Abstract
Morphologic and histochemical characteristics were noted for three spontaneous tumors with eosinophilic cytoplasmic granules that occurred in aged Fischer 344 rats. Macroscopic lesions were widely distributed in the body, mainly involving the intra-abdominal adipose tissue, pancreas, and mesenterium. These lesions were generally hard swellings with nodular and sclerosing areas. Bloody ascites was a concomitant finding. Histologically, the tumor cells were round, from 9 to 30 μm in diameter with one or two round to oval nuclei, and characterized by eosinophilic granules (0.5–2.0 μm) that stained definitely to weakly positive with the periodic acid-Schiff reaction and demonstrated no metachromasia with toluidine blue stain. Furthermore, the granules were characterized by a positive reaction with lectin histochemistry for concanavalin A (Con A), wheat germ agglutinin (WGA), phaseolus vulgaris agglutinin (PHA-E4), lens culinaris agglutinin (LCA), and recinus communis agglutinin (RCA-I) in all tumors and for ulex europaeus agglutinin (UEA-I), peanut agglutinin (PNA), and soybean agglutinin (SBA) in one tumor. Positive reactions for anti-rat mast cell protease II and CD8 were not demonstrated immunohistochemically. Abundant glycogen was noted in the large tumor cells from one rat. With electron microscopy, the cytoplasmic granules were identified as electron-dense homogenous bodies bounded by a single unit membrane. These characteristics are similar to those of granulated metrial gland cells, but further study is needed to clarify the cell of origin for these tumors.
Neoplasms derived from cells containing cytoplasmic eosinophilic granules are extremely rare. Since the first feline case described in 1972, 3 several similar feline cases have been reported and are referred to as globule leukocyte (GL) tumors, 3,5,12 granulated round cell tumors, 11 or large granular lymphocytic (LGL) lymphomas. 4,9 Recently, the first case in rats was reported, 13 but its features and derivation were not fully discussed. A database search of long-term studies conducted at our laboratories since 1990 revealed three similar cases in Fischer 344 (F344) rats. The purpose of the present report is to provide a more detailed description of the morphologic and histochemical characteristics of the tumors, with comparison to those of GLs, mucosal mast (MM) cells, and granulated metrial gland (GMG) cells for determination of cell origin.
All animals studied were F344 rats used in different long-term studies. Animals were found dead or were euthanatized in moribund condition at 58, 95, or 97 weeks of age (Table 1). After complete necropsy, all tissues were fixed in 10% neutral buffered formalin and embedded in paraffin. Sections were stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), phosphotungstic acid and hematoxylin (PTAH), toluidine blue (TB), and Giemsa. Immunohistochemistry for rat mast cell protease II (RMCPII, Moredun Scientific, Scotland) and CD8 (Serotec, England) and lectin immunohistochemistry for concanavalin A (Con A), wheat germ agglutinin (WGA), phaseolus vulgaris agglutinin (PHA-E4), lens culinaris agglutinin (LCA), ulex europaeus agglutinin (UEA-I), peanut agglutinin (PNA), dolichos biflorus agglutinin (DBA), soybean agglutinin (SBA) and recinus communis agglutinin (RCA-I) (Honen, Japan) were evaluated using an avidin–biotin–peroxidase complex method. Small pieces of tumor tissue originally fixed in formalin were processed for electron microscopy. Smears of blood and ascites from rat Nos. 3 and 1, respectively, were stained with Giemsa stain. Hematologic examination was also performed for rat No. 3. For comparison purposes, GLs and MM cells from the intestinal mucosa and GMG cells from the uterus of the pregnant rat were examined.
Profiles and gross findings in three F344 rats with spontaneous eosinophilic granulated tumors.
No characteristic clinical signs were observed in any of the rats, except for gradual decreases in body weight for 2 weeks before death.
Macroscopic findings are summarized in Table 1. The most prominent changes were observed in the abdominal cavity, mainly involving the adipose tissue, pancreas, and mesenterium, in which there were many nodular and sclerosing areas. Bloody ascites was a concomitant finding in all rats, and similar fluid was also observed in the thoracic cavity in rat Nos. 2 and 3. Although the macroscopic lesions were widely distributed in the body, no lymph-splenomegaly was noted.
The tumor cells infiltrated into the various tissues, markedly involving the abdominal adipose tissue, pancreas (Fig. 1), and mesenterium (Table 2). Numerous tumor cells were present in the smears of ascites from rat No. 1, and a few were observed in the hepatic sinusoids and/or pulmonary capillaries and bone marrow in rat Nos. 1 and 3 but not in the blood smears from rat No. 3. Erythrophagia was occasionally seen in all rats.
Distribution∗ of tumor cells in rats.
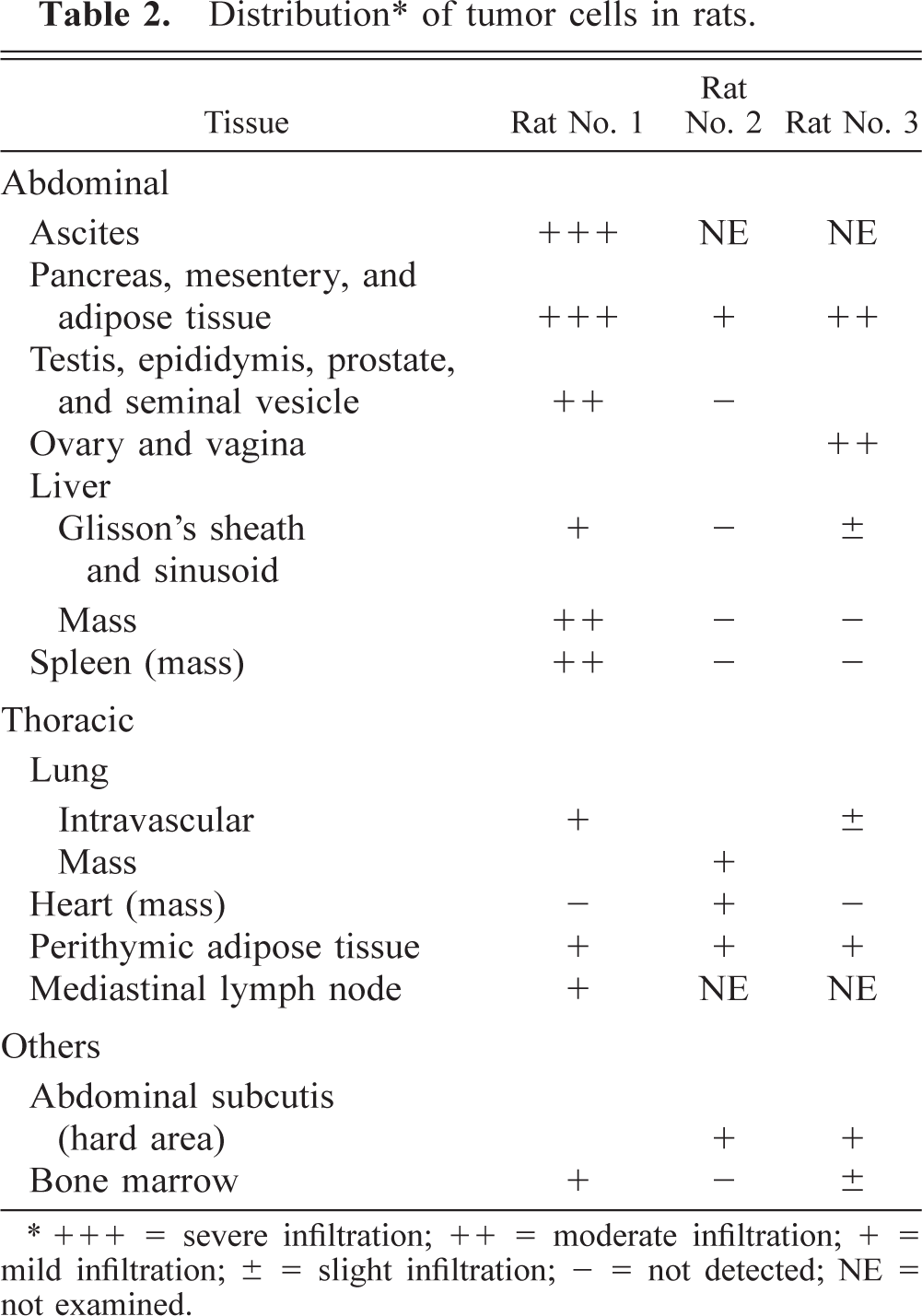
+++ = severe infiltration; ++ = moderate infiltration; + = mild infiltration; ± = slight infiltration; - = not detected; NE = not examined.
