Abstract
The aim of this study was to check the balance between tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-10 (IL-10) in well-developed end-stage disk disease in the disk itself as well as in paradiskal spine. In 6 domestic pigs the cranial bony end plate of the L4 vertebra was perforated to the nucleus pulposus. At 3 months the degenerated experimental and contiguous control disks, together with the adjoining bony and cartilaginous vertebral end plates, bone marrow, and spinal ligaments, were excised and used for immunohistochemical analysis. In general, there were more TNF-α and in particular IL-10 positive cells in the degenerated disks than in the control disks, whereas the number of IL-6 labeled cells did not differ among sites or between control and experimental intervertebral disks. These results suggest that TNF-α and IL-10 are involved in the late reparatory phases of the experimental disk lesion. Use of an experimental model showed that strictly disk-directed manipulation and degeneration are also reflected in the contiguous vertebrae, including adjoining cartilage, bone, marrow, and ligaments.
Introduction
Intervertebral disk degeneration (IDD) continues to be an unsolved problem in veterinary and human medicine. Tumor necrosis factor-α (TNF-α) is the principal mediator of inflammation as has perhaps most clearly been exemplified by the very success of anti-TNF therapies.12, 31 In degenerated and herniated lumbar intervertebral disks, TNF-α has been identified in tissue extracts20 and in histiocytes, fibroblasts, endothelial cells, and chondrocytes.41 Lack of TNF-α in tissue culture medium of explants of such tissues8, 22 might indicate that TNF-α remains bound to the cell surface and acts in a juxtacrine manner or that it is solubilized and subsequently effectively bound to TNF receptors type I (p55) and type II (p75). TNF-α was not released from healthy mouse intervertebral disks, and was not induced upon stimulation with the strong pro-inflammatory lipopolysaccharide.35 According to immunohistochemical analysis TNF-α is produced in acute situations soon after intervertebral disk herniation.50 TNF-α locally applied to spinal nerve root or dorsal root ganglion induced radicular pain20, 33 and proteinases.38
Interleukin-6 (IL-6) production is significantly increased by stimulation with TNF-α.5 In human articular cartilage, IL-6 inhibits synthesis of proteoglycans,15 which normally maintain high nucleus pulposus tissue hydration and prevent blood and lymphatic vessel ingrowth. If IL-6 has a similar effect on intervertebral disk (repair) tissue, increased IL-6 production would facilitate the degenerative process. IL-6 stimulates the formation of osteoclast precursors from colony-forming unit granulocyte macrophage and increases osteoclast numbers in vivo, leading to increased bone resorption,29 which may contribute to the spondyloarthrosis changes often accompanying degenerative intervertebral disk disease. In contrast to TNF-α, IL-6 is a pleiotropic cytokine,45 which is spontaneously produced by human and murine disk tissues7, 22, 35, 41 and which, in addition to its pro-inflammatory properties, can also suppress inflammation and inhibit bone destruction.2, 34, 37, 43, 49 IL-6 fails to directly induce proteinase expression2 but instead up-regulates tissue inhibitor of metalloproteinases-1 (TIMP-1).39 Many acute-phase proteins induced in the liver by IL-6 have anti-inflammatory properties.42 Finally, IL-6 has been reported to down-regulate IL-137 and up-regulate IL-1 receptor antagonist (IL-1Ra) expression.44
Interleukin-10 (previously known as cytokine synthesis inhibitor factor, or CSIF) is known as an anti-inflammatory and immunosuppressive cytokine,32 which, together with TNF-α and IL-6, was lumped together in one recent review as “the good, the bad, and the ugly.”40 It was emphasized that both inflammatory and anti-inflammatory reactions are normal components of a coordinated response, and the aim is to eradicate the noxious stimulus with minimum damage to the host.32 Therefore, IL-10 can be produced, apart from regulatory T cells, by a large number of other cells, including macrophages.11 IL-10 is extremely potent at suppressing the ability of macrophages to release TNF-α.6, 11 Murine nucleus pulposus-derived cells spontaneously produced and released IL-6 and IL-10, but not TNF-α, into the culture medium. Pro-inflammatory lipopolysaccharide stimulation increased IL-6 75-fold and increased IL-10 150-fold, but no TNF-α was detectable after stimulation, although some other cytokines (IL-1 and GM-CSF) were induced.35 In spite of this apparently important anti-inflammatory (and anti-TNF) role of IL-10, its localization has received little attention in degenerative intervertebral disk disease, although one study described IL-10 messenger RNA in human disk specimens, but only in 2 of 23 (9%) herniated human disk samples.1
Thus, it seems that cytokines work in a complex interacting manner.4, 10, 16 The aim of present study was to analyze the late stage of an experimental porcine model of disk degeneration to determine the interrelation of the pro-inflammatory TNF-α, the pleiotropic IL-6, and, in particular, the anti-inflammatory IL-10, in the affected disk and in the bony and cartilaginous end plate, vertebral body bone marrow, and spinal ligaments.
