Abstract
A conventional nonmutant animal that could be experimentally infected with Helicobacter pylori isolates would be a useful animal model for human H. pylori-associated gastritis. Gnotobiotic and barrier-born pigs are susceptible to H. pylori infection, but attempts to infect conventional pigs with this bacterium have been unsuccessful. In the present study, a litter of eight 20-day-old crossbreed piglets were purchased from a commercial farm. Six of them were orally challenged two to five times at different ages, between 29 and 49 days, with doses of H. pylori inoculum containing approximately 109 bacterial cells. Two animals served as controls. The inoculation program began 2 days postweaning when the piglets were 29 days of age. Prior to every inoculation, the piglets were fasted and pretreated with cimetidine, and prior to the first and second inoculation each piglet also was pretreated with dexamethasone. The challenged piglets were euthanasized between 36 and 76 days of age. H. pylori colonized all six inoculated piglets. The pathology of the experimentally induced gastritis was examined macroscopically and by light and electron microscopy. H. pylori induced a severe lymphocytic gastritis in the conventional piglets and reproduced the large majority of the pathologic features of the human disease. Therefore, the conventional piglet represents a promising new model for study of the various pathogenic mechanisms involved in the development of lesions of the human H. pyloriassociated gastritis.
The development of animal models is an important aspect in the study of Helicobacter pylori pathogenesis. The need for suitable animals for this purpose has been clearly stated. 18,31 To date, persistent colonization of the gastric mucosa and infection with H. pylori has been induced experimentally in gnotobiotic piglets, 32,34 barrier-born pigs, 17 nonhuman primates, 11,50 gnotobiotic dogs, 46 conventional dogs, 47 specific-pathogen-free (SPF) cats, 19 mice (SPF, germ-free, athymic, and transgenic), 22,28,29,37,40 SPF Mongolian gerbils, 24 rats, 38 and guinea pigs. 49,52
The lesions induced by H. pylori in a candidate animal model should always be examined thoroughly. The resemblance of these lesions to those observed in humans can indicate whether the usage of this model will contribute to the understanding of the various pathogenic mechanisms involved in the development of the human H. pylori-associated disease. Generally, the ideal animal model of the human H. pylori infection should reproduce the essential features of the human disease, should be widely available to investigators, should be easily manipulated (e.g., endoscopy, therapy), and should not be expensive or a species that is endangered. 31 Furthermore, the animal should have dietary habits and gastrointestinal physiologic characteristics similar to those of humans. This animal should be conventional and nonmutant, and the glandular region of its stomach should be colonized by original human isolates and not only by animal-adapted strains. Nonetheless, the oral challenge of the ideal animal model with H. pylori should result in high levels of colonization, demonstrated by convincing pictures of bacteria in the gastric mucosa. 37
The consistent experimental establishment of gastric H. pylori infection in the gnotobiotic piglet, 13,14,32,34 the induced lesions, 4,32 the demonstration of bacterial adherence in the epithelium of the gastric glandular mucosa, 48 and the fact that the piglet fulfills many of the other criteria indicate that this animal could be a very useful animal model of the human H. pylori infection. However, serious disadvantages, such as the technical difficulties of maintaining and handling the gnotobiotes and the time limitations in their use, undermine the usefulness of this animal model. 13,14,16 To date, conventional pigs have not been considered susceptible to H. pylori infection 31 ; all attempts to establish H. pylori colonization and infection in conventional pigs have been unsuccessful. 30,32,57
The present study was designed with two related aims. The first was to establish H. pylori colonization in the gastric glandular region of orally challenged conventional piglets using an original human H. pylori isolate. The second goal was to examine the pathologic findings of the experimentally induced H. pylori-associated gastritis macroscopically, and by both light and electron microscopy. The comparison of these findings with the well-known pathologic features of the human gastritis caused by H. pylori was used to evaluate the conventional piglet as an animal model for the study of the pathogenesis of H. pylori-associated diseases.
Materials and Methods
Animals
A litter of eight (five male and three female) 20-day-old crossbreed (cross between Large White and Pietrain) piglets were purchased from a commercial farm. They were divided into two groups, one group of six piglets inoculated with H. pylori and a second group of two piglets that served as controls. The two groups were housed separately in appropriate cement boxes. The housing temperature and the diet throughout the experiment were always in accordance with the standards of pig rearing. The diet did not contain antibiotics. The piglets were weaned at the age of 27 days. All animal manipulations were in conformity with the guidelines for animal care of the Faculty of Veterinary Medicine of the Aristotle University of Thessaloniki.
H. pylori strain
H. pylori strain 31A/93 used in this study had been isolated 2.5 years before from a human with pyloric ulcer and had been stored at −70 C in a storage medium prepared from a mixture of a 10% glycerol (Glycerin dopplet destilierd, Merck, Darmstadt, Germany) solution and a 4% proteose peptone No. 3 (Difco Laboratories, Detroit, MI) solution at a 4:1 ratio. The strain was highly motile, urease, catalase, and oxidase positive, and resistant to nalidixic acid.
Preparation of bacterial inoculum
The H. pylori strain was grown on blood agar plates in anaerobic jars (Merck) with gas generating kits for reduced oxygen atmospheres (Oxoid, Hampshire, England). The cultures were incubated at 37 C for 3 days. The morphology and motility of the bacteria were checked by light microscopy and the ability to produce urease was confirmed. Then the strain was subcultured. At the end of every subculture period (3 days), the plates were harvested and suspended in 1 ml of sterile saline. The density of bacterial suspensions was adjusted approximately to 109 colony-forming units/ml according to McFarland's turbidity standards. These suspensions were sedimented at 2,500 × g for 10 minutes, the supernates were decanted, and 4 ml of sterile saline were added and mixed with the bacterial pellet. A dose of 4 ml of sterile saline containing approximately 109 bacterial cells was used in every oral H. pylori challenge of the piglets.
Inoculation protocol
Piglets were deprived of any access to food and had free access only to water for 15 hours before H. pylori inoculation. On the days that the piglets were challenged for the first and second time and 2.5 hours before the inoculation, each animal received an intramuscular (IM) injection of 0.1 ml/kg dexamethasone TAD (TAD Pharmazeutisches Werk Gmbh, Cuxhaven, Germany) equivalent to 0.2 mg/kg of dexamethasone. At 1.5 hours before every inoculation of H. pylori, 60 mg/kg of cimetidine (Tagamet, Smith Kline and French Laboratories, Herdfordshire, England) was administered orally to each piglet. All steps of the procedure were applied similarly to the controls, which received 4 ml of sterile saline orally every time a bacterial inoculation was given to the treatment piglets. The six piglets were infected with H. pylori for the first time at 2 days postweaning. The timetable of H. pylori inoculation and the age of piglets at euthanasia are summarized in Table 1.
Age of piglets at time of oral H. pylori inoculations and at euthanasia.
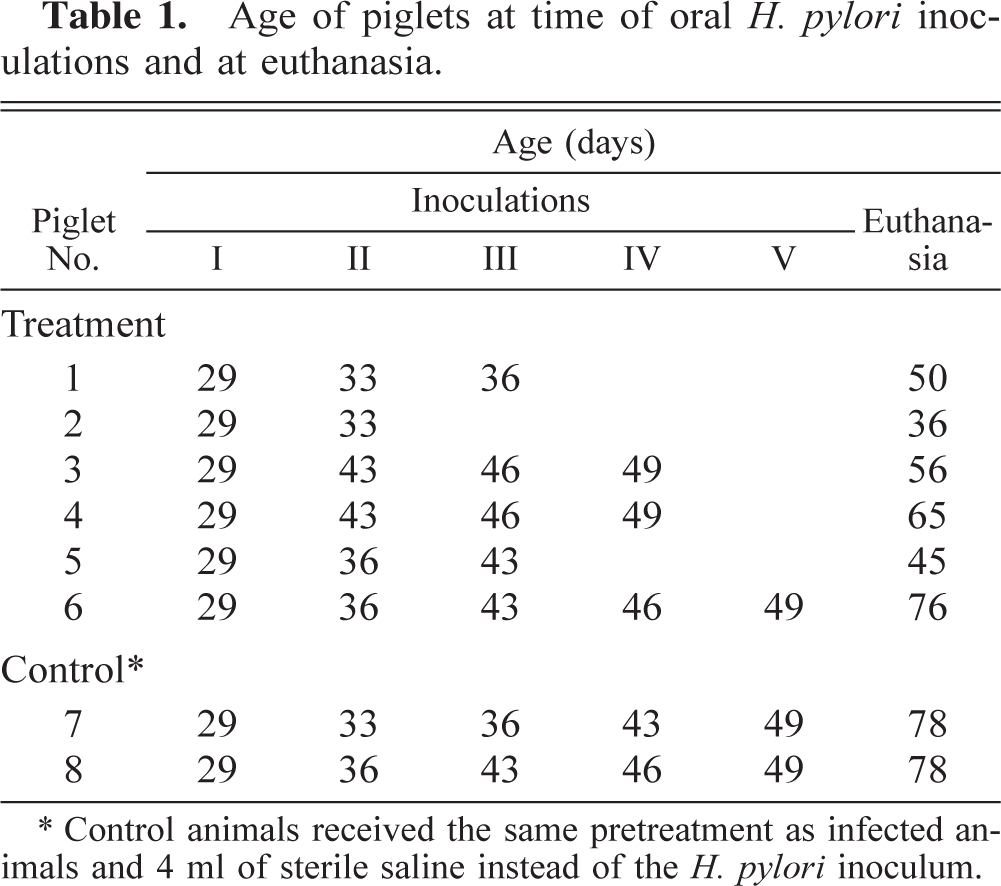
∗ Control animals received the same pretreatment as infected animals and 4 ml of sterile saline instead of the H. pylori inoculum.
