Abstract
Idiopathic chondrolysis is a human clinical entity typically reported in adolescent individuals. In this brief communication, we report 2 cases of presumptive idiopathic chondrolysis of the femoral head in Cynomolgus macaques and discuss the clinical symptomatology and pathology of the disease. In detail, we describe the histomorphologic changes of idiopathic chondrolysis and compare these findings with those typically observed in the primary differential diagnoses of Legg-Calve-Perthes disease and nonspecific osteoarthritis. Consideration of this entity among differential diagnoses in young Cynomolgus macaques with unilateral osteoarthritis could be important both for laboratory animal veterinarians and pathologists.
Keywords
In this report, we describe 2 cases of presumptive idiopathic chondrolysis of the femoral head in young female Cynomolgus macaques. The animals were of Vietnamese origin and were received into the commercial supplier/breeding facility on 13 June 2006 for standard Centers for Disease Control and Prevention (CDC) quarantine. After clearing quarantine, they were group housed from July 2006 until January 2007, at which time they were sent to the National Institutes of Health (NIH) Animal Center. The animals were housed at the NIH Animal Center for biomedical research purposes in developing animal models for vaccination against hemorrhagic fever. Here, the standard 90-day quarantine period included 5 tuberculosis tests, 2 physical examinations, and blood collections for complete blood count/chemistry and serology. No abnormalities were noted on either animal's entry or exit quarantine physical exam (17 January 2007 and 12 April 2007, respectively). Both monkeys tested negative for B virus and simian immunodeficiency virus. These animals were singly housed from receipt on 11 January 2007 until they were euthanatized on 2 July 2007. The monkeys had ad libitum access to water via an automatic watering system, were fed 8 Purina 5045 biscuits twice a day, and received various enrichment treats (apples, bananas, prima treats, etc.) twice a week.
On 12 June 2007, during a routine quarterly physical examination, 2 female 3-year-old Cynomolgus macaques presented with muscle atrophy of the right leg. Both cases favored the affected right leg, while supporting most of their weight with the left leg. Because of these abnormal observations, the animals were never placed on study.
Physical examination of the affected limbs revealed approximately 40% reduction in muscle mass of the proximal thigh. Tape measurements were taken mid-femur. Macaque No. 1 measured 9.2 cm around the right leg and 15.7 cm around the left leg. Macaque No. 2 measured 10.2 cm around the right leg and 15.7 cm around the left leg. The right legs of both animals were also shorter, and significant crepitus was noted in the hip joint when the legs were manipulated. Macaque No. 2 was non–weight bearing on the leg when moving around the cage, whereas macaque No. 1 was using the affected leg for stability and to grasp objects.
Radiographs were taken of both animals and were reviewed by a human radiologist (N. Safdar) and 2 veterinary surgeons (J. Bacher and R. Herbert). Deformity/degeneration of the femoral head with increased radiodensity, changes in the acetabular wall, and thinning of the cortices were noted in both series of radiographs (Fig. 1). Both animals also had an increase in density of the acetabulum and significant muscle atrophy of the right leg (Fig. 2). The soft tissues on the affected (right) side were atrophic when compared with the unaffected (left) side. Although radiographically the physes of the femoral head and greater trochanter were clearly delineated on the unaffected side, these were less well defined or even fused on the affected side. The joint space of the right hip was narrowed superiorly, with remodeling of the acetabulum. Asymmetry of the femoral heads was significant, with areas of subchondral sclerosis and contour deformity of the articular surface. In both cases presented, there was significantly more coverage of the femoral head on the affected side than on the normal side.
