Abstract
The monoclonal antibody AM-3K, which was developed using human pulmonary macrophages as the immunogen, immunocytochemically labels most human macrophages except for blood monocytes and dendritic cell populations. AM-3K also shows cross-reactivity in some animal species. To evaluate the usefulness of AM-3K, the present study investigated the detailed distribution of AM-3K–immunopositive macrophages in normal and diseased tissues of dogs, cats, horses, cattle, pigs, and rabbits. Zamboni's solution-fixed, paraffinembedded sections were the most available for the immunocytochemistry with AM-3K. In all animal species examined, AM-3K labeled most macrophages in splenic red pulp, lymph node sinuses and thymus, and tissue macrophages in the interstitium of various organs and sites such as the kidneys, lungs, heart, pancreas, intestines, and skin. Alveolar macrophages and perivascular microglial cells were also immunoreactive for AM-3K. Interestingly, Kupffer cells of dogs, cats, and horses were labeled for AM-3K, but those of cattle, pigs, and rabbits were not. Furthermore, in tumor tissues and inflammatory lesions such as liver fibrosis and encephalomalacia that were obtained from dogs, infiltrating macrophages were stained with AM-3K, but not all infiltrating macrophages reacted to AM-3K. In addition, only 30–50% of pulmonary and peritoneal macrophages obtained from cats and dogs were reactive for AM-3K. AM-3K did not react with blood monocytes, dendritic cell populations, and osteoclasts. These observations indicate that AM-3K specifically labels most exudate and tissue macrophages in the animal species examined. However, the expression of antigens recognized by AM-3K on macrophages may be dependent on differential maturation stages or different functions evoked by some conditions. AM-3K immunoreaction products were seen on the cytoplasmic membrane of macrophages by immunoelectron microscopy. AM-3K would be useful for detection of macrophage populations in the animal species examined here.
Macrophages play an important role in inflammation and tumor immunology. 23 , 29 The roles of macrophages in pathologic conditions remain to be evaluated. Immunocytochemical staining with monoclonal antibodies specifically recognizing macrophages is very useful for detection of these cells appearing in diverse lesions. 24 , 33 , 35
In the last decade, monoclonal antibodies specific to macrophages have been generated. In particular, in humans and laboratory animals, several well-characterized antibodies recognizing macrophages have been reported, and these antibodies are commercially available including CD68 for humans; 7 , 10 ED1, ED2, and ED3 for rats; 5 , 6 and F4/80, Mac-1 (M1/70), Mac-2, and Mac-3 for mice. 1 , 21 , 29 , 30 In contrast, to the best of our knowledge, few useful monoclonal antibodies specifically stain monocytes and macrophages in domestic animals such as dogs, cats, horses, cattle, and pigs. Recently, M. Takeya and his colleague developed a novel monoclonal antibody, named AM-3K, by using human pulmonary macrophages as the immunogen. 35 AM-3K immunocytochemically stains most macrophages in human tissues except for blood monocytes and dendritic cell populations. 35 More interestingly, AM-3K was found to react to macrophages present in the spleen and lymph nodes of dogs, cats, horses, cattle, pigs, monkeys, guinea pigs, and rabbits, suggesting broad interspecies reactivity of this antibody. 34 , 35 AM-3K seems to be useful for detection of macrophages in such animal species. However, macrophage populations recognized by AM-3K and the distribution of positive cells in the whole body have not been established in these animals species. In the present study, in order to evaluate the usefulness of AM-3K, the detailed distributions of AM-3K–immunopositive cells in normal and diseased tissues in dogs, cats, horses, cattle, pigs, and rabbits were investigated.
Materials and Methods
AM-3K
Production of AM-3K has been described. 34 , 35 The molecular weights of the antigen recognized by AM-3K in human macrophage lysates were 120 kd and 70 kd, and the isotype of AM-3K was immunoglobulin (IgG)1, kappa.
Tissue and cell preparation
The following normal tissues without any histopathologic lesions were prepared from autopsied or surgically resected cases of dogs, cats, horses, cattle, pigs, and rabbits: liver, kidneys, lungs, heart, thymus, spleen, lymph nodes, pancreas, small and large intestines, skin, brain (cerebrum and cerebellum), spinal cord, and bone marrow. Inflammatory or tumor lesions obtained from dogs at autopsy or biopsy were examined as pathologic tissues. The inflammatory lesions involved liver fibrosis (n = 4), nonsuppurative interstitial nephritis (4), encephalomalacia (2), cutaneous granulomas (2), and arthritis (1). Tumor tissues examined consisted of malignant melanomas (3), malignant fibrous histiocytoma (MFH) (1), hemangiopericytomas (3), malignant schwannomas (2), fibrosarcoma (1), leiomyosarcoma (1), rhabdomyosarcoma (1), osteosarcoma (1), urinary bladder transitional cell carcinoma with bone metaplasia (1), mast cell tumors (2), and cutaneous histiocytomas (3); the criteria of diagnosis was based on those described by Moulton. 16 These samples were fixed in 10% neutral buffered formalin or in Zamboni's fixative (0.21% picric acid, 2% paraformaldehyde, and 130 mM phosphate buffer, pH 7.4) for 1–3 days, embedded in paraffin, and sectioned at 3–5 µm. Sections were stained with hematoxylin and eosin for histopathologic examinations. They were also stained immunocytochemically with AM-3K as described below. To identify osteoclasts, selected sections from transitional cell carcinoma with bone metaplasia and osteosarcoma were histochemically stained by the tartrate-resistant acid phosphate staining. 8 , 22
Smears of peripheral blood cells from healthy dogs were made on slide glasses. The slides were fixed with cold acetone for 5 minutes, and stained for AM-3K immunocytochemistry.
Pulmonary macrophages were collected by bronchoalveolar lavage of healthy dogs and cats, and peritoneal macrophages were obtained from dogs with heart failure. These cells were incubated in culture flasks (75 mm2 in surface area) for 3 hours at 37 C in a humidified 5% CO2 incubator. The medium used was RPMI 1640 (Nissui, Tokyo, Japan) supplemented with 10% bovine fetal serum (Bioserum, UBC, Japan), 0.03%
Immunocytochemistry
According to previous papers, 34 , 35 cryostat tissue sections that were fixed with 2% periodate–lysine–paraformaldehyde solution, or tissue sections that were fixed in buffered formalin solution and then processed routinely for paraffin embedding, were recommended for use for immunocytochemistry with AM-3K. First, in the present study, formalin-fixed, paraffin-embedded tissue sections were evaluated for AM-3K immunostaining. However, the immunostaining results on such sections for AM-3K were unstable, as mentioned by Zeng et al. 35 Because our laboratory has used Zamboni's solution as a fixative for immunostaining, 27 we tried to fix tissue samples in Zamboni's solution, and found that this fixative is more appropriate for immunocytochemistry using the AM-3K antibody.