Sacrifice and tissue sampling
The piglets were fasted for 12 hours before euthanasia. The animals were anesthetized with an IM injection of 1.4 mg/kg xylazine (Rompun 2%, Bayer, Leverkusen, Germany) plus 0.04 mg/kg atropine and 28 mg/kg ketamine (Imalgen 1000, Rhône Mérieux, Lyon, France). The animals were euthanatized by an intravenous injection of 100 mg/kg pentobarbital sodium (Sigma, St. Louis, MO), and their stomachs were removed, cut open along the greater curvature, emptied, and observed macroscopically. Multiple tissue samples were collected immediately from the mucosa of the cardiac, fundic, and pyloric regions for electron and light microscopic studies and microbiologic isolation of H. pylori.
Ultrastructural studies
Tissue specimens for electron microscopic examination were fixed in 2.5% glutaraldehyde solution in sodium phosphate buffer (pH 7.3) and then postfixed in 1% osmic tetroxide with veronal acetate buffer (pH 7.2). Tissues were then dehydrated in acetone and propylene oxide and embedded in Epon 812, and sections were cut using a Reichert Om U2 ultramicrotome. Sections were stained with tolouidine blue and examined, and ultrathin sections (80 nm thick) of selected areas were stained with uranyl acetate and lead citrate and examined and photographed using a Zeiss EM-9S-2 and a Jeol JEM 2000FXII electron microscope.
Light microscopic studies
Tissue specimens for light microscopy were fixed in 10% neutral buffered formalin, embedded in paraffin and cut in sequential 4–5 µm sections. These sections were stained with hematoxylin and eosin (HE) for histopathologic examination and with Warthin-Starry (WS) silver stain and peroxidase-antiperoxidase (PAP) complex immunohistochemical detection of H. pylori. The WS staining procedure was modified because highly purified liquid chromatography grade water from a D7402 EasyPure UV compact ultrapure water system (Barnstead/Thermolyme Corp., Dubuque, IA) was used instead of triple-distilled water for the preparation of the acidulated water.
Immunohistochemistry
Tissue sections were deparaffinized in xylene and rehydrated through a graded alcohol series. After proteolytic predigestion in a thrypsin (Thrypsin, Sigma) solution (0.25% thrypsin, 0.02% CaCl2) for 45 minutes, sections were treated with a 30% H2O2–methanol solution for the removal and quenching of endogenous peroxidase activity. Sections were then preincubated with normal swine serum in a 1:5 solution for 20 minutes at room temperature and incubated with the rabbit anti-H. pylori polyclonal antibody (Dako, Glostrup, Denmark) in a 1:5 solution for 45 minutes at 37 C, the link antibody (swine anti-rabbit immunoglobulins, Dako) in a 1:50 solution for 45 minutes at 37 C, and the PAP complex (rabbit PAP, Dako) in a 1:50 solution for 18 hours at 4 C. Tris-treated phosphate-buffered saline (PBS) was used for intervening washes of the sections. Visualization of the complex was accomplished with 3,3′-diaminobenzidine terahydrochloride chromogen (Sigma). Mayer's hematoxylin was used as a counterstain. Control slides were incubated in PBS in the first step instead of the first specific antibody and examined for nonspecific staining. Sections of human gastric mucosa from biopsies that had been previously diagnosed as positive for H. pylori were always included as positive controls.
Bacterial isolation
Tissue specimens for microbiologic examination were placed in 2 ml of transport medium (brain-heart infusion agar, Oxoid) and were transported within 10 minutes to the microbiologic laboratory. The gastric mucosa was scraped off the samples and homogenized. An aliquot of the homogenized suspension was spread on blood agar supplemented with H. pylori-selective supplement (Oxoid). The cultures were incubated for 3–4 days.
Results
Macroscopic lesions
With the exception of piglet No. 2, the stomachs of the remaining infected piglets (Nos. 1, 3–6) were filled with undigested food. Submucosal edema and enlargement of gastric folds were evident in all infected piglets. The gastric glandular mucosa of piglet No. 2 exhibited mild congestion, but the same region of the gastric mucosa of the remaining infected piglets exhibited severe congestion, microerosive lesions, and hemorrhages. In these five animals, multiple micronodules (lymphoid follicles) were observed in the gastric glandular mucosa, especially along the lesser curvature of the stomach. There were no grossly visible gastric lesions at necropsy in the control animals (Nos. 7, 8).
Light microscopic findings
Detection of H. pylori
The bacteria were faintly visible and practically unidentifiable by HE staining. The WS silver stain was more effective for detecting H. pylori, which was found in the cardiac, fundic, and pyloric mucosa of all infected piglets. The bacteria were easily identifiable in gastric mucosa sections by virtue of their characteristic morphology and positioning. Bacteria were S-shaped or curved, but some straightened forms were also observed. H. pylori were located in gastric mucus and were closely associated with gastric epithelium (Figs. 1–3). Although with light microscopy it was not possible to determine the nature of this close association, the positioning of the bacteria in some cases implied adhesion or even possible intracellular or intercellular localization (Figs. 1, 3). Most commonly the bacteria were found in groups of two to four bacterial cells, but foci of high bacterial density were also observed. The bacteria were distributed mainly in the surface and the upper half of gastric pit epithelium but were also found in smaller numbers deep in gastric pits and in the lumina of gastric glands. H. pylori were never found below the basal lamina of gastric epithelium. The same results were obtained by the immunohistochemical method. This method allowed a more specific identification of H. pylori. The bacteria were labeled with a characteristic brown color and were easily identifiable. Nonspecific staining was not observed. The bacterial morphology and positioning in the gastric mucosa were the same as those observed with the WS stain (Figs. 2, 3). Although the gastric mucosa of the control animals was thoroughly examined, no bacterial cells were stained by the WS stain or the PAP method.