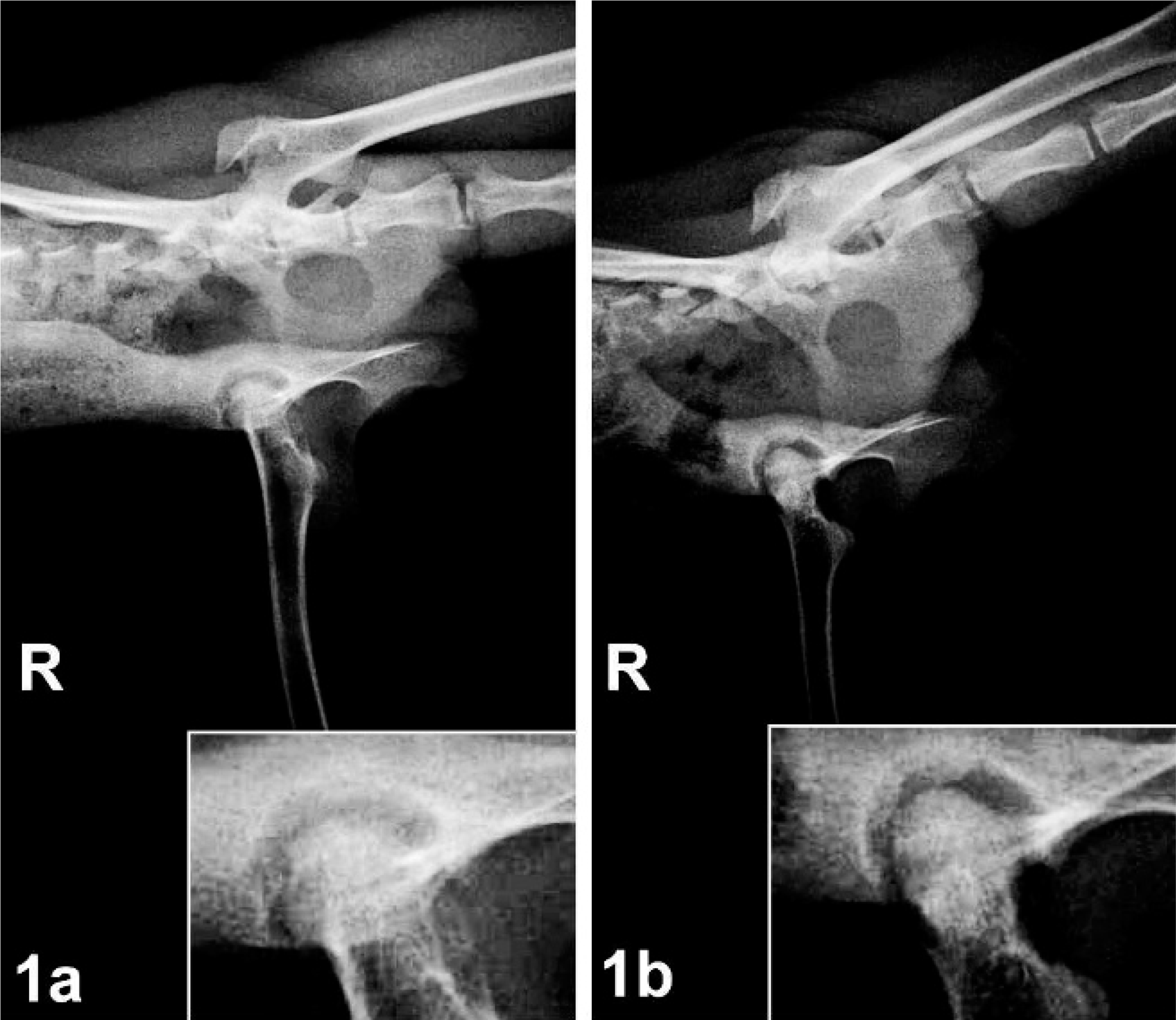
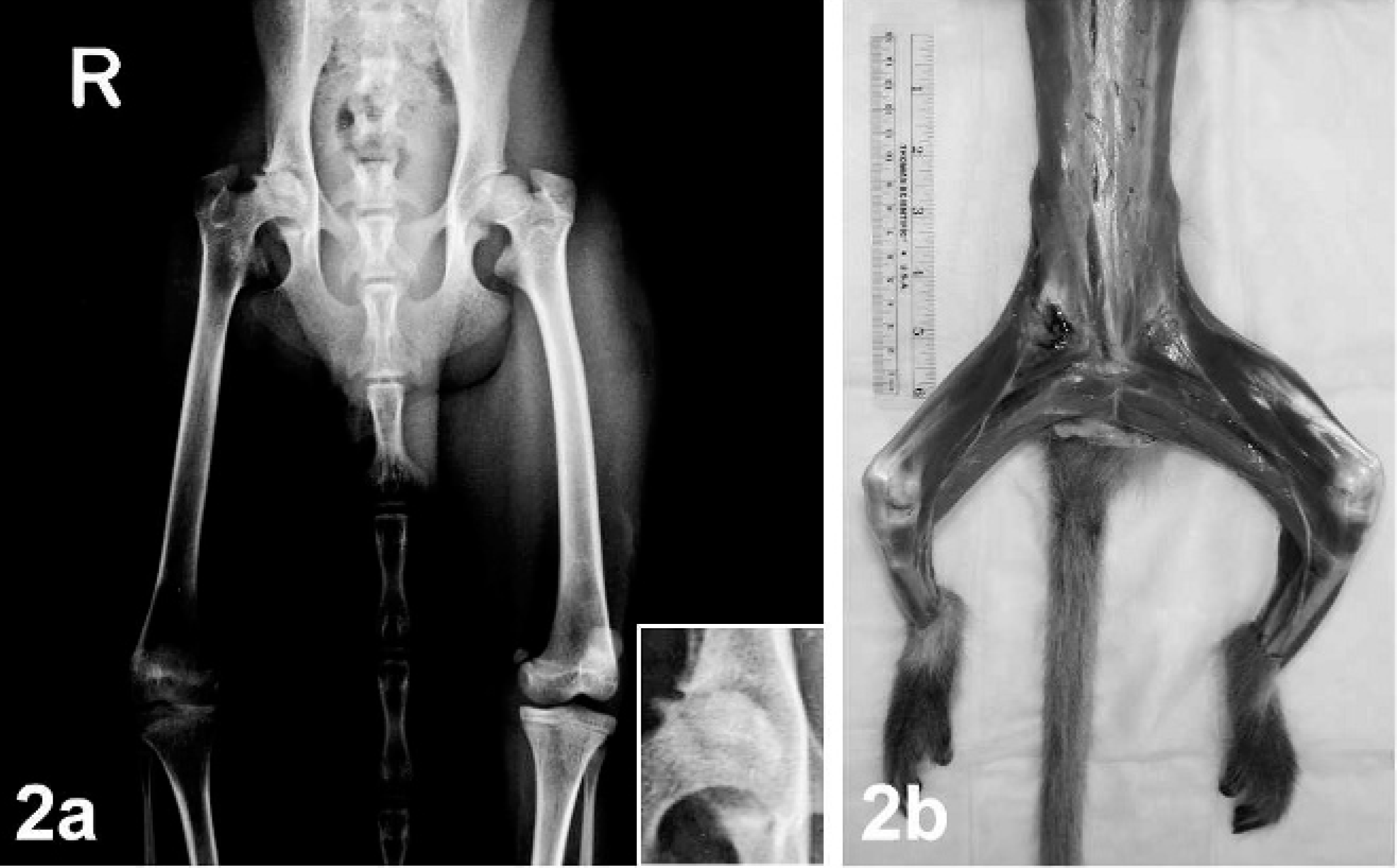
Radiograph. Pelvis; 3-year-old female Cynomolgus macaque No. 1. Fig. 2a. VD view showing diminished muscle mass and thinning of the femoral cortices of the right leg compared with the left leg; inset: right coxofemoral joint. Fig. 2b. Severe muscle atrophy of the right leg, with approximately 40% reduction in skeletal muscle mass of the proximal thigh.
After radiographic evaluation, the affected animals were placed on nonsteroidal anti-inflammatory medication to decrease pain and discomfort associated with the illness. Naproxen was given at 5 mg/kg PO, BID for 5 days. No significant changes were noted in the animals' condition while on treatment. Femoral head and neck osteotomy was not considered a viable treatment option for these animals, so they were euthanatized on 2 July 2007.
