Abstract
We describe a case of large granular lymphocyte (LGL) leukemia in a dog that we followed over a period of 2 years. Analysis of a hematological profile revealed lymphocytosis (19,500 lymphocytes per microliter; reference values, 1,000–4,800 lymphocytes per microliter), with a majority of LGL on the blood smear. LGL is defined as a lymphoid subset comprising 10% of peripheral blood mononuclear cells and corresponding to either CD3- CD8- NK cells or CD3+ CD8+ T cells. The cells are characterized by abundant basophilic cytoplasm containing distinct granules of variable size and number. The characteristic phenotype of our leukemic LGL is of a cytotoxic T cell, CD3+ and CD8+. A new cell line, DLC 02, was established from the peripheral lymphocytes of the leukemic dog. Particles with type C retroviral morphology were found in ultrathin sections of DLC 02 cell pellets. These particles were found to have a sucrose gradient density of 1.17 g/liter and a reverse transcriptase activity with an Mn21 preference, suggesting that they correspond to a mammalian type C oncovirus.
Keywords
Large granular lymphocytes (LGLs) are heterogenous cells, some of which are able to lyse certain tumors or virus-infected cells. They constitute a morphologically recognizable lymphoid subset with abundant cytoplasm containing azurophilic granules and represent only a minority (around 10%) of normal peripheral blood lymphocytes. LGL leukemia can be divided into two classes depending on the cell lineage of the leukemic cells: a CD3+ T cell subtype (T-LGL) and a CD3− natural killer subtype (NK-LGL). 9 In humans, T-LGL leukemia has an indolent course with prominent autoimmune features and can occur in association with other autoimmune diseases, particularly rheumatoid arthritis. NK-LGL leukemia has an acute clinical presentation marked by massive hepatosplenomegaly and systemic illness. In this report, we describe a canine LGL leukemia belonging to the T subset and a cell line derived from it that was found to produce type C retrovirus.
Material and Methods
Case history
An 11-year-old female husky was presented for consultation because of a decline in general condition that was accompanied by diarrhea and moderate polydipsia. The physical examination showed moderate splenomegaly, without hepatomegaly or lymph node enlargement. Analysis of the hematological profile revealed a leukocytosis (27,800 leucocytes per microliter; reference values, 5,000–17,000 leucocytes per microliter) caused by a lymphocytosis (19,500 lymphocytes per microliter; reference values, 1,000–4,800 lymphocytes per microliter), with a majority of LGL on the blood smear. There was no anemia, thrombocytopenia, or neutropenia. Two weeks later, when the lymphocytosis was still present (26,000 lymphocytes per microliter), a myelogram was carried out. The bone marrow was characterized by a definite but moderate (16%) medullary lymphocytosis with well-differentiated lymphocytes, including a contingent with clear cytoplasm, of which a large number contained azurophilic granules. The general condition of the dog was good, and it was not presented for another consultation until 6 months later, following another episode of diarrhea with anorexia. Once again, no clinical signs were noticed other than those that had led to the consultation. Further hematological examinations revealed the persistence of a peripheral lymphocytosis (27,250 lymphocytes per microliter) associated with normocellular bone marrow, without medullary insufficiency but characterized by lymphocytosis (23.5%). The lymphocytes were well differentiated (relatively small, with cytoplasm that was moderately extended, and, in rare cases, azurophilic granulations). The degree of the concomitant plasmocytosis (8.5%) and monocytosis (12.5%) suggested a reactive lymphocytosis. A rickettsial origin (Ehrlichia canis) was ruled out by serology and by the fact that tetracycline treatment did not result in any improvement in the hematological signs. Clinical and biological examination have been carried out regularly since that time, but the only clinical sign that has been noted is intermittent diarrhea, most often originating in the large intestine and sometimes accompanied by warm and painful joints without lesions, as determined by articular radiographs. The persistence of the diarrhea resulted in repeated fecal examinations, biochemical examinations, endoscopy with staged biopsies, and histological examinations. These revealed a subacute to chronic colitis, without ulceration. Histopathological analyses revealed a discrete lymphoplasmocytic enteritis.