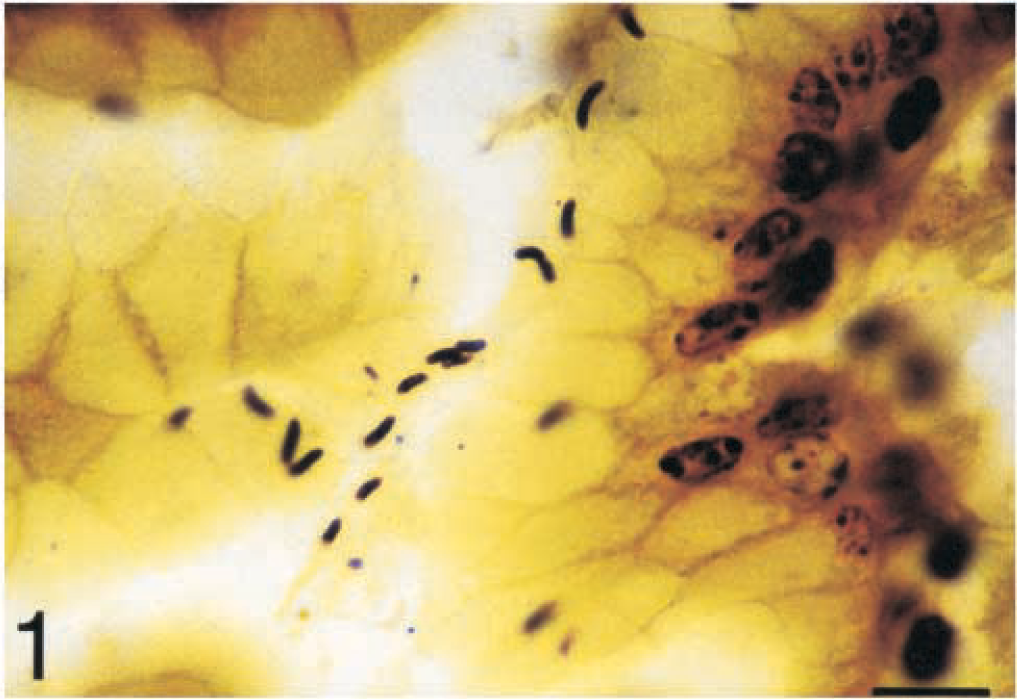
Cardiac mucosa; piglet No. 4. Characteristic appearance of H. pylori in gastric mucus and closely associated to surface epithelium. The positioning of H. pylori is suggestive of bacterial attachment to gastric epithelium and of possible intercellular or intracellular penetration. Warthin-Starry. Bar = 10 µm.
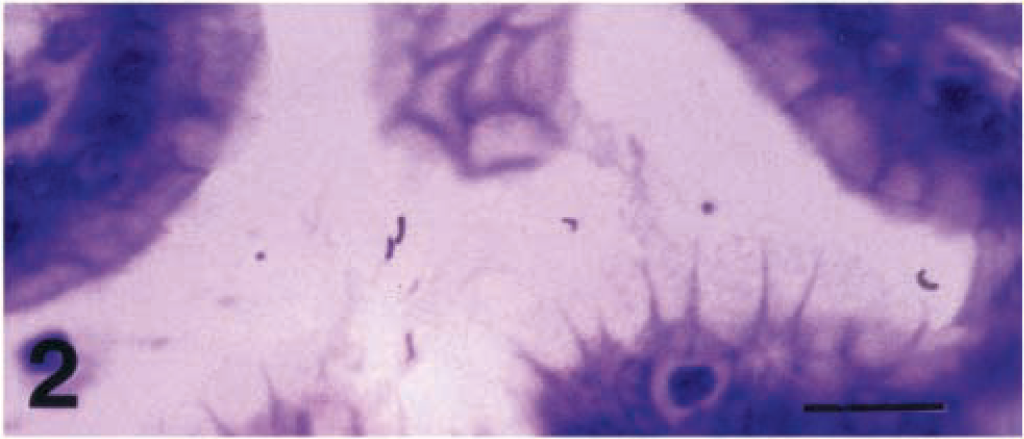
Cardiac mucosa; piglet No. 4. Immunohistochemical labeling of H. pylori. Peroxidase-antiperoxidase complex method, Mayer's hematoxylin counderstain. Bar = 16 µm.
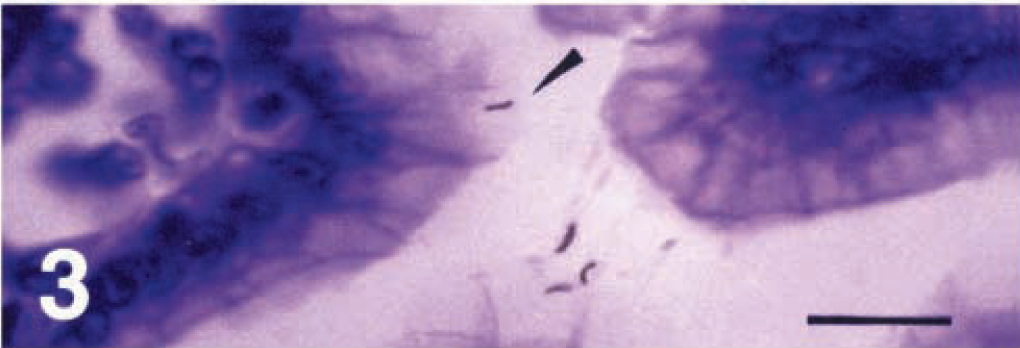
Pyloric mucosa; piglet No. 6. Possible intracellular localization of H. pylori within the cytoplasm of a gastric mucous cell (arrow). Peroxidase-antiperoxidase complex method, Mayer's hematoxylin counterstain. Bar = 16 µm.
Histopathologic changes
Microscopic lesions indicative of severe focal to diffuse H. pylori-associated gastritis were found in the cardiac, fundic, and pyloric mucosa of all six infected piglets. The submucosal edema was prominent, and the mucosal edema characterized mainly the superficial part of the mucosa. In the fundic and pyloric mucosa, severe congestion and hemorrhages were found, especially in the upper half (Figs. 4, 5).
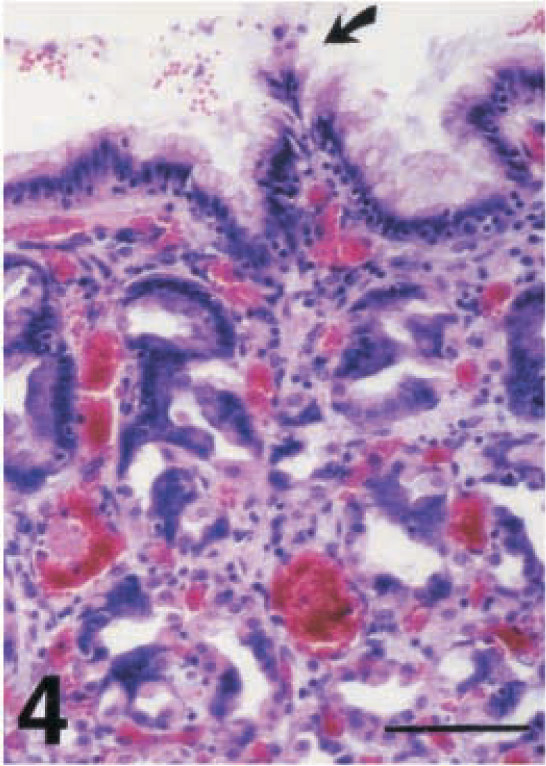
Fundic mucosa; piglet No. 4. Superficial gastritis with congestion and hemorrhages. Note the formation of the so-called cellular tuft in surface epithelium (arrow). HE. Bar = 64 µm.
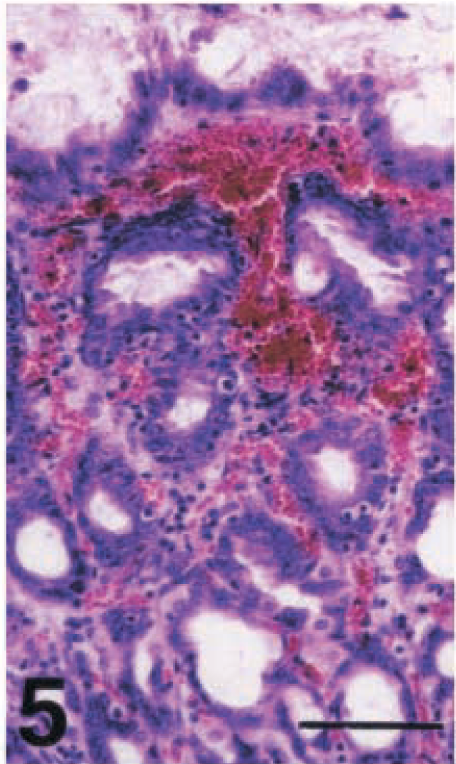
Fundic mucosa; piglet No. 5. Severe hemorrhage and congestion in the lamina propria of the upper half of gastric mucosa. HE. Bar = 64 µm.
The epithelial cells most strikingly affected by the H. pylori infection were the gastric mucous cells. The microscopic lesions observed in these cells included degeneration, necrosis, destruction and loss of their apical cytoplasmic portion, intercellular edema, and detachment from the basal lamina. The severe degeneration and loss of gastric mucous cells resulted in epithelial erosions (Fig. 6). Gastric mucous cells also showed depletion of their apical cytoplasmic portion and therefore a high nucleus:cytoplasm ratio. Thus, the gastric glandular epithelium in many cases gave the impression more of a cuboidal than a columnar epithelium. In the surface epithelium, gastric mucous cells formed cellular tufts (Fig. 4). Great numbers of exfoliated epithelial cells were detected in the gastric mucous layer. Another striking histologic feature was the significantly increased epithelial cell proliferation. The number of mitotic figures observed in the epithelial cells in many areas of the gastric mucosa exceeded 10 per 40× field (Fig. 7).