Zamboni's solution–fixed, paraffin-embedded tissue sections were immunostained by using the indirect immunoperoxidase method. Briefly, after treatments with 0.1% trypsin solution at 37 C for 15 minutes, sections were incubated with 3% H2O 2 to quench endogenous peroxidase and then with 1.5% skim milk for 40 minutes at room temperature to inhibit nonspecific reactions. Thereafter, the sections were reacted for 24–48 hours at 4 C with culture supernatant (1:1) of AM-3K or ascites diluted 1:100 as a primary antibody. 34 The peroxidase-conjugated sheep anti-mouse Ig (Fab′) 2 fragment diluted 1:100 (Amersham, Poole, UK) was reacted to sections for 1.5 hours at 37 C as a secondary antibody. The positive reactions resulted in brown staining with the substrate 3,3′-diaminobenzidine tetrahydrochloride (DAB), and the sections were counterstained with hematoxylin. Similar immunostaining results could be obtained by using the avidin–biotin complex (ABC) method, in which labeled streptavidin biotin (LSAB) kit (DAKO, Santa Barbara, CA, USA) was used. 26 Briefly, the sections were incubated with AM-3K as described above, followed by the application of ABC reagents for 30 minutes at room temperature; the products were visualized with DAB. Pulmonary and peritoneal macrophages fixed in acetone were also immunostained by the ABC method. Tissue sections that were incubated with phosphate-buffered saline (PBS) or mouse nonimmune sera instead of primary antibody were used as negative controls.
An antibody against lysozyme has been used to detect macrophages in domestic animals. 18 , 25 In order to compare AM-3K–positive cells with lysozyme-immunopositive cells, the double immunocytochemical stainings were carried out on hepatic fibrosis sections. The sections were first incubated with AM-3K, processed by the ABC method, and then immersed in DAB to yield a brown reaction product. Sections were then reacted with anti-lysozyme polyclonal antibody (undiluted; DAKO) for 14 hours at 4 C, and incubated with a secondary antibody of peroxidase-conjugated affinipure goat anti-rabbit IgG (H+L) (1:5,000; Jackson Immunoresearch Laboratory, West Grove, PA, USA) for 1 hour at 37 C. The positive reactions for anti-lysozyme antibody were stained blue with true blue-peroxidase substrate (Kirkegaard & Perry Laboratories, Gaithersburg, Maryland, USA). An antibody against S-100 protein labels dendritic cells in lymph nodes as well as neural tissues such as neural, glial, and Schwann cells. 4 , 35 The double immunocytochemistry with AM-3K and rabbit anti–cow S-100 protein antibody (undiluted; DAKO) was conducted on lymph nodes obtained from dogs and cattle. After the immunostaining with anti–S-100 protein antibody by ABC methods, the immunostaining with AM-3K was carried out using the alkaline phosphatase anti-alkaline phosphatase (APAAP Kit, DAKO) methods. Thus, the positive products for anti–S-100 protein antibody were stained brown, and those for AM-3K were red.
Immunoelectron microscopy
Pulmonary macrophages collected as described above were fixed in 2.5% buffered glutaraldehyde for 30 minutes, and immunostained by the indirect immunoperoxidase method. Briefly, after treatment with 1.5% skim milk for 3 hours at room temperature, cell pellets were incubated with AM-3K for 2 days at 4 C, and subsequently with the secondary antibody described above for 3 hours at room temperature. They were immersed in DAB solution, and the DAB reaction was performed by adding 3% H2O2. The reacted cells were then postfixed with 1% osmium tetroxide for 1 hour. The cell pellets were dehydrated with alcohol and embedded in epoxy resin, according to the usual procedures for electron microscopy. 32 The ultrathin sections were observed in an electron microscope (Hitachi H-600, Tokyo, Japan) at 75 kV.
Protein extraction and Western blot analysis
Protein extraction and Western blot analysis were carried out according to the methods described by Tsukamoto et al. 27 Freshly isolated pulmonary or peritoneal macrophages of dogs and cats were homogenized in PBS with a polytron homogenizer. The homogenate was centrifuged at 15,000 rpm for 10 minutes at 4 C. The pellets were solubilized by incubation in 10 mM Tris-acetate buffer (pH 8.0) (including 1 mM ethylenediaminetetraacetic acid and 0.5% Nonidet P-40) for 2.5 hours at room temperature on a rotating shaker and then centrifuged at 40,000 rpm for 90 min. The resultant solubilized membrane fractions were analyzed by Western blotting. The samples were separated in 10% sodium dodecyl sulfate–polyacrylamide gels under reducing or nonreducing conditions, and electrotransferred to a polyvinyl difluoride (PVDF) membrane. The blots were blocked with 2% skim milk in 10 mM Tris-HC1 buffer (pH 8.0) containing 150 mM NaCl (TS buffer), and incubated with AM-3K supernatant (1:2) for 1 hour at room temperature. After washing with TS buffer containing 0.05% Tween 20 four times and then with TS buffer three times, the blots were incubated in biotinylated anti-mouse IgG and in avidin-biotinylated peroxidase complex (LSAB Kit). Peroxidase activity was visualized using DAB as a substrate. For molecular size in kd, a prestained marker was electrotransferred on the PVDF membrane.
Results
Distribution of AM-3K–immunopositive macrophages
The distribution of AM-3K–immunopositive macrophages in lymphoid organs is shown in Table 1. The distribution patterns were very similar among animal species examined. In the spleen, AM-3K–positive cells were frequently seen in the red pulp, whereas the number of AM-3K–positive cells was very small in the white pulp (germinal center, mantle zone, and marginal zone) (Fig. 1). In the lymph node, many AM-3K–positive cells were present in the subcapsular and medullary sinuses (Fig. 2), and infrequently in the lymphoid follicle (germinal center and paracortical zone). These AM-3K–positive cells in the splenic red pulp and subcapsular and medullary sinuses in the lymph node were large and round or oval in shape. In the thymus, AM-3K–positive cells were more frequently seen in the medulla than in the cortex. In these organs, spindle-shaped tissue macrophages reacting to AM-3K were sporadically seen in the capsule and interstitial connective tissues (trabecules in the spleen and lymph node). In the lymph node, the double immunostainings revealed that S-100 protein–positive interdigitating cells in the paracortex and sinuses did not react to AM-3K (Fig. 2).