Establishment and characterization of cell line
The lymphocytes isolated from the peripheral blood were separated on Ficoll gradients and incubated at a final concentration of 106 lymphocytes per milliliter in an RPMI 1640 culture medium supplemented with 20% heat-inactivated fetal calf serum (GibcoBRL, Cergy Pontoise, France),
Phenotyping of canine lymphocytes
Flow cytometry analysis of cells was performed on a FACScan equipped with Consort-30 and Lysis software (Becton Dickinson Immunocytometry Systems, Sunnyvale, CA, USA) using a standard indirect immunofluorescent technique. A polyclonal rabbit anti-human CD3 (Dako, Glostrup, Denmark) that recognizes intracytoplasmic canine CD3∊ chain in lymphocyte cells was used to detect T lymphocytes. The polyclonal antibodies and monoclonal antibodies (mAbs) used in this study were the following: anti-human HLA-DQ/IOT2d mAb (SPVL3, Immunotech, Marseille, France) and rat anti-canine mAbs directed against CD4, CD5, CD8, and CD45 molecules (Dr. S. P. Cobbold, Sir William Dunn School of Pathology, University of Oxford, UK). FITC-conjugated goat anti-mouse immunoglobulin G (IgG; Silenus, Melbourne, Australia) and immunoglobulin M (Jackson Immunoresearch, PA, USA), anti-rabbit IgG (Pharmingen, CA, USA) and anti-rat IgG1 (Interchim, Paris, France) were used to detect the binding of primary antibodies.
Detection of retroviral particles and morphological evaluation
After induction with 0.5% dimethyl sulfoxide (DMSO) for 1 day, the cells were centrifuged at 400 × g for 10 minutes. The cells were resuspended at 106 cells per milliliter, fixed with 2% glutaraldehyde and 0.5% paraformaldehyde in 0.1 M phosphate buffer, and then postfixed in 1% OsO4. After dehydration in alcohol, the samples were embedded in epoxy resin and stained ultrathin sections of samples were examined by electron microscopy at the Centre de Microscopie Electronique Appliquée à la Biologie et à la Géologie, Villeurbanne, France, with a CM120 electron microscope (Philips, Eindhoven, The Netherlands). Cytocentrifuge smears of peripheral blood mononuclear cells from the dog or of DLC 02 cells were prepared and stained with May-Grünwald-Giemsa for observation by light microscopy.
Virus isolation and sucrose density gradient analysis
After induction with 0.5% DMSO for 1 day, the DLC 02 cells were centrifuged at 400 × g for 10 minutes. Supernatants were clarified by filtration using 0.45-µm filters, and viral particles were concentrated through a ribonuclease-free, 25% sucrose cushion by centrifugation at 30,000 rpm for 3 hours (Beckman, TI 45). The pelleted material was resuspended in 10 mM Tris-HCl (pH 7.5), 100 mM NaCl, and 1 mM EDTA; it was then loaded on 8-ml gradients of 20 to 60% sucrose. Gradients were centrifuged in an SW41 rotor (Beckman) at 80,000 × g for 4 hours at 4 C. Two hundred fifty microliter fractions were carefully collected from the bottom with a Pasteur pipette connected to a peristaltic pump. Densities were calculated on 10 fractions from each gradient using a refractometer, and the density of the intermediate fractions was extrapolated from the linear curve obtained. Samples of 10 µl from each fraction were used to quantify reverse transcriptase (RT) activity.
RT assays
RT assays were performed according to published procedures. 4 RT was performed on 30 µl of culture supernatant by the incorporation of thymidine from α-[32P]-dTTP (10 µCi/ml) into a poly (rA):oligo dT(15) primer template substrate in a final volume of 50 µl of buffer containing 50 mM Tris-Cl, pH 8.3, 20 mM DTT, 0.05% Nonidet P40, and 0.6 mM MnCl2. After 1 hour's incubation at 37 C, samples were dot-blotted on a diethylaminoethyl 0.45-µm membrane using a Biorad dot-blotter apparatus, extensively washed with 3 M NaCl and 0.3 M Na citrate, pH 7.0. Spot intensity was measured with a PhosphoImager (Storm 860). The NIH/3T3 murine cell line infected by Friend murine leukemia virus strain 57 was used as positive control for Mn2+-dependent RT activity.