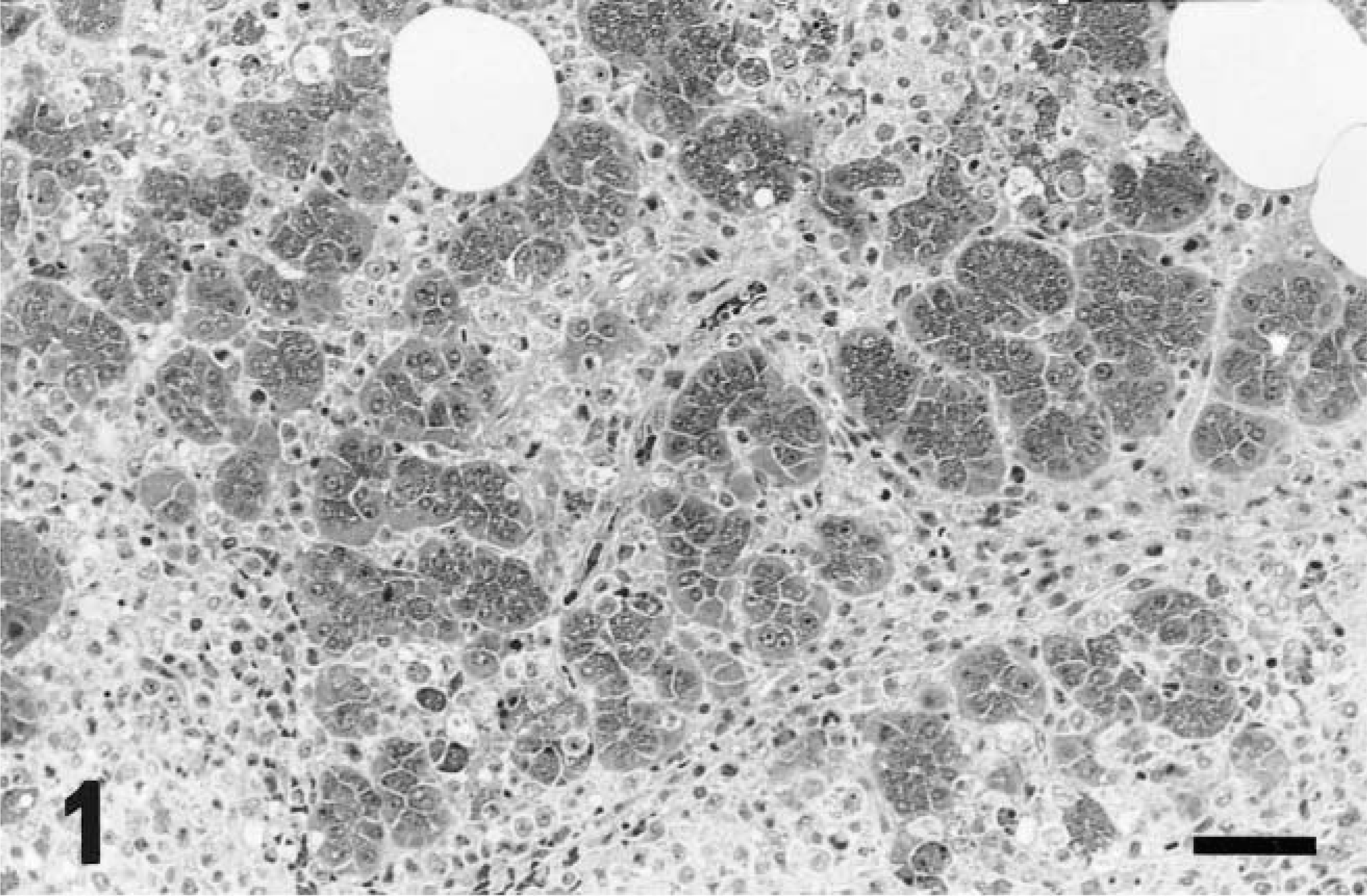
Pancreas; rat No. 1. Extensive infiltration of tumor cells in the interstitium. HE. Bar = 53 µm.