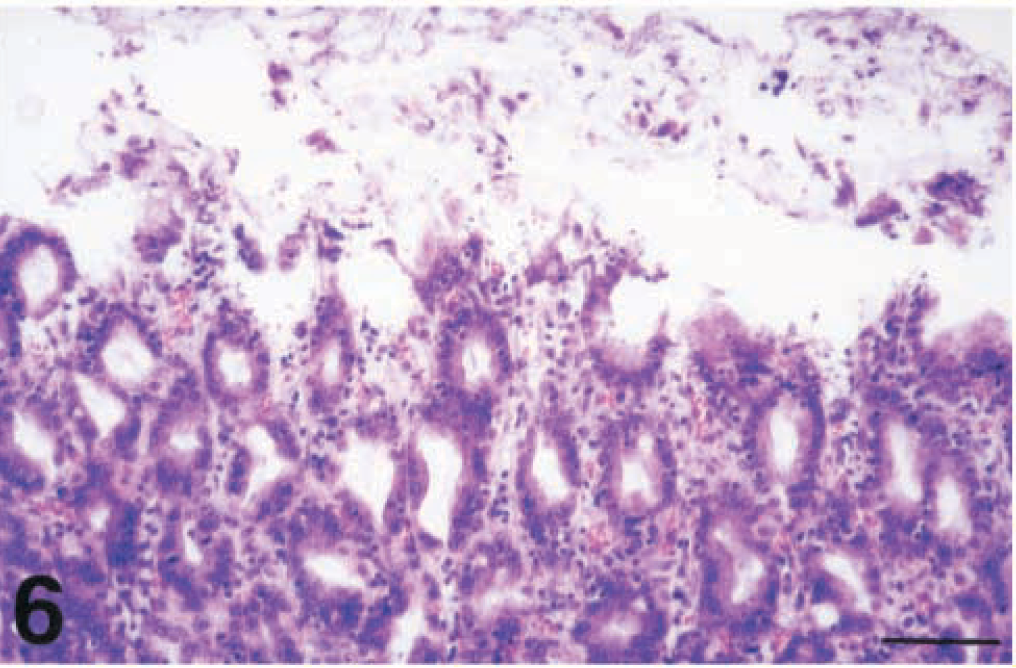
Fig. 6 Fundic mucosa; piglet No. 5. Extensive epithelial erosion. HE. Bar = 64 µm.
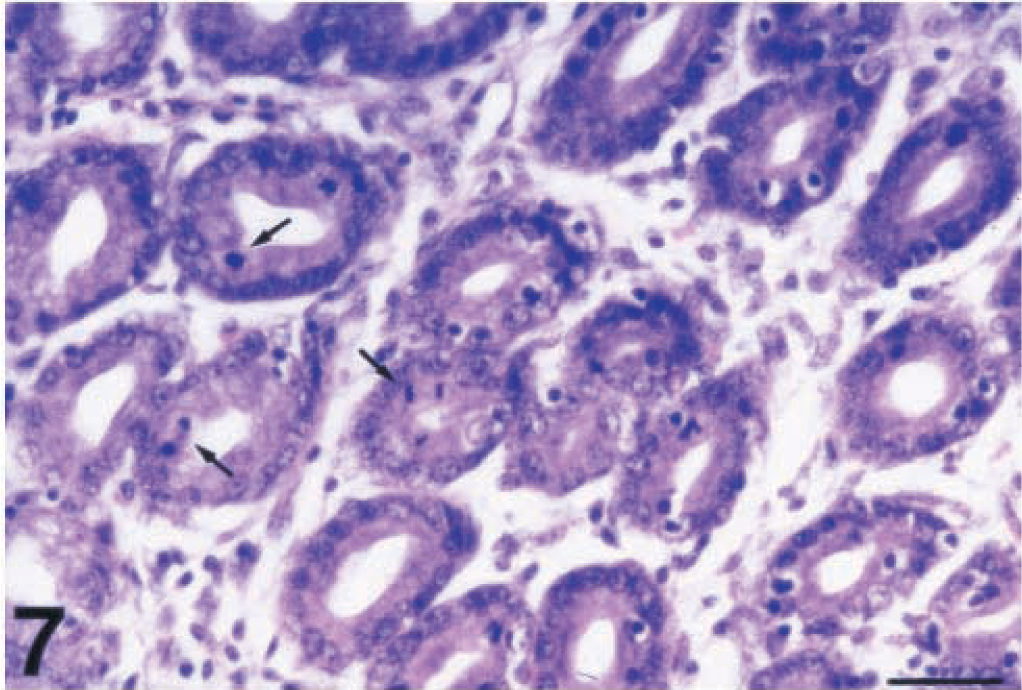
Cardiac mucosa; piglet No. 3. Markedly increased epithelial cell proliferation. A large number of mitotic figures (arrows) can be counted in the field. HE. Bar = 40 µm.
The lamina propria of the gastric glandular epithelium was severely infiltrated by lymphocytes (Fig. 8), which also insinuated extensively in the surface and gastric glands epithelium (Fig. 9). Lymphocytes tended to accumulate in small aggregates as single foci in the mucosa of piglet No. 2 and as confluent foci that matured into large lymphoid follicles in the mucosa of the remaining infected piglets (Nos. 1, 3–6). These enlarged lymphoid follicles caused the displacement and destruction of gastric glands and in many cases the destruction of surface epithelium (Fig. 10). The lymphoid hyperplasia often included both submucosa and lamina propria. The gastric mucosa-associated lymphoid tissue (MALT), which was observed mainly in tissue samples derived from areas along the lesser curvature of the stomach, was also hyperplastic (Fig. 11) in all infected piglets except piglet No. 2. Except for lymphocytes, the only other inflammatory cells found in significantly increased numbers were the mast cells. In the fundic mucosa of piglet Nos. 4 and 6, a focal replacement of the parietal and chief cells by gastric mucous cells of the antral gland type was detected at the base of individual fundic glands above the muscularis mucosa (Fig. 12).
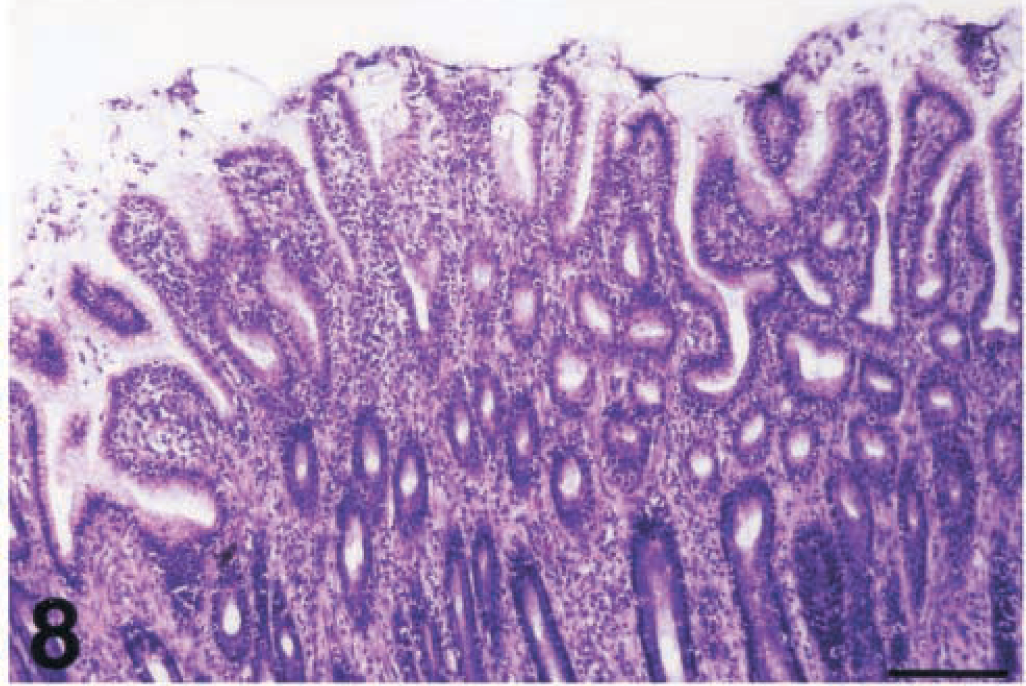
Pyloric mucosa; piglet No. 6. Severe diffuse lymphocytic infiltration of the gastric mucosa. HE. Bar = 160 µm.
