Abstract
Meningiomas are common primary brain tumors in dogs; however, little is known about the molecular genetic mechanisms involved in their tumorigenesis. Several tumor suppressor genes have been implicated in meningioma pathogenesis in humans, including the neurofibromatosis 2 (NF2), protein 4.1B (4.1 B), and tumor suppressor in lung cancer-1 (TSLC1) genes. We investigated the expression of these tumor suppressor genes in a series of spontaneous canine meningiomas using quantitative real-time reverse transcription polymerase chain reaction (RT-PCR) (NF2; n = 25) and western blotting (NF2/merlin, 4.1B, TSLC1; n = 30). Decreased expression of 4.1B and TSLC1 expression on western blotting was seen in 6/30 (20%) and in 15/30 (50%) tumors, respectively, with 18/30 (60%) of meningiomas having decreased or absent expression of one or both proteins. NF2 gene expression assessed by western blotting and RT-PCR varied considerably between individual tumors. Complete loss of NF2 protein on western blotting was not seen, unlike 4.1B and TSLC1. Incidence of TSLC1 abnormalities was similar to that seen in human meningiomas, while perturbation of NF2 and 4.1B appeared to be less common than reported for human tumors. No association was observed between tumor grade, subtype, or location and tumor suppressor gene expression based on western blot or RT-PCR. These results suggest that loss of these tumor suppressor genes is a frequent occurrence in canine meningiomas and may be an early event in tumorigenesis in some cases. In addition, it is likely that other, as yet unidentified, genes play an important role in canine meningioma formation and growth.
Keywords
Introduction
Meningiomas are one of the most common central nervous system (CNS) tumors in dogs and account for approximately 40% of all primary intracranial neoplasms in this species. 30 Canine meningiomas bear striking similarities to their human tumor counterparts, particularly in their histopathologic characteristics, although an increased incidence of higher grade (specifically grade II/atypical) tumors has been reported in dogs compared with humans. 31 Location and magnetic resonance imaging characteristics of canine meningiomas are also similar to their human tumor counterparts, although canine intraspinal meningiomas predominantly arise in the cervical rather than thoracic region as is reported in humans. 26,31 Molecular and genetic characteristics of canine meningiomas have been poorly defined at this time, and it is unclear whether the similar histopathologic and imaging characteristics of canine and human meningiomas will be reflected in similar findings at the molecular level. Preliminary data suggest that this may be the case; initial microarray analysis of canine meningiomas identified several genes with similar expression patterns to human tumors, 35 and expression of progesterone receptors in canine meningiomas has been associated with a lower proliferative index and improved prognosis as reported in humans. 22,33 Telomerase activity, as assessed by expression of telomerase reverse transcriptase (TERT), has also been demonstrated in canine meningiomas, 19,23 and mirrors findings seen in human tumors. A variety of growth factors and their receptors have been shown to have a significant role in meningioma pathogenesis in humans, including vascular endothelial growth factor (VEGF) and epidermal growth factor (EGF), and while data in canine meningiomas are incomplete, findings appear to be similar to human reports. 4,5,27,28 In addition to many pro-oncogenic factors such as those listed above, meningioma pathogenesis has been associated with altered or loss of function of a variety of tumor suppressor genes. Following on from initial studies investigating the association of familial neurofibromatosis type 2 (NF2) and meningiomas, a family of putative tumor suppressors (protein 4.1 family) and associated proteins have been identified and implicated in the development and progression of meningiomas in humans. The protein 4.1 superfamily contains several proteins, including merlin (also called schwannomin, neurofibromin 2), 4.1B, and 4.1R, that have growth regulatory functions and are thought to act as tumor suppressors, possibly by association at the cell surface with transmembrane proteins such as CD44 and TSLC1 (tumor suppressor in lung cancer-1). Merlin is the protein product of the NF2 gene, and familial loss of the gene is associated with the NF2 inherited cancer syndrome. 29 Individuals affected with NF2 are prone to the development of schwannomas and multiple meningiomas. 21 In humans, loss of NF2 expression has been observed in 30–80% of sporadic meningiomas, usually associated with loss of heterozygosity (LOH) on chromosome 22q and bi-allelic inactivation of the NF2 gene. 6,11,15,17,18,25,36,38 In addition, some reports have suggested that NF2 inactivation defines distinct molecular variants of meningioma, with NF2 protein loss being less common in the meningothelial subtype.∗ Similarly, loss of expression of 4.1B (also termed DAL-1, “differentially expressed in adenocarcinoma of lung”) also has been reported in up to 76% of sporadic meningiomas, 7,24,25 and loss of TSLC1 expression, a protein that has been shown to interact with 4.1B, 40 has been found in 30–85% sporadic meningiomas. 32 Data are not available regarding expression of these tumor suppressor genes in spontaneous canine meningiomas. Comprehensive characterization of similar molecular pathways involved in canine meningioma pathogenesis may provide valuable information relating to tumor classification and prognosis beyond standard histopathologic criteria, potential therapeutic targets, and validation of canine spontaneous meningiomas as a model for human disease. The purpose of this study was to examine NF2 gene expression, both at the mRNA and protein levels, using real-time reverse transcription polymerase chain reaction (RT-PCR) and western blotting, in a series of spontaneous canine meningiomas. Additionally, western blotting was used to assess protein expression of the related 4.1B tumor suppressor gene and the associated TSLC1 gene. As reported for human meningiomas, some canine meningiomas in the current study also exhibited loss of 4.1B and TSLC1 protein expression; however, the consistent loss of merlin protein associated with specific subtypes of human meningiomas was not seen in canine tumors. These results suggest that some of the tumor suppressor genes implicated in human meningioma pathogenesis are also conserved in dogs and may perform analogous growth regulatory functions.
