Abstract
An adult horse was euthanatized following a clinical diagnosis of cauda equina neuritis. Significant gross postmortem and histopathologic findings were limited to the sacral spinal cord and cauda equina. The sacral spinal cord, meninges, and spinal nerve roots were expanded and partially effaced by sclerosing granulomatous inflammation with necrosis. The lesion contained numerous nematode larvae and fewer adults with a rhabditiform esophagus having a corpus, isthmus, and valved bulb. Female nematodes were amphidelphic and didelphic with reflexed ovaries. These morphologic features confirm Halicephalobus gingivalis as a novel cause of clinical signs in this case of cauda equina neuritis.
A 14-year-old gelded American paint horse presented to the Colorado State University Veterinary Teaching Hospital for ataxia and urinary incontinence. Ten days prior to presentation, the animal was vaccinated for Equine Herpes Virus-1 (EHV-1). Physical examination findings included normal vital signs and mentation, a flaccid tail, urinary incontinence, and severe hind-limb proprioceptive deficits. Palpation revealed poor rectal tone with impaction of feces. The bladder was atonic and distended and was manually expressed without difficulty. Hematology, serum chemistry, and urinalysis were within normal limits with the exception of hyperglobulinemia (6.5; normal 2.4–3.5 g/dl), elevated creatine kinase (658; normal 102–442 IU/liter), and mild elevations of creatinine (2.2, 2.3, 2.1; normal 1.1–2.0 mg/dl). Cerebral spinal fluid (CSF) had a white blood cell count of 60 cells/µl (normal 0–8/µl) and markedly elevated protein (710; normal 20–80 mg/dl). Titers of serum and CSF for EHV-1 measured 1:256 and 1:16, respectively. A clinical diagnosis of cauda equina neuritis was made. The horse became recumbent over the next 48 hours and was euthanatized upon the owner's request.
At postmortem examination the significant gross lesions were limited to the sacral spinal cord, spinal rootlets, and cauda equina (Figs. 1–4). The spinal cord was expanded by an approximately 3.5- × 3- × 18- cm cylindrical, tan, lobulated mass involving the nerve rootlets, spinal