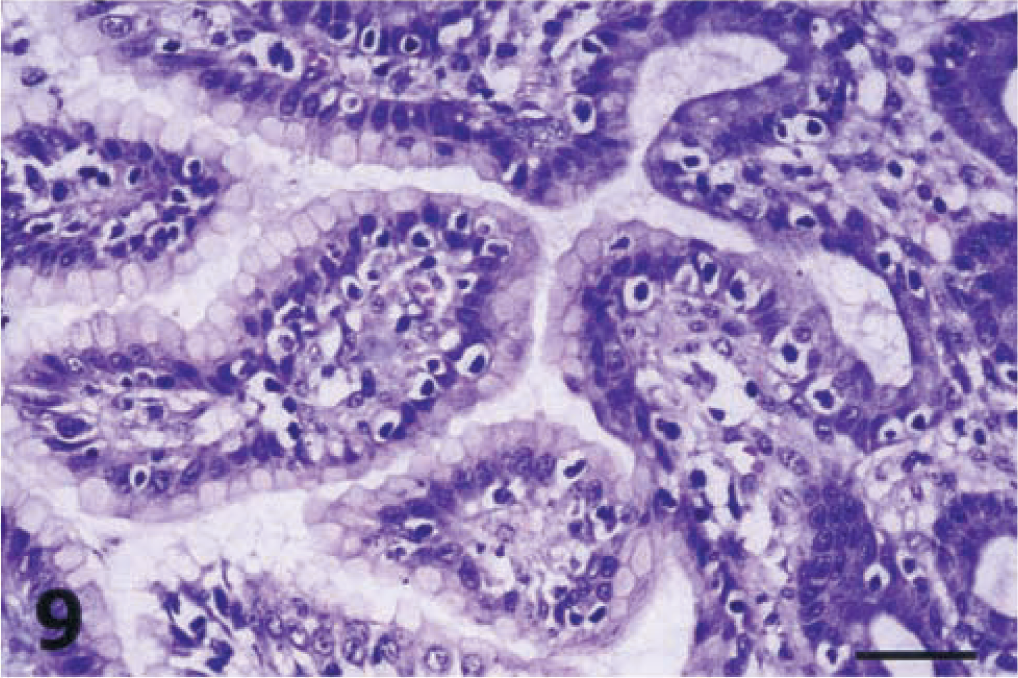
Cardiac mucosa; piglet No. 1. Marked lymphocytic infiltration of the gastric epithelium. Note the characteristic surrounding haloes of the intraepithelial lymphocytes. HE. Bar = 40 µm.
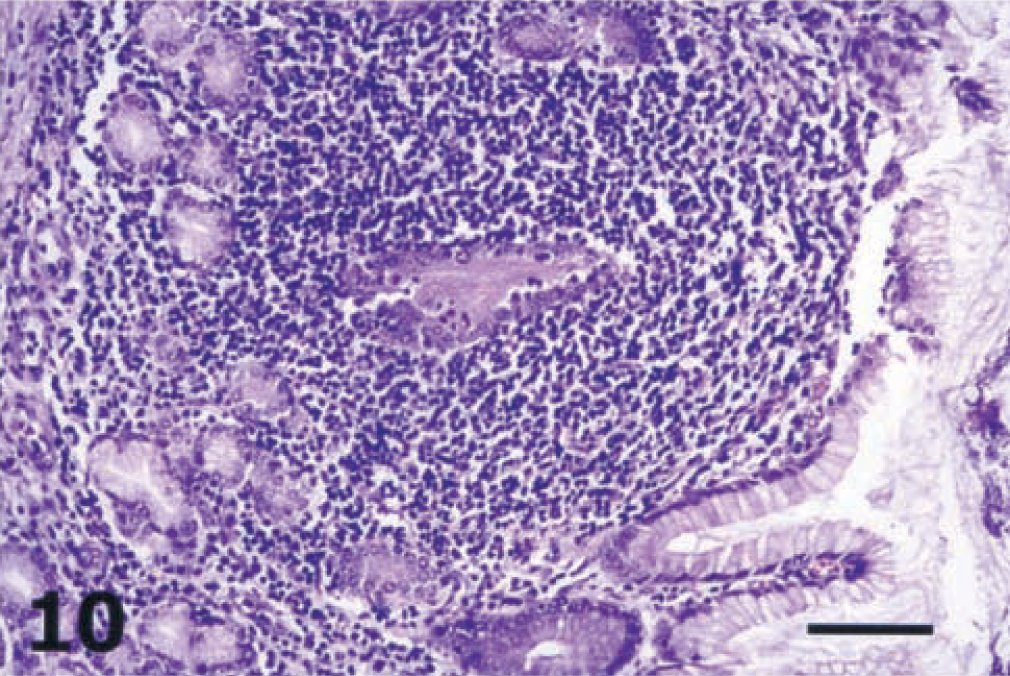
Cardiac mucosa; piglet No. 5. Large lymphocytic aggregate causing the displacement and destruction of cardiac glands in the area involved. The overlying surface epithelium is disrupted. HE. Bar = 64 µm.
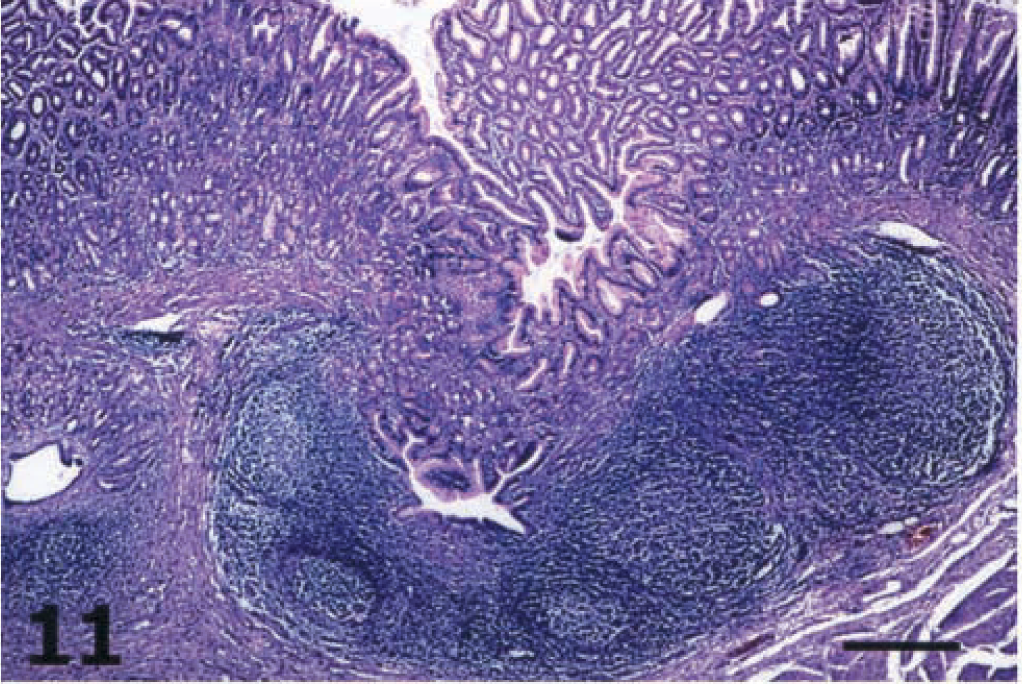
Cardiac mucosa; piglet No. 6. Hyperplasia of gastric MALT. HE. Bar = 400 µm.
