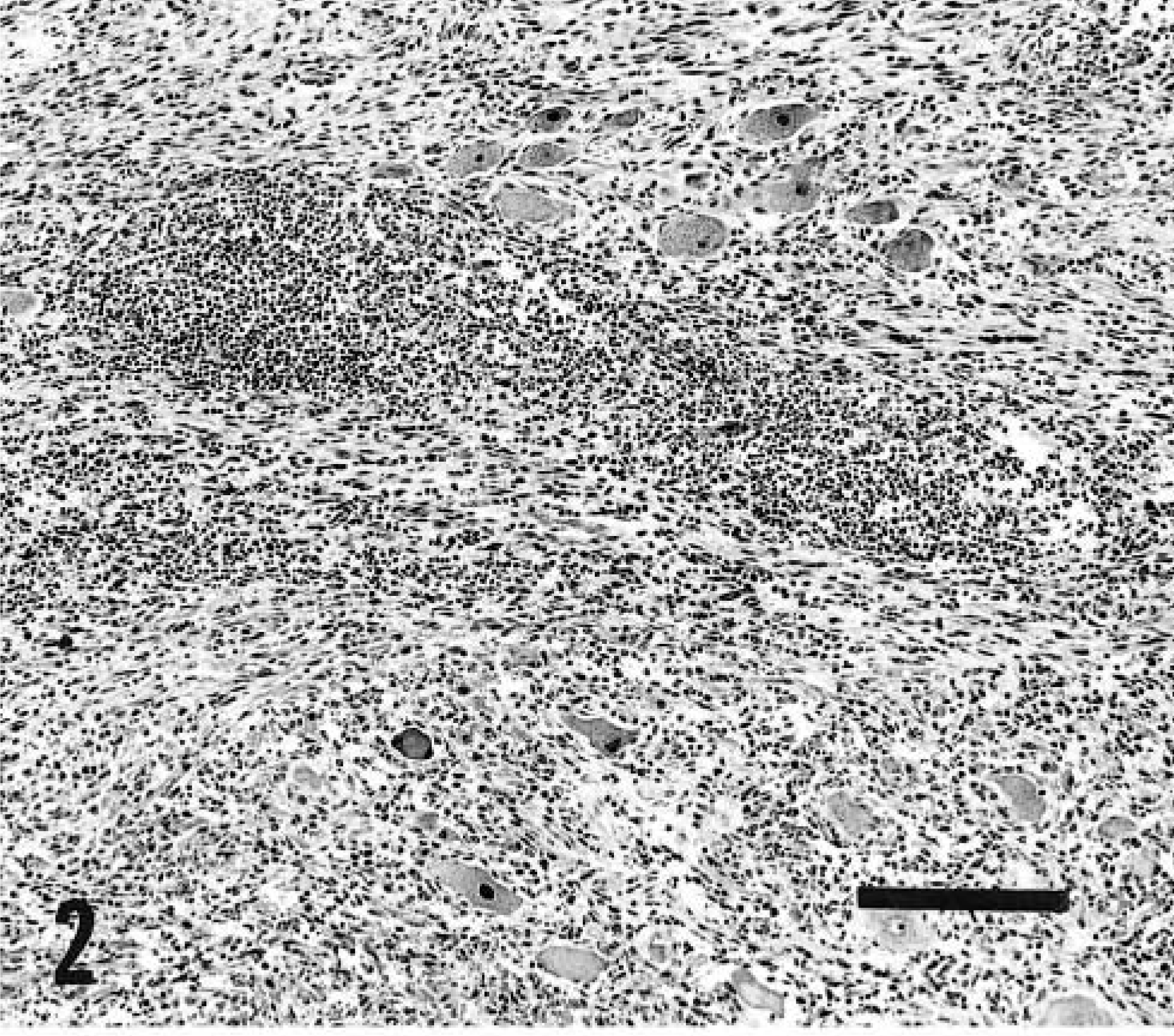
Left trigeminal ganglion; dog. Severe ganglionitis characterized by variably intense inflammatory cell infiltrate. Many ganglion cells have central chromatolysis and nuclear displacement. HE. Bar = 200 µm.
