Abstract
Linear IgA disease (LAD) is an acquired autoimmune subepidermal blistering dermatosis that affects human children and adults. In contrast to bullous pemphigoid, in which autoantibodies recognize transmembrane type XVII collagen (BP180, BPAG2), LAD is associated with skin-fixed and circulating IgA autoantibodies that target LAD-1, the processed extracellular form of type XVII collagen. An immunologic homologue of LAD in humans was identified in two dogs according to the following criteria: 1) erosive, ulcerative, and crusted lesions seen on the face, in the oral cavity, and on the extremities, 2) dermoepidermal clefting present in the basement membrane lamina lucida without inflammation or with mild neutrophilic infiltration, 3) basement membrane-fixed IgG and/or IgA antibodies, and 4) circulating IgA and IgG autoantibodies that target the 120-kd soluble protein LAD-1. The present study establishes unequivocally the existence of a naturally occurring canine model of LAD of humans.
Keywords
Blistering dermatoses of humans frequently occur because of autoimmunity against cell–cell or cell–matrix adhesion structures. 14 Humoral and cellular immune responses specific for keratinocyte–keratinocyte adhesion molecules induce an acantholytic separation distinctive of pemphigus. Similarly, autoantibodies directed against keratinocyte–basement membrane attachment proteins result in subepidermal vesiculation. 14 Currently, the classification of the principal autoimmune subepidermal blistering dermatoses of humans is based upon the clinical signs exhibited by the patients and the molecular characterization of targeted antigens. For example, autoantibodies against membrane-bound type XVII collagen (BP180, BPAG2) define either bullous pemphigoid (BP) or a superficial variant of mucous membrane pemphigoid (MMP). 14 Deeper forms of MMP (e.g., cicatricial pemphigoid sensu stricto) also occur in association with autoantibodies directed against the carboxy terminus of type XVII collagen 2 or against epitopes on laminin-5 chains, another protein of the lamina densa. 4 , 6 , 15 Scarring dermatoses associated with collagen VII autoantibodies are grouped under the denomination of epidermolysis bullosa acquisita (EBA). 5 , 27
Linear IgA disease (LAD), also known as linear IgA bullous disease, is an acquired autoimmune blistering dermatosis of humans that affects the skin and sometimes mucosae. LAD is characterized by linear deposits of IgA autoantibodies at the dermal–epidermal basement membrane zone. In many but not all human patients with LAD, IgA autoantibodies recognize either LAD-1, a 120-kd protein present in keratinocyte conditioned medium, 13 or a 97-kd protein that is a degradation product of LAD-1. 33 Rare human individuals exhibit both circulating IgA and IgG autoantibodies against the 97-kd degraded form of LAD-1. 3 , 31 Recent studies suggest that LAD-1 likely represents a proteolytic product containing the extracellular segment of collagen XVII (Fig. 1). Proteolysis of collagen XVII appears to create new epitopes on the ectodomain that are recognized by autoantibodies from patients with LAD. 34 These epitopes also are recognized by several monoclonal antibodies (mAbs) such as mAb 123. 13