meninges, and sacral spinal cord (Fig. 1). There was no evidence of nasal discharge or granulomatous inflammation within the oral cavity. The maxillary sinuses were not examined; however, the maxilla, mandibles, and gingiva were symmetrical and unremarkable on external examination. Tissues were fixed in 10% buffered formalin for >48 hours. Sections of brain, spinal cord, kidney, lung, heart, adrenal gland, intestine, stomach, and liver were embedded in paraffin, sectioned at 5 µm, stained with hematoxylin and eosin, and examined.
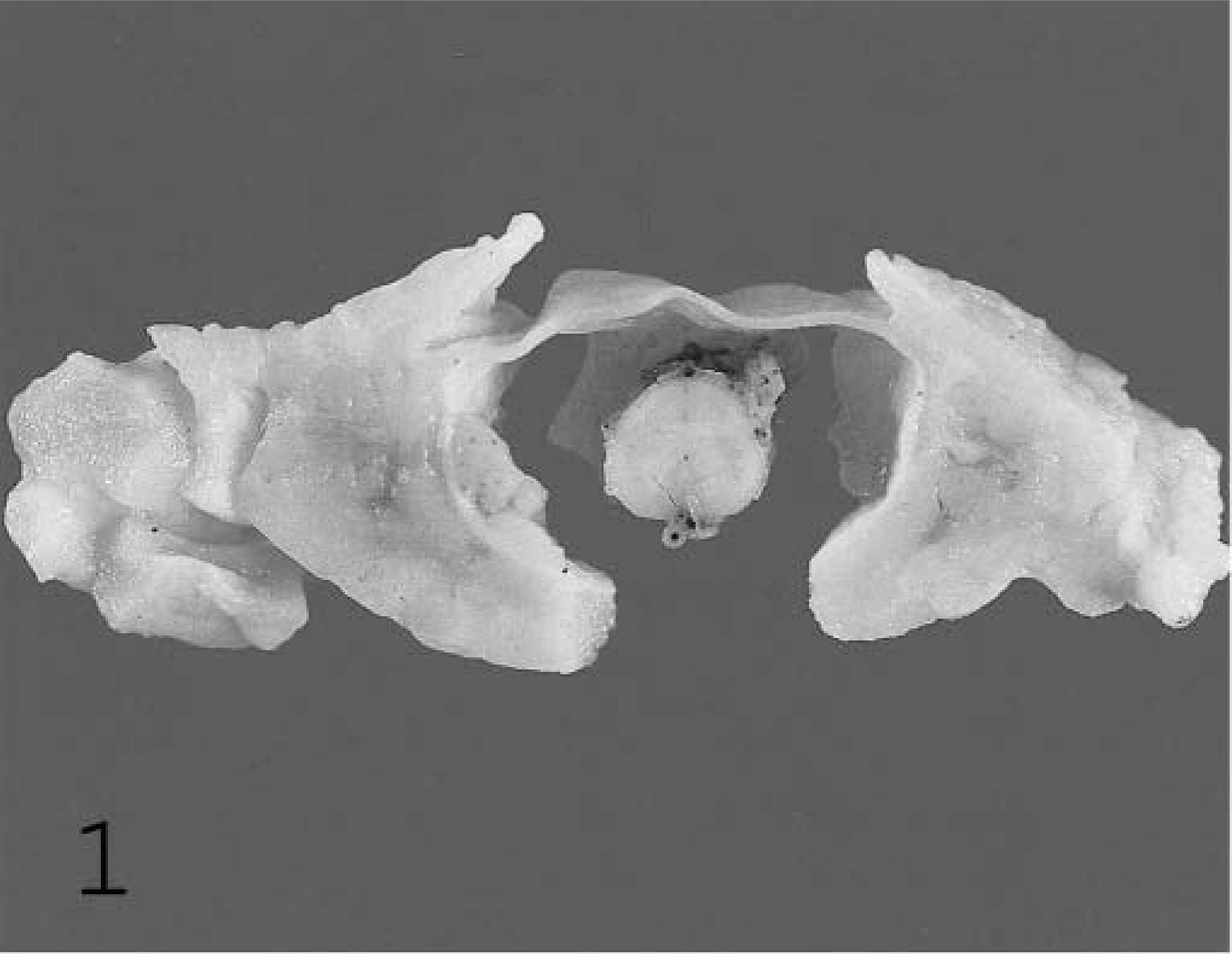
Sacral spinal cord; equine. Expansile and destructive mass encompassing spinal cord, meninges, and spinal nerve roots. Formalin-fixed tissue.
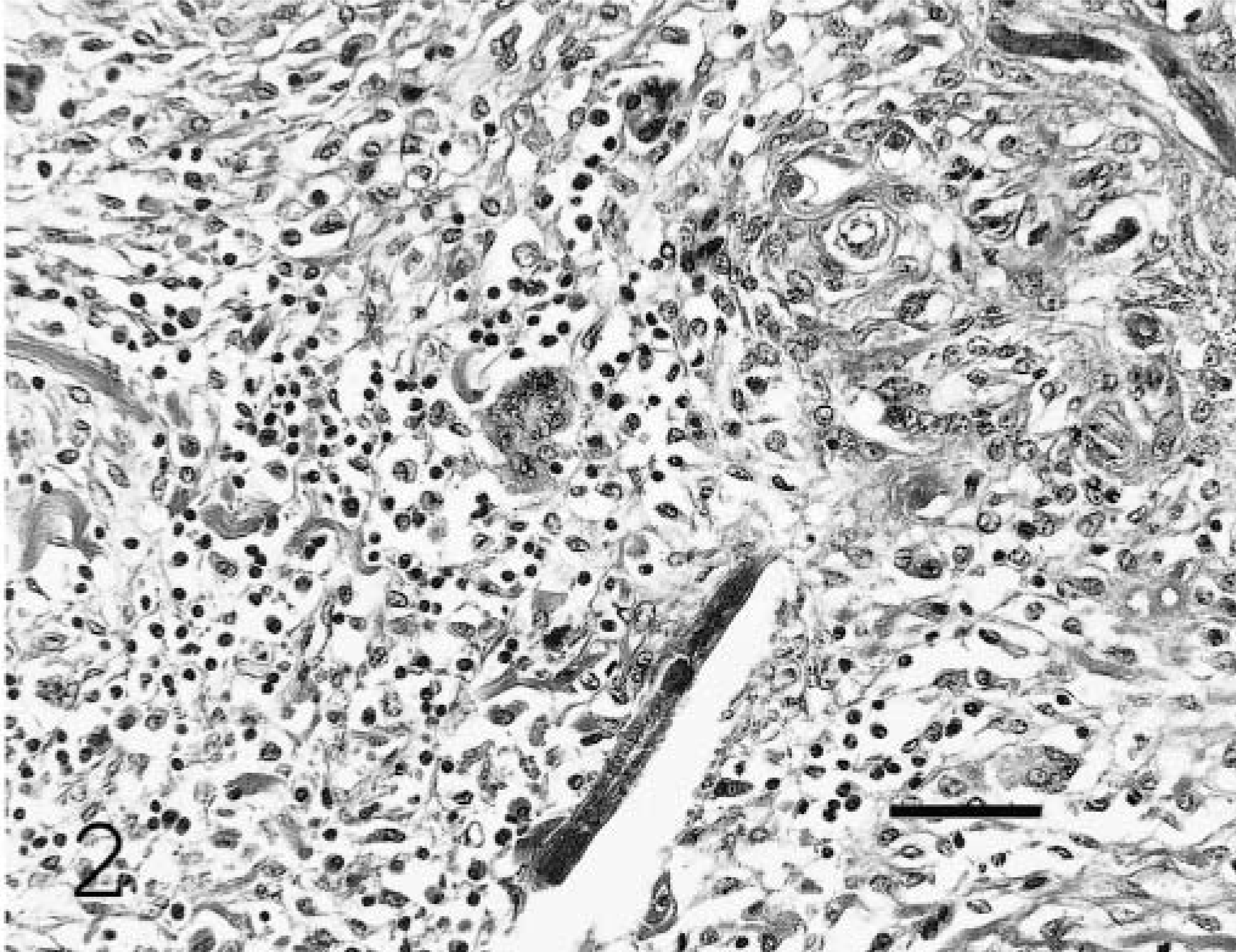
Sacral spinal nerve root; equine. Nematodes admixed with macrophages, lymphocytes, plasma cells, and multinucleated giant cells. HE. Bar = 80 µm.
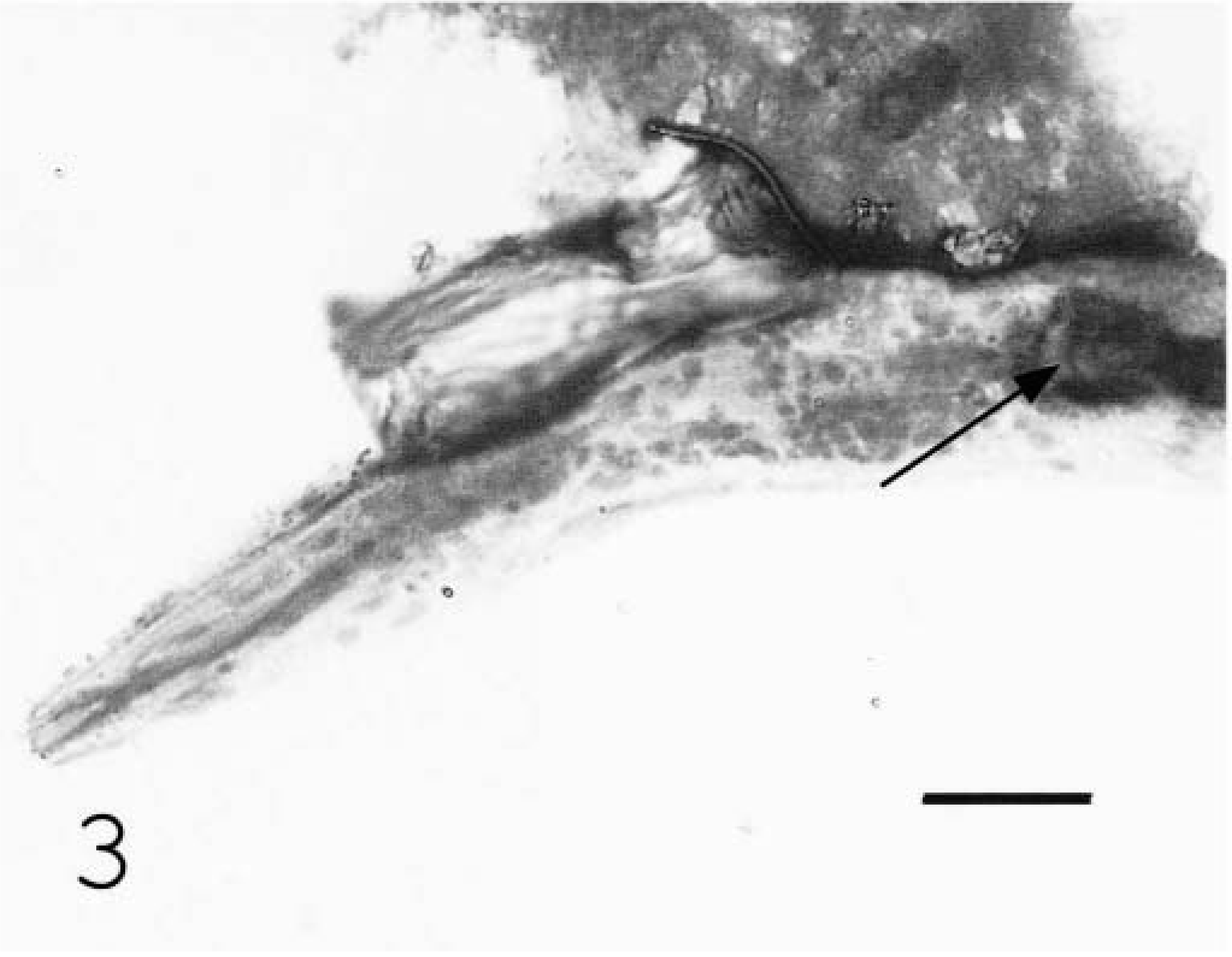
Cytocentrifuge preparation; equine. Nematode larvae with valve in anterior portion of bulb (arrow). Wright-Giemsa. Bar = 15 µm.