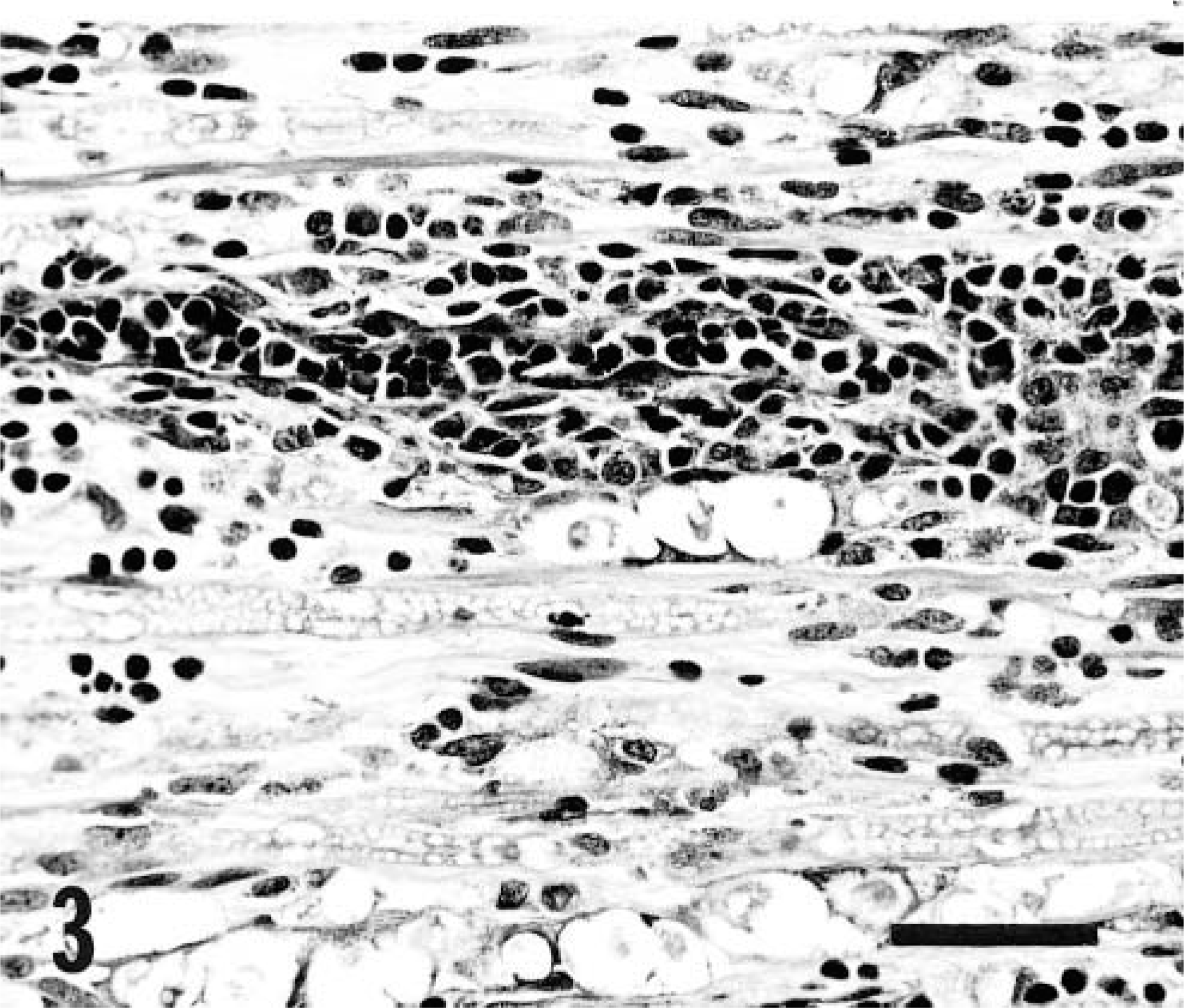
Left mandibular nerve; dog. Note digestion chambers and predominantly perivascular, lymphocytic and plasmacytic inflammatory cell infiltrate. HE. Bar = 50 µm.
In addition to trigeminal nerve and ganglion involvement, limited inflammatory foci comparable in quality to those in the trigeminal were present in spinal nerve roots at T5, T9, and S1, and in dorsal root ganglia at T3 and T8. Similar, though limited, lesions were present in both the sciatic and radial nerves. Inflammatory lesions were not observed in sections of thoracic spinal cord segments 1, 2, and 3, or in the cervicothoracic ganglion, or in either the sympathetic or vagosympathetic trunks or the cranial cervical ganglion. However, moderately numerous degenerate fibers in white matter were present at virtually all levels of the spinal cord and rootlets and in the brainstem. A small unidentified nerve trunk adjacent to the emergence of the right optic nerve from the globe contained several degenerate myelinated fibers. Ocular lesions were not observed.