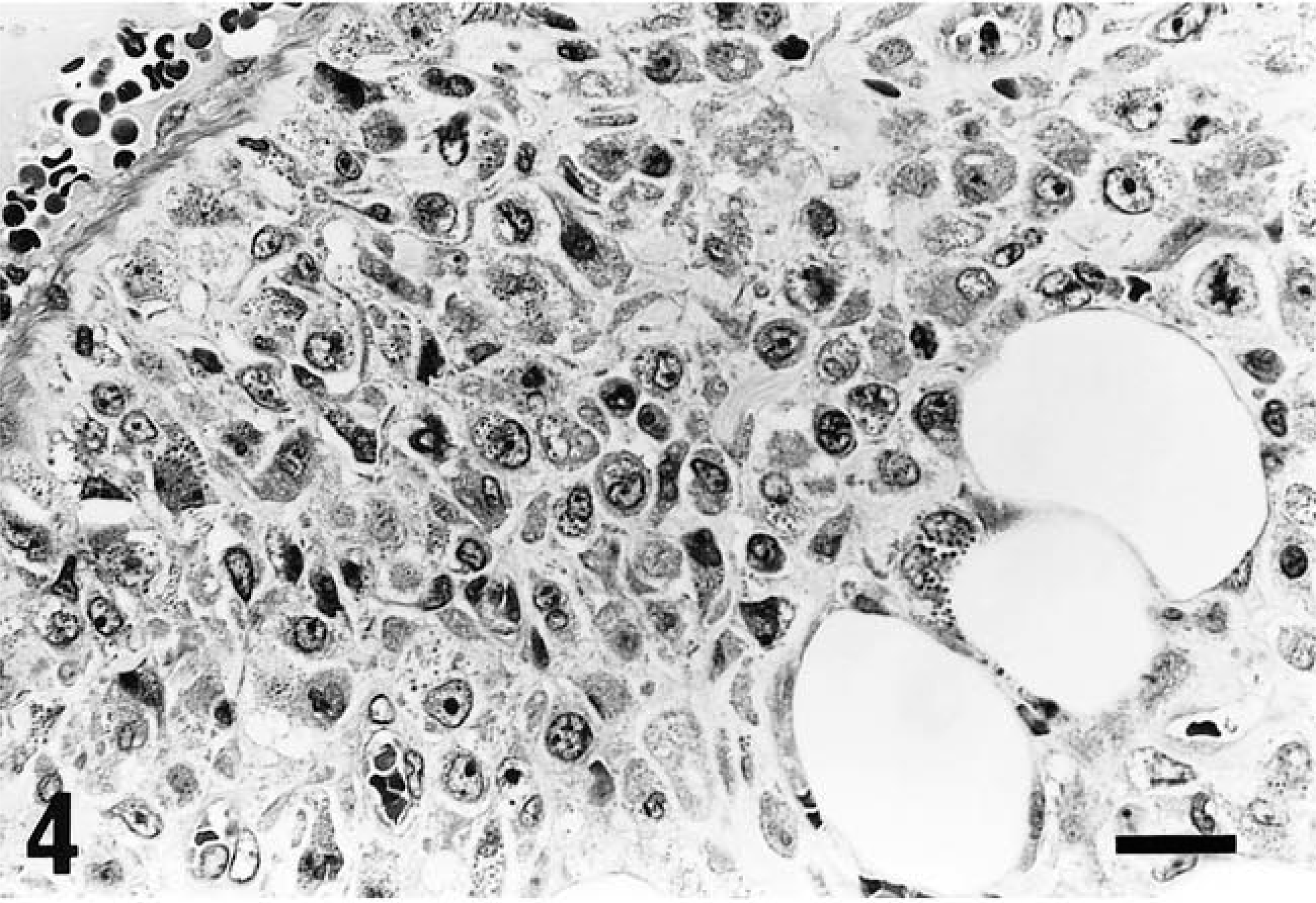
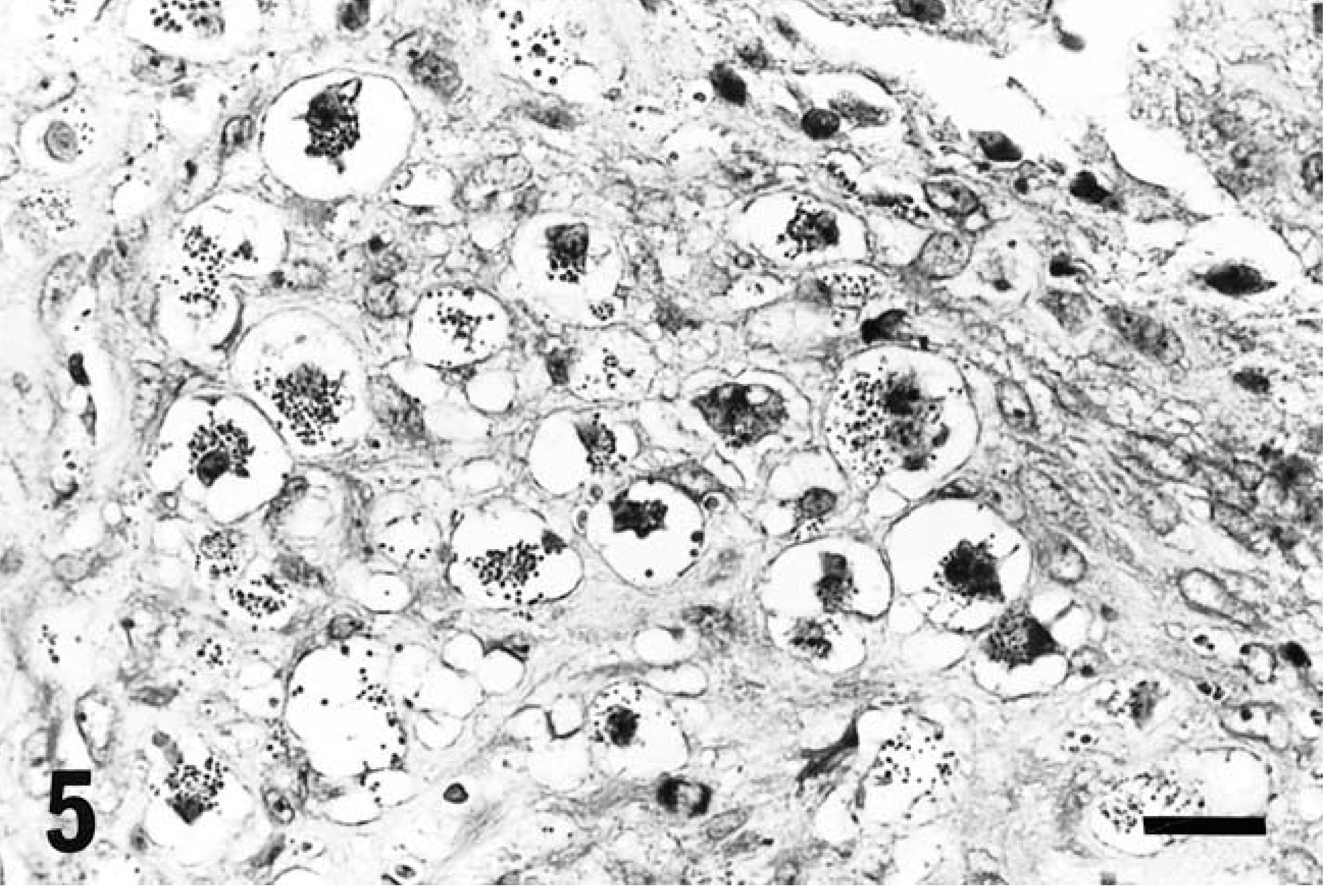
The tumor cells were round to oval in shape, contained one or two round, reniform, or pleomorphic nuclei, were characterized by distinct eosinophilic granules in clear cytoplasm, and were roughly divided into two types: large and small. The large type, approximately 20–30 µm in diameter, had abundant pale cytoplasm containing various numbers of cytoplasmic granules (Fig. 2) and one or two nuclei located eccentrically. In addition, some of these cells contained abundant glycogen granules even after prolonged fixation with formalin solution (Fig. 3a, b). The small type, approximately 10 µm in diameter, had scant basophilic cytoplasm with few or no cytoplasmic granules (Fig. 4). Cells that contained no cytoplasmic granules closely resembled lymphocytes. These two types of cells intermingled in different proportions in each case, the large type predominantly in rat No. 1 and the small type in rat Nos. 2 and 3. These morphologic features, especially in the large type of cells, closely resemble those of GMG cells (Fig. 5).