Material and Methods
End plate and intervertebral disk injury surgical procedures
Ethical approval for the use of animals in this study was received from the Animal Research Ethics Committee of Gothenburg. Domestic pigs (n = 6), 4–5 months old, weighing 50–60 kg, were sedated by an intramuscular injection of Ketalar (ketamine, 15–20 mg/kg; Parke-Davis, Gwent, UK) and after 10 minutes were anesthetized with intravenous Hypnodil (methomidate chloride, 3–5 mg/kg of body weight; AB Leo, Helsingborg, Sweden) and Stresnil (azaperone, 0.1 mg/kg of body weight; Janssen-Celag, Sollentuna, Sweden). With the pig lying on its right side, the L3–L4 disk was exposed using a left retroperitoneal approach. The cranial end plate of the L4 vertebra was perforated using a 3.5-mm drill with its tip inserted at the middle point of the lateral cortex of the vertebra (at its mid-height) and directed cranially toward the center of the end plate and nucleus pulposus. Angulated at 45° to reach the central part of the end plate, a single hole was drilled into the nucleus pulposus and the drill was withdrawn.18
Postoperatively, the pigs recuperated in an animal facility for 3 months. This model has been validated in an earlier study using gross morphology, histology, biochemistry, and magnetic resonance imaging at 3 months.18 After the animals were euthanized, the spine was excised en bloc, samples for histologic examination were fixed in formalin using routine procedures, and the injured and degenerated disks and paradiskal spine were later subjected to histologic and immunohistochemical examinations. Nonmanipulated vertebrae with associated disks and ligaments located contiguous to the experimentally traumatized segments were used as normal control tissues. Histologic sections were cut in sagittal orientation to the remnants of nucleus pulposus and annulus fibrosus (or in the control sample the disk itself) together with cartilaginous and bony vertebral end plates, vertebral body bone marrow, and spinal ligaments.
Histologic analysis
Samples were fixed for 24 hours in 4% neutral formaldehyde, decalcified over 3 weeks in 10% aqueous ethylenediaminetetraacetic acid (EDTA), dehydrated, and embedded in paraplast. Sections 3–4 μm thick were cut, mounted on l-polylysine-coated slides, and stained with HE for general evaluation of the tissues under study and for immunohistochemical analysis.
Immunohistochemical staining
Tissue sections of 5 μm were deparaffinized, rehydrated in descending ethanol series, and washed in 10 mM phosphate buffered 140 mM saline (pH 7.4). Endogenous peroxidase was blocked with 0.3% H2O2 in methanol for 25 minutes at +22°C. Antigen retrieval was performed in 0.01 M citrate buffer, pH 6.0, at +65°C for one hour. Nonspecific binding was blocked using 3% normal blocking serum (Vector Laboratories, Burlingame, CA, USA) diluted in 1% bovine serum albumin in phosphate buffer for 1 hour at +22°C. Primary antibodies used were affinity purified goat anti-porcine TNF-α antibody (optimal dilution 0.67 μg/ml), mouse anti-porcine IL-6 IgG1 (optimal dilution 1 μg/ml), and affinity purified goat anti-porcine IL-10 antibody (optimal dilution 0.67 μg/ml; R&D Systems, Minneapolis, MN, USA). They were diluted in 1% bovine serum albumin in phosphate buffer and used for overnight incubations at +4°C. Incubation in secondary antibodies (Vector Laboratories), diluted 1:200 in 0.1% bovine serum albumin in phosphate buffer, was performed at +22°C for one hour. Finally, the slides were incubated in freshly prepared avidin-biotin-peroxidase complex (ABC complex, Vector Laboratories) for one hour at +22°C. Color was developed using 0.03% H2O2 and 3,3′-diaminobenzidine (DAKO A/S, Glostrup, Denmark) for 3 minutes. All incubations were performed in humid atmosphere. The slides were rinsed 2 × 5 minutes after each step. Counterstaining was performed for 15 seconds in hematoxylin (Merck, Darmstadt, Germany), followed by dehydration through graded ethanol series, clearing in xylene and mounting in Histomount (Histolab, Gothenburg, Sweden). Negative staining controls for all antibodies included substitution of the primary antibody with irrelevant IgG of the same species, class, and concentration as the primary antibody.
Evaluation of the results
General histopathologic evaluation was done using all slides and the whole sample areas. After that semiquantitative microscopic assessment of immunohistochemical staining was performed under ×400 (high power fields) scoring 5 random fields per slide in 2 representative slides from each animal, using four grades: − = no immunoreactivity; + = only a few immunoreactive profiles; ++ = some immunoreactive profiles; and +++ = many immunoreactive profiles. Two histopathologists evaluated the results of histologic and immunohistochemical staining independently using this predefined and very simple scoring system. Readouts were very similar and were followed by a consensus session where the few and slight discrepancies were discussed. Stained specimens were analyzed and photographed using a Leitz Diaplan microscope coupled to a 5MP digital Leica DFC420 camera and a semiautomatic Leica Application Suite 3.0 image analysis and processing system.
Results
Histopathologic findings
The drill hole could not be seen in histologic sections, and the lesion was interpreted to be healed. General histologic analysis of the tissues stained with HE disclosed intervertebral disk degeneration characterized by disorganization of the lamellar structure of the annulus fibrosus, fissures, cystic or myxomatous changes, cloning of chondrocyte-like cells, and clefts within the cartilaginous endplate and cartilaginous metaplasia of the annulus. Granular degeneration of nucleus pulposus was also found. Spinal ligaments had in part lost their strict fiber orientation and packaging. Newly formed blood vessels were seen in disorganized compact connective tissue.
In control samples, annulus maintained its dense lamellar structure of collagen fibers without disorganization or the lamellar structure was slightly disorganized. Nucleus pulposus underwent some slight degree of fibrosis, but no granular degeneration was seen in experimental samples (Table 1). In all 6 pigs, histologic changes in experimental samples were rather similar.
Histopathologic changes in the experimental lumbar disc degeneration. ∗

BM = bone marrow; VB = vertebral bone; Ep = end plate; NP = nucleus pulposus; AF = annulus fibrosus; Lig = spinal ligament. Score: − = no change; ± = occasionally changes; + = slight changes; ++ = moderate changes; +++ = strong changes.