To phenotypically identify the infiltrating inflammatory cell populations, tissues were immunohistochemically stained for T lymphocytes (CD3, DAKO, Carpinteria, CA), B lymphocytes (CD79a, DAKO), and macrophages (MAC387, DAKO). The majority of infiltrating mononuclear cells were immunohistochemically identified as either B lymphocytes, T lymphocytes, or macrophages. Positive and negative control sections stained as expected. Approximately equal numbers of B and T lymphocytes were found within the lesions. The T lymphocytes were rather consistently arranged as a disseminated infiltrate, whereas the B lymphocytes, although scattered throughout the inflammatory infiltrate, were frequently concentrated in cellular aggregates. MAC-positive cells were fewer in number and, like the T lymphocytes, they were disseminated throughout all regions of the inflammatory infiltrate.
The primary lesion in this dog was a disseminated, nonsuppurative ganglioradiculoneuritis that was particularly intense in the trigeminal nerves. Based on the absence of histologic lesions in upper and lower motor and preganglionic components of the sympathetic system of the eye, we conclude that the severe inflammation that involved the trigeminal ganglion and nerve resulted in both mandibular nerve deficit and dysfunction of sympathetic fibers to the left eye. We presume that Horner's syndrome occurred as a result of secondary or bystander damage to postganglionic fibers where those fibers were enmeshed in the severely inflamed trigeminal nerve and its ophthalmic branch. Regrettably, neither clinical nor histomorphologic assessment was made of sensory branches of the trigeminal.
Horner's syndrome associated with trigeminal neuritis has been reported occasionally in dogs 11 but most accounts do not specifically record association with trigeminal neuritis and we have not encountered a single original report that histopathologically documents trigeminal neuritis in association with Horner's syndrome.
The present case closely resembles in both clinical and pathologic features cases of trigeminal neuritis with Horner's syndrome mentioned by de Lahunta. 11 Carmichael and Griffiths 2 studied a case of trigeminal neuritis with loss of sensory function in the trigeminal distribution; Horner's syndrome was not recorded. Carpenter et al. 3 documented trigeminal neuropathy due to neoplastic infiltration of the trigeminal nerve that caused motor dysfunction and Horner's syndrome. Several other reports have been made of dysfunction of the mandibular branch of the trigeminal nerve leading to paralysis and atrophy of muscles of mastication. Most were based on clinical criteria without documentation as to the basis for trigeminal disease. 14,18,19
Alhough inflammation of trigeminal nerves was diffuse and intense, inflammatory changes in other neural elements were limited in both distribution and intensity. The latter lesions involved peripheral nerves, ventral roots, and dorsal root ganglia; thus, although trigeminal inflammation was the prominent pathologic feature of this case, the dog actually had a polyradiculoneuritis and ganglionitis. Immunohistochemical stains demonstrated the presence of B and T lymphocytes as predominant components of the inflammatory infiltrate. This suggests activation of both the humoral and cell-mediated arms of the immune response, which we believe were primarily responsible for the neural damage present and the resultant development of clinical signs.
The classification of the neuritides in animals and humans is incomplete; the pathogenesis of neuritides and the relationships among neuritic syndromes are not well established. 5,20 For example, the definition of the relatively well-characterized GBS in humans is based upon multiple criteria, one of which is “no other known cause.” 22 GBS (peracute to acute) and a morphologically similar syndrome, chronic inflammatory demyelinating neuropathy, are distinguished primarily by differences in their time course. 5 Evidence regarding the etiology of those syndromes for which pathogenesis has been posited rests with an immune-mediated mechanism, a response initiated by one or several environmental antigens that share epitopes with myelin or neural substances. 13 In addition to the widely distributed neuritis and well-defined lesions in the motor nucleus of the left trigeminal nerve, we interpret the degenerate fibers distributed throughout the spinal cord and caudal brain stem to represent evidence of a degenerative myelopathy unrelated to the polyradiculoneuritis that is the focus of this report.
Trigeminal neuritis was the major component of a disseminated polyradiculoganglioneuritis, clinically expressed with dysfunction of the trigeminal nerve and trigeminal-associated segments of the sympathetic innervation to the eye resulting in Horner's syndrome. The neural inflammatory infiltrate was characterized both morphologically and immunocytochemically. The character of the inflammatory response suggests that the lesions were immune-mediated and similar to that present in certain other neural inflammatory syndromes in humans and animals.