Materials and Methods
Sample collection
Tumor tissue was obtained from surgical biopsy/resection specimens or at necropsy from clinical cases presented to the Veterinary Medical Teaching Hospital, University of California, Davis. Samples of tumor from necropsy were collected within 20 minutes of death and snap frozen in liquid nitrogen. Surgical samples were similarly stored following collection. Samples of adjacent tumor tissue were processed for routine paraffin embedding and histology whenever fresh tissue was collected in liquid nitrogen. Normal brain samples were collected at necropsy from 8 clinical patients with nonneurologic disease. Normal samples included cortex/arachnoid and dura. All tumors were graded by a board certified pathologist (RJH, AWB) essentially according to the international World Health Organization classification of human tumors of the nervous system. 20,31 Meningiomas were graded as either grade I (benign), grade II (atypical), or grade III (malignant); Histologic subtypes were determined for benign (grade I) tumors.
Quantitative real-time RT-PCR
Total RNA extraction, cDNA preparation, and quantitative real-time RT-PCR were done as previously described. 4 NF2 and housekeeping gene PCR primers (glyceraldehyde-3-phosphate dehydrogenase GAPDH; heat shock 90 kD protein 1; alpha HSPCA) were designed based on published canine sequence data using Primer Express software (Applied Biosystems, Foster City, CA) and validated as previously described. 4 PCR products were designed with either one of the primer pairs or internal probe placed over an exon-exon junction to allow discrimination between cDNA and gDNA. NF2 (GenBank accession # XM534729) (specific for exon junction 2/3; called NF2/3) forward primer (exon 1) 5′ CGGTGTCCTTGATCGTGTACTG 3′, NF2 reverse primer (exon 3) 5′ TCAATTGCGAGATGAAGTGGAA 3′, NF2/3 probe 5′ CTGGGGCT 3′ (Universal probe library [UPL, Roche Applied Science, Indianapolis, IN] #45; product length 266 bp). GAPDH (AB038240) forward primer 5′ GATGGGCGTGAACCATGAG 3′, GAPDH reverse primer 5′ TCATGAGGCCCTCCACGAT 3′, GAPDH probe 5′ CCCTCAAGATTGTCAGCAATGCCTCCT 3′ (product length 131 bp). HSPCA (BI395999) forward primer (exon 10) 5′ ACCGCATTTACCGCATGATC 3′, HSPCA reverse primer (exon 11) 5′ CTCCGCTGCCACTTCATCTT3′, HSPCA probe 5′ TAGGCCTGGGCATCGA3′ (product length 621bp). NF2 PCR products were validated by agarose gel electrophoresis and direct sequencing. Transcript quantitation was done using the comparative CT method and reported as relative levels, or the n-fold difference relative to the mean value for individual normal arachnoid samples (n = 8). Tumor samples that had GAPDH CT values weaker than 3 times the average GAPDH CT value were considered low-quality cDNA samples and were not included in the analysis.