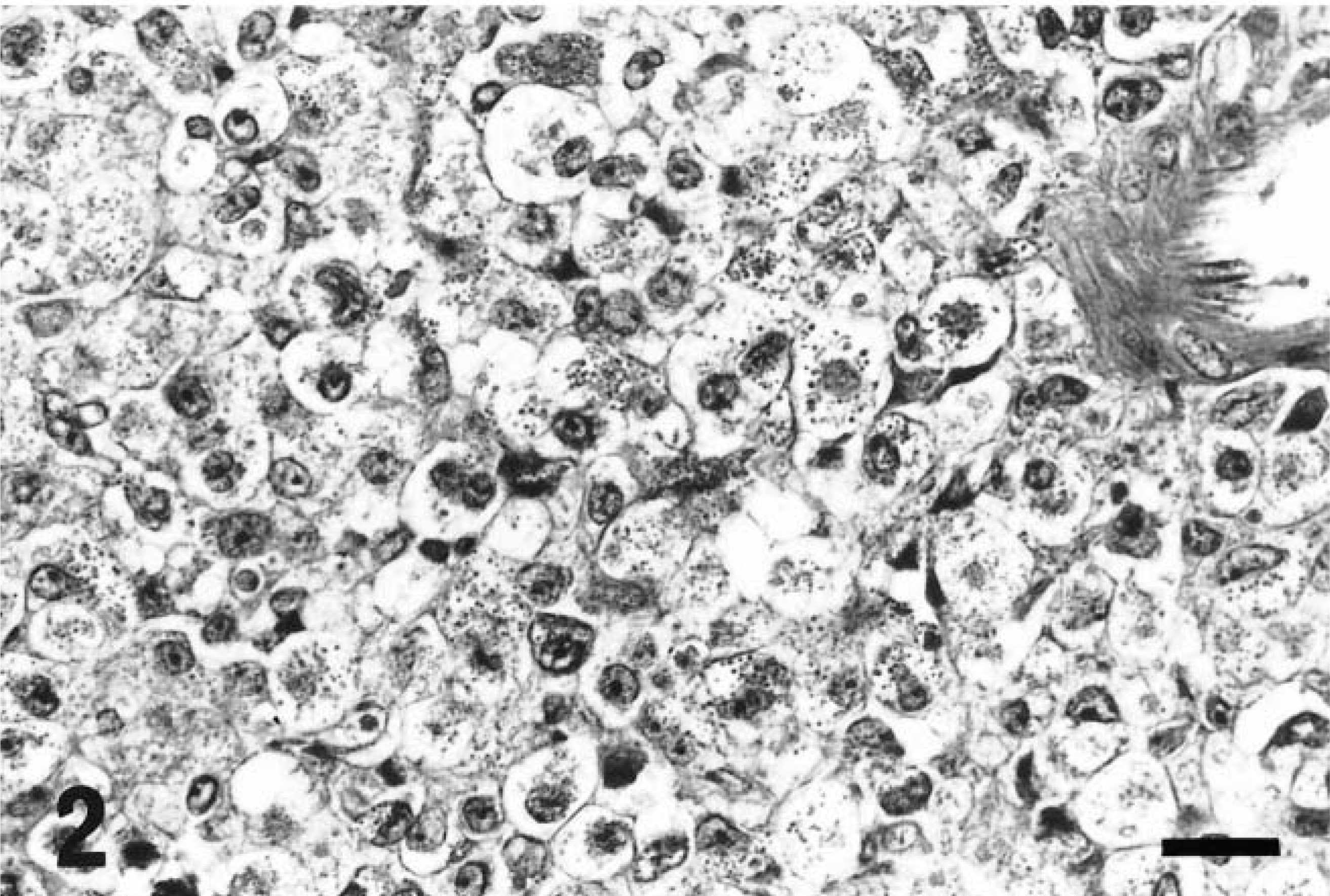
Abdominal mass; rat No. 1. Large tumor cells. Scattered cytoplasmic granules in abundant cytoplasm. HE. Bar = 20 µm.
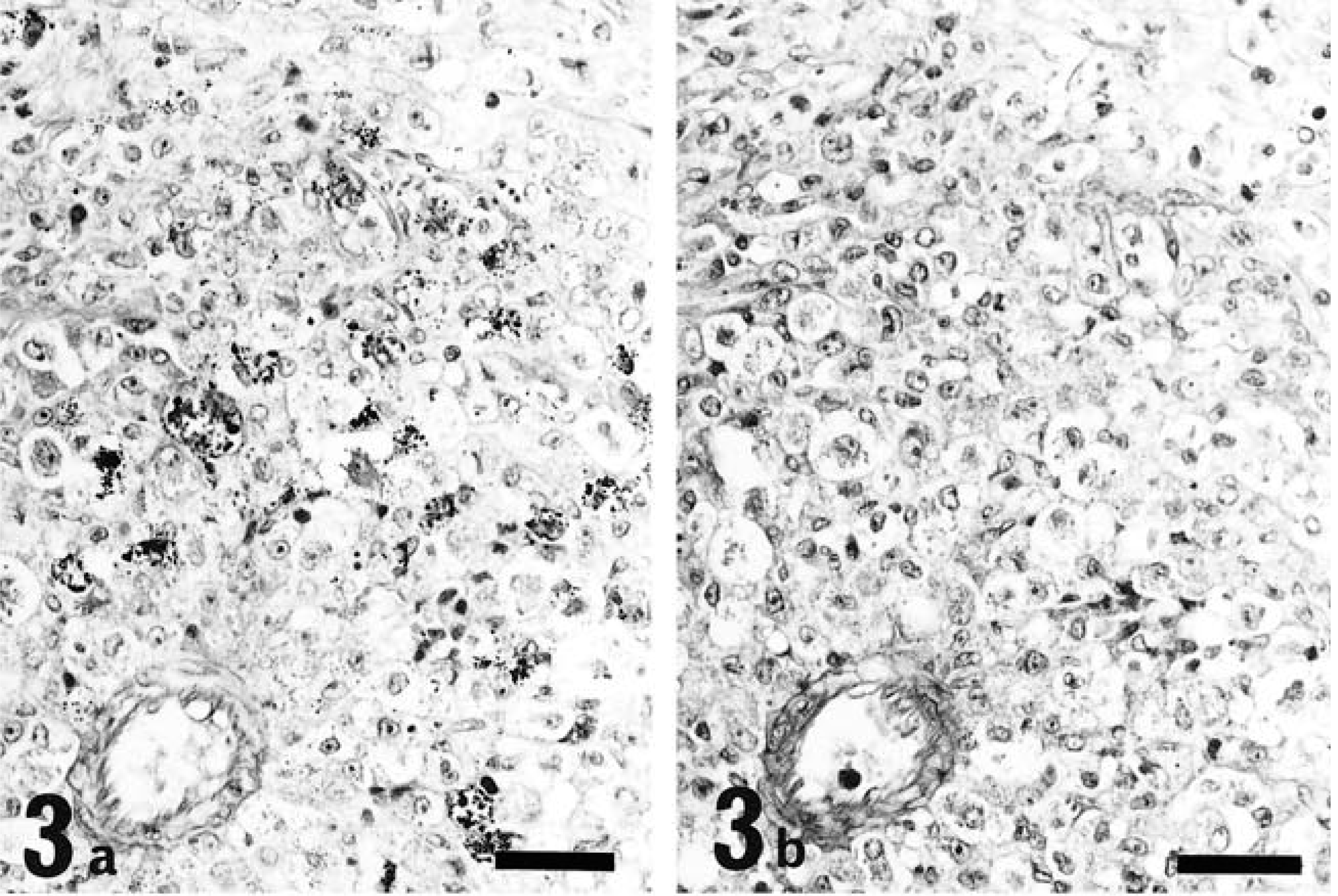
Abdominal mass; rat No. 1. Small tumor cells. Scanty cytoplasm with few or no granules. HE. Bar = 20 µm.