Results
The values of the peripheral lymphocyte count over the 2-year study period are summarized in Table 1. A regular increase in CD8+ lymphocytes can be observed. This corresponds to an increase in the number of LGLs; LGLs were identified by their large size, abundant pale cytoplasm, and prominent azurophilic granules (Fig. 2A). In cytospin slides of peripheral blood mononuclear cells (PBMC), the cells with the same typical LGL morphology expressed CD3 but did not express CD4 molecules. A more extensive phenotypic characterization was carried out by FACScan analysis. In a typical experiment, the peripheral blood cells were 81.8% CD8+, 0.9% CD4, 97.4% CD5+, and 99% CD45+ (Fig. 1A).
Follow-up of number of peripheral lymphocytes over a 2-year period. Only the most significant values are represented.
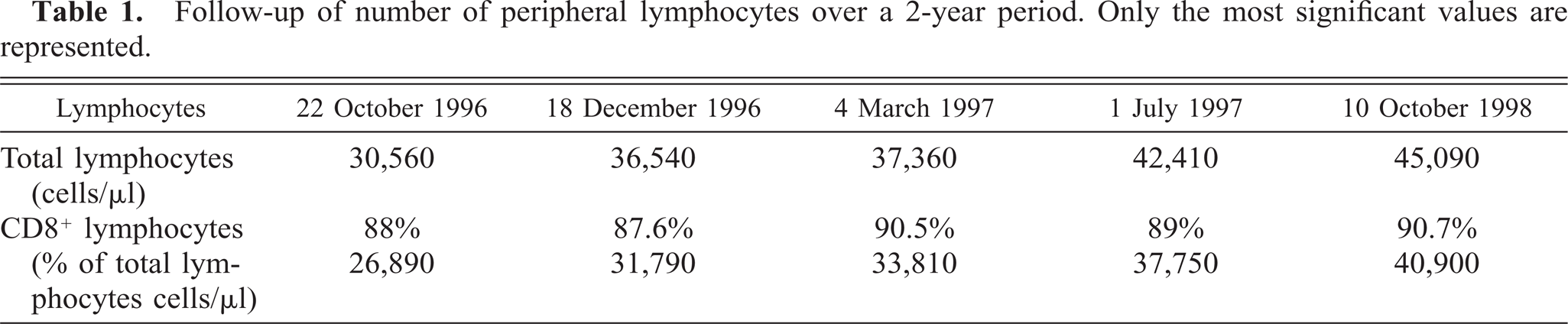
A representative FACS analysis of peripheral blood lymphocytes of the LGL leukemic dog (A) and from DLC 02 cells line after 2 months of culture (B). Normal peripheral blood mononuclear cells (PBMC) isolated from normal dog are stained as a positive control (C). The cells were stained with a panel of uncoupled mAbs revealed by FITC-conjugated anti-mouse immunoglobulins. The controls were stained with FITC-conjugated anti-mouse immunoglobulins (see Material and Methods). A total of 10,000 events were acquired. Results are representative of 10 experiments. The phenotype of primary, uncultured lymph node cells showed 96.5% CD8+, and these cells did not express the CD4 molecule (A). By contrast, the percentage of CD4 cells in normal peripheral blood lymphocytes is 45 (C). After 2 months of culture, DLC 01 cells lacked CD8+ molecule (B).