Western blotting
Canine meningioma specimens and normal control tissues were homogenized in NP40 lysis buffer containing protease inhibitors. Protein concentrations were determined by the BCA method (Biorad Laboratories, Hercules, CA). Electrophoresis was performed using either 100 µg or 50 µg of each sample separated on 10% SDS-PAGE gels. Proteins were transferred onto Immobilon membranes (Millipore, Bedford, MA) for western blotting with antibodies against TSLC1 (ES1; dilution 1 : 20,000) 32 , merlin (C18; dilution 1 : 1,000; Santa Cruz Biotechnology, Santa Cruz, CA), protein 4.1B (dilution 1 : 2,000) 16 , α-tubulin (dilution 1 : 40,000; Sigma-Aldrich, St. Louis, MO), and β-actin (dilution 1 : 4,000; Sigma-Aldrich). Western blots were developed using horseradish peroxidase-conjugated secondary antibodies (dilution 1 : 20,000) and ECL chemiluminescence (Amersham Biosciences, Piscataway, NJ). Reduced or absent protein expression was determined by scanning densitometry and defined as >75% reduced expression compared with normal canine cortex/arachnoid tissue for 4.1B and TSLC1. Densitometric values of expression for individual samples were calculated relative to control genes to correct for loading. Because minimal expression of merlin was seen in control tissue on western blots, determination of reduced protein expression relative to normal tissue was not done. Corrected merlin densitometric values were used to compare expression between different tumor grades and subtypes, and expression was compared with the mean of the total samples for each blot. One sample from blot 2 had a signal value >3 standard deviations from the mean and was excluded for calculation of the mean of the total tumor samples.