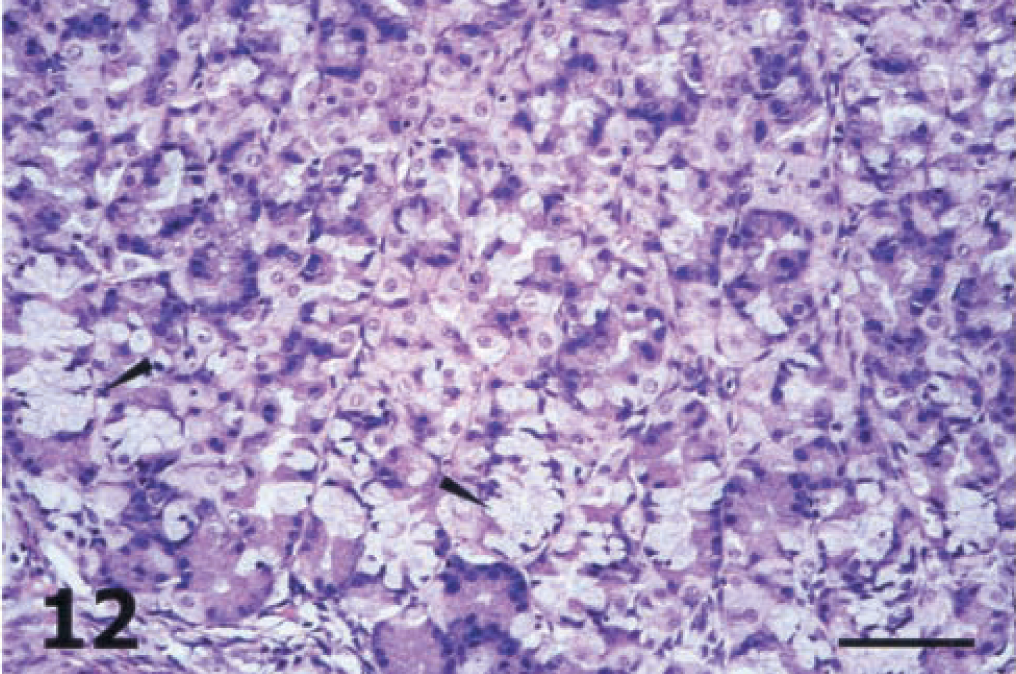
Fundic mucosa; piglet No. 6. Pseudopyloric metaplasia of the fundic glands above the muscularis mucosa. Note focal replacement of the normal epithelial cells that line the fundic glands by gastric mucous cells of the antral gland type (arrowheads). HE. Bar = 64 µm.
The histologic examination of the stomachs of the control animals revealed no abnormalities.
Electron microscopic findings
Detection of H. pylori
H. pylori was detected ultrastructurally in the stomachs of all infected piglets. The presence of these bacteria in the gastric glandular epithelium was confirmed on the basis of unique ultrastructural features. Bacteria were 2.5–3 µm long and 0.5 µm wide. The so-called double unit membrane of the bacterium was composed of the smooth outer cell wall and the inner bacterial cell membrane. The ends of the bacterium were hemispherical and did not taper. Bacterial unipolar flagella were of the sheathed type (Fig. 13). Flagella sheaths were in continuity with the unit membrane of the cell wall, and there was no terminal concavity at the origin of the flagella. In bacterial cytoplasm, pale areas containing nuclear fibrils and electron-dense structures, probably corresponding to metalloprotein complexes, were observed. Electron microscopy also revealed H. pylori cells during their conversion from bacillary to coccoid forms. Disc-shaped structures were found within the cytoplasm of one H. pylori cell. These structures might represent the oblique-sectioned discs of flagella (Fig. 14).
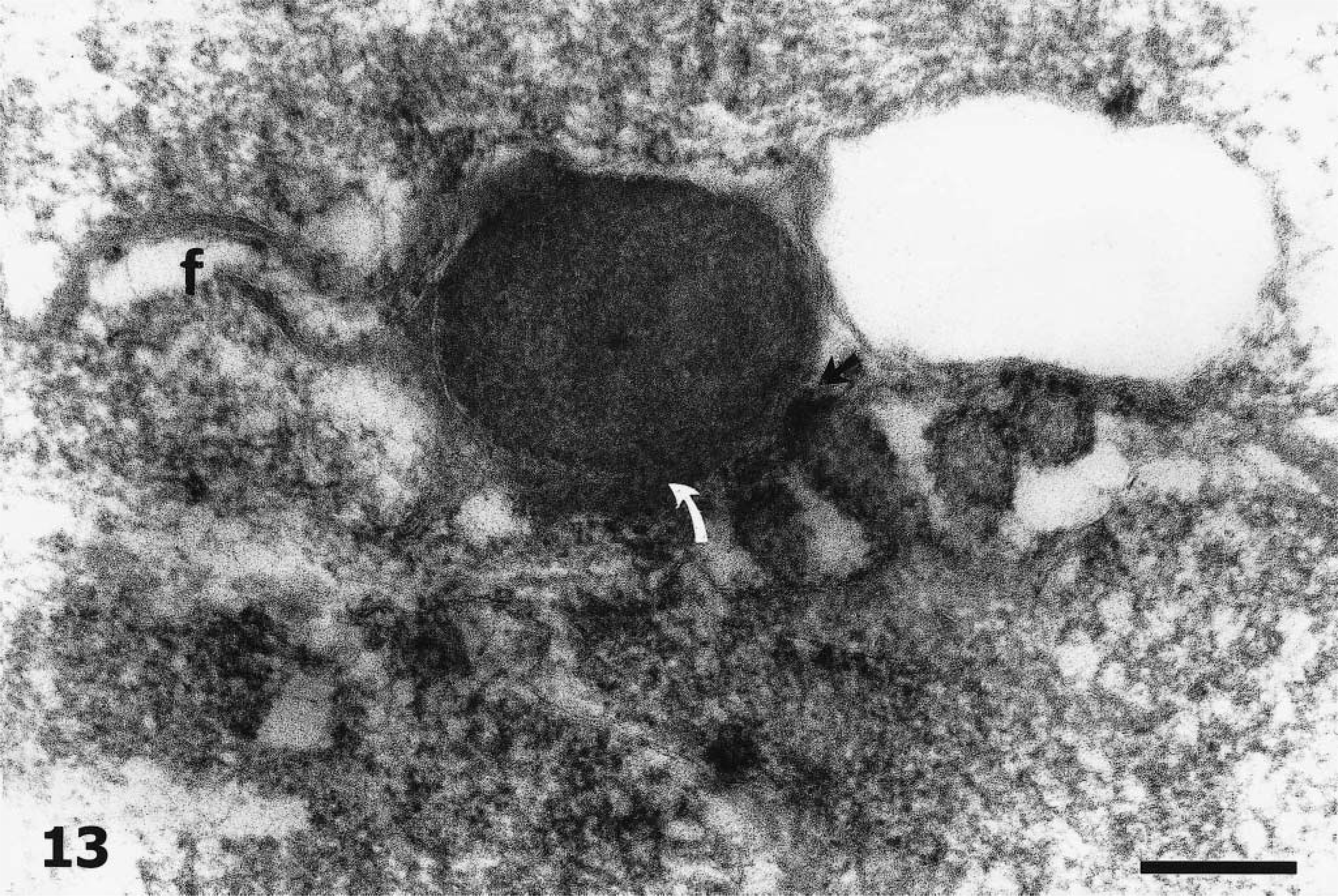
Transmission electron micrograph. Fundic mucosa; piglet No. 3. Bacterial adhesion to microvilli. At one side of intimate contact (black arrow), the bacterial cell wall seems to be fused with the gastric mucous cell membrane, and at another site (white arrow) the bacterial fibrillar adhesins are connecting the H. pylori cell with the cell plasma membrane. Note the sheathed flagella (f) of the bacterium. Bar = 200 nm.