Immunohistochemical findings
TNF-α
TNF-α immunoreactivity was found in the fibrotically changed end plate of experimental disks (Fig. 1) and in degenerated disks themselves (Fig. 2). Additionally, a few TNF-α positive profiles were seen in blood vessel endotheliocytes and pericytes of degenerated intervertebral disks and in some fibroblasts of the spinal ligaments. Some TNF-α positive hematopoietic cells were found in bone marrow adjacent to the degenerated intervertebral disk (Table 2). In control samples, disk and spinal ligaments were negative, but a few TNF-α positive cells were found in bone marrow and in chondrocytes of the cartilaginous vertebral end plates.
Expression of tumor necrosis factor-α, interleukin-6, and interleukin-10 in immunohistochemical analysis in an experimental model of lumbar disk degeneration. ∗
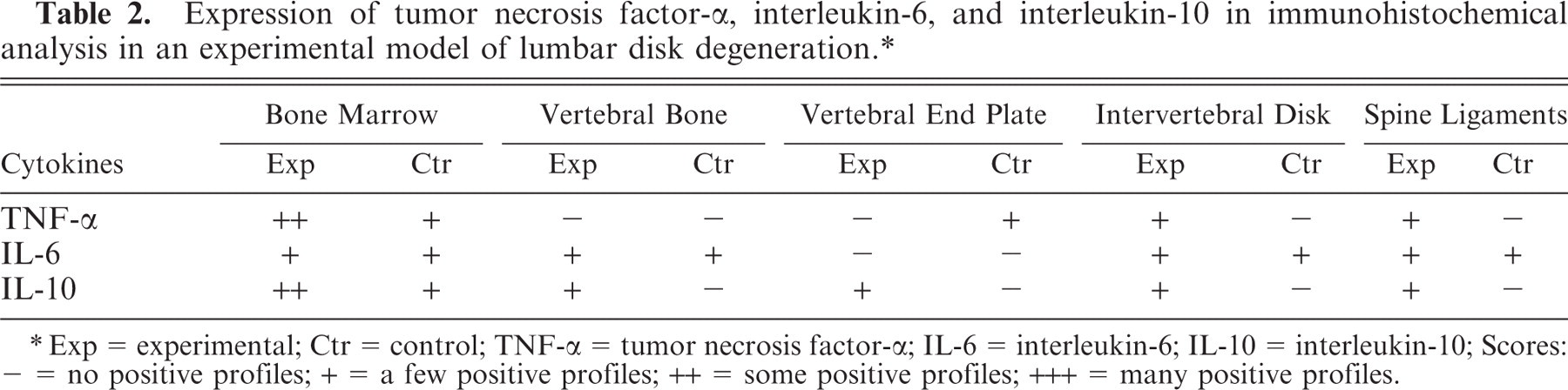
Exp = experimental; Ctr = control; TNF-α = tumor necrosis factor-α; IL-6 = interleukin-6; IL-10 = interleukin-10; Scores: − = no positive profiles; + = a few positive profiles; ++ = some positive profiles; +++ = many positive profiles.
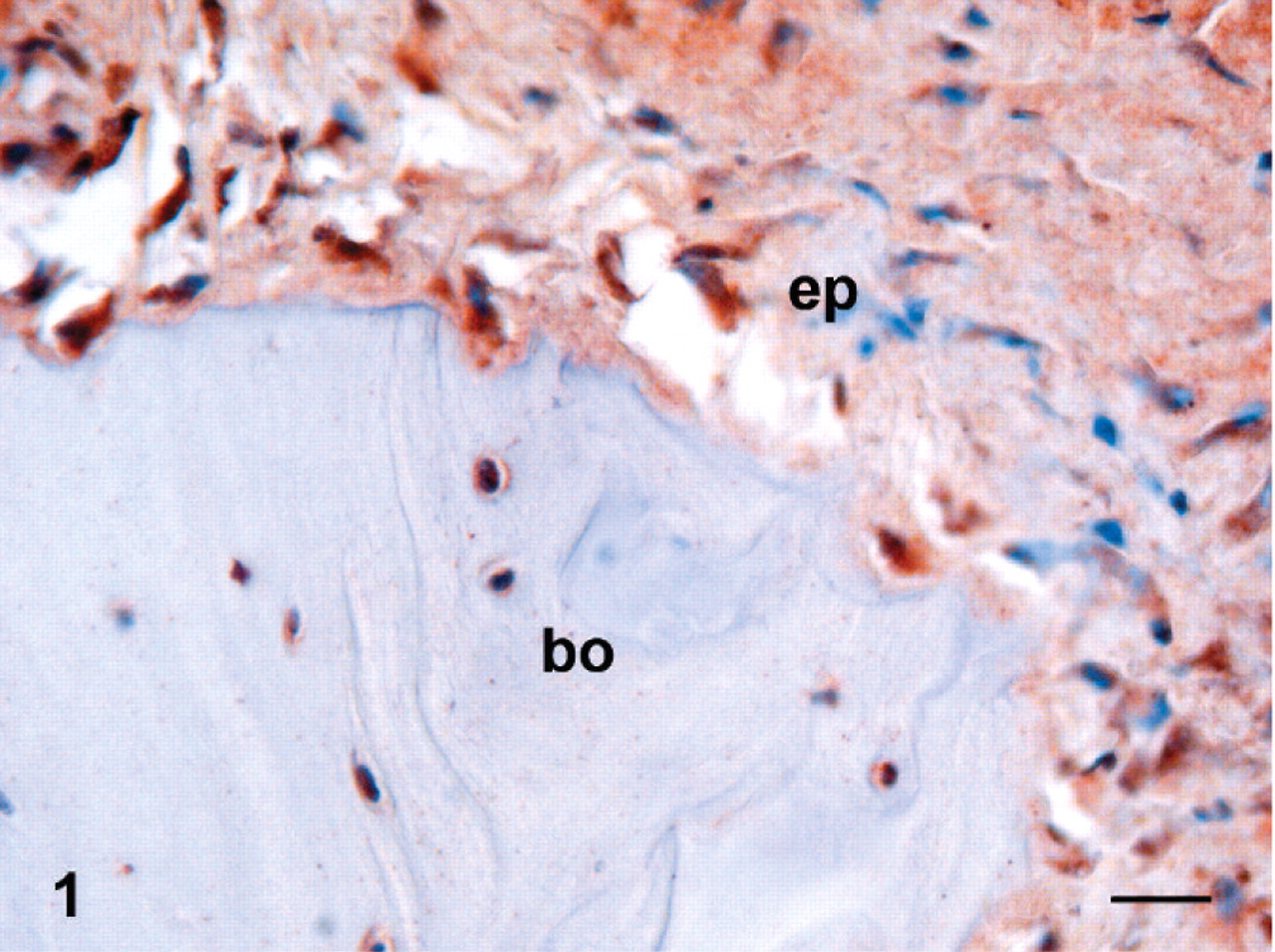
Tumor necrosis factor-α in the end plate and bone in an experimental porcine disk lesion model.