Statistical analysis
Reduced or absent expression of TSLC1 or 4.1B on western blotting was assessed in relation to tumor grade, meningothelial versus other grade I meningiomas, meningothelial versus all other meningiomas, and intraspinal versus intracranial tumors using Fisher's exact test. Relative densitometric values for merlin expression on western blotting were assessed for the same variables using the Wilcoxon-Mann-Whitney test stratified for the 2 separate blots. Relative levels of NF2 gene transcripts determined by RT-PCR were assessed for the same variables using the Wilcoxon-Mann-Whitney test. The Kruskal-Wallis test was used to determine if there was a significant association between tumor grade and number of tumor suppressor genes (TSLC1, 4.1B) that had reduced or absent expression. Numbers of tumors for specific breeds or specific locations other than intracranial or intraspinal were not sufficient to allow statistical evaluation of these variables. Significance was defined as P < .05.
Results
A total of 30 meningiomas were analyzed by western blotting (Figs. 1, 2). Twenty-five tumors also were assessed for NF2 levels by quantitative RT-PCR (Fig. 2). Twenty-one tumors were intracranial, 7 tumors were intraspinal, and 2 tumors involved both the brainstem and cranial spinal cord. For the intracranial tumors, 13 were grade I (9 meningothelial, 2 microcystic, 1 psammomatous, 1 transitional), and 8 were atypical (grade II). For intraspinal tumors, 2 were grade I (1 meningothelial, 1 transitional), and 5 were atypical (grade II) (Table 1). Both tumors traversing the foramen magnum were grade I (meningothelial) and were analyzed as intracranial tumors.