Uterus with decidual reaction; GMG cells. Scattered cytoplasmic granules in abundant clear cytoplasm. Cells resemble tumor cells of Fig. 2. HE. Bar = 20 µm.
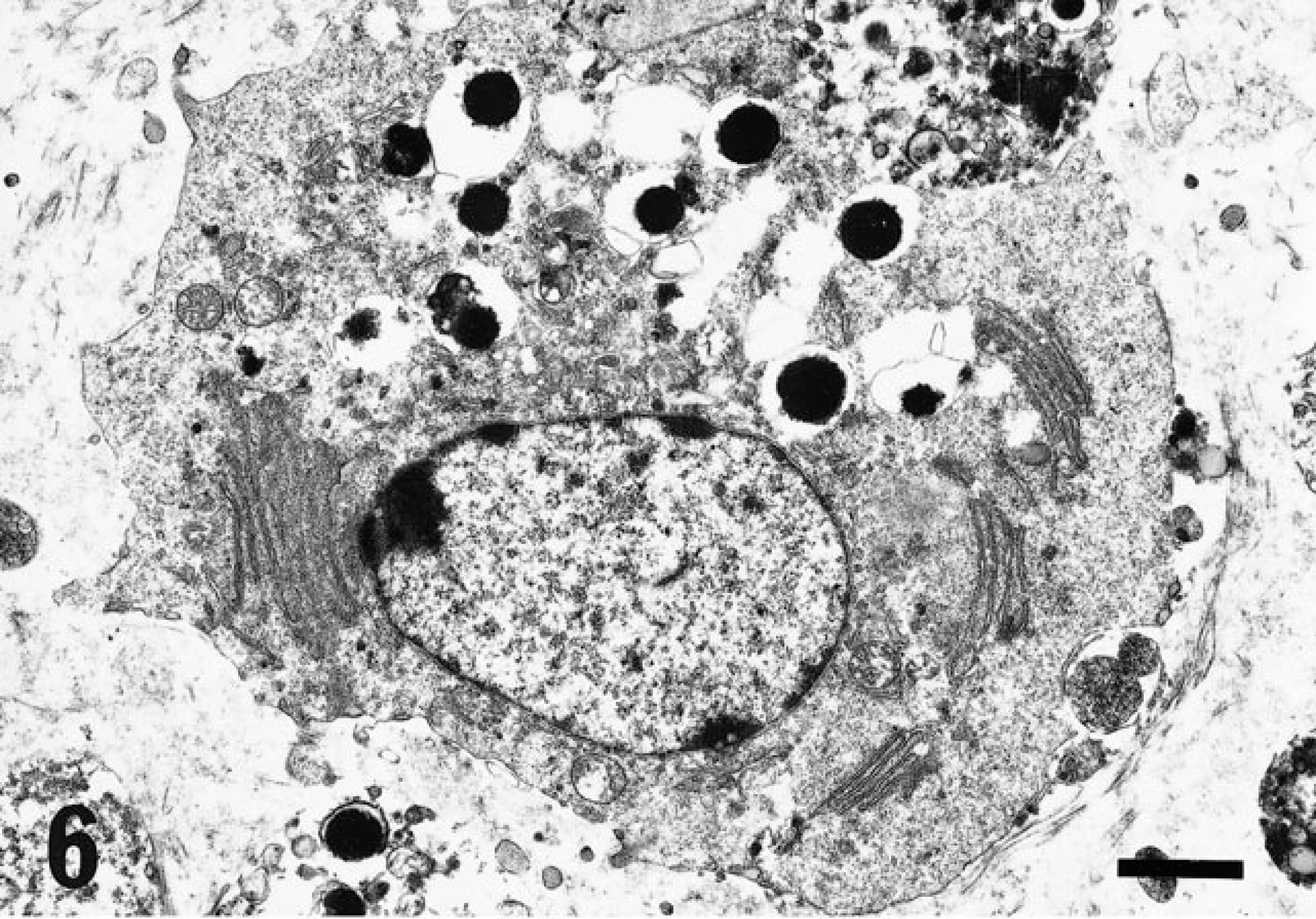
Ultrastructurally (Table 3), the cytoplasmic granules were identified as electron-dense amorphous bodies, up to 2 µm in diameter, bounded by a single unit membrane (Fig. 6), and located around a well-developed Golgi complex. A parallel array of rough-surfaced endoplasmic reticulum was occasionally noted. In general, except for ribosomes, the cytoplasmic organelles were poorly developed. No evidence of interdigitation with adjacent cells or basement membrane was observed.
Comparison of ultrastructural characteristics of the tumor cells, granulated metrial gland cells, globule leukocytes, and mucosal mast cells in rats.