Pathologic evaluation after euthanasia was conducted on both Cynomolgus macaques. On gross examination, the right rear leg was smaller in size than the left rear leg in both animals because of decreased skeletal muscle tissue (Fig. 2b). Similarly, the right rear leg was shorter than the left rear leg in both animals. On exposing the right coxofemoral joint, the joint capsule was noted to be markedly thickened (Fig. 3). In both cases, more than 80% of the right femoral head was ulcerated, the articular cartilage was diffusely absent, and the right femoral head was smaller than the left and was light yellow-white in color. Most of the acetabular surface was ulcerated or eroded in both animals (Fig. 4). The left coxofemoral joint in both animals appeared normal (Fig. 3). Gross abnormal findings were not apparent in the remainder of the tissues from these animals.
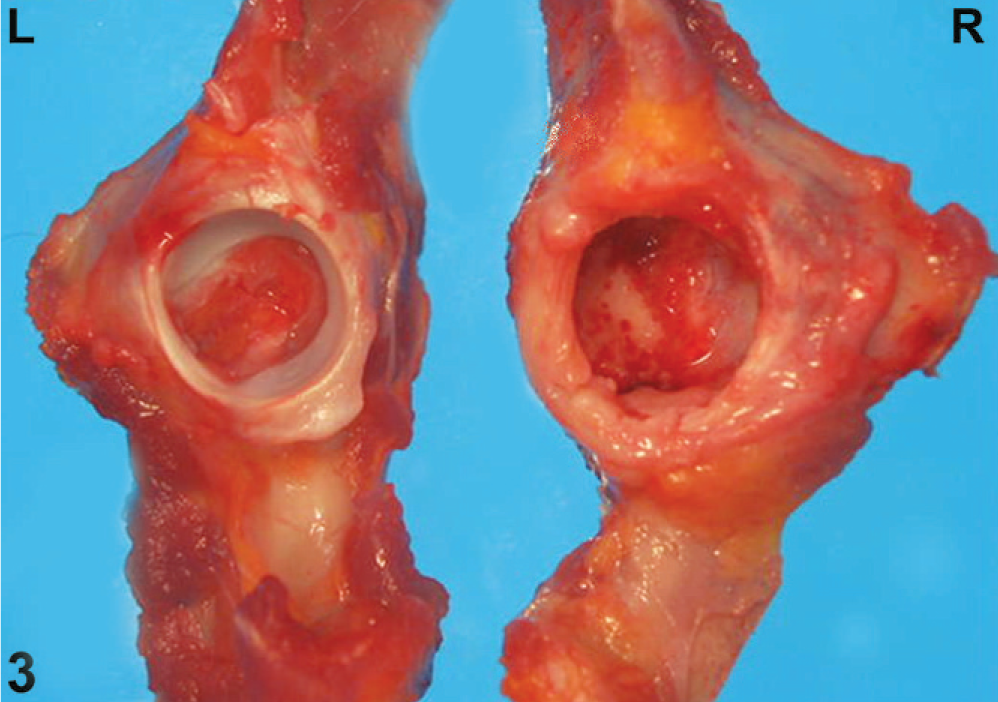
Acetabulum; macaque No. 1. Left and right side; the right acetabulum is severely ulcerated with loss of the articular cartilage and thickening of the joint capsule.