Distribution of AM-3K–immunopositive macrophages in lymphoid organs. ∗

∗ + = infrequently; 2 + = occasionally/less frequently; 3 + = frequently.
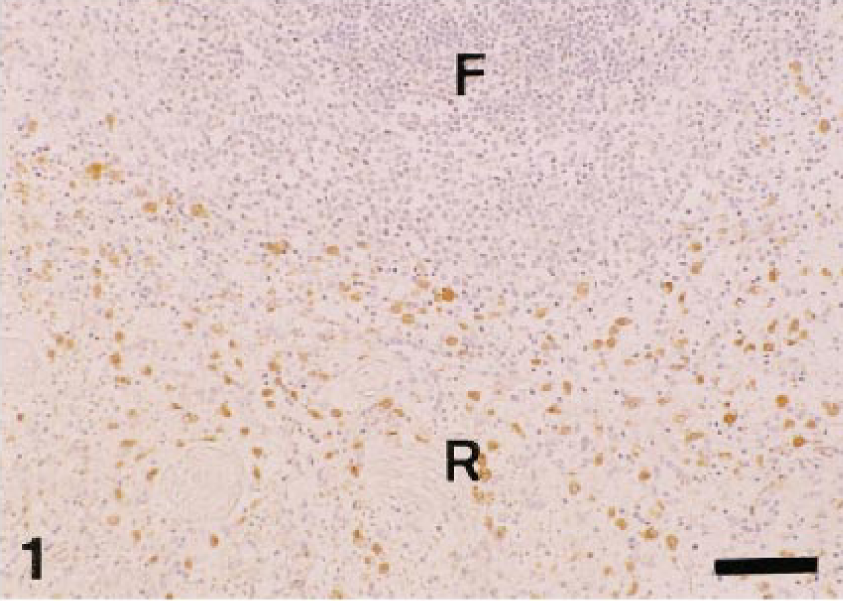
Dog. Spleen; many AM-3K–positive macrophages (brown reaction) are seen in the red pulp (R), whereas very few positive cells are present in the lymphoid follicle (F). AM-3K immunostaining, counterstained with hematoxylin. Bar = 60 µm.
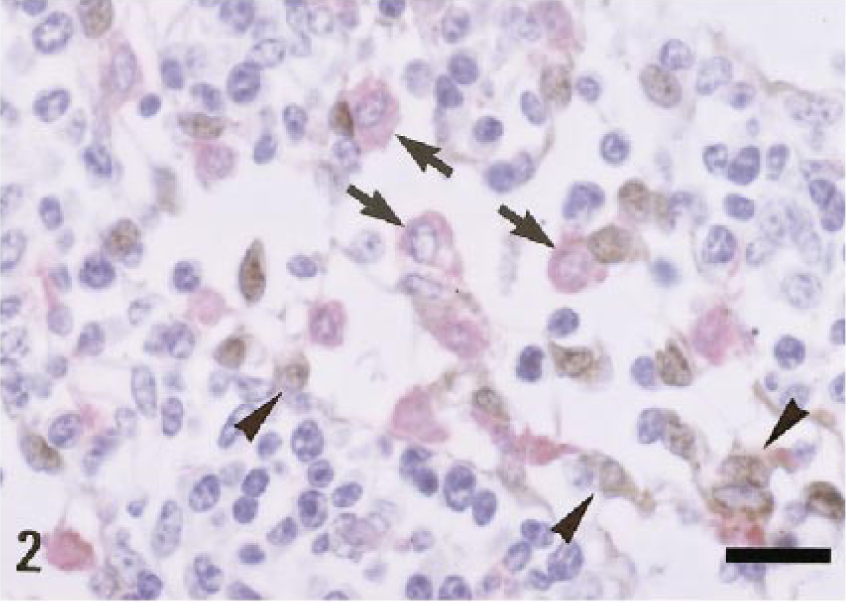
Cattle. Lymph node; many AM-3K–positive macrophages are seen in the medullary sinuses (arrows, red reaction), but anti–S-100 protein antibody–positive cells (arrowheads, brown reaction) do not correspond to AM-3K–positive macrophages. Lymphocytes are negative for AM-3K or anti–S-100 protein antibody. Double immunostaining with AM-3K and anti–S-100 protein antibody, counterstained with hematoxylin. Bar = 20 µm.
Table 2 shows the distribution of AM-3K–positive macrophages in nonlymphoid organs and sites. In the liver, AM-3K–positive cells were seen frequently in hepatic lobules of dogs and cats, and less frequently in horses; these cells were spindloid, round, or stellate in shape, and were present along the sinusoids, indicating that the positive cells were Kupffer cells (Fig. 3). On the other hand, no cells reacting to AM-3K were detected in hepatic lobules of cattle, pigs, and rabbits (Table 2). In the portal tract, a few AM-3K–positive cells were seen in all animals examined. In other organs and sites listed in Table 2, no differences were found in distribution and staining intensity of AM-3K–positive cells among animal species examined. Tissue macrophages in the interstitium of the kidneys and pancreas, in the intestinal mucosa (particularly in the lamina propria mucosa) and muscularis, in the meninx of the brain and spinal cord, and in the dermis were reactive for AM-3K. AM-3K–positive cells were also present among cardiac muscle fibers (Fig. 4). The majority of these tissue macrophages were spindle-shaped, but occasional cells were round. No cells were reactive for AM-3K in the cortex and medulla (parenchyma) of brain and spinal cord, whereas occasional cells reacting to AM-3K were seen around blood vessels in the brain and spinal cord (Fig. 5); these cells were considered to be perivascular microglial cells (Mato's fluorescent granular perithelial cells). 15 Alveolar macrophages in alveoli (Fig. 6) and interstitial macrophages around bronchioles were positive for AM-3K. No cells reacting to AM-3K were detected in the epidermis and glomerulus.