Canine meningioma samples, signalment, location, and histologic classification.∗
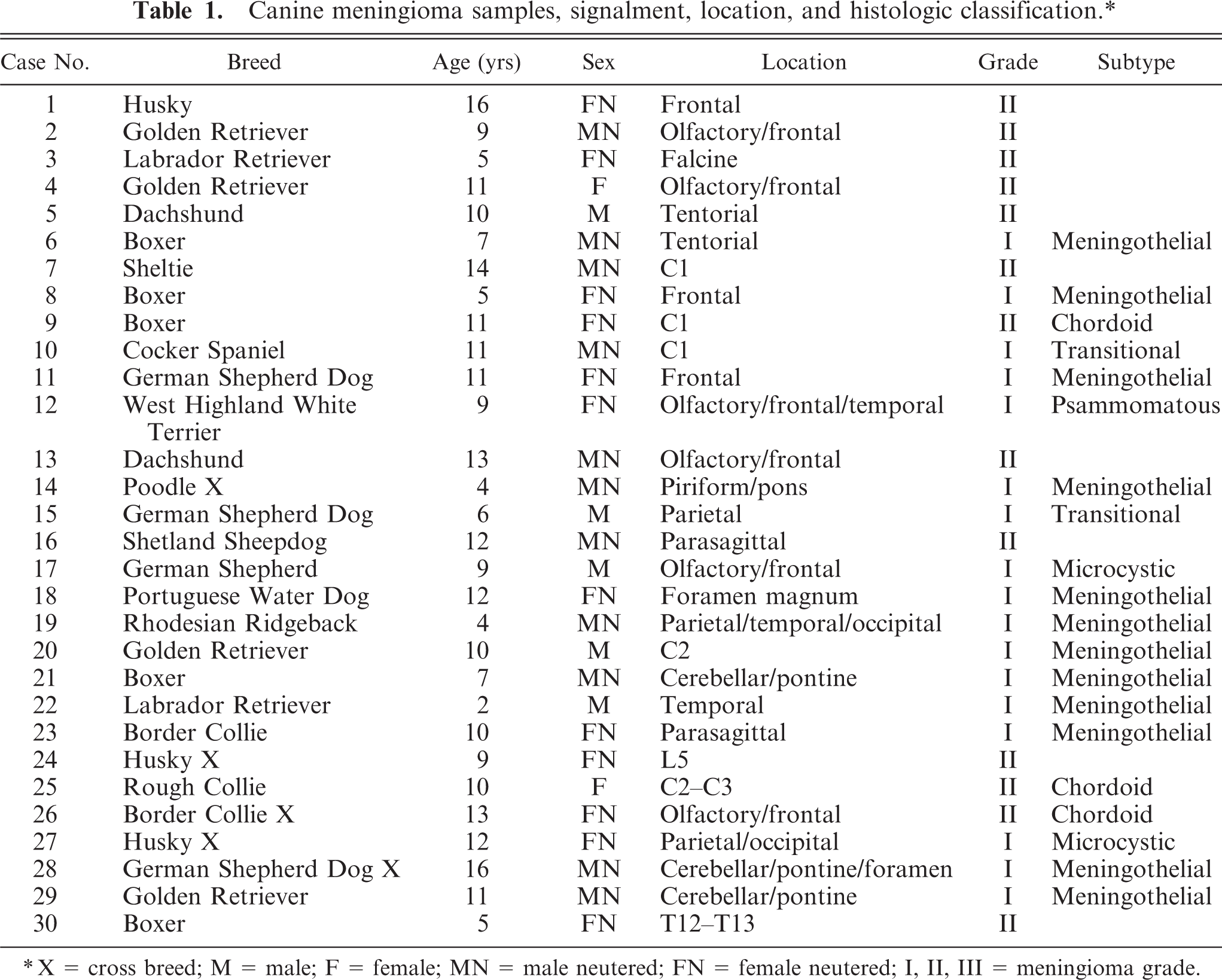
∗X = cross breed; M = male; F = female; MN = male neutered; FN = female neutered; I, II, III = meningioma grade.
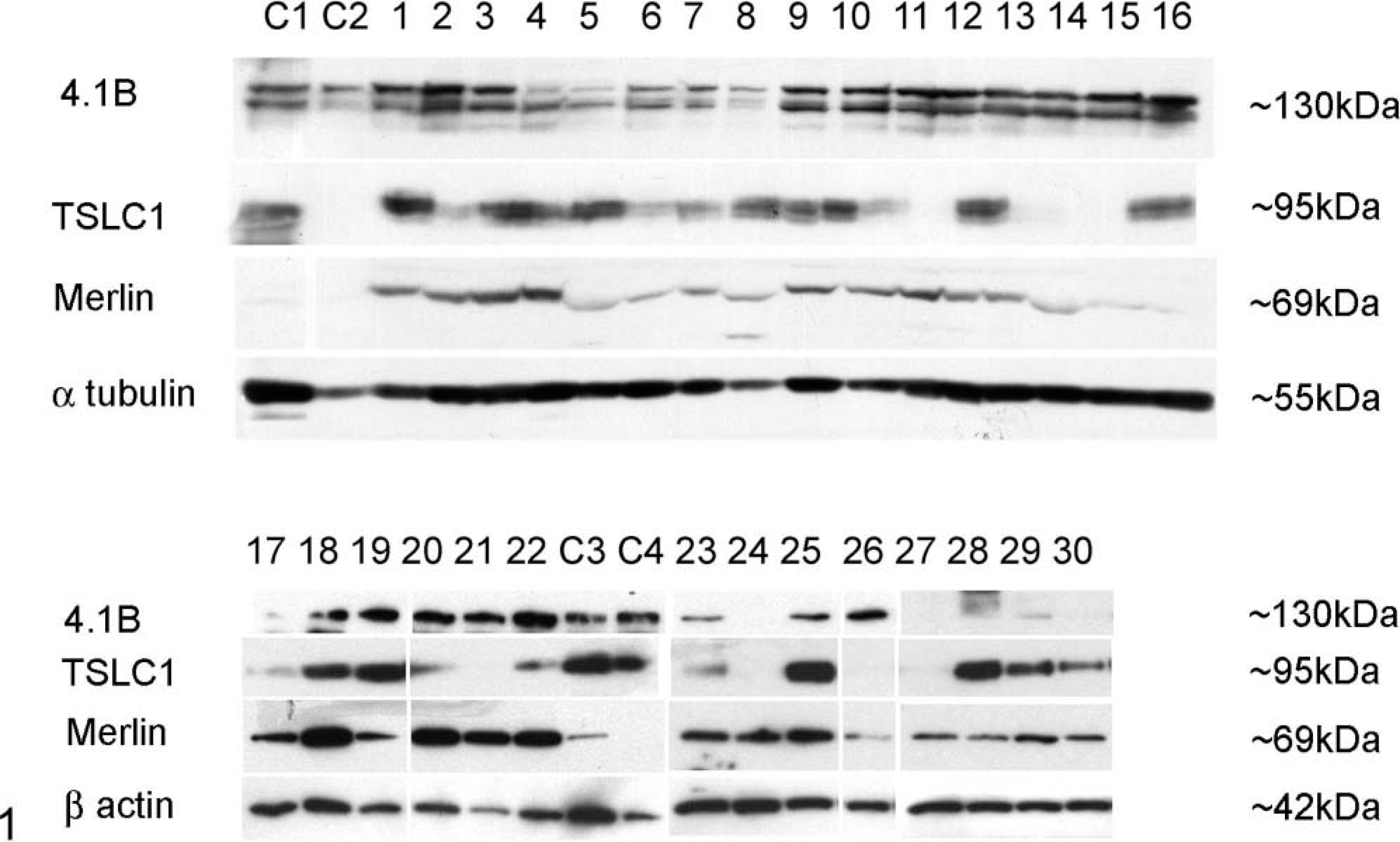
Western blots of TSLC1, 4.1B, and merlin from spontaneous canine meningiomas (blot 1: samples 1–16, blot 2: samples 17–30). Strong staining for 4.1B and TSLC1 was seen in normal brain cortex/arachnoid samples (C1, C3, C4), with 4.1B staining only in normal dura (C2). Staining for merlin was weak or absent in all control samples. Marked loss (<25% normal cortex/arachnoid) or absent signal for 4.1B or TSLC1 was seen in several tumor samples. Presence of staining for merlin was variable but present in all samples, although several samples (6, 15, 16, 26) had low signal (<25% mean value for tumor samples combined).
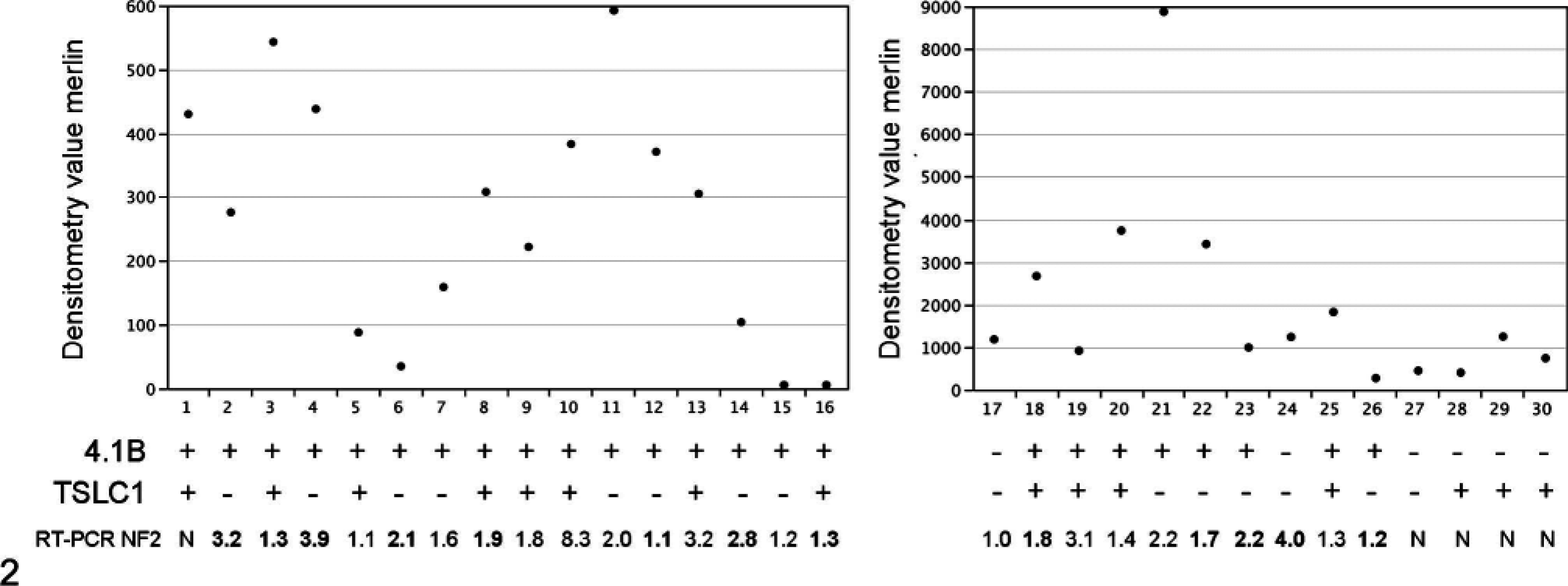
Summary of 4.1B, TSLC1, and merlin western and RT-PCR expression data for all canine meningioma samples. Densitometric values are plotted for the merlin western blots, corrected for loading relative to control genes (α tubulin/blot 1 or β actin/blot 2). For each sample expression of 4.1B and TSLC1 are denoted as +/− with − representing absent or reduced signal to <25% normal cortex/arachnoid. Quantitative RT-PCR data for each sample represents the n-fold increase or decrease in NF2 mRNA relative to normal cortex/arachnoid tissue (increased levels are in bold). N = not done. Several tumors can be seen to have decreased or low expression of 1 or more genes, No specific pattern of 4.1B, TSLC1, or merlin/NF2 expression was seen relating to tumor grade or subtype.