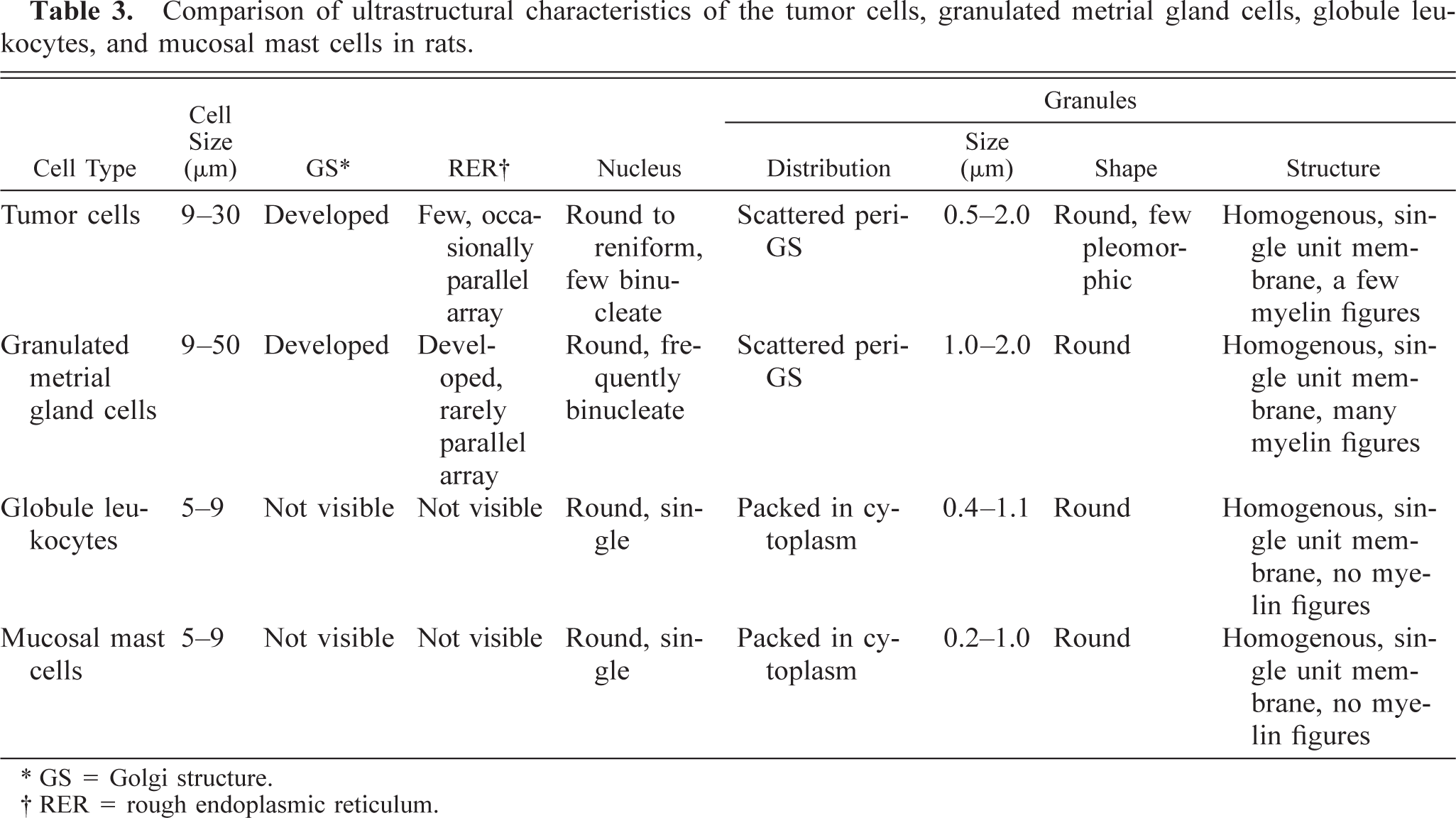
GS = Golgi structure.
RER = rough endoplasmic reticulum.
Electron micrograph. Tumor cell; rat No. 1. Parallel rough endoplasmic reticulum and several electron-dense bodies in the cytoplasm. Bar = 1.9 µm.
The immunohistochemistry and lectin histochemistry staining results are summarized in Tables. 4 and 5, respectively. The cytoplasmic granules from all rats typically stained deep blue with PTAH, pale blue to purple with Giemsa, definitely to weakly positive with PAS reaction, and nonmetachromatic with TB. No positive reaction was demonstrated immunohistochemically with anti-RMCPII, a marker of mast cells, and anti-CD8. In the lectin immunohistochemistry, ConA, WGA, PHA-E4, LCA, and RCA-I were positive in all rats, but positive reactions with UEA-I, PNA, and SBA were limited to rat No. 1. The many similarities of growth, invasive characteristics, and morphology indicate that these three tumors represent an identical type.
Histochemical characteristics∗ of the cytoplasmic granules in the rat tumor cells, granulated metrial gland cells, globule leukocytes, and mucosal mast cells.

- = negative (no metachromasia with TB); ± = weakly positive; + = definitely positive; ND = not done.
HE = hematoxylin and eosin; PTAH = phosphotungstic acid-hematoxylin; TB = toluidine blue; PAS = periodic acid-Schiff.
Cells from ascites.
Lectin histochemistry∗ of the cytoplasmic granules in the rat tumor cells, granulated metrial gland cells, globule leukocytes, and mucosal mast cells.

− = negative; ± = weakly positive; + = definitely positive; ND = not done.