Distribution of AM-3K–immunopositive macrophages in nonlymphoid organs/sites. ∗
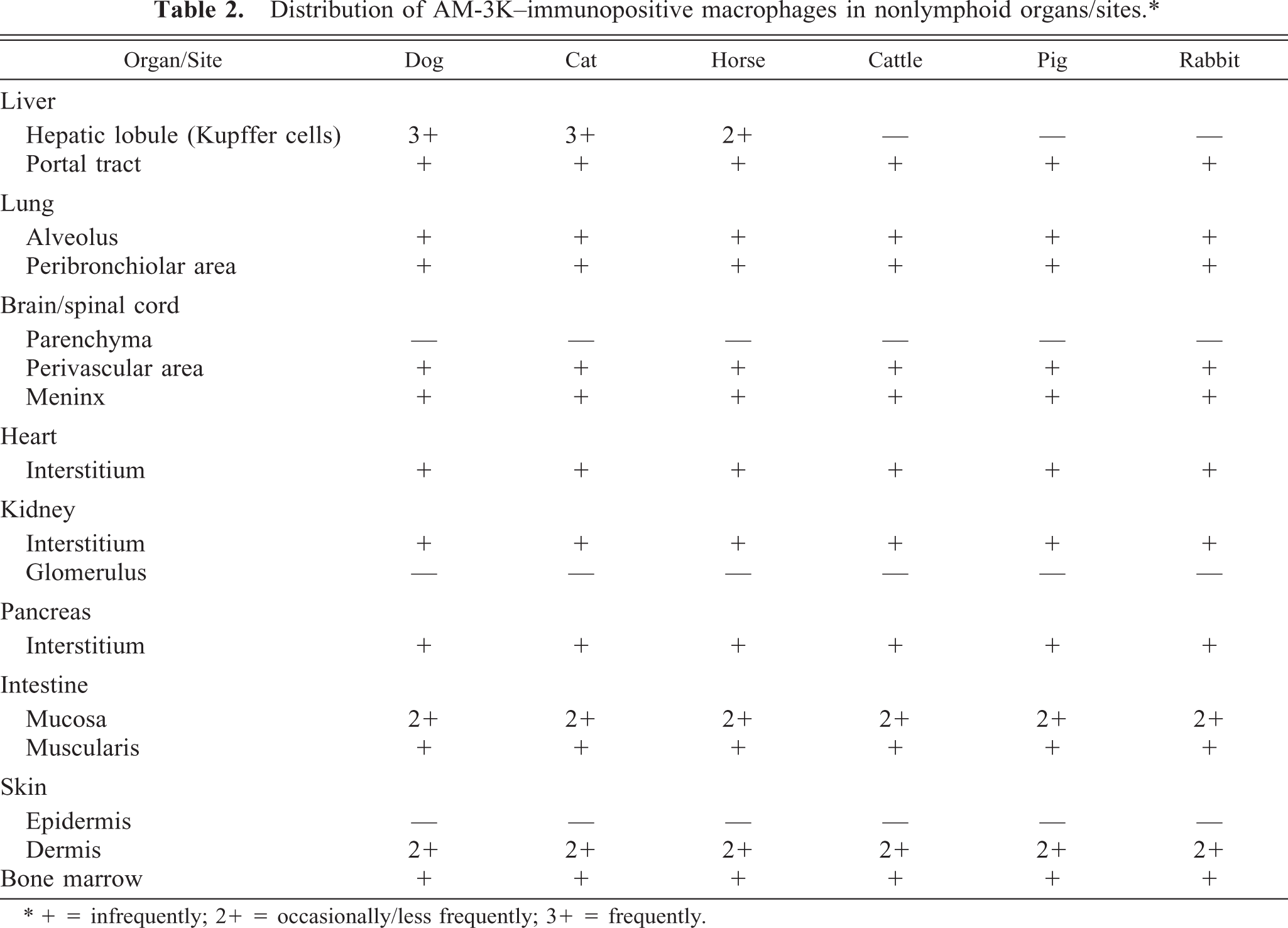
∗ + = infrequently; 2+ = occasionally/less frequently; 3+ = frequently.
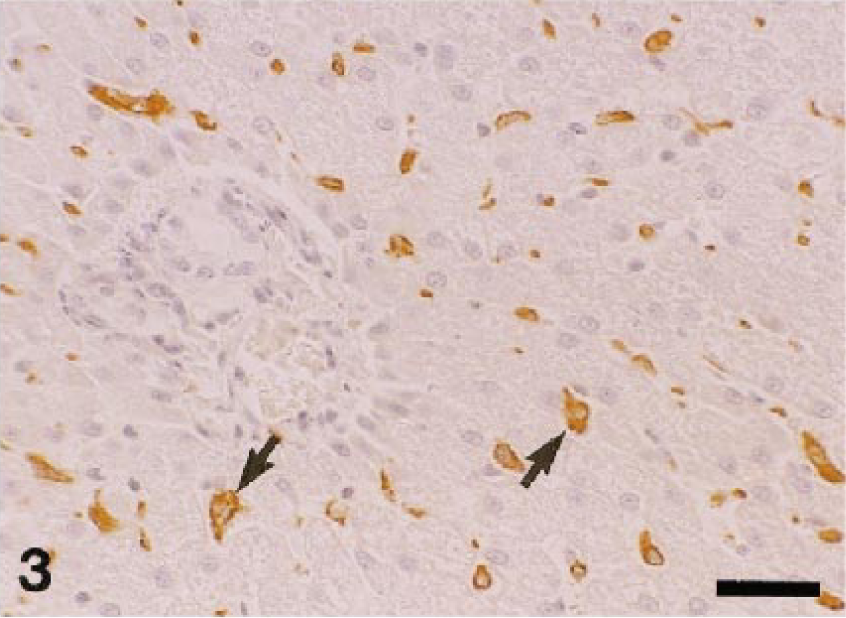
Dog. Liver; Kupffer cells present along the sinusoids are reactive for AM-3K (arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 40 µm.
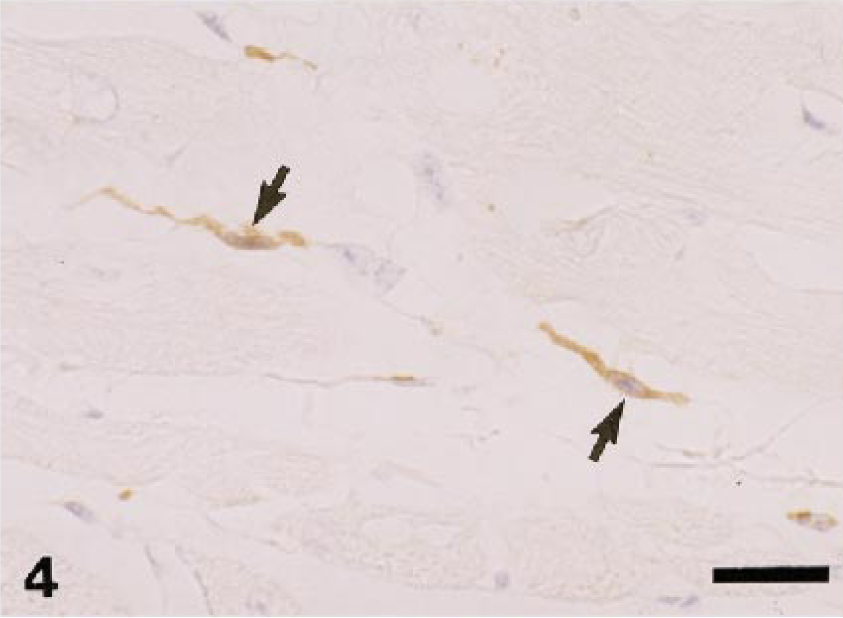
Dog. Heart; spindle-shaped, interstitial macrophages in the myocardium react to AM-3K (arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 20 µm.