Merlin expression was seen in all tumors on western blots, although level of expression varied markedly, with some tumors having very low levels. Control samples of cortex/arachnoid tissue had absent or very low levels of merlin expression below that seen in the tumor samples. Calculation of expression of merlin protein relative to cortex/arachnoid control tissue, as for TSLC1 and 4.1B, was therefore not done. Four samples (6, 15, 16, 26) had levels of expression that were less than 25% of the mean for the total meningioma samples as a group. Absent or reduced TSLC1 expression was seen in 15/30 (50%) tumors (2/7, 28% spinal cord; 13/23, 57% brain; 10/17, 59% grade I, 5/13, 38% atypical/grade II). Absent or reduced 4.1B expression was seen in 6/30 (20%) tumors (2/7, 28% spinal cord; 4/23, 17% brain; 4/17, 24% grade I, 2/13, 15% atypical/grade II). Eighteen out of 30 tumors (60%) had absent or reduced expression of either TSLC1 or 4.1B, and 3 of these tumors had absent or reduced expression of both genes.
Relative transcription of NF2 mRNA for all samples was within +/− fourfold difference relative to the mean value for normal cortex/arachnoid except for 1 sample (−8.3).
Loss or reduced expression of TSLC1, merlin, or 4.1B on western blotting was not significantly associated with histologic grade (P = .46, P = .52, P = .67), meningothelial versus other grade I subtypes (P = .34, P = .38, P = .54), or meningothelial versus all other subtypes (P = 1, P = .28, P = 1). Loss or reduced expression of TSLC1 or 4.1B on western blotting was not significantly associated with intracranial versus intraspinal location (P = .39, P = .60). Similarly, relative expression of NF2 gene transcripts was not significantly associated with histologic grade, meningothelial versus other grade I subtypes, meningothelial versus all other tumors, or anatomical location (P = .65, P = .30, P = .72, P = .18) (Fig. 2). Loss or reduced expression of more than one protein (TSLC1 or 4.1B) was not associated with increased histologic grade (P = .25).
Discussion
Inactivation of one or more members of the protein 4.1 super family including 4.1B (DAL-1), 4.1R, and the NF2 gene product merlin, as well as inactivation of interacting proteins such as TSLC1, has been implicated in the pathogenesis of human meningiomas. Although 4.1 proteins are thought to play a major role in the maintenance of cell shape by linking cell surface glycoproteins to actin and actin-binding proteins, some may also act as negative growth regulators and tumor suppressors, possibly through cell contact or extracellular matrix–mediated mechanisms. 7,9,25,32 Results from the current study suggest that loss or reduced expression of these related tumor suppressor gene products also may occur in up to 60% of spontaneous canine meningiomas, although specific patterns of gene expression previously reported in different grades and subtypes of human tumors were not identified.