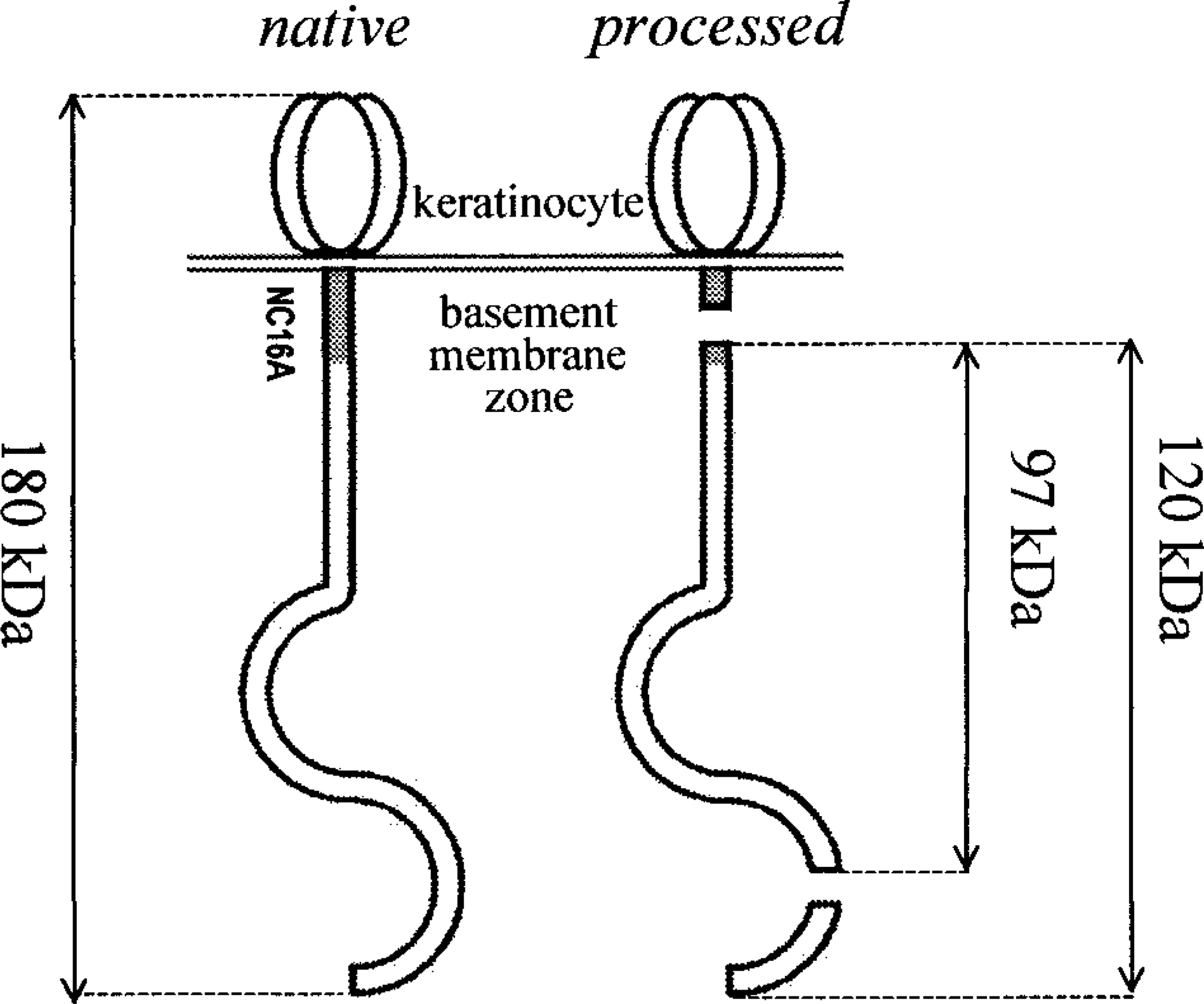
Native and processed type XVII collagen. The gray area illustrates the location of the NC16A domain, the segment encompassing the major epitopes recognized by autoantibodies in human and animal BP. Proteolytic cleavage within the NC16A domain of type XVII collagen yields LAD-1, a soluble fragment of 120 kd (per collagen XVII monomer). 7 , 13 A later degradation at the C-terminus of BP180 yields a shorter 97-kd soluble peptide. 7 , 34
Since 1976, 1 animals affected with erosive and/or blistering dermatoses associated with basement membrane–specific autoantibodies have been given the “generic” diagnosis of BP. In none of these cases, however, were the self-antigens determined. In dogs, cats, pigs, and horses, blistering skin diseases associated with circulating anti-collagen XVII IgG autoantibodies, hence analogous to BP in humans, have been characterized recently. 9 , 16 , 17 , 19 The immunologic homology of canine, feline, porcine, equine, and human BP has been strengthened by the recent demonstration that circulating IgG autoantibodies target similar antigenic epitopes in the NC16A ectodomain of type XVII collagen. 17 , 19 , 28 Additional canine subepidermal autoimmune blistering dermatoses have been shown to represent natural homologues of diseases seen in humans. Recently, dogs with vesicles, erosions, and autoantibodies against type VII collagen have been identified. These dogs were shown to be affected with canine homologues of either human EBA or bullous systemic lupus erythematosus (BSLE). 18 , 20 , 21
The purpose of the present report is to describe the clinicopathologic characteristics of a novel canine autoimmune subepidermal blistering dermatosis. This canine skin disease, analogous to LAD of humans, is characterized by IgA/IgG autoantibodies that recognize epitopes on the processed extracellular segment of type XVII collagen (LAD-1).
Material and Methods
Histopathology
Lesional skin biopsy specimens were obtained from two adult dogs exhibiting ulcerative lesions located preferentially in the oral cavity and on the face and extremities (Table 1). The biopsy specimens were placed in neutral buffered formalin and processed for routine histopathologic examination. Additional perilesional skin biopsies were obtained from dog No. 1. These biopsies were deposited in a holding medium (OCT Tissue Tek, Baxter Diagnostics, McGaw Park, IL), snap-frozen in isopentane cooled to its freezing point in liquid nitrogen, and kept at −70 C until further processed.
Signalment and clinical data for two dogs with ulcerative lesions.