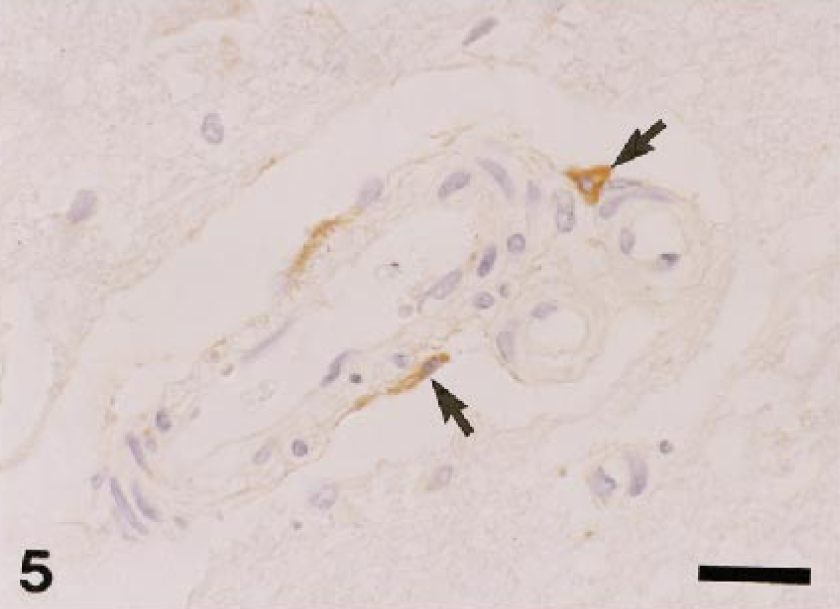
Dog. Spinal cord; perivascular microglial cells react to AM-3K (arrow). AM-3K immunostaining, counterstained with hematoxylin. Bar = 20 µm.
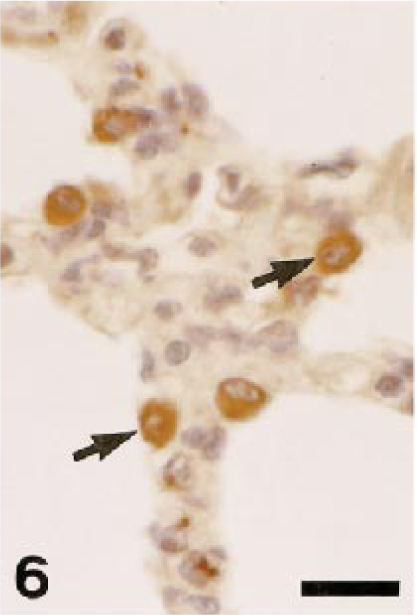
Pig. Lung; alveolar macrophages are positive for AM-3K (arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 20 µm.
On smears from canine peripheral blood, neither monocytes nor other white blood cells were positive for AM-3K. Thirty to 50% of pulmonary and peritoneal macrophages from dogs and cats were labeled with AM-3K; some pulmonary macrophages reacting to AM-3K contained phagocytized coal-dust–like particles (anthracosis) (Fig. 7).
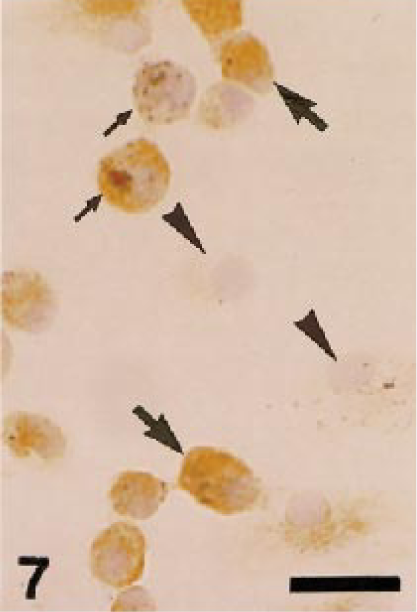
Dog. Pulmonary macrophages collected by bronchoalveolar lavage; macrophages react to AM-3K (large arrows), but some are negative for AM-3K (arrowheads). These macrophages phagocytize coal-dust–like particles (small arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 20 µm.
In inflammatory lesions (liver fibrosis, interstitial nephritis, encephalomalacia, cutaneous granuloma, and arthritis), many infiltrating macrophages were intensely stained with AM-3K. Interestingly, gitter cells seen in encephalomalacia were reactive for AM-3K, some of which were rod-shaped; however, gitter cells that did not react to AM-3K were also present in the lesion (Fig. 8). In fibrotic livers, infiltrating macrophages labeled with both AM-3K and anti-lysozyme antibody, or macrophages reacting to either AM-3K or anti-lysozyme antibody were present (Fig. 9). Neoplastic cells of malignant melanoma, hemangiopericytoma, malignant schwannoma, fibrosarcoma, leiomyosarcoma, rhabdomyosarcoma, osteosarcoma, mast cell tumor, and transitional cell carcinoma were negative for AM-3K, but reactive macrophages in and around tumor tissues were positive for AM-3K. Neoplastic cells of MFH and canine cutaneous histiocytomas were also negative for AM-3K, but a small number of infiltrating macrophages reacted with AM-3K. Multinucleated osteoclasts seen in a osteosarcoma and metaplastic bone formation in a transitional cell carcinoma were negative for AM-3K (Fig. 10); these osteoclasts were positively stained by the tartrate-resistant acid phosphatase staining, indicating that these cells are osteoclasts. 8 , 22
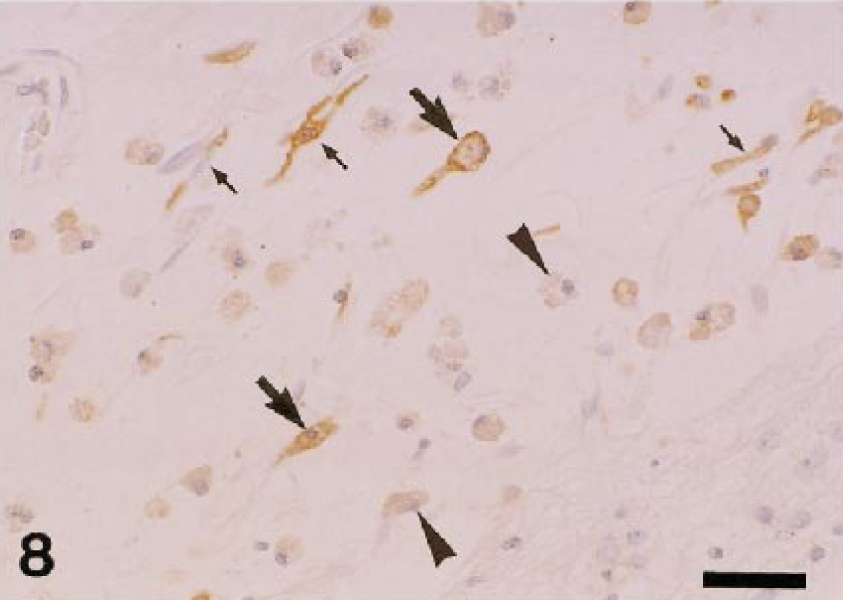
Dog. Encephalomalacia; gitter cells react to AM-3K (large arrows), but some gitter cells also do not react to AM-3K (arrowheads). Rod-shaped macrophages are reactive for AM-3K (small arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 40 µm.