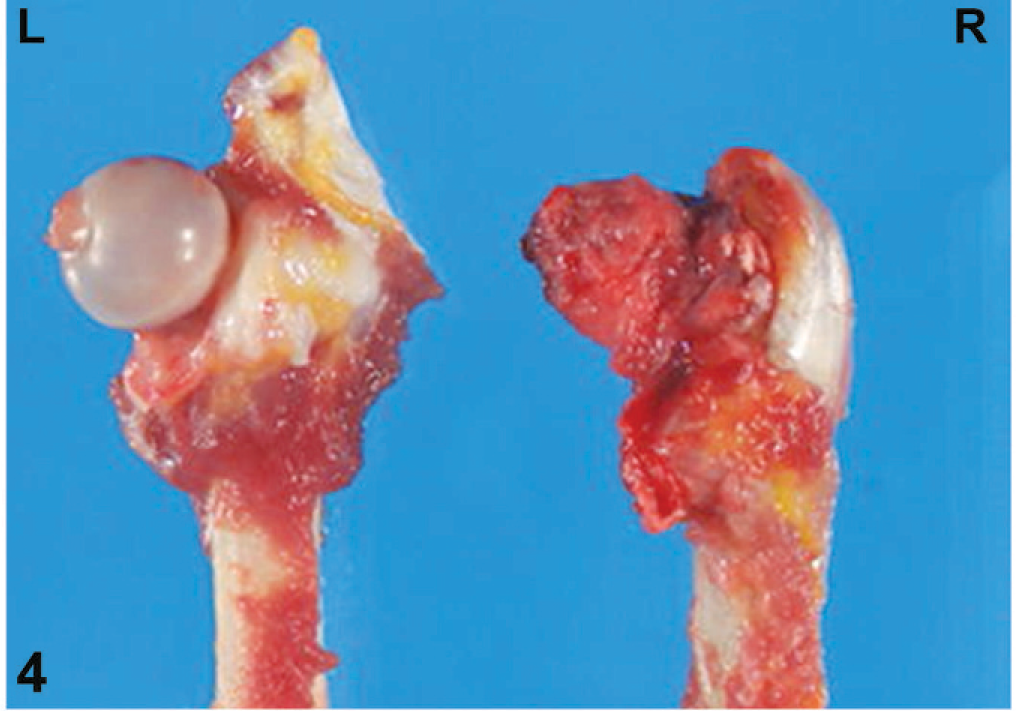
Femoral head; macaque No. 2. Left and right sides; the right femoral head is severely ulcerated with loss of the articular cartilage.
Histologically, in both cases, there was full thickness loss of the articular cartilage of the right femoral head and right acetabulum, and the articular surfaces of these 2 bones were partially covered with fibrous connective tissue that extended only into the superficial subchondral bone marrow (pannus) (Figs. 5,6), and in some areas of this connective tissue, small bone fragments were undergoing resorption (Fig. 7). In other areas, exposed bone was present at the articular surface. Also, some portions of the subchondral bone were undergoing resorption and remodeling. The hematopoietic tissue in the bone marrow of the subchondral bone was hypocellular, and the residual bone marrow stroma and vasculature appeared intact. The medullary bone was viable, with osteocytes present in lacunae (Fig. 7 insert); necrotic medullary and cortical bone was not present in the femoral head in the examined sections (Fig. 6). The physes, although still present histologically, were thinner and less active than the physes in the unaffected femoral heads.
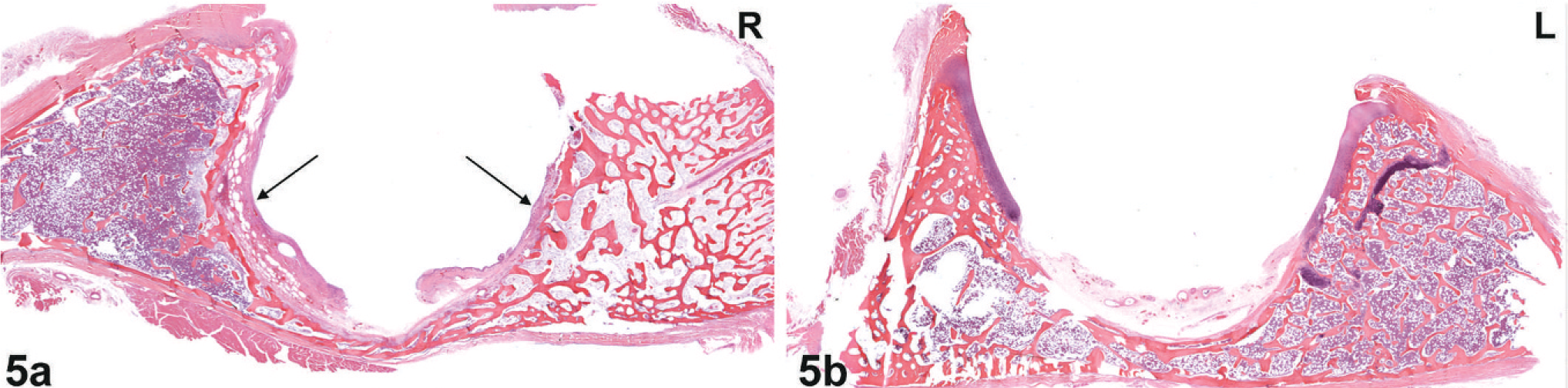
Histology. Acetabulum; macaque No. 1. Fig. 5a. Right side; abnormal acetabulum with ulceration (black arrows), loss of articular cartilage, and replacement with fibrous connective tissue (pannus). Demineralized HE. Fig. 5b. Left side; normal acetabulum with articular cartilage present on shoulders of acetabulum. Demineralized HE.