In rodents, three kinds of wandering cells containing eosinophilic cytoplasmic granules, such as GLs (Fig. 7), MM cells (Fig. 8), and GMG cells (Figs. 5, 9), are well known. GLs are present in the epithelial layer of the mucosa of the gastrointestinal, respiratory, and urogenital tracts, 1 and their origin and function are still unclear. MM cells differ from mast cells in the connective tissue and are distributed in the lamina propria of the intestinal mucosa. 10 GLs may represent nothing more than MM cells migrating in the epithelial layer 6,7,14 or they may be common precursors of mast cells. 8 GMG cells appear in the uterus during pregnancy and decidual reactions 15 and exhibit natural killer (NK) activity. 2 MM cells and GLs are similar in morphology, being round to oval in shape and 5–9 µm in diameter, with the cytoplasm almost completely occupied by eosinophilic granules. GMG cells are larger than GLs and MM cells, ranging from 9 to 50 µm in diameter with eosinophilic granules dispersed in clear cytoplasm with glycogen. GMG cells of the small type have scant basophilic cytoplasm containing few or no cytoplasmic granules and little glycogen. The large GMG cells are characterized by many eosinophilic granules and abundant glycogen and frequently form binucleated cells. Thus, plentiful glycogen is a typical finding in GMG cells. 15
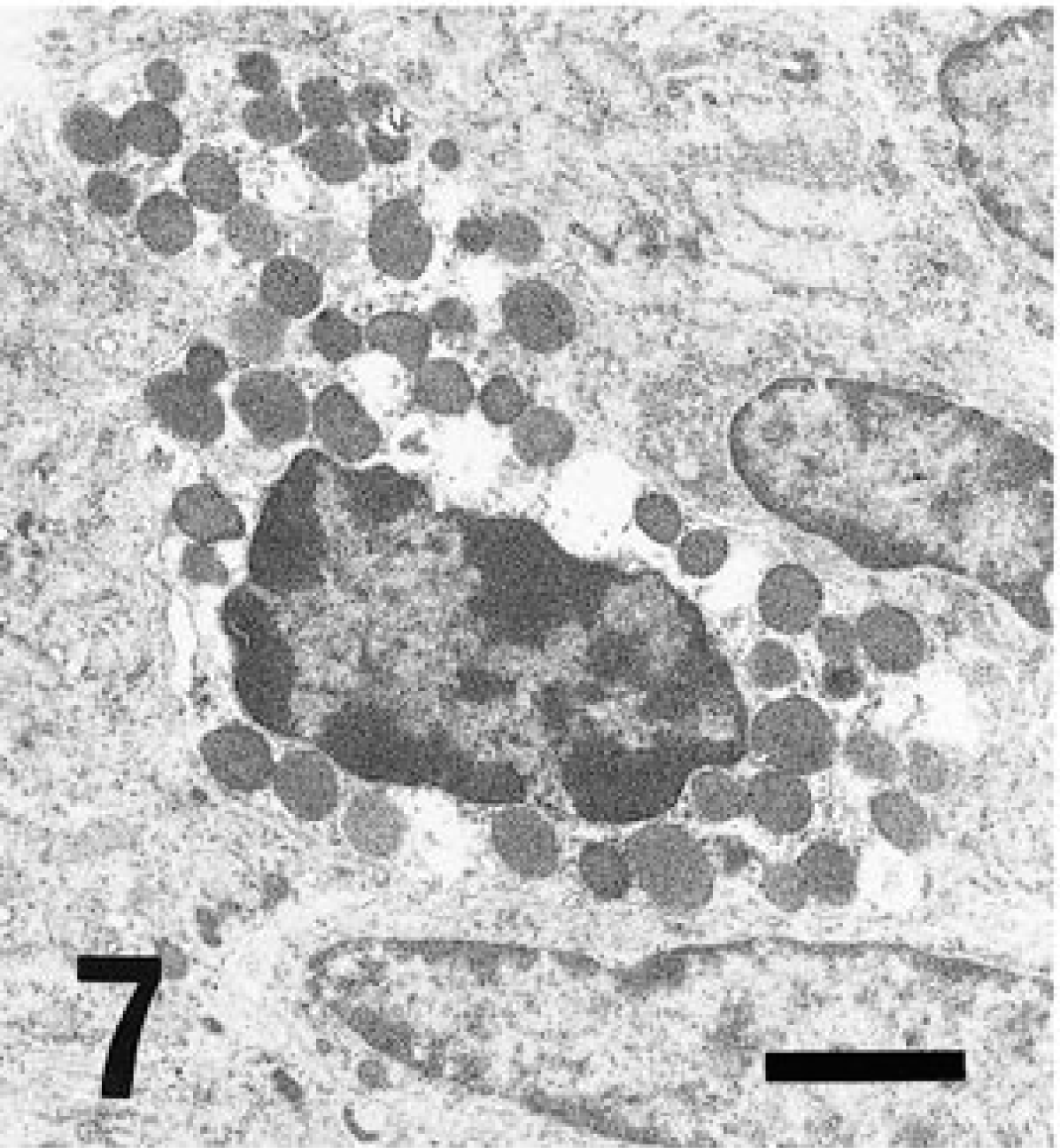
Electron micrograph. Cecum; GL. Many electron-dense bodies packed in the cytoplasm. Bar = 1.8 µm.
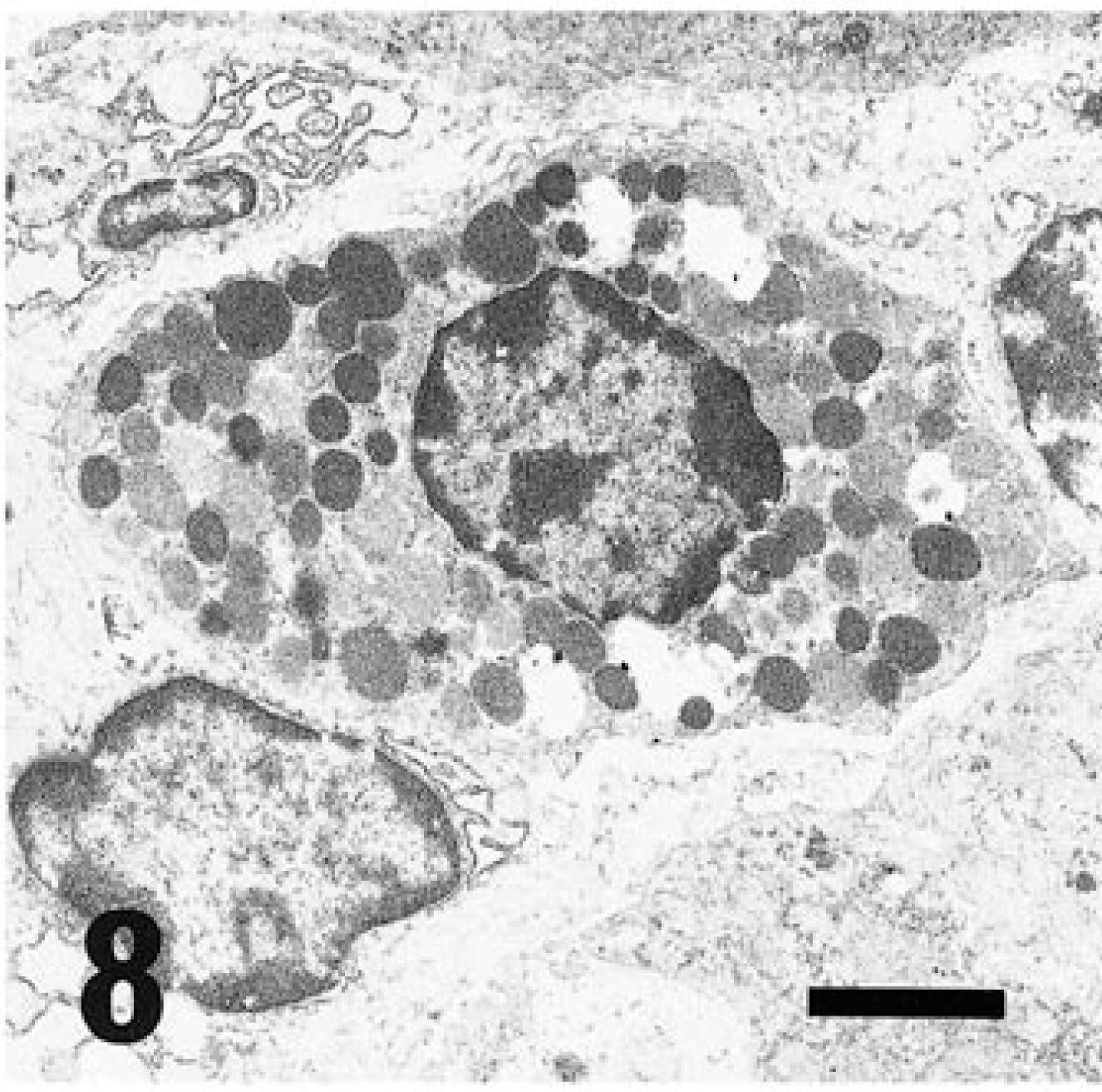
Electron micrograph. Cecum; MM cells. Many electron-dense bodies packed in cytoplasm. Bar = 1.8 µm.