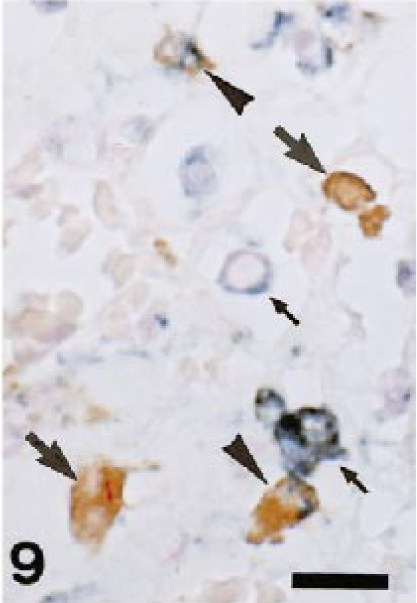
Dog. Liver fibrosis; infiltrating macrophages reacting to both AM-3K and anti–lysozyme-antibody (arrowheads; they have blue and brown reactions in the cytoplasm), or macrophages reacting to either AM-3K (large arrows, brown reaction) or anti–lysozyme-antibody (small arrows, blue reaction) are present. Double immunostaining with AM-3K and anti–lysozyme-antibody, counterstained with nuclear fast red (Kernechtrot). Bar = 20 µm.
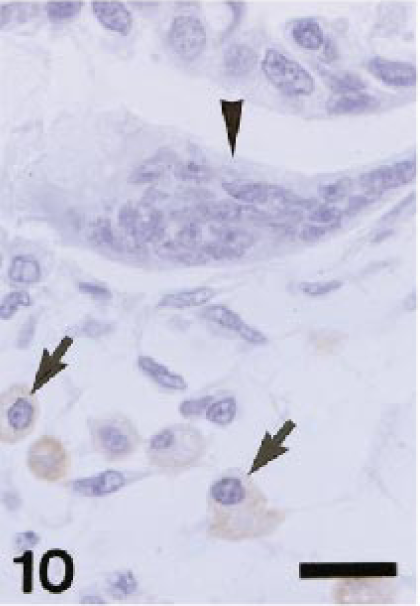
Dog. A transitional cell carcinoma with bone metaplasia; in area with bone metaplasia, multinucleated osteoclasts are negative for AM-3K (arrowhead), but infiltrating macrophages are positive for AM-3K (arrows). AM-3K immunostaining, counterstained with hematoxylin. Bar = 20 µm.
Immunoelectron microscopic localization of AM-3K
AM-3K immunoreaction products were seen on the cytoplasmic membrane of canine pulmonary macrophages (Fig. 11).
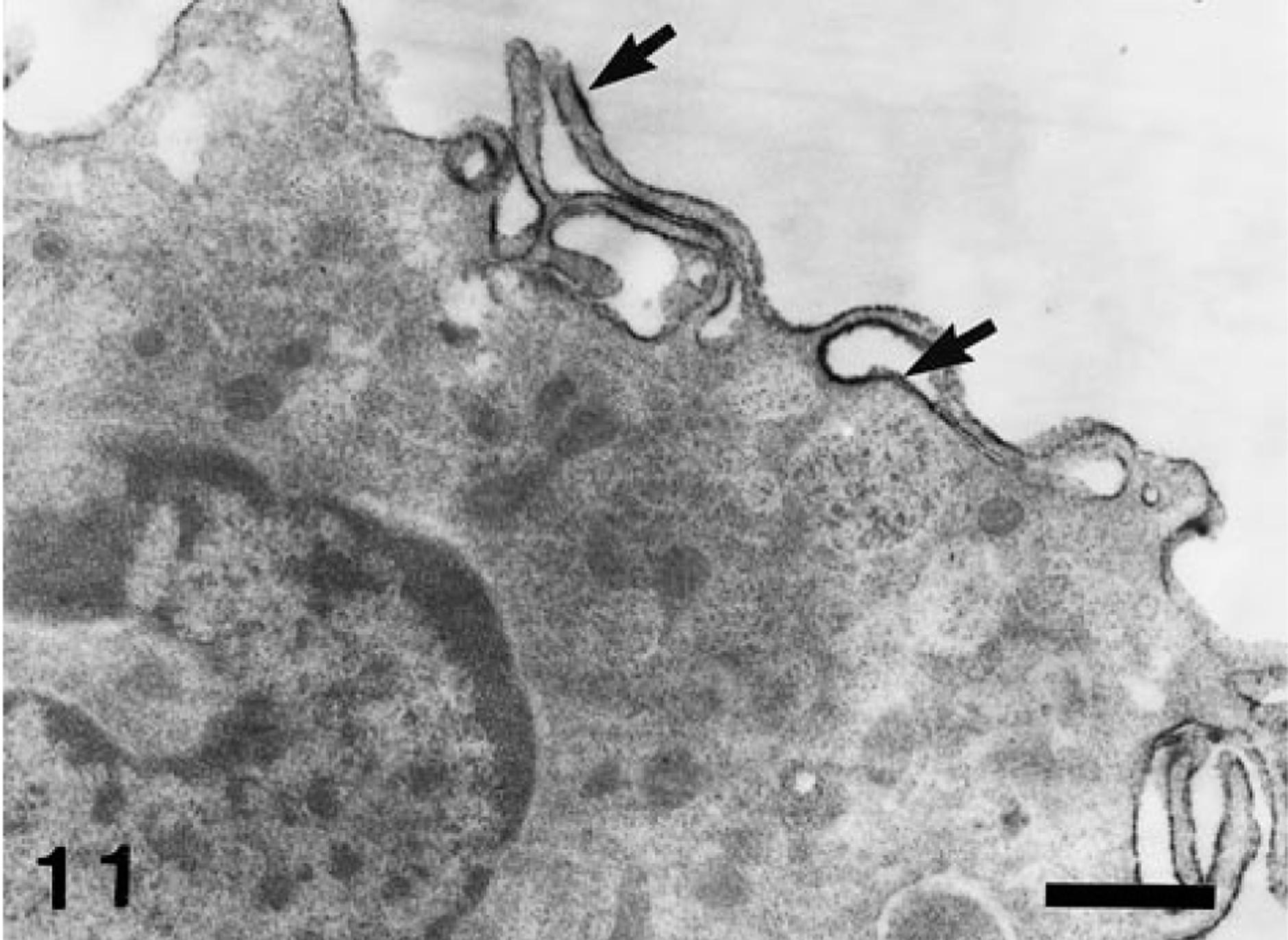
Dog. Pulmonary macrophages; AM-3K–positive products are located on the cell membrane (arrows). Immunoelectron microscopy for AM-3K. Bar = 0.5 µm.