Antigen immunomapping
To specify the location of separation within the epidermal basement membrane zone, antigen immunomapping was performed as previously described. 18 Immunoperoxidase staining was accomplished, using paraffin-embedded skin sections, with polyclonal antibodies specific for type IV collagen (PU079-UP, Biogenex, San Ramon, CA; dilution 1:10) and appropriate secondary antiserum (biotinylated goat anti-rabbit IgG; EA-1000, Vector Laboratories, Burlingame, CA; dilution 1:40). Antigen retrieval was achieved by proteolytic digestion with pepsin (0.25% for 35 minutes at 37 C). The red chromogen was amino-ethyl-carbazole (Biogenex, San Ramon, CA).
Immunofluorescence studies
Detection of tissue-bound autoantibodies
Skin-fixed IgG, IgA, and IgM autoantibodies and activated complement (C3 fraction) were detected by direct immunofluorescence (IF) testing of either frozen or paraffin-embedded skin sections using fluorescein-labeled antibodies as previously described. 18 When formalin-fixed specimens were utilized, antigen retrieval was performed with trypsin proteolysis (Sigma, St. Louis, MO; 0.1% for 45 minutes at 37 C). Normal canine lip was used as a negative control, and skin from a dog affected with BP served as a positive control.
Detection of circulating autoantibodies
Initial screening for circulating autoantibodies was performed with an indirect IF method using normal canine lip and haired-skin substrates with serial dilutions of the dogs' sera. The extinction titer of basement membrane–specific antibodies was determined using fluorescein-labeled anti-dog IgA and IgG antisera as previously described. 18
Additional indirect IF studies were carried out using both canine and human salt-split epithelial substrates. This method utilizes a normal skin specimen in which the epidermal basement membrane has been artificially cleaved after a 24-hour incubation in 1 M sodium chloride. After incubation, gentle friction separates the epidermis from the underlying lamina densa. This technique has been validated for dogs. 8 Additionally, it has proven useful in differentiating BP from other canine autoimmune subepidermal blistering dermatoses. 18 , 21 In all indirect IF studies, negative controls consisted of the pooled sera collected from 10 normal healthy dogs. Serum from a dog with BP served as a positive control. In this dog, the autoantibodies were shown by immunoblotting and enzyme-linked immunosorbent assay to target membrane-bound type XVII collagen (NC16A segment). Supplementary indirect IF studies were performed using mouse mAb 123 directed against the processed soluble ectodomain of type XVII collagen. 13
Immunoblotting analyses
Immunoblotting was performed with conditioned serum-free medium and cell fractions from the human SCC-25 cell line as previously described. 12 , 13 Membrane-transferred proteins were incubated with sera from the two dogs with presumptive LAD and the one dog with BP. As positive controls, the serum from a human patient with LAD and anti-LAD-1 mAb 123 was selected. The negative control consisted of sera from one normal dog and one normal human.
Results
Histopathology
In both canine patients, microscopic examination of hematoxylin and eosin (HE)-stained sections revealed subepidermal vesiculation in the absence of basal keratinocyte cytolysis (Table 2). In dog No. 1, subepidermal vesicles contained low numbers of neutrophils (Fig. 2a). In dog No. 2, there was no inflammation in the vesicle lumen (Fig. 2b). In one specimen of oral mucosa, a mild lymphocytic and plasmacytic superficial dermal infiltrate was seen. In both dogs with LAD, the microscopic vesicles were markedly different from those seen in the dog with BP in which many neutrophils and eosinophils were observed in the vesicles and superficial dermis. 9 Microscopic examination of Luna-stained sections 11 corroborated the absence of intact or degranulated eosinophils in the vesicles and superficial dermis of these two dogs with LAD. These findings are in marked contrast with the eosinophil-rich canine BP skin sections.
Histopathological summary ∗ for skin biopsy sections from two dogs with ulcerative lesions.

∗ 0 = no infiltration; + = mild infiltration; ++ = moderate infiltration; +++ = marked infiltration.
† BP = dog with bullous pemphigoid, used as positive control.
‡ NEUT = neutrophil infiltration; EOS = eosinophilic infiltration.
§ MONO = mononuclear infiltration.