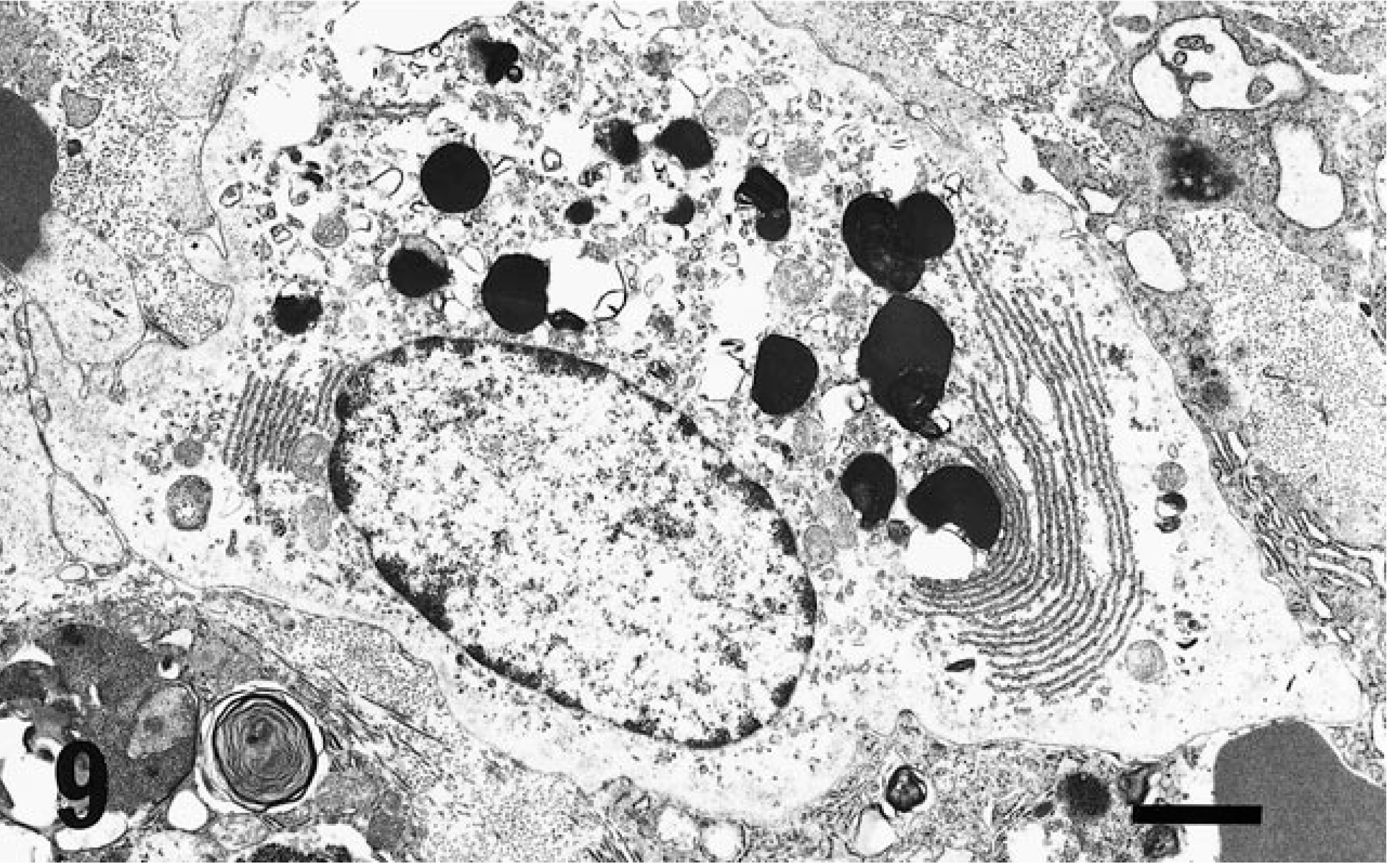
Electron micrograph. Uterus with decidual reaction; GMG cells. Parallel arrayed rough endoplasmic reticula and several electron dense bodies with myelin figures in the cytoplasm. Bar = 1.9 µm.
Regarding histologic, histochemical, lectin, and immunohistochemical features, GMG cells are more comparable to the tumor cells in these rats than are GLs and MM cells, especially in terms of their abundant glycogen and reaction with anti-RMCPII. Regarding the lectin histochemistry, except for the MM cells, the other cells and the tumor cells revealed almost the same characteristics. In another study, 18 GMG-like cells in mice showed a specific reaction for DBA, SBA, and PNA, in contrast to the results from the present study (Table 5). The basis for this discrepancy is unclear, although it may represent a species difference.
Feline GL tumors have been reported to arise mostly within the intestinal mucosa and to grow out of the intestinal wall. 11 However, the tumors in these rats arose primarily from the abdominal adipose tissue and not from the intestinal mucosa. In addition, GMG cells or GMG-like cells have been observed in the uterus in certain pathologic conditions in virgin rats 21 and in malignant fibrous histiocytic-like sarcoma in mice of both sexes. 19 These findings support the premise that the present tumors may not originate from GLs or MM cells but from GMG or GMG-like cells. There is also the possibility of extrauterine occurrence of these types of tumors, even in males.
Others have postulated that feline GL tumors are derived from LGLs, based on the immunoreactivity for perforin demonstrated in the cytotoxic T cells and NK cells. 9 In contrast, rodent neoplasms from LGLs (LGL leukemias) are known to occur spontaneously in some strains of rats, and these tumor cells exhibit NK activity. 17,20 In F344 rats, LGL leukemia is one of the common spontaneous tumors. 17 However, apparent differences were observed between LGL leukemias and the tumors in the present report; the most consistent attributes of LGL leukemias are marked infiltration and proliferation of tumor cells in the red pulp of the spleen, which shows extensive enlargement with congestion, and diffuse proliferation of tumor cells in the hepatic sinusoids. Moreover, erythrophagia and hemolytic anemia are characteristic of LGL leukemias. Between LGL leukemias and the present tumors, no similarity was observed except erythrophagia. In addition, no visible granules are seen in LGL leukemias or in normal LGLs. A tumor composed of cells with eosinophilic cytoplasmic granules that was observed recently in an F344 rat might have originated from GLs. 13 The cellular morphology and other characteristics of the present tumors are very similar to those reported previously. 13 However, the derivation of GLs appears definitely to differ between rodents and cats; those in rodents are mast cells, 6,7,14 and those in cats are LGLs. 9 These rat tumor cells show greater resemblance to GMG cells than to GLs and MM cells. Feline GLs, LGLs, and GMG cells are thought to exhibit NK activity. If so, the rat tumors in the present report originated from the same cell lineage. Further studies on the histogenesis of this kind of tumor and of the morphologic and biologic characteristics of NK cells in rats are needed.
Present observations suggest that spontaneous eosinophilic granulated round cell tumors in F344 rats are of the same lineage as those of GMG cells.
Footnotes
Acknowledgements
We thank Dr. A. Maekawa (Department of Pathology, Sasaki Institute) for his review of the manuscript and valuable suggestions.