Loss of expression or mutational NF2 inactivation 1,12,15,25,38 and loss of 4.1B expression 7,25 have been reported in all grades of human meningiomas, suggesting that these genetic changes represent early events in meningioma pathogenesis. However, some studies have shown significantly decreased expression of NF2 in grade III meningiomas relative to other grades, 1,3 and loss of heterozygosity of 4.1B has been reported more commonly in malignant tumors. 24 Loss of TSLC1 protein, determined by immunohistochemistry, has been associated with increasing tumor grade, 32 and a trend of combined alterations of merlin and 4.1B associated with tumor grade has also been reported. 24,25 Analysis of tumor suppressor gene expression across all grades of canine meningiomas in this study was not possible, as no grade III canine meningiomas were available for analysis. However, approximately 43% of the tumors in our series were atypical (grade II) meningiomas. Canine meningiomas have been reported to differ from their human counterparts with respect to a higher incidence of grade II tumors (43%), although the incidence of malignant grade III tumors appears similar (1%). 31 In contrast to some human studies, significant association was not seen between tumor grade and NF2, 4.1B, or TSLC1 protein expression in the canine meningiomas, and loss of multiple tumor suppressor proteins was also not associated with increasing grade. These results suggest that loss of merlin and 4.1B and TSLC1 are more likely associated with tumor initiation, rather than progression, in canine meningiomas. Further studies on a larger series of high grade (III) tumors will be necessary to strengthen these observations.
Ten of the canine tumor samples studied were of grade I meningothelial subtype. Although the majority of studies defining NF2 status in human meningiomas have reported a consistently lower frequency of NF2 inactivation associated with the meningothelial subtype, and possibly an anterior skull base location, 1,6,12,15,17,36,38,39 a statistically significant difference in protein levels was not observed in the canine meningiomas when comparing meningothelial tumors with other grade I tumors or to all other tumors as a group. Genetic aberrations involving NF2 have been defined for the majority of spontaneous meningiomas in humans, the majority being nonmeningothelial. For the majority of the remaining tumors, no obvious genetic, epigenetic, or karyotypic abnormalities have been found, 37 and it has been suggested that as yet undefined oncogenic pathways may underlie the development of this group of tumors. 1,6,15,17,37–39 The reason for the low merlin signal in normal (cortex/arachnoid) control tissues in the current study is not known, and it is possible that comparison of protein expression levels with the group of tumors as a whole may have underestimated the degree of merlin abnormalities. Regardless of the nominal value chosen to assess individual expression of merlin, complete loss of protein or expression levels related to specific tumor subtype were not seen. It is also possible that reduced expression of merlin protein in the current study has been underestimated as a result of normal stromal and vascular elements contaminating tumor tissue samples resulting in non-tumor-related signal. Immunohistochemistry may provide additional data in future studies, allowing expression to be localized spatially to tumor versus normal tissues; however, based on the western data, the incidence of altered gene function of NF2 and 4.1B appears to be lower in dogs