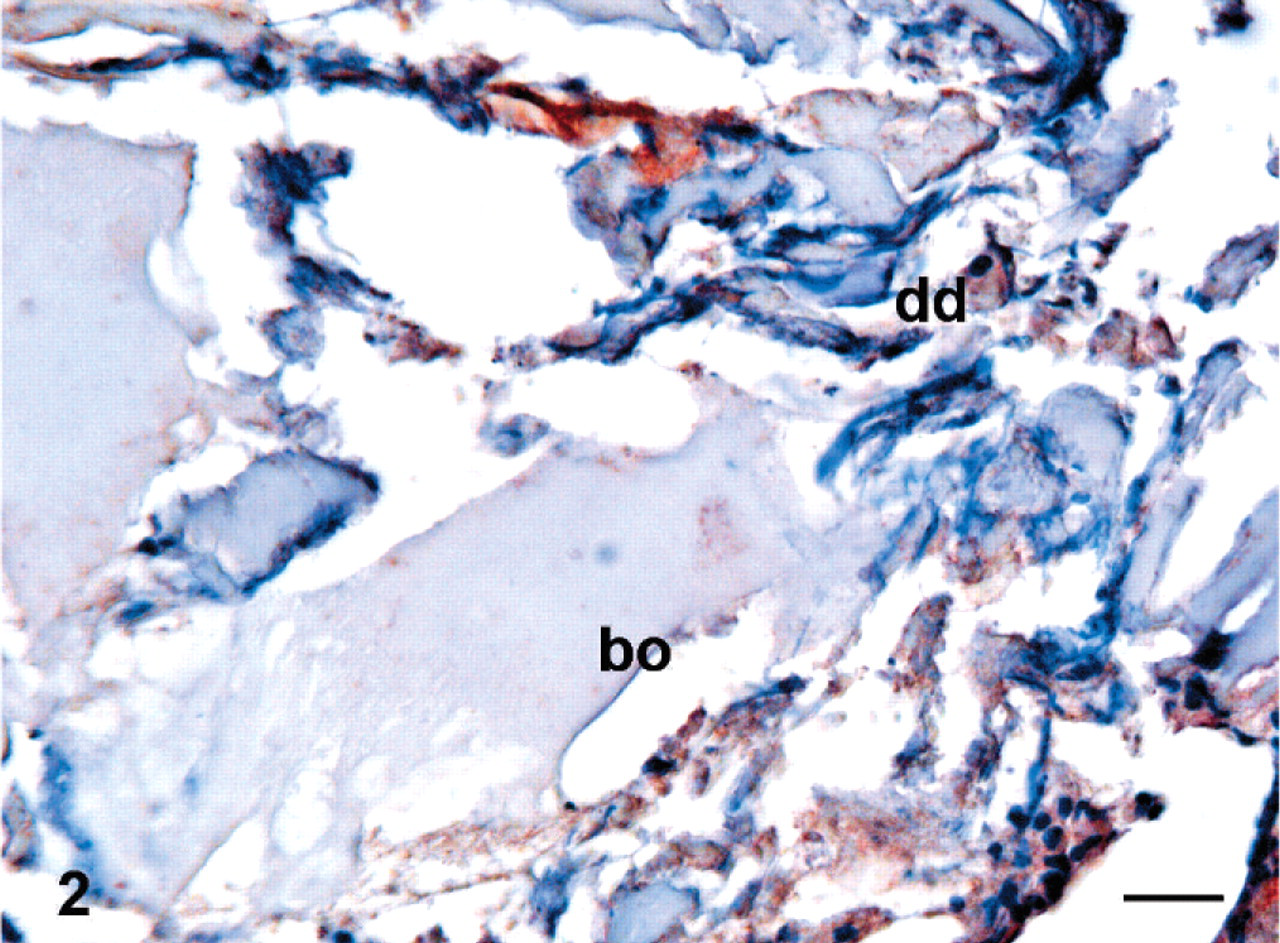
Tumor necrosis factor-α in an experimental porcine disk lesion model.