Western blot analysis of the AM-3K antigen
Under reducing conditions, AM-3K stained one band with a molecular weight of approximately 70 kd in membrane fractions from peritoneal and pulmonary macrophages of dogs and cats (Fig. 12). Under nonreducing conditions, no positive band against AM-3K was detected (data not shown).
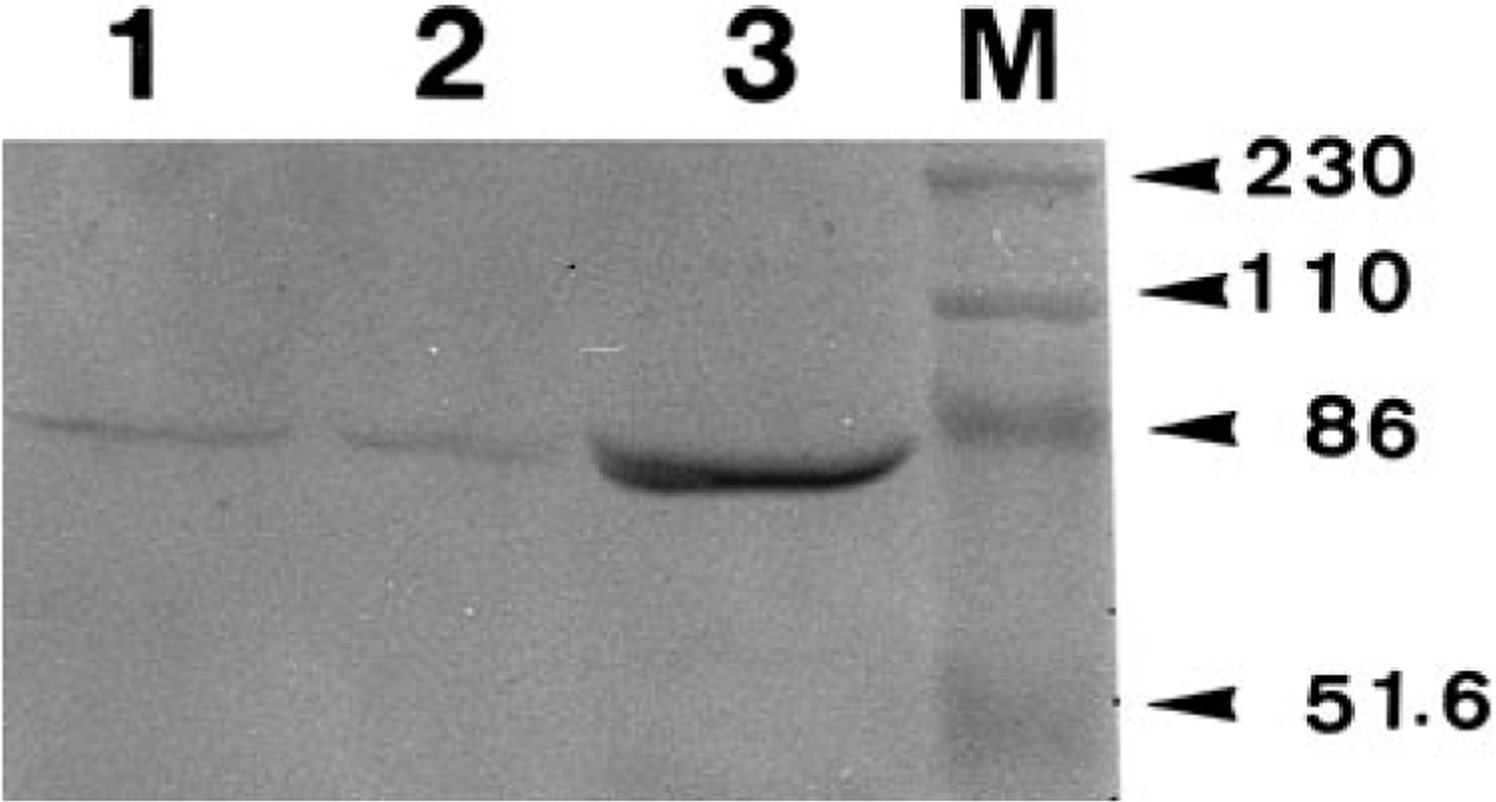
Western blot analysis. AM-3K reacts to a band approximately 70 kd in molecular weight. Lanes 1, 2, and 3 are samples from canine peritoneal macrophages, canine pulmonary macrophages, and feline pulmonary macrophages, respectively. Lane M is standard molecular markers of kd (arrowheads).
Discussion
Western blot analysis for AM-3K revealed a 70-kd band on pulmonary and peritoneal macrophages of dogs and cats. A 70-kd band has been also detected on human pulmonary macrophages and spleen cells of guinea pigs and rabbits. 34 , 35 In human macrophages, a 120-kd band was also seen. 35 Such a band could not be detected in dogs, cats, guinea pigs, and rabbits. 34 AM-3K immunoreaction products were located on cytoplasmic membrane of both human 35 and canine pulmonary macrophages, indicating that AM-3K recognizes an epitope of membrane antigens. 34 , 35 The function of antigens recognized by AM-3K and the difference in the 120-kd band between humans and the animal species examined are now under study.