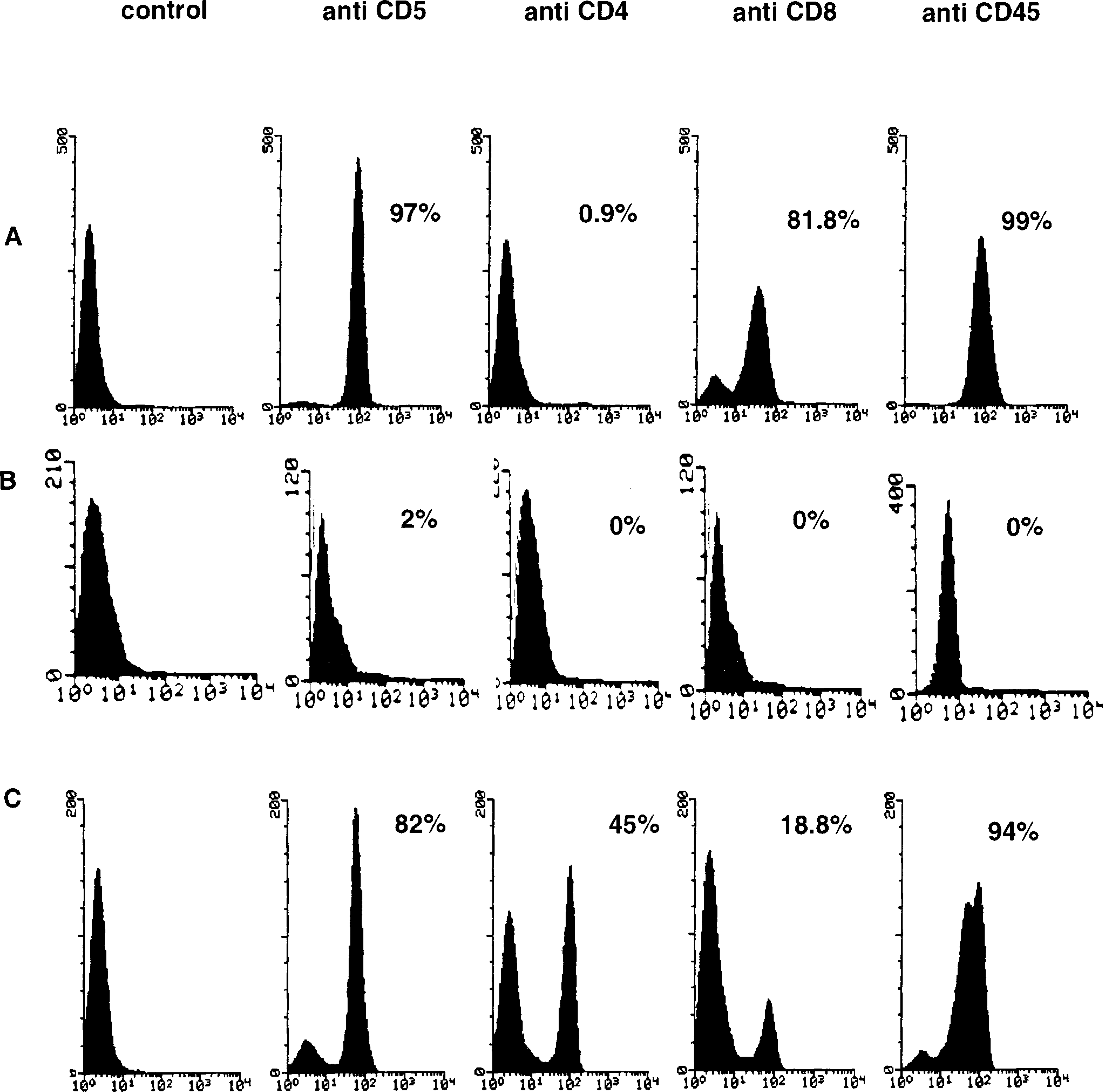
Morphological analysis of leukemic cells. Fig. 2A. Cytological characteristics of a fresh lymph node smear after cytospin preparation of suspension cells. May-Grünwald-Giemsa stain. Bar = 10 µm. Arrowhead: a large leukocytes with a few cytoplasmic granules. Fig. 2B–D. Ultrathin section of DLC 02 cell line. (B) Note the nuclear segmentation and the abundance of ribosomes and mitochondria (thin arrow), which indicate activated cells. Many granules with electron-dense cores (asterisk) are present (lysosomes). Bar = 2.5 µm. (C) Extracellular virus around the cell (thin arrow) with mature pentagonal and centrally located core. Note the mitochondrion (m). Bar = 250 nm. (D) Virion budding at the plasma membrane. Bar = 100 nm. For electron micrographs, representative sections were contrasted with uranyl acetate and lead nitrate.
In culture, the cells grew as suspended clusters with similar morphological characteristics to the fresh tumor cells, although more frequent mitoses were observed. After 2 months of primary culture, cells were still proliferating. Cells were subcloned by the limiting dilution method, and the DLC 02 cell line was established. DLC 02 cells proliferated rapidly, with a doubling time of 24 hours. However, after 2 months of culture, DLC 02 cells had lost the expression of the CD8, CD5, and CD45 antigens (Fig. 1B). DMSO treatment can induce cell differentiation 2 , 3 ; however, 0.5% DMSO did not induce re-expression of CD8, CD5, and CD45 molecules in DLC 02 cells.