IL-6
A few IL-6 immunoreactive macrophages and fibroblasts were found in degenerated intervertebral disks (Fig. 3) and spinal ligaments. A few blood vessels in degenerated intervertebral disk and spinal ligaments also stained positively for IL-6. A few IL-6 immunoreactive cells were found in bone marrow and, similarly, a few periosteal cells, osteoblasts, and osteocytes of bone tissue were immunoreactive in the degenerated samples. In contiguous control samples, IL-6 was similarly distributed, as also shown in figures of bone marrow cells, osteocytes, blood vessel endotheliocytes and pericytes, and some fibroblasts in the disk (Fig. 4, Table 2).
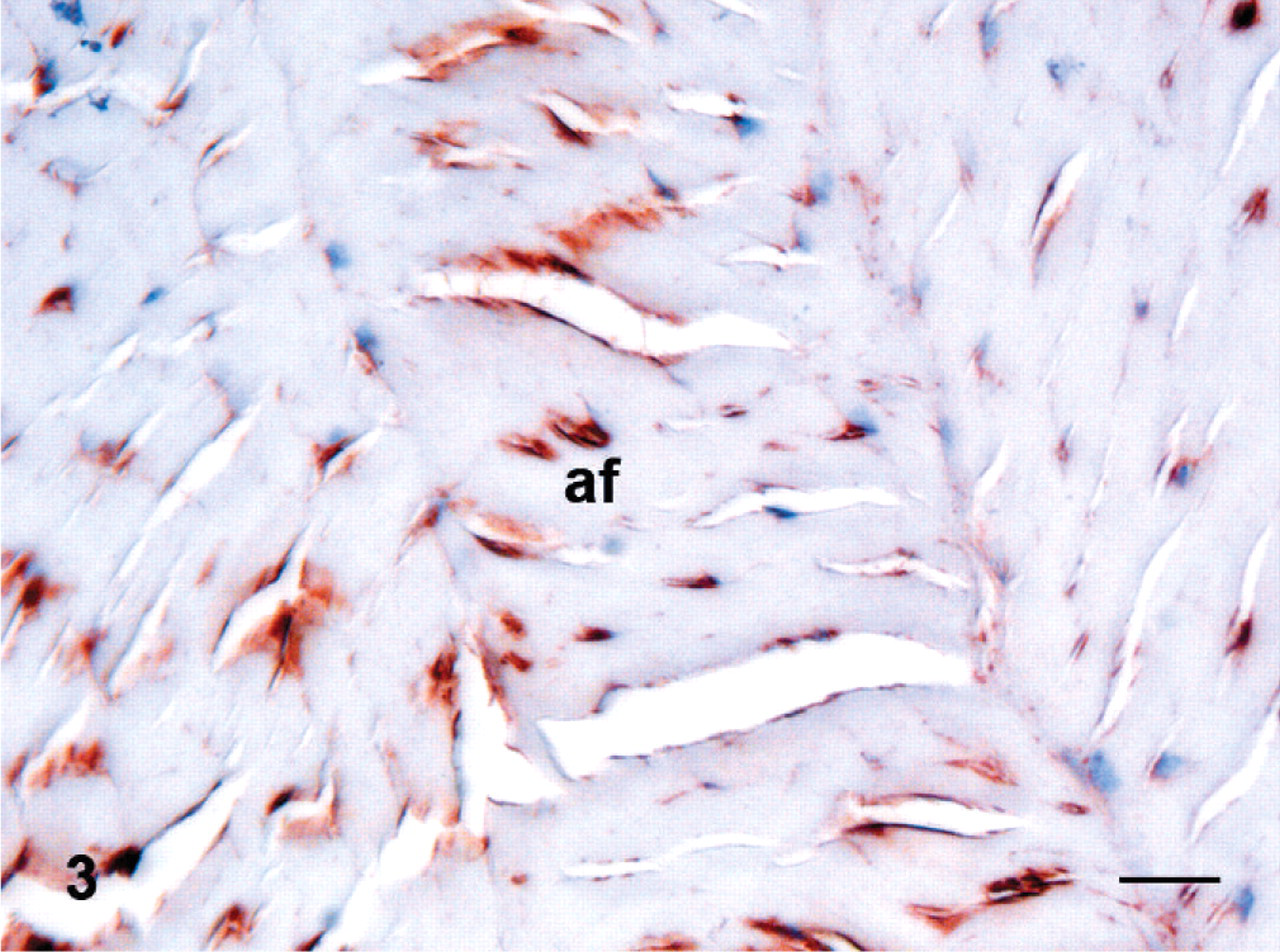
Interleukin-6 in the annulus fibrosus in an experimental porcine disk lesion model.