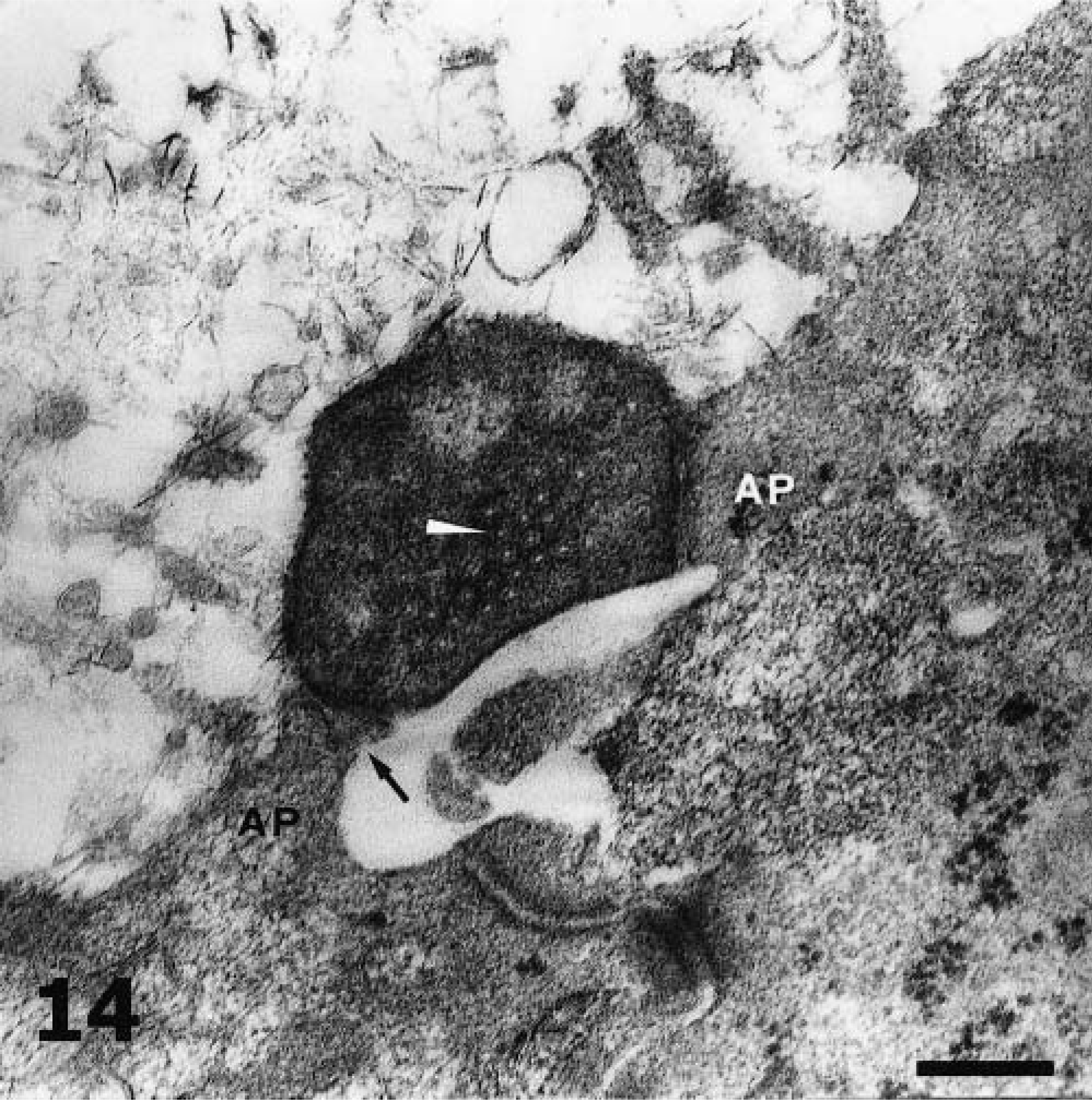
Fundic mucosa; piglet No. 5. H. pylori adhering to the plasma membrane of two adjacent gastric mucous cells. At the sites of attachment the characteristic adherence pedestals (AP) have been formed. Note the bacterial fibrillar adhesins (arrow) and the destruction of microvilli. The plasma membrane underlying the bacterium is disrupted, and the tight junction is unsealed. The disc-shaped structures within the bacterial cytoplasm (white arrowhead) might represent the oblique-sectioned basal discs of a bacterial flagellum. Bar = 200 nm.
Adherence of H. pylori
H. pylori was found adhered to the apical cell membrane of gastric mucous cells (Figs. 13, 14). At the sites of adhesion, cell membrane alterations such as adherence pedestals (Fig. 14) or cuplike invaginations were formed. The abutting type of adhesion was also observed. With this type, the contour of the epithelial membrane was unaltered at the point of contact. Sometimes, at sites of intimate contact the bacterial cell wall seemed to be fused with the cell plasma membrane (Fig. 13). The bacterial adhesins observed during our ultrastructural studies were always fibrillar in nature (Figs. 13, 14). No fibriae or pilli were found.
Ultrastructural lesions
The gastric mucous cell was indeed the main target cell of the bacterial infection. Intracellular and intercellular edema was found in these cells. A striking feature was the destruction and loss of microvilli. However, this loss was not correlated with the presence of closely associated bacteria. Gastric mucous cells also showed depletion and dissolving of mucin granules, apical membrane disruption, destruction and loss of the apical mucin-containing cytoplasmic portion, and characteristic irregularities of the luminal surface. The late alterations included indentation, clublike protrusions, and ragged flaplike protrusions. Dome-shaped bulging of gastric mucous cells was frequently observed. Endocytic vacuolation occurred but was not common.
Degenerative changes were also found in parietal cells. These changes consisted of intracellular edema, degeneration of mitochondria, and the presence of intracytoplasmic myelinic figures.
Lymphocytes were not the only inflammatory cells insinuating into gastric epithelium. Mast cells also were frequently seen within the epithelium of gastric glands.
Microbiologic studies
Although a selective medium was used for the isolation of H. pylori from gastric mucosa specimens of the infected piglets, an overgrowth of contaminants such as Bacillus and Lactobacillus occurred. Material from colonies that looked like H. pylori colonies was stained with Gram's stain, and bacillary and coccoid forms of the bacterium were observed. However, all efforts at H. pylori subculture and isolation were unsuccessful because of the persistent overgrowth of the contaminants.
Discussion
Most of the proposed animal models for H. pylori infection have been unconventional (germ free, gnotobiotic, athymic, transgenic) or conventional but infected with an animal-adapted H. pylori strain. However, the immunologic status of these unconventional animals and the mutations that the H. pylori isolate might have undergone during the animal-adaptation procedure both raise questions in terms of the reproduction of the pathogenic mechanisms of the human H. pylori associated disease. The results of the present study demonstrate that an original human H. pylori isolate instead of an animal host-adapted H. pylori strain can colonize conventional animals.
The conventional piglet is a much more practical option for most investigators than nonhuman primates and gnotobiotic, germ-free, athymic, or transgenic animals. The conventional piglet as an animal model of the human H. pylori infection offers advantages of a functional monogastric animal with gastric anatomic and physiologic characteristics similar to those of humans. 31 The large size of the piglet might be considered a disadvantage, 17 but it makes feasible the taking of repeated gastric biopsy samples by endoscopy, 17 which is not possible with other small animal models such as rodents. Repeated samples make it easy to follow the infection of an individual animal over a long period. The conventional piglets do not have the disadvantages of their gnotobiotic counterparts, such as the time limitations in their use and the difficulties of maintenance under gnotobiotic conditions. However, because of the florid gastric microbiota of the conventional pig it is difficult to isolate H. pylori following experimental infection. 18
Previous attempts to establish gastric H. pylori colonization and infection in conventional pigs have been unsuccessful, 30,32,57 and most reasearchers have concluded that these animals are not susceptible to H. pylori infection. 16,31 Therefore, we designed the present experiment to optimize the survival of H. pylori in the stomachs of piglets during the first hours postinoculation. In that way, we hoped to give the bacterium time to use its sophisticated mechanisms 35 to colonize the gastric glandular epithelium of the challenged animals. We presumed that the temporary suppression of gastric acid secretion with the oral cimetidine treatment before every H. pylori inoculation in all piglets was not sufficient because this manipulation had been used in every similar animal model experiment. Therefore, dexamethasone was also used in anti-inflammatory doses prior to the first and second H. pylori inoculation. This manipulation was used to delay the immune response of the piglets against the bacterium. However, corticosteroids promote protein synthesis in the liver; causing increased levels of circulating amino acids. Amino acids oxidize into urea, and urea tranverses the gastric epithelium via the paracellular route. 44 Given the vital role of urea for H. pylori, 12,23 the increased levels of this substance at the luminal side of gastric mucosa might have facilitated bacterial survival and colonization. Furthermore, the time of the first H. pylori inoculation was carefully selected to be the second day postweaning. At that time, lactobacilli practically disappear temporarily from the piglet's stomach. 39 These bacteria inhibit the growth of H. pylori in vitro and exhibit antagonistic activity against it in vivo. 7,27 Further studies are needed to elucidate the role that both corticosteroids and the absence of lactobacilli might have played in the establishment of H. pylori colonization in the conventional piglets. Such studies may produce novel evidence for the association of the increased corticosteroid secretion observed under stressful conditions with H. pylori-associated disease. In addition, they may add information about the preventive role of lactobacilli against H. pylori.