Macrophages encompass exudate macrophages, resident macrophages (so-called histiocytes), and cells differentiating into dendritic cells. 6 , 23 , 28 Although controversy still remains over the derivation and the relationship between these macrophage subpopulations, the development, differentiation, and maturation of these types of macrophages are different from each other. 6 , 23 , 28 The exudate macrophages are derived from blood monocytes through monocytic lineage from hematopoietic stem cells. Ontogenetically, the precursors of resident macrophages migrate from bone marrow into connective tissues and then differentiate into resident macrophages. The precursors of dendritic cells are still unclear. The dendritic cell populations include interstitial dendritic cells, interdigitating cells in the lymph node, and Langerhans cells of the epidermis. 23 , 28
In humans, AM-3K reacted with most macrophages in the lymph node, thymus and red pulp of the spleen, and with interstitial macrophages in connective tissues of various organs and sites. 35 Analysis of our data showed that the distribution of AM-3K–immunopositive macrophages in lymphoid organs and nonlymphoid organs and sites of dogs, cats, horses, cattle, pigs, and rabbits was generally similar to that in humans (Table 1, 2). In humans, infiltrating macrophages in atherosclerosis, Gauchers's disease, and some tumors were also positive for AM-3K. 35 In the present study, macrophages infiltrating in inflamed sites and neoplastic lesions were reactive for AM-3K. These findings suggest that AM-3K reacts with most resident and exudate macrophages in humans as well as the animal species examined here.
Although infiltrating macrophages are considered to be recruited from blood monocytes and/or resident macrophages, 23 AM-3K did not react with blood monocytes of dogs. In humans, blood monocytes were also negative for AM-3K. 35 Only 30–50% of pulmonary and peritoneal macrophages from dogs and cats were labeled with AM-3K. In fibrotic livers, macrophages labeled with anti-lysozyme antibody did not always correspond to AM-3K–positive cells; some macrophages were positive for both anti-lysozyme antibody and AM-3K. The expression of antigens recognized by AM-3K on macrophages may be dependent on the differentiation stage or be evoked by microenvironmental conditions. AM-3K has been speculated to label an epitope of macrophages at the late maturation stage. 35
Microglial cells are a special kind of macrophage. 23 , 29 Perivascular (Mato's cells) and perineural resting (ramified) microglial cells exist in the brain and spinal cord; these cells may be derived from circulating monocytes or precursor cells related to the monocyte–macrophage lineage that originate in the bone marrow and settle in the brain tissue during embryonic life. 3 , 9 , 14 , 20 In the present study, perivascular microglial cells were positive for AM-3K, but no reactivity for AM-3K was found in perineural resting microglia in the parenchyma. Gitter cells in encephalomalacia gave positive reactions for AM-3K, but not all cells reacted to AM-3K. Gitter cells may be derived from resting microglial cells. 3 , 12 Reactive microglia in diseased brain develop elongated shapes and are called rod cells. 12 AM-3K–positive, rod-shaped cells were seen in encephalomalacia. These findings reflect the heterogeneity of microglial cells, only some of which express the AM-3K antigen.
Kupffer cells in the liver are a type of resident macrophage. 23 , 28 Kupffer cells of humans 35 as well as in dogs, cats, and horses in this study reacted to AM-3K. On the contrary, Kupffer cells of cattle, pigs, and rabbits did not react with AM-3K, a finding confirmed by Zeng et al. 34 for rabbits and guinea pigs. Heterogeneity seemed to occur in AM-3K immunoreactivities of Kupffer cells between animal species. In cattle, rabbits, and guinea pigs that revealed a negative reaction of Kupffer cells to AM-3K, most macrophages in the lymph node and spleen and interstitial macrophages in various organs and sites were reactive for AM-3K (Table 1, 2). Macrosialin, which is located on the cell membrane of mouse macrophages, is a homologue of human macrophage glycoprotein CD68. The expression of CD68 antigen was found to be more restricted in dendritic cells than in tissue macrophages in the mouse. 11 Therefore, the expression of antigens recognized by AM-3K on Kupffer cells might be restricted in cattle, pigs, and rabbits, presumably depending on biochemical or functional differences of these antigens.
Anti–S-100 protein antibody is used for detecting interdigitating cells in the lymph node. 4 , 32 , 35 The present double immunostaining of lymph nodes failed to demonstrate that anti–S-100 protein antibody–positive interdigitating cells correspond to AM-3K–positive cells. Our findings that no cells were reacting to AM-3K in the epidermis suggest negative reactions of Langerhans cells to AM-3K. Likewise, in humans, AM-3K did not react with dendritic cell populations. 35 In addition, osteoclasts showed no reaction to AM-3K, although osteoclasts are generated from blood monocytes. 8 , 23 Neoplastic cells of MFH and canine cutaneous histiocytoma have been considered to have a histiocytic nature; 16 , 18 , 25 however, these tumor cells were negative for AM-3K.
In short, the present study demonstrated that AM-3K reacts with most exudate and resident macrophages, alveolar macrophages, and perivascular microglia in dogs, cats, horses, cattle, pigs, and rabbits, and that the expression of antigens recognized by AM-3K may depend on functional differences or different maturation stages. Furthermore, AM-3K characteristically reacts with Kupffer cells of dogs, cats, and horses. In contrast, blood monocytes, dendritic cell populations, and osteoclasts were not labeled by AM-3K immunostaining.
In humans, CD68 antibodies such as EBM/11, KP1, and Ki-M6 are considered to be the most reliable markers for detection of monocytes and macrophages. 7 , 10 , 13 , 17 , 19 However, the tissue distribution of CD68-positive cells is different from that of AM-3K–positive cells; the CD68 antibodies recognize dendritic cell populations and blood monocytes as well as exudate and tissue macrophages. 35 Several antibodies including PM-2K, 24 X4, 31 X14, 31 Ber-MAC3, 2 and EBM/11 13 showed various interspecies reactivities for macrophages in domestic and laboratory animals. 34 Most of these antibodies must be applied on cryostat sections, and distribution of positive cells throughout the body in domestic animals has not been determined. Immunocytochemical stainings with AM-3K can be performed on Zamboni's solution–fixed, paraffin-embedded sections; this method provides more well-conserved histology than cryostat sections. AM-3K may become a useful antibody to detect exudate and resident macrophages in various pathologic conditions in the animal species examined in the present study.