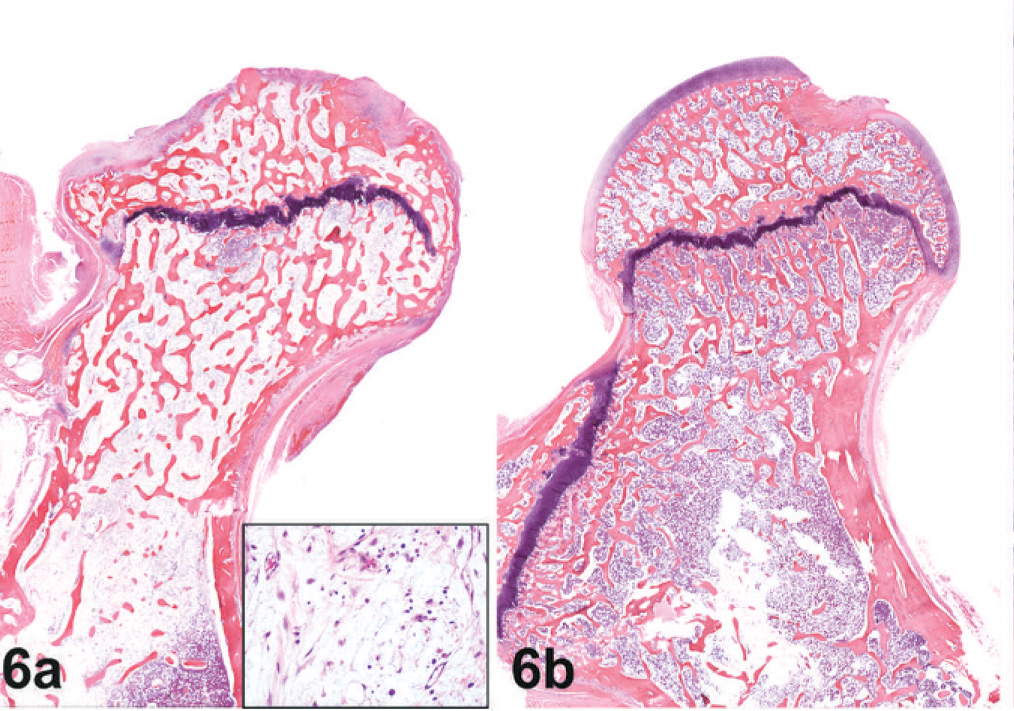
Histology. Femoral head; macaque No. 1. Fig. 6a. Right side; the femoral head is severely ulcerated with loss of articular cartilage and replacement by fibrous connective tissue. In between bony trabecula is pre-existing stroma (see insert) with loss of hematopoietic cells. Demineralized HE. Fig. 6b. Left side; normal femoral head and articular cartilage. Demineralized HE.