It has been stated recently that “none of the published H. pylori models has shown convincing pictures of bacteria in the gastric mucosa which presumably means they could not be easily observed.” 37 The results obtained from the present experiment are indicative of a high H. pylori colonization density in all infected piglets. The pictures shown illustrate the fact that the bacteria were readily observed during microscopic studies. The observation of these bacteria, especially in WS silver stain preparations, was further enhanced by the lack of artifacts. The silver precipitation, which is described as a disadvantage of this method in the detection of H. pylori, 56 was not observed probably because of the modification made to the staining protocol.
H. pylori was not isolated microbiologically from the infected piglets in the current study because of aberrant species overgrowth (mainly Lactobacillus). This result is similar to that of a previous study in H. pylori-infected gnotobiotic piglets that were transferred under conventional conditions. 14 However, the successful establishment of H. pylori colonization in all infected piglets was confirmed on the basis of characteristic morphology and positioning demonstrated by the WS stain, identification by immunohistochemistry, and characteristic ultrastructural features 33,43,56 observed by electron microscopy. Moreover, the stomachs of the control animals, which were completely normal macroscopically and histologically, were thoroughly and extensively examined and no H. pylori or Helicobacter-like bacterial cells were observed. Unlike cats 18,19 and nonhuman primates, 1,18 which may be naturally infected with H. pylori, pigs may harbor “Canditatus Helicobacter suis” (formally named Gastrospirillum suis and H. heilmannii type 1) in their stomachs. 9,18,45 However, this tightly coiled bacterium, which cannot be confused morphologically with H. pylori, was not found in the numerous histologic sections of piglet gastric mucosa examined with both light and electron microscopy. Generally, if the conventional piglet is to be used as an animal model of H. pylori infection, a thorough histologic examination of multiple gastric mucosa tissue specimens should always follow an experiment. This examination can be used to rule out the presence of “Canditatus Helicobacter suis” or to evaluate its putative contribution to the induced inflammation in animals where it is present. 45 Alternatively, the presence of this uncultured bacterium can be excluded by a 16S ribosomal DNA-based polymerase chain reaction assay that was developed recently. 9
The histopathologic and ultrastructural findings of human H. pylori gastritis have been described in numerous studies and are well known. 4,5,8,51,55 The detailed comparison of these lesions with those described in the present study reveals a remarkable similarity. The experimentally induced lesions in the gastric glandular mucosa of the infected conventional piglets are strikingly similar to those observed in humans. The only lesions not reproduced in the conventional piglet model were the neutrophilic inflammation and the intestinal metaplasia of the gastric mucosa. Although the lack of intestinal metaplasia may be attributed to the relatively short time of this experiment, the absence of the so-called active component is yet to be explained. The answer to why H. pylori induces a lymphocytic, nonactive gastritis in both piglets and children 4,41 may provide significant insights into the pathogenesis of H. pylori infection.
Gnotobiotic piglets infected with H. pylori developed a lymphocytic reaction of the cardia, and little if any inflammation was present in the fundus and antrum. 4 The lymphocytic inflammation observed in conventional piglets involved not only the cardiac region but also the fundic and pyloric regions. Neutrophils were occasionally found in the gastric glandular region, but generally their presence was unremarkable. Similarly, in H. pylori-infected barrier-born pigs, neutrophils were not a component of the gastritis induced. 17 The gnotobiotic piglet studies also showed that neutrophils are present sporadically only in low numbers, mainly during the first week postinoculation, and are located in and near the squamous rather than the glandular portion of the gastric mucosa. 13,32 However, the hypothesis that the lack of neutrophilic inflammation in the gnotobiotic piglets is due to their different immune status in comparison with conventional animals 13,32 is not supported by the results of the present study. The repeated oral inoculations of piglets with H. pylori would be expected to favor the presence of a more acute component in the gastritis, but in another study no neutrophilic infiltrates were present in H. pylori-challenged gnotobiotic piglets that had been orally immunized previously against the bacterium. 13
The severity of the lymphocytic inflammation and the insinuation of lymphocytes into the gastric epithelium were much more pronounced than those found in other animal models of H. pylori infection. Although the enlargement of the lymphoid follicles of the gastric mucosa is considered a hallmark of H. pylori infection in both humans and animals, the opinions about the hyperplasia of the gastric MALT differ. In humans, the normal gastric mucosa is devoid of organized lymphoid tissue, and several researchers have suggested that infection with H. pylori is associated with the accumulation of gastric MALT. 54 However, normal adult sows exposed to a normal microbe-rich environment possesed a well-developed gastric MALT. 21 In the present study, although there was some evidence of the presence of gastric MALT in the control animals, the hyperplasia of this specific, organized lymphoid tissue was observed only in the H. pylori-infected piglets.
Mast cells seem to play an important role in the pathogenesis of gastritis and especially in H. pylori-associated peptic ulcer. 42 Yet, little attention has been given to these inflammatory cells in all previously published animal models of H. pylori infection, with the exception of an SPF CD1 mouse model. 53 In the model described here, the number of mast cells detected in the gastric mucosa of the infected piglets was significantly increased in comparison to the controls. In addition, many intraepithelial mast cells were detected by electron microscopy.
The histopathologic finding of increased gastric epithelial cell proliferation is of special interest. The hyperproliferation of gastric epithelial cells increases the risk of the development of H. pylori gastritis to gastric adenocarcinoma, 2 and the focal displacement of the epithelial cells that line the fundic glands by gastric mucous cells of the antral gland type is an indication of incipient fundic gland metaplasia (pseudopyloric metaplasia), which is a common finding in human atrophic gastritis. 55 The formation of epithelial cellular tufts observed in these infected piglets has not been described in H. pylori-infected animals, although it has been found in humans with H. pylori-associated gastritis. 6 The cellular tufts probably represent a regenerative process, 6 but the lesion is so peculiar that it can be misinterpreted as a precancerous dysplastic change. 10
The severity of the experimentally induced gastritis in the current study seemed to progress with the length of time postinoculation. This progression explains why the gastritis was milder in piglet No. 2, which was euthanatized earlier than the others, and why lesions such as the epithelial erosions, the enlargement of lymphoid follicles, and the gastric MALT hyperplasia were not found in this animal. In addition, probably for the same reason, evidence of incipient pseudopyloric metaplasia was detected only in piglet Nos. 4 and 6.
Light microscopic results revealed a possible intracellular localization of H. pylori in the cytoplasm of gastric mucous cells in a few animals. However, the gold standard for the determination of such localization is electron microscopy. Hence, electron microscopic studies focusing in the intracellular localization of H. pylori in the gastric epithelium of the infected conventional piglets are needed, especially in light of the hypothesis that the sanctuary where this bacterium avoids antibacterial treatment is intracellular. 15
It is difficult to determine whether the disc-shaped structures detected ultrastructurally in the cytoplasm of an H. pylori cell were indeed the oblique-sectioned basal discs of the bacterial flagellum. If they were, this would be their first observation in a section; these discs have been demonstrated previously but only in negatively stained preparations of either intact or sarcosinate-treated H. pylori cells. 20,25,26,36
The ultrastructural detection of H. pylori undergoing transformation into coccoid forms indicates that the conventional piglet model may be useful for further studies that have to be done to determine whether the coccoid forms are viable or are simply a morphologic manifestation of cell death. 33 This model also may address open questions about the role of the coccoid form in the transmission of the bacterium and its resistance to antibacterial drugs. 3,6
None of the studies of animal models for H. pylori infection suggested so far have included a complete ultrastructural study of the H. pylori-induced lesions. In the present study, a thorough electron microscopic examination of the gastric glandular epithelium of the infected piglets documented complete agreement of these findings with the ultrastructural lesions observed in humans.
Furthermore, electron microscopy revealed that many H. pylori cells were adhered to gastric epithelium, whereas in other, otherwise promising, animal models (e.g., SPF Mongolian gerbils) the bacterium was rarely seen attached in gastric mucous cells. 24 This fact indicates that the conventional piglet may be an ideal model for the study of H. pylori adhesion in vivo, which is considered to be an important aspect of H. pylori pathogenesis. 22
Conventional piglets were colonized by an original human H. pylori isolate, and the H. pylori-associated experimentally induced gastritis reproduced the large majority of the pathologic features of the human H. pylori disease. The conventional piglet is a convenient and widely available animal model that could prove to be extremely valuable for long-term H. pylori-associated disease studies.
Footnotes
Acknowledgements
We thank Dr. J. G. Fox for his support, encouragement, and critical reading of the manuscript. We also thank Dr. H. Holcombe and Dr. V. P. Rao for their discussion and help during manuscript preparation.