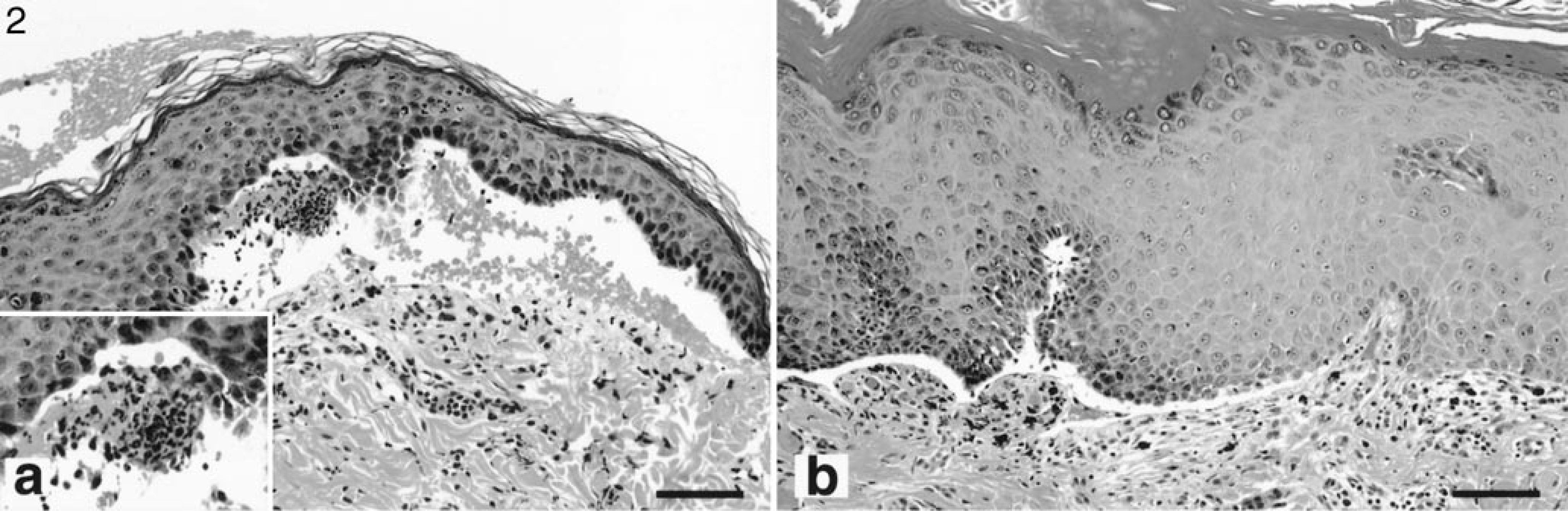
Haired skin; dog No. 1. Neutrophils (inset) and red blood cells can be observed in subepidermal vesicles. HE. Bar = 100 µm. Fig. 2b. Planum nasale; dog No. 2. Dermoepidermal separation without basal keratinocyte cytolysis. HE. Bar = 100 µm.
Antigen immunomapping
To determine the level of vesiculation within the epidermal basement membrane zone, immunostaining for type IV collagen was performed on paraffin-embedded sections exhibiting microscopic vesicles. In sections from both dogs with LAD, subepidermal vesicles occurred above collagen IV (Fig. 3). This finding corroborated that dermoepidermal separation invariably took place in the lamina lucida. Similar findings were seen with sections from the dog with BP.
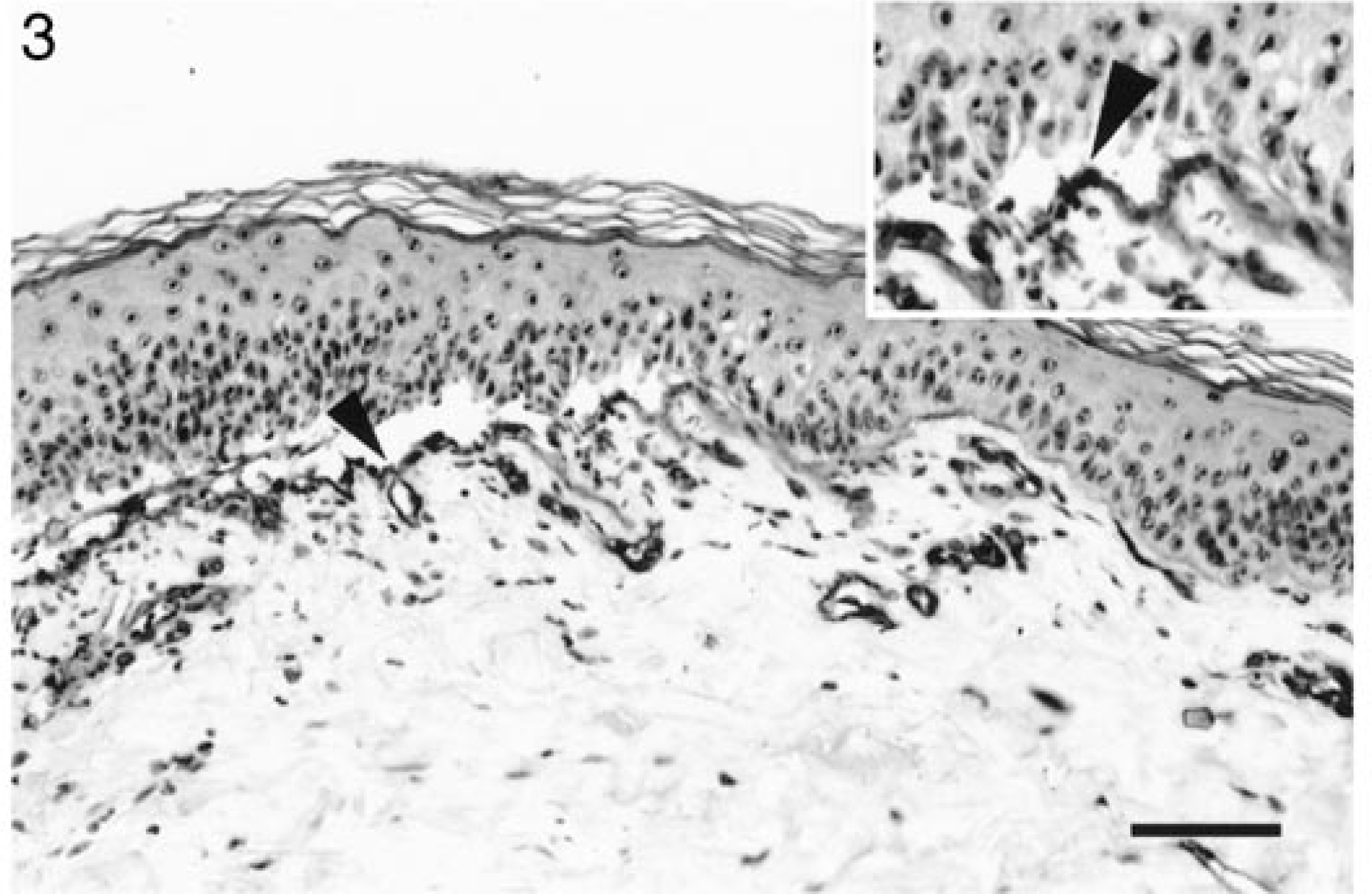
Haired skin; dog No. 1. Dermoepidermal separation occurs above collagen IV. Immunoperoxidase, collagen IV-specific mAbs. Bar = 75 µm.
Immunofluorescence studies
Direct immunofluorescence testing performed with skin biopsy specimens of both dogs with LAD and the dog with BP revealed a strong linear and continuous deposit of IgG along the epidermal basement membrane zone (Table 3, Fig. 4a). In biopsies from dog No. 1, basement membrane deposition of IgA (Fig. 4c), IgM, and C3 also was detected. Immunoglobulin staining of the normal canine lip biopsy was unremarkable.
Immunofluorescence summary ∗ for skin biopsy sections from two dogs with ulcerative lesions.

∗ + = linear and continuous immunostaining at the basement membrane zone. DIF = direct immunofluorescence testing; IIF = indirect immunofluorescence testing.