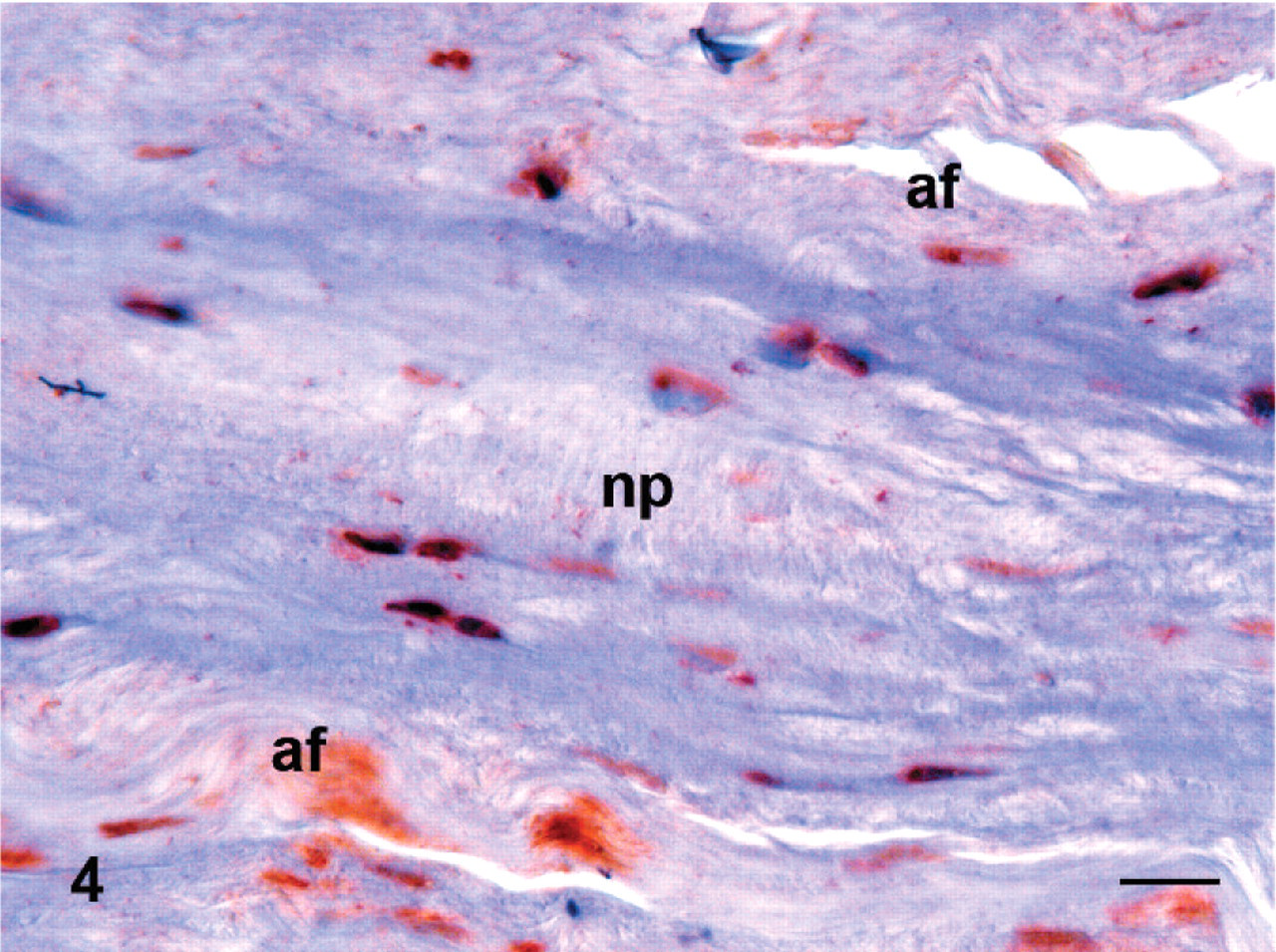
Interleukin-6 in the disk of the contiguous control vertebra. Immunoperoxidase staining, counterstained with hematoxylin. Bar = 20 μm. af = annulus fibrosus; bo = bone; dd = degenerated intervertebral disc; ep = endplate; np = nucleus pulposus.
IL-10
A few IL-10 positive cells, predominantly fibroblasts (Fig. 5), were found in damaged and degenerated intervertebral disks (Fig. 6) and adjoining spinal ligaments. Some IL-10 positive cells were found in hematopoietic cells in bone