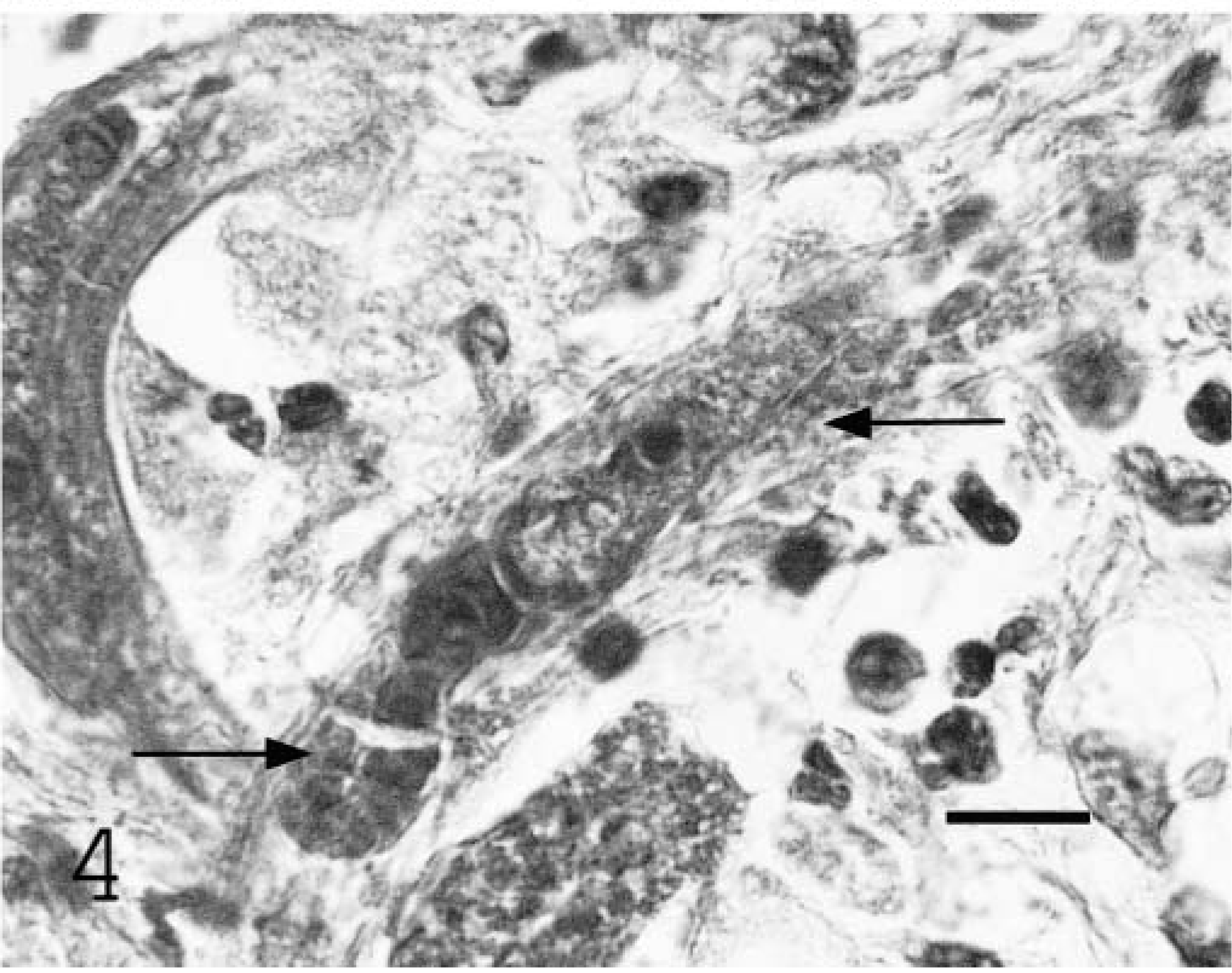
Cauda equina; equine. Female nematode with reflexed ovary (arrows). HE. Bar = 20 µm.