† BP = dog with bullous pemphigoid, used as positive control.
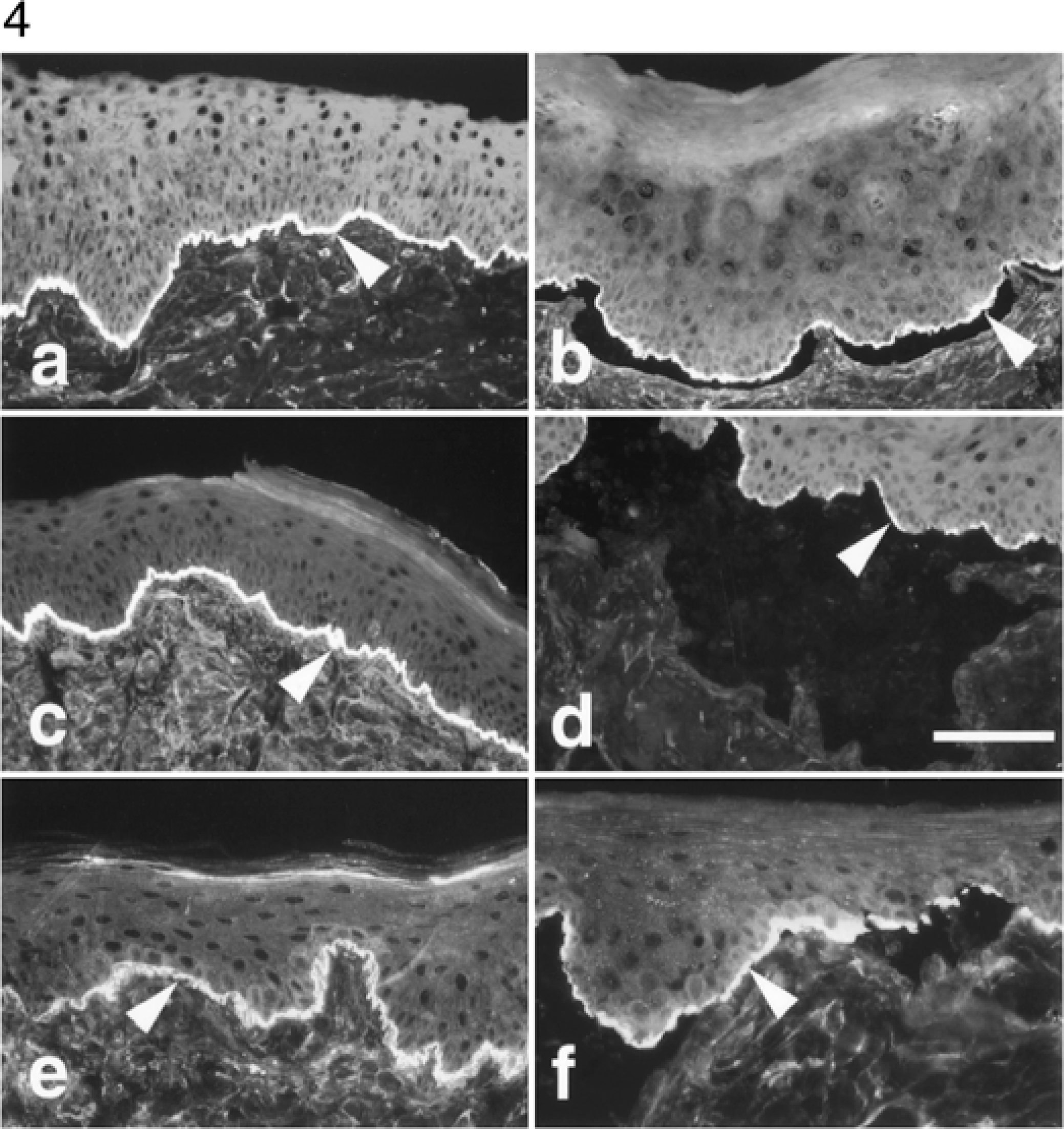
Oral mucosa; dog No. 1. Skin-fixed IgG (Fig. 4a) and IgA (Fig. 4c) are deposited linearly at the epidermal basement membrane zone (arrowheads). Direct IF, anti-dog IgG or IgA-fluorescein. Circulating IgG (Fig. 4b) and IgA (Fig. 4d) autoantibodies label the epidermal side of salt-split normal canine lip. Indirect IF, anticanine IgG or IgA-fluorescein. Anti-LAD-1 monoclonal antibodies label the epidermal basement membrane zone of intact (Fig. 4e) or salt-split (Fig. 4f) epithelium. Indirect IF, mAb 123 antibodies. Bar = 300 µm (Fig. 4a, 4c, 4e) or 150 µm (Fig. 4b, 4d, 4f).
Indirect immunofluorescence testing using normal canine or human lip substrates yielded similar results in both dogs with LAD (Table 3). Circulating IgG and IgA autoantibodies were bound to the epithelial basement membrane in a fine linear and continuous pattern in dog Nos. 1 and 2. In both dogs, the extinction titers were much higher for IgG than for IgA autoantibodies. When canine and human salt-split substrates were used, the IgG or IgA autoantibodies labeled antigen(s) located on the basal aspect of the epidermal side of the artificial clefts (Fig. 4b, 4d). In contrast, the IgG from the dog with BP bound not only to the basal aspect but also faintly to the lateral and apical sides of the stratum basale keratinocytes. Indirect IF testing using the pooled normal canine sera was negative at a 1:20 dilution. The indirect IF pattern seen with the sera