marrow (Fig. 5). In addition, a few chondrocytes in the end plates and osteocytes and osteoblasts in vertebral bone were IL-10 immunoreactive (Table 2). In control samples, a few IL-10 positive cells were found in the end plate (Fig. 7) and some hematopoietic cells in bone marrow were IL-10 positive.
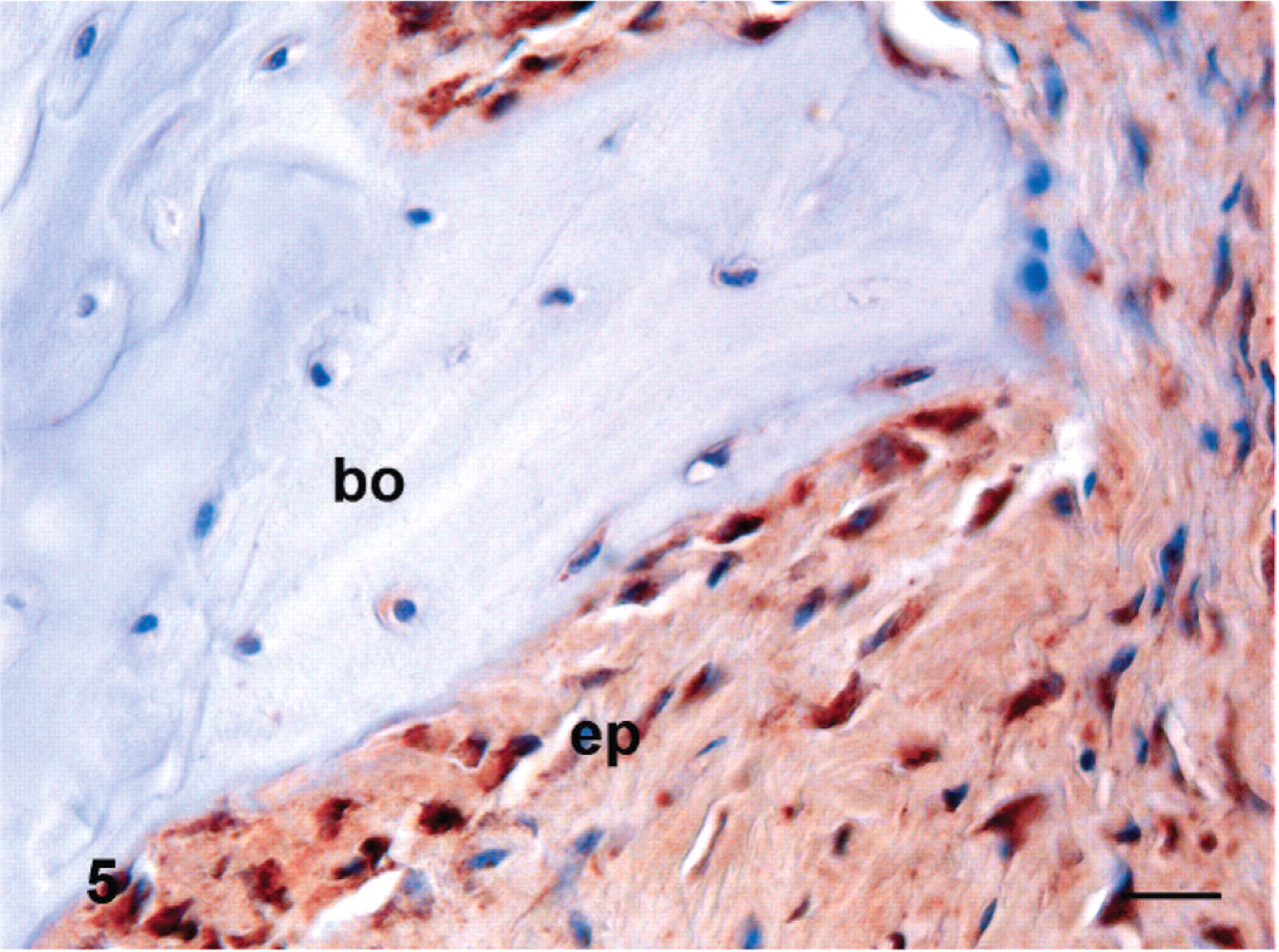
Interleukin-10 in the end plate in an experimental porcine disk lesion model.
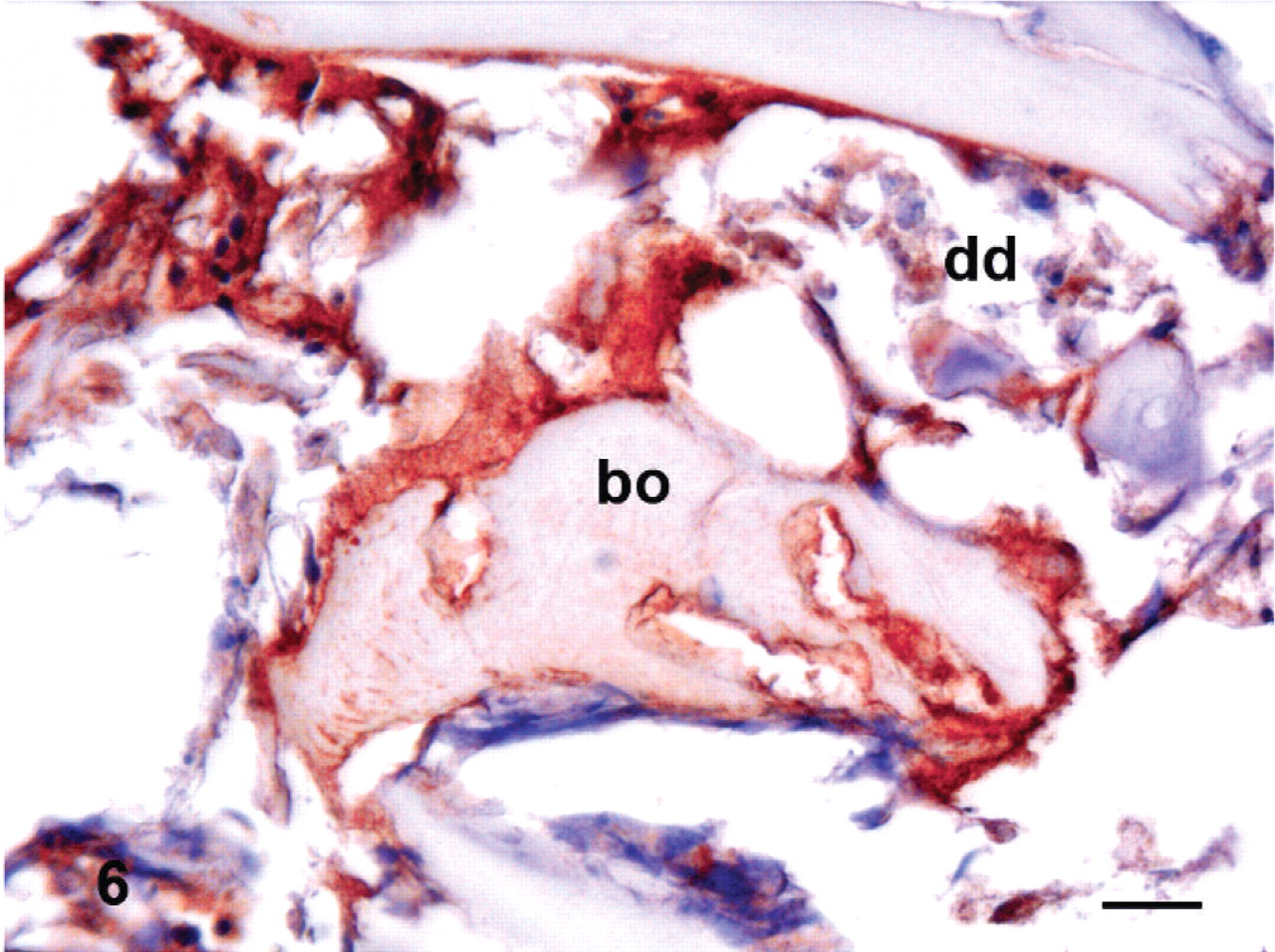
Interleukin-10 in an experimental porcine disk lesion model.