compared with previously published human studies (see Table 2). Although TSLC1 data in both species are limited, the frequency of decreased TSLC1 protein expression appears to be similar. Decreased merlin expression in human meningiomas is often associated with loss of heterozygosity on chromosome 22q and bi-allelic inactivation of the NF2 gene. 11,36,38 Consistent with the apparently low incidence of abnormal NF2 gene expression in the current study, preliminary analysis of 3 canine meningiomas did not find deletions of the canine orthologue of human chromosome 22q (CFA 26), although the histologic subtype was not reported. 2 Analysis of 30 canine meningiomas by genomic microarray has also failed to document any deletions of CFA 26 in either meningothelial or nonmeningothelial tumors, 34 further supporting the view that NF2 may not play the major role in canine meningiomas that it does in human meningiomas.
Expression of 4.1B, TSLC1, and NF2/merlin in spontaneous meningioma: Comparison of human and current canine studies.∗
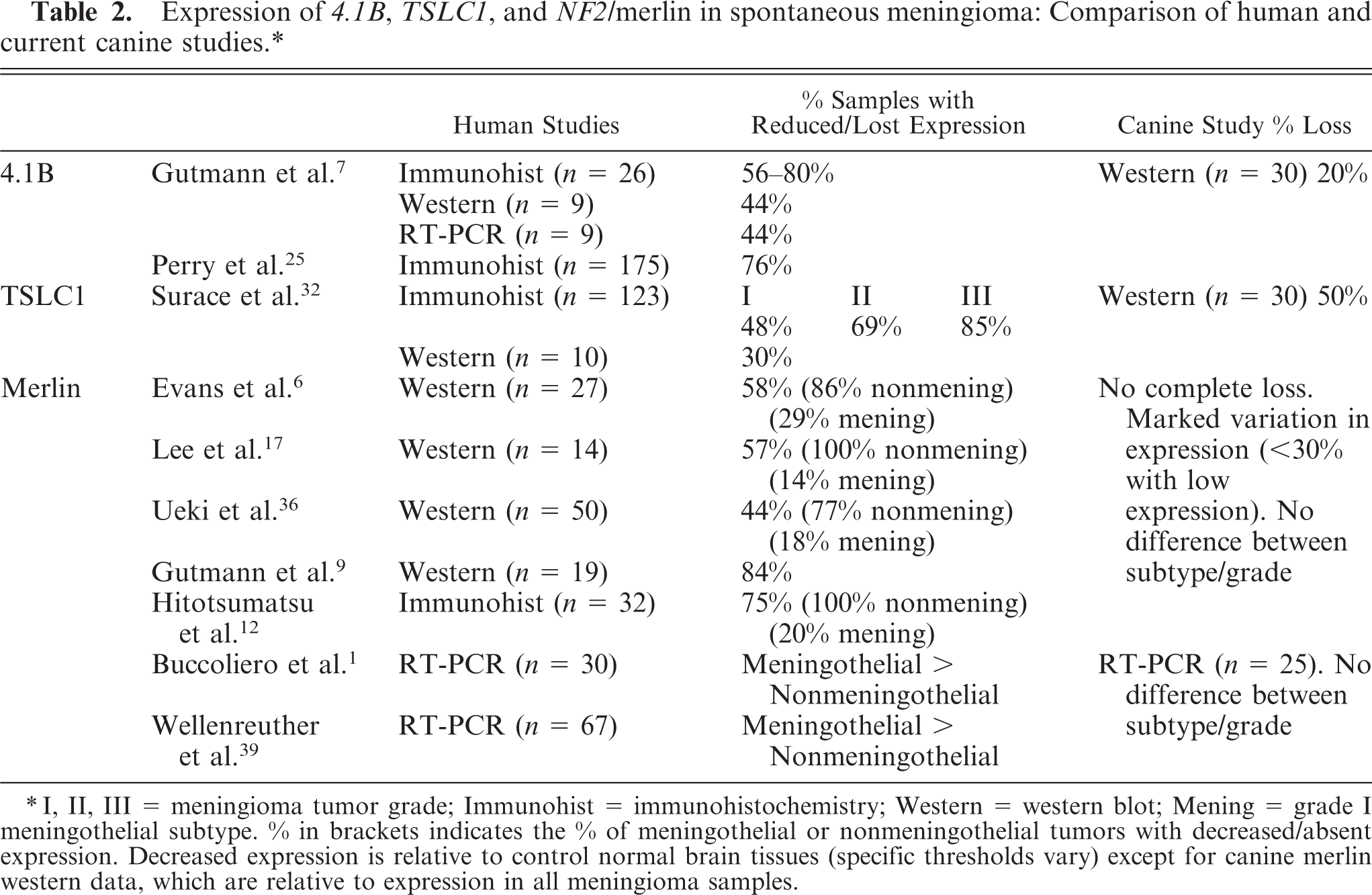
∗I, II, III = meningioma tumor grade; Immunohist = immunohistochemistry; Western = western blot; Mening = grade I meningothelial subtype. % in brackets indicates the % of meningothelial or nonmeningothelial tumors with decreased/absent expression. Decreased expression is relative to control normal brain tissues (specific thresholds vary) except for canine merlin western data, which are relative to expression in all meningioma samples.
As with previously reported NF2 RT-PCR studies in human meningiomas, 1,39 there was variability in transcription levels in individual tumors within specific groups, and conclusions should therefore be drawn from trends between groups rather than between individual tumors. Direct correlation was not seen between RT-PCR for NF2 mRNA and merlin western blotting for all samples where both studies were done (Fig. 2); however, samples from archival material for the western and RT-PCR studies were small, and differences may reflect heterogeneity within the archival sample. In addition, differences in mRNA and protein levels may also represent posttranslational regulation of merlin expression, such as the cleavage of merlin by activation of the calpain-mediated proteolytic system, 14 or the presence of missense or nonsense mutations that result in potentially normal mRNA levels but unstable or defective merlin protein. 8 In contrast to human NF2 transcriptional data (Table 2), and consistent with the protein expression seen on the canine meningioma western blots, no correlation was seen between meningioma grade or subtype and NF2 mRNA expression in canine meningiomas.
The current study demonstrates that reduced or absent expression of tumor suppressor genes 4.1B and TSLC1 is present in some canine meningiomas, and that aberrant NF2 gene expression does not appear to follow the same pattern seen in human tumor counterparts. The observations above raise the intriguing possibility that the pathogenesis of canine meningiomas in general may more closely resemble the pathogenesis of human meningothelial meningioma or that additional mechanisms of tumorigenesis not dependant on merlin, 4.1B, and TSLC1 perturbation may be present in canine tumors. Breed and morphologic predispositions to the development of meningiomas are known to occur in dogs, 31 and the limited degree of genetic variation present in highly inbred dog breeds may more readily allow for the identification of these underlying genetic factors that may also be important in human tumors.