of these two dogs with LAD was indistinguishable from that seen in sections immunostained with mAb 123, which is specific for the processed ectodomain of type XVII collagen (Fig. 4e, 4f). This similarity of fluorescence pattern suggested close spatial proximity between 123 epitopes and those targeted by autoantibodies from these canine patients with LAD.
Immunoblotting studies
To further verify the identity of the antigen recognized by autoantibodies from both dogs, the sera were tested by immunoblotting using SCC-25 cell conditioned medium, which has been shown previously to contain LAD-1. 13 The second substrate tested was an SCC-25 cellular extract that contains unprocessed membrane-bound collagen XVII. As shown in Fig. 5, circulating IgA from both dogs recognized a soluble 120-kd antigen (white arrowhead) in the SCC-25 culture medium (lanes 2 and 4) but not in cellular membrane-rich fractions (lanes 1 and 3). A second band of 97 kd was visualized using the serum of dog No. 2 (lane 4). Similar results were obtained for both dogs using the anti-dog IgG secondary reagent (data not shown). The 120-kd band represents LAD-1, the soluble LAD antigen that was also recognized by mAb 123 (lane 6) and the serum of a human individual with LAD (lane 8). The 97-kd band likely is degraded LAD-1. In contrast, circulating IgG from the serum of a dog with BP bound to 180-kd unprocessed collagen XVII (black arrowhead) in the SCC-25 cell fraction (lane 9) but did not recognize any soluble autoantigen (lane 10). Bands were not visualized using the normal canine and human sera.