Electron microscopic examination of the DMSO-treated DLC 02 cells showed indented nuclei, numerous ribosomes, abundant endoplasmic reticulum, and mitochondria with the presence of many lysosomes characteristic of cytotoxic cells (Fig. 2B). Surprisingly, we observed extracellular spherical particles (Fig. 2C). These particles were closely associated with the plasma membrane of the cells, but they were also found in cellular debris and in vacuoles that may have been the result of internalization. These particles were approximately 100 to 120 nm in diameter and were limited by a bilayered envelope. A centrally located, electron-dense core was usually separated from the envelope by an electron lucent space. We also observed incomplete structures of similar diameter and morphology still associated with the plasma membrane. This strongly suggested that these particles assembled and budded at the plasma membrane (Fig. 2D). The shape of the central core and the budding morphology were extremely suggestive of type C retroviral particles.
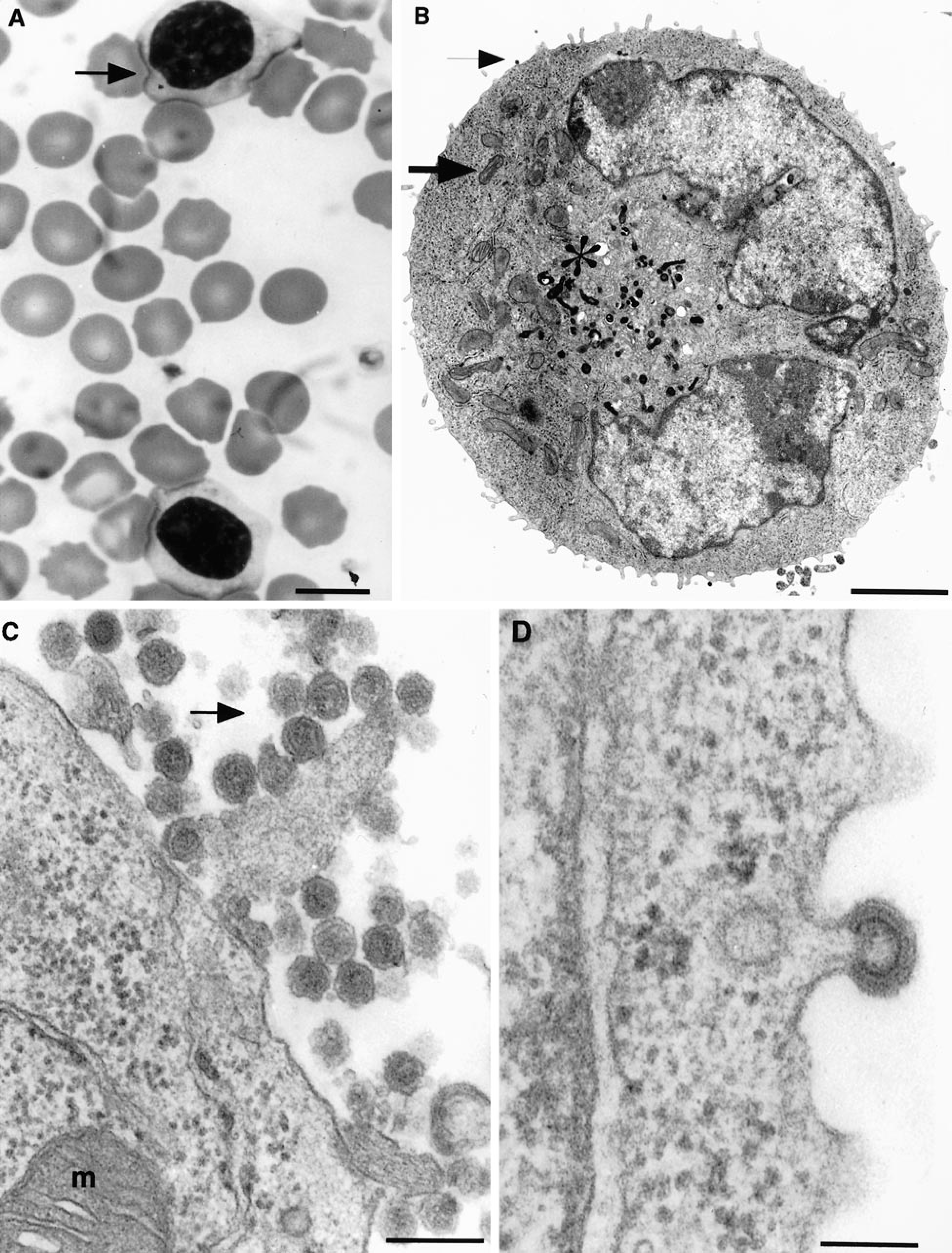
To confirm the retroviral origin of these particles, we tested the supernatant of DLC 02 cells for RT activity. When compared with PBMC from a healthy dog as a negative control, DLC 02 supernatant was strongly positive for RT activity in the presence of manganese (compare spots 3 and 4 in Fig. 3A). No significant RT activity was detected in the presence of magnesium. As shown in Fig. 3B, RT activity was not free in the supernatant but sedimented in a sucrose gradient at a density of 1.17 g/ml, typical for retroviral particles.