Histologically, inflammatory and necrotizing changes affected the spinal cord parenchyma adjacent to the ventral paramedian sulcus, spinal nerve roots and cauda equina, meninges, and perineurium, resulting in Wallerian degeneration and fiber loss. These lesions contained multinucleated giant cells, macrophages, lymphocytes, plasma cells, and eosinophils with fibrosis, dystrophic mineralization, and intralesional nematodes. Numerous nematode larvae and fewer adult forms were present. All organisms identified were female. Nematodes had a rhabditiform esophagus with a corpus, isthmus, and bulb. Adult females were didelphic and amphidelphic with reflexed ovaries that curved ventrally and dorsally at the posterior and anterior aspects, respectively (Fig. 4). Organisms were 15–20 µm in diameter and 100–300 µm in length.
Nematodes were recovered from formalin-fixed tissue to better characterize morphologic features. Two-centimeter- long sections of formalin-fixed sacral spinal cord were digested in 5 ml hydrochloric acid, 5 g pepsin, and 500 ml H2O for 48 hours at room temperature. Approximately 10 ml of fluid was collected after mixing and centrifuged at 120 × g for 2 minutes. Sixty- milliliters of the sediment was then collected and cytocentrifuged onto a glass slide at 100 × g for 5 minutes. Slides were airdried, stained with Wright-Giemsa, and examined. Recovered larvae had an anterior valve in the bulb portion of the esophagus (Fig. 3). Morphologic features lead to identification of the parasite as Halicephalobus gingivalis. 1
Rhabditiform nematodes were the only agent identified in histologic sections. Hyperglobulinemia was supportive of chronic inflammation; elevated creatine kinase levels were interpreted as muscle injury as a result of recumbency; and mild elevations in creatinine, with the absence of dehydration, were considered the result of postrenal obstruction due to lower motor neuron disease as a result of cauda equina neuritis. Elevated EHV-1 titers in serum and CSF were interpreted as a postvaccinal response with compromise of the blood–central nervous system barrier due to cauda equina neuritis. Polymerase chain reaction for EHV-1 and EHV-4 in serum, CSF, liver, and lung resulted in no amplification product. Because EHV was eliminated as a potential cause of clinical signs and no other agents were identified, the clinical signs, localizable to the sacral spinal cord, were due to H. gingivalis.
Halicephalobus gingivalis (formerly Micronema deletrix) is a small nematode of the Order Rhabditata and Family Rhabditidae. Most species inhabit decaying organic matter and are common in soil, foul water, and decaying humus. 3 Halicephalobus gingivalis has been reported to invade tissues and cause disease in horses and man 6,7 but has yet to be identified outside the host 2 and therefore is considered a facultative parasite of horses and humans. 11 Tissues from previous reports have identified female adults, larvae, and ova but not male organisms, 2,5,8,11 supporting an asexual cycle where only parthenogenetic females exist and a hypothesized sexual cycle with free-living male and female worms. 2
Common postmortem findings in cases of H. gingivalis in horses are typified by proliferative, firm, gray-white, granulomatous lesions that may involve the brain, kidneys, oral and nasal cavities, lymph nodes, lungs, spinal cord, and adrenal gland. 11 Infrequent sites of granuloma formation in horses with H. gingivalis include the heart, liver, stomach, ganglion, bone, eye, testicle, and prepuce. 5,8,9,11 Histologic findings may include replacement of normal tissue architecture by dense collagen and fibroblasts with infiltration of tissue by lymphocytes, plasma cells, epithelioid macrophages, multinucleate giant cells, eosinophils, and intralesional adult and larval nematodes.
The pathogenesis, lifecycle, and route of infection are poorly understood. Potential routes of invasion include entrance through manure-contaminated cutaneous wounds 6,7,10 or via ingestion or inhalation. 2 Organisms likely disseminate hematogenously based on identification of intravascular larvae 12 and perivascular inflammation. The unique distribution of lesions in this case produced clinical signs and gross spinal cord lesions similar to the syndrome cauda equina neuritis. 4 This report confirms Halicephalobus gingivalis as a novel cause of cauda equina neuritis.
Footnotes
Acknowledgements
We thank Glenda Taton-Allen (Veterinary Diagnostic laboratory, Colorado State University) for technical assistance in recovering organisms from formalin fixed tissues.