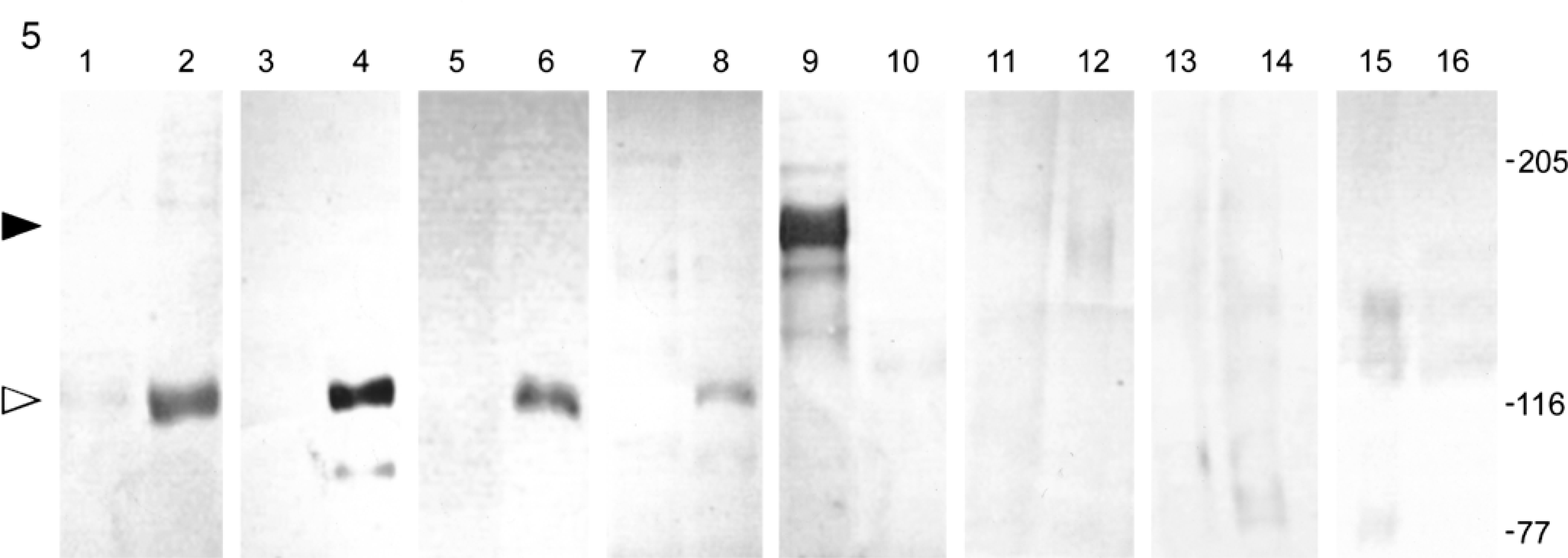
Immunoblot analysis of canine IgA autoantibodies with SCC-25 conditioned medium (lanes 2, 4, 6, 8, 10, 12, 14, 16) or SCC-25 cell fractions (lanes 1, 3, 5, 7, 9, 11, 13, 15) separated on a 7% acrylamide gel. Immunoblotting was performed with the following primary antibodies: lanes 1, 2: 1:40 dilution of serum from dog No. 1; lanes 3, 4: 1:40 dilution of serum from dog No. 2; lanes 5, 6: LAD-1 mAb 123 (10 µg/lane); lanes 7, 8: serum from a human patient with LAD (1:40); lanes 9, 10: serum from a dog with BP (1:40); lanes 11, 12: normal canine serum (1:40); lanes 13, 14: normal human serum; lanes 15, 16: mouse IgA (10 µg/lane). Lanes 1–4, 11, and 12 were incubated with a secondary anti-dog IgA antiserum, and lanes 9 and 10 were incubated with anti-dog IgG antibodies. Lanes 7, 8, 13, and 14 were incubated with anti-human IgA antibodies. Lanes 5, 6, 15, and 16 were incubated with anti-mouse IgG antibodies. Markers to the right represent the estimated molecular mass in kilodaltons. The black arrowhead marks the site of membrane-bound collagen XVII (lane 9), and the white arrowhead marks its processed form (lanes 2, 4, 6, and 8).