Analysis of RNA-dependent DNA polymerase activity in DLC 02 supernatant. Viral particles purified from cell supernatant were monitored for RT activity in the presence of 6 mM MnCl2, using an exogenous poly(rA)-oligo(dT) primer template substrate and α-[32P]-dTTP. Polymerization products were filtered on a DEAE membrane. Fig. 3A. Viral particles were purified from negative controls, culture medium (1) and normal peripheral blood cells stimulated with IL2 and PHA (3), positive control NIH/3T3 mouse cell line infected with F-MuLV FB29 viral strain (2) and DLC 02 cell line (4). Fig. 3B. Density gradient analysis of retroviral particles produced by DLC 02 cell line. Virus pelleted from cell supernatant were centrifugated through 8 ml 20 to 60% sucrose density gradient, as described in Material and Methods. Each fraction of 250 µl encompassing the expected density peak of 1.17 were monitored for the level of RT as in A, and spot intensities were measured with a PhosphorImager. The experiment was repeated twice with similar results.
Discussion
We describe a canine LGL lymphoproliferative disease that progressed as chronic leukemia, with lymphocytosis persisting for more than 2 years and only mild and nonspecific clinical manifestations. Although human LGL lymphoproliferations have been extensively studied, very little information is available concerning canine LGL leukemias.
In humans, LGL lymphoproliferation is a rare condition. It is usually a heterogeneous disorder with manifestations ranging from spontaneous regression to progressive malignant disease. A level of LGL higher than 2,000 lymphocytes per milliliter that lasts for more than 6 months is currently considered a key criterion for diagnosis 5 , 17 ; however, it is not essential if clonal proliferation of LGL can be demonstrated. 18 Distinction between various subgroups of these malignancies is based mostly on the expression of leukocyte differentiation (CD) antigens. In humans, both T cell–derived (CD3+) and natural killer (NK) cell–derived (CD3−) forms exist, along with several subtypes. 9 T-LGL leukemias are the most frequent (85%). Their manifestations usually include chronic neutropenia, and they are frequently associated with rheumatoid arthritis. They are generally characterized by chronic indolent evolution and dominated by recurrent infections caused by severe neutropenia. NK-LGL leukemia, on the other hand, is an aggressive disease characterized by massive hepatosplenomegaly, and manifestations of systemic disease are common. Severe neutropenia is unusual, but anemia and thrombocytopenia are more common and pronounced.
We have found only two reports of LGL leukemias in dogs. Clinical signs were relatively nonspecific. The first report describes three cases of canine LGL leukemias. 22 Two of those dogs had a rapidly progressive form of the disease, with hepatosplenomegaly or mediastinal mass, anemia, and thrombopenia. The third had only mild enlargement of peripheral lymph nodes, and lymphocytosis was the only hemogram abnormality. In this latter case, the clinical course was prolonged over more than 4 years. Unfortunately, an immunophenotyping of peripheral lymphocytes was not performed. The second study reports a single case of LGL leukemia associated with cutaneous lymphoma. 6 The physical examination showed an alert, afebrile dog with generalized cutaneous lesions. The neoplastic cells in the blood and the skin were CD3, CD4, and CD8 negative; however, when stained with MoAb 1A1, the neoplastic cells appeared to recognize T cells and NK-like cells in the dog. Other phenotypic analyses were suggestive of the NK lineage.