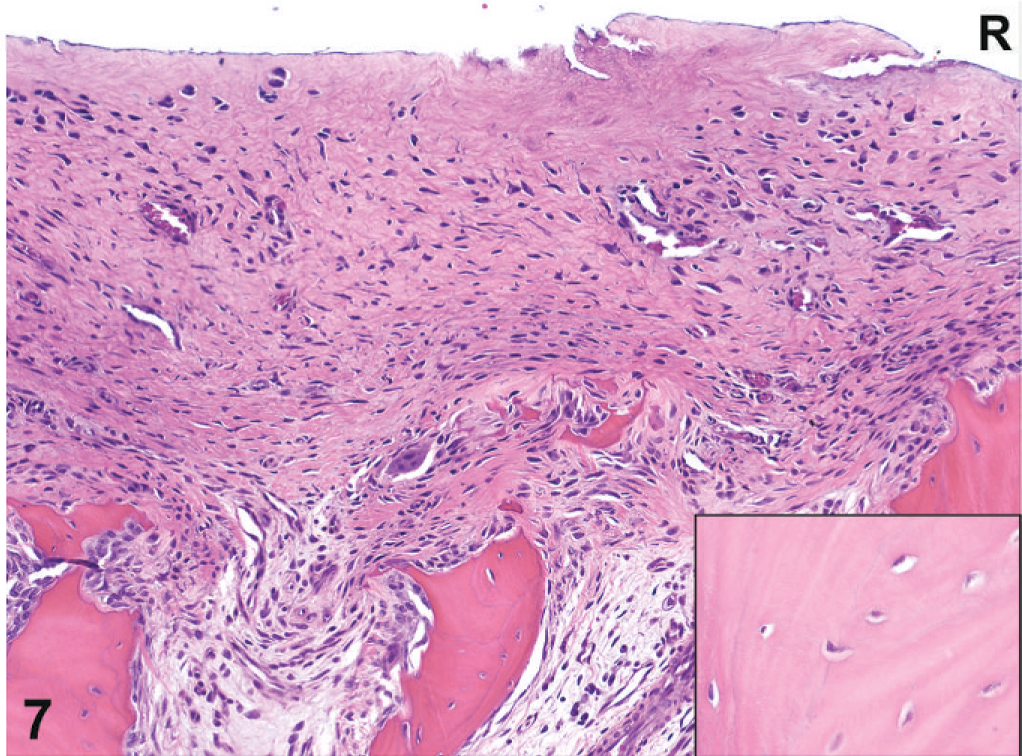
Histology. Femoral head; macaque No. 1. Right side; the femoral head is severely ulcerated with loss of articular cartilage and replacement by fibrous connective tissue. Small fragments of medullary bone entrapped in the fibrous connective tissue are undergoing resorption. Viable osteocytes are present in lacunae in the medullary bone of the femoral head (see insert). Demineralized HE.
The initiating cause for the above discussed findings was not found. In diagnosing these 2 cases, the gross and histologic changes in the right coxofemoral joints of both macaques are morphologically consistent with osteoarthritis (OA), also known as degenerative joint disease, although we could not determine a cause. Osteoarthritis is a naturally occurring entity in young female Cynomolgus macaques; however, the disease is typically presented in the knee joint, with primary presenting symptoms including thickening of the subchondral plate in the medial and lateral tibial plateaus, with progression to the femoral condyles, and articular cartilage fibrillation and loss. 3, 11
Alternative diagnoses also include idiopathic chondrolysis of the femoral head, Legg-Calve-Perthes (LCP) disease, and idiopathic pigmented villonodular synovitis. Initial reports of idiopathic chondrolysis of the femoral head in people indicate that this condition occurs more often in adolescent females of African descent 6, 9, 13 ; however, subsequent reports have included individuals of other races and a higher incidence in adolescent males than was previously reported. 1, 4, 5, 10 The typical presenting clinical sign is pain in the hip joint, knee joint, or groin region, or a combination of these symptoms, of long-term duration. Initially patients are affected unilaterally, but as the condition progresses, bilateral involvement is not uncommon. 1, 4– 6, 8– 10, 13 Although these bilateral cases have been reported, the majority of cases in the largest case series are unilateral. 1, 6 Over time, passive range of motion is lost in the affected hip joint; also, the affected limb is shorter with diminished muscle mass compared with the unaffected limb. This disuse atrophy of the affected limb was noted in both cases presented here. In idiopathic chondrolysis, clinical pathology tests are usually normal, and laboratory examinations for infectious disease, tuberculosis, and juvenile rheumatoid arthritis are negative. A history of trauma is absent, and radiographic changes are not consistent with LCP disease. On radiographic examination, these individuals typically have a narrowed joint space in the hip from loss of articular cartilage, lateral overgrowth of the femoral head on the neck, early closure of the femoral capital physis, regional osteoporosis, protrusio acetabuli, and erosions of the articular cartilage of the acetabulum or femoral head or both with subchondral cyst formation. 1, 4, 10 In some individuals, surgically collected joint tissues were evaluated grossly and histologically. In these individuals, the synovial membrane was thickened, a nonspecific synovitis was present, there was a villous proliferation of the synovium with fibrillation and thinning of the articular cartilage, and in some areas bare bone was exposed. 6 In some sections of evaluated bone, cystic areas had formed that were filled with synovial tissue, but necrotic areas were not observed. 6 Premature closing of the physis and early OA are often the end results in those affected. 1 Although possible causes were investigated in these reports, the exact etiology for chondrolysis is currently unknown.