Discussion
The present study provides convincing evidence for the existence of a novel canine autoimmune subepidermal blistering dermatosis that is similar to LAD in humans. Canine and human LAD patients have in common the presence of microscopic subepidermal vesicles without inflammation or with neutrophilic infiltration and skin-fixed and/or circulating IgA and IgG autoantibodies that target epitopes situated on LAD-1, the soluble processed form of type XVII collagen. Canine LAD should be added, therefore, to the expanding list of autoimmune blistering dermatoses that can be proposed as animal models of cutaneous diseases of humans.
LAD can affect humans during childhood or adulthood. 26 Most patients afflicted with chronic bullous disease of childhood (CBDC), the juvenile form of LAD, exhibit lesions before 5 years of age. 26 In contrast, adult LAD mostly commences in old age with a peak of prevalence in the 60s. 26 Both of the present dogs with LAD experienced skin lesions in young adulthood. This observation is dissimilar from the two peaks of onset of the human counterpart.
Diversity of clinical features has been reported among humans with either childhood or adult forms of LAD. 26 This phenotypic variation is seen in the extent, distribution of cutaneous versus mucosal involvement, and depth (scarring) of the lesions. 26 Most children with CBDC exhibit large and turgid bullae arising from noninflamed or erythematous underlying tegument. 24 The lesions have a predilection for the extremities, lower abdomen, perioral zone, and oral cavity. 24 Clustering of vesicles commonly is observed. 24 Remarkably, these two dogs with LAD did not exhibit macroscopic vesicles at the time of presentation to the veterinarian. In fact, erosive, ulcerated, and crusted lesions predominated. Oral involvement was present in both of these dogs. Skin lesions seemed to have a predilection for friction or pressure areas, and repeated microtraumas could possibly explain an ephemeral duration of primary cutaneous vesicles. Unfortunately, the clinical signs observed in dog No. 1 resembled those seen in previously reported cases of EBA and type-1 BSLE. 18 , 21 Clinical observations alone therefore cannot reliably distinguish between the different canine autoimmune subepidermal blistering dermatoses.
In both humans and dogs with LAD, histopathologic examination of lesional skin biopsy specimens reveals similar findings. Subepidermal vesicles arise without basal keratinocyte damage and in the presence of absent to moderate inflammation. 3 , 24 , 26 In most human patients with LAD and in dog No. 1, neutrophil emigration into subepidermal vesicles can be detected. 3 , 24 , 26 Unfortunately, neutrophil accumulation in subepidermal microabcesses also is observed in canine cases of EBA and type-1 BSLE. 18 , 21 Skin biopsies of lesional skin, albeit indispensable to the diagnostic investigation of canine autoimmune subepidermal blistering dermatoses, consequently cannot be utilized to differentiate among the various immunologic entities.
For many years, the diagnosis of LAD in humans has been based on the sole observation of either skin-fixed or circulating IgA autoantibodies along the epidermal basement membrane zone. In-depth immunologic investigations recently proved, however, that the antigens targeted by IgA autoantibodies were heterogeneous. 10 , 25 , 29 Circulating IgA autoantibodies were shown to target BP or EBA antigens or epidermal proteins of 120 or 97 kd. 30 Whereas the diagnosis of IgA BP or IgA EBA should be used when autoantibodies target BP or EBA antigens, the term LAD is now reserved for those human patients, either children or adult, who exhibit IgA and/or IgG autoantibodies that recognize the 120/97-kd processed extracellular segment of type XVII collagen. 7 , 13 , 22 , 32–34
Canine patients with IgA deposition at the basement membrane zone have been recognized for almost 20 yr. 23 Because these animals differed in their clinical signs and cutaneous histologic lesions, they were given the diagnosis of BP, discoid lupus erythematosus, or subcorneal pustular dermatitis. 23 More recently, a dog with EBA was shown to exhibit in vivo and circulating IgG and IgA antibodies that targeted type VII collagen. 18 Hence, basement-membrane IgA autoantibodies can be seen in a variety of canine dermatologic entities.
For comparative purposes, we therefore propose to restrict usage of the name LAD to the dermatosis that affects dogs with cutaneous macro- or microvesicles and IgA and/or IgG circulating autoantibodies that target LAD-1, the 120/97-kd processed extracellular type XVII collagen. Prospective observational case studies of canine and feline autoimmune subepidermal blistering dermatoses are presently underway to determine whether or not a classification scheme based on targeted autoantigens will yield pertinent clinical, prognostic, or therapeutic information.
Footnotes
Acknowledgements
We are grateful to Drs. Thelma Gross and Michael Goldschmidt for initial histopathologic diagnosis and referral of the two dogs and to Dr. James Jeffers for providing clinical information on dog No. 2. This study was funded in part by the Office of Research and Development at the VA Palo Alto Health Care System and National Institutes of Health grant AR44-012.