In our case, the diagnosis of LGL proliferation was based on the typical morphology of the cells. Although we did not study the clonality of these cells, the great number of LGLs (about 100-fold the normal value) that persisted for more than 2 years is highly suggestive of neoplastic proliferation. The immunophenotype of the tumoral cells (CD3+, CD4−, CD8+) is similar to that of most human T-LGL leukemias. Unfortunately, we could not test other informative markers of differentiation, such as CD16, CD56, or CD57, because of the lack of specific antibodies for canine homologues. Clinical similarities between this case and human T-LGL leukemia were also striking. The absence of hepatomegaly, lymph node enlargement, severe anemia, or thrombocytopenia and the prolonged indolent evolution were similar. The arthralgia noticed in the dog is reminiscent of the high incidence of rheumatoid arthritis in human T-LGL leukemias in which intestinal infiltration has been observed. 5 , 7 , 8 However, this dog did not have the neutropenia usually observed in human cases. The case we report is similar to dog 3 described by Wellman et al., 22 whereas dogs 1 and 2 were more reminiscent of the clinical presentation of human NK-LGL. Accordingly, the more aggressive form associated with cutaneous lymphoma described by Helfand et al. 6 belonged to the NK lineage. However, we recently reported a similar case of canine LGL leukemia associated with cutaneous lymphoma in which neoplastic cells expressed CD3 and CD8 molecules. 3 The clinical course was rapid, and the dog was euthanized because of its poor response to therapy and the rapid deterioration of its condition.
We have established a cell line, DLC 02, from the peripheral LGL of the leukemic dog described in this report. Established cell lines are important tools for the study of molecular mechanisms involved in the transformation process. Electron microscopy showed that the DLC 02 cell line produced extracellular particles typical of a type C retrovirus. The demonstration that these particles contained Mn2+-dependent RT activity confirms the finding. There have been several independent reports of putative retroviral infections in dogs, 1 , 14 , 20 which were detected either by electron microscopy or by RT assays. For example, retroviral particles associated with a magnesium-dependent RT activity were detected in indicator cells cocultivated with peripheral blood mononuclear cell from a leukemic dog, 16 and a manganese-dependent RT activity has been reported in an immunosuppressed dog. 13 However, no cell line has been derived from the corresponding animals, and these reports remained isolated and were never followed by any further characterization of the viruses. As a consequence, the existence of canine retroviruses is not currently considered to be established. Obtaining the DCL 02 cell line will allow us to study this virus, and its full characterization is in progress.
Of the seven genera of retroviruses, two show an association of a type C morphology with manganese-dependent RT activity: the mammalian type C oncoviruses and the spumaviruses. The virus produced by DCL 02 may belong to the genus Spumavirus. However, we did not observe the prominent spumous cytopathic effect that characterizes these viruses. Thus, we consider that the virus produced by DCL 02 is more likely a mammalian type C oncovirus. Furthermore, we have previously established another T CD8+ cell line from the dog suffering from an LGL leukemia associated with a cutaneous lymphoma (see above). Interestingly, this cell line, termed DLC 01, also produces a type C retrovirus associated with a manganese-dependent RT activity. 3 This retrovirus has now been fully characterized (in preparation) and belongs to the mammalian type C oncovirus genus. Preliminary results indicate that the viruses isolated from the DLC 01 and DLC 02 cell lines are related but distinct.
Mammalian type C retroviruses are etiologically associated with leukemias in numerous mammals. 21 Notably, the feline leukemia virus, responsible for feline leukosis, is a member of this genus. The isolation of such viruses in two independent CD8+ T-LGL leukemic dogs suggests that these viruses might be involved in this pathology. Interestingly, an important proportion of human T-LGL leukemias, immunophenotypically and clinically related to the case presented here, are associated with weak seroreactivities against human T leukemia virus (HTLV) antigens. 11 , 19 Most of these cases do not correspond to true HTLV infections, 12 and seroreactivity is believed to be due to cross-reactivity with an unknown retrovirus. 10 The possibility that both human and canine T-LGL leukemias might be associated with related retroviruses would be of considerable interest.
Footnotes
Acknowledgements
We are grateful to CMEABG for their assistance in electron microscopy, C. Bonnefont and I. Goy-Thollot for technical assistance, and Edmund Derrington for corrections of manuscript. Supported in part by the Ligue Contre le Cancer, Région Rho∘ne-Alpes, and the Association pour la Recherche Contre le Cancer.