Alternatively, LCP disease and osteonecrosis of the femoral head can have clinical presentations similar to the 2 cases presented here; however, the radiologic, gross, and histologic findings support a different diagnosis. LCP disease can, in some cases, have cartilage overgrowth that causes an increase in the joint space. Also, in early stages of LCP and osteonecrosis of the femoral head, histologically the articular cartilage is viable, and there are areas of necrotic subchondral bone. In later stages, the subchondral bone can fracture and collapse, with buckling of the overlying articular cartilage. 2 In the cases presented here, articular cartilage was absent, and the subchondral bone was viable and not necrotic. On radiographic evaluation of these 2 monkeys, there appears to be more coverage of the femoral head by the acetabulum and narrowing of the joint space on the affected side compared with the unaffected side, indicating a decrease or absence of articular cartilage. In addition, these cases demonstrate premature fusion of the physes of the femoral head and greater trochanter, which are not features typical of LCP or osteoarthritis. Although frank protrusio acetabuli, defined as a condition in which the femoral head lies further in the pelvis than normal, of the affected hips is not present, the medial acetabular walls have narrowed. 7 Neither LCP nor OA is associated with protrusion or such antecedent narrowing of the medial acetabulum. The combination of the increased acetabular coverage of the femoral head, premature physeal fusion, and narrowing of the medial acetabulum makes the diagnoses of LCP or OA exceedingly unlikely.
Idiopathic pigmented villonodular synovitis was also ruled out because it typically affects people later in life and is characterized by joint pain, swelling, and stiffness, accompanied by inflammatory signs. Periarticular soft-tissue swelling, joint effusion, and erosions of the articular margins with preservation of joint space and lack of osteoporosis are the classic radiographic finding for this disease and did not fit the parameters seen in these 2 monkeys. 12
Because of the unusual presentation seen in these 2 cases—degenerative joint disease confined to the hip in a young animal associated with almost complete loss of the articular cartilage in the joint, no known precipitating traumatic event, and no necrosis in the femoral head—we think that these cases are remarkably similar to a human syndrome known as idiopathic chondrolysis of the hip. To the best of our knowledge, this syndrome has never been reported before in a nonhuman primate species. Unfortunately, as in the human case, the exact pathogenesis of the syndrome is unknown and warrants additional investigation.
Footnotes
Acknowledgements
We gratefully acknowledge the contributions of Ms. Alyse Zajac for manuscript coordination and preparation. We also gratefully acknowledge the contributions of Mr. Alphie Cisar for providing animal care; Ms. Brenda Hartman for figure formatting; Ms. Alida Ault, Mr. John-Paul Todd, and Ms. Linda Bessacque for scheduling and technical assistance; Mr. Jorge Chavez and Mr. Erik Lasker for providing gross photographs; Ms. Annie Merriweather and Ms. Kathy Dukes for preparation of histologic tissue sections; and Mr. Rick Dreyfuss for preparation of histologic photographs. This research was supported in part by the Intramural Research Program of the NIH, Vaccine Research Center.