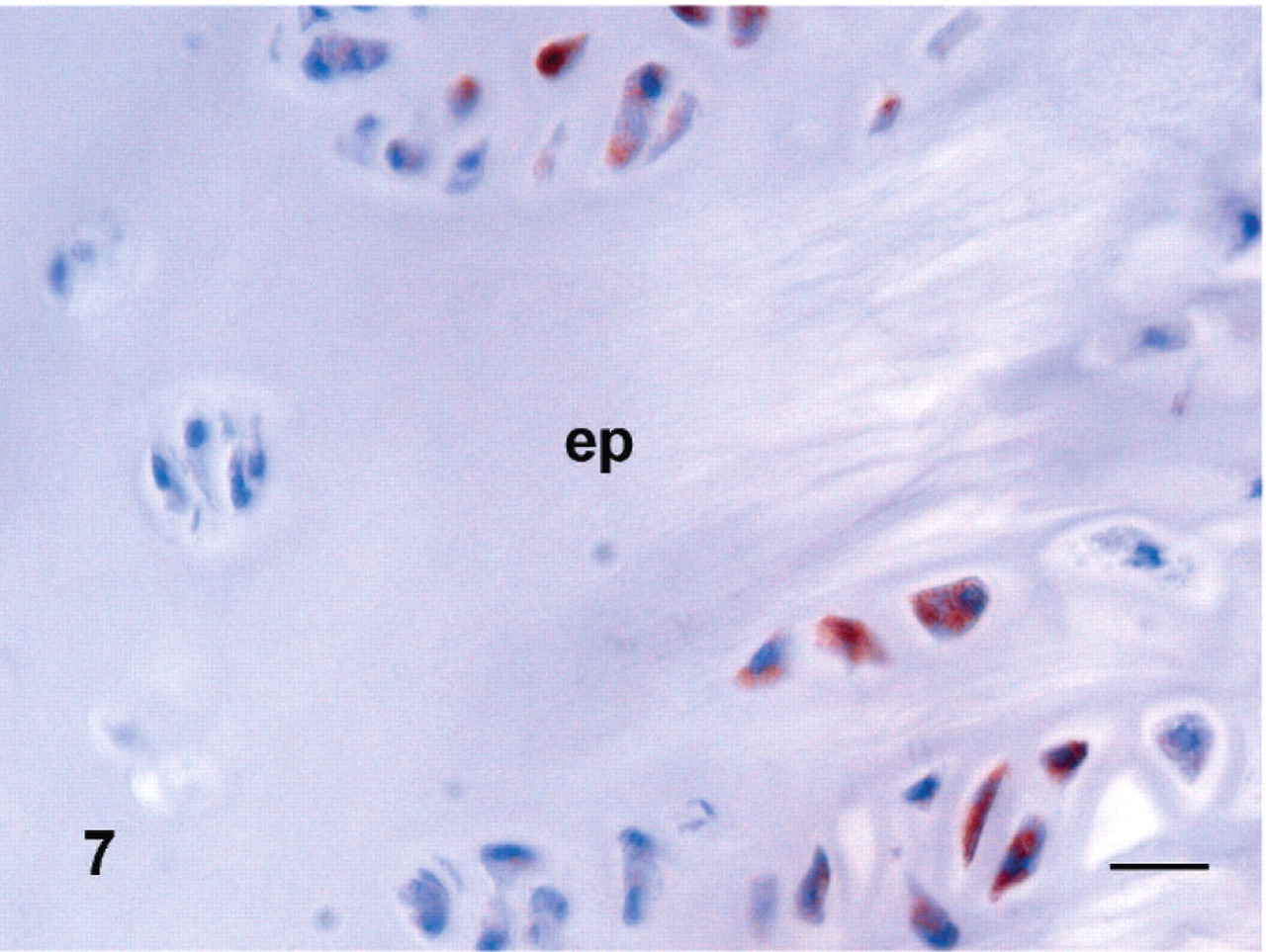
Intereleukin-10 in the end plate of a contiguous control vertebra.
Negative staining controls using goat or mouse IgG were always negative (Fig. 8).
Negative staining control with normal goat IgG of an intervertebral disk. Immunoperoxidase staining, counterstained with hematoxylin. Bar = 20 μm. af = annulus fibrosus; bo = bone; dd = degenerated intervertebral disc; ep = endplate; np = nucleus pulposus.
Discussion
This study disclosed that the clearest changes occurred with IL-10, modest changes occurred with TNF-α, but no clear changes occurred in IL-6 labeling. In spite of an increase in TNF-α, this experimental model does not induce any apparent pain at 3 months, so that the pigs did not disclose any pain behavior and were not receiving any analgesics at the time of sacrifice. Therefore, this model provides mainly information on the subclinical histologic and molecular events (cytokines) that, if excessive and when advanced, may also contribute to diskogenic pain. Neurons in lumbar intervertebral disk may play a role in inflammation via their neuropeptides and other nonadrenergic-noncholinergic (NANC) transmitters, and in more severe cases of intervertebral disk disease they may be at least partly responsible for diskogenic pain.3 TNF-α in higher quantities stimulates synthesis of molecules, which sensitize such primary afferent nociceptive nerves.23, 25 In humans it was demonstrated that the levels of pro-inflammatory mediators are higher in the degenerated disks from patients with chronic low back pain than in those with asymptomatic disk damage.8 During disk inflammation, proteolytic processes are activated and blood vessels and accompanying nerves grow into the disk.9, 13, 14, 17, 23 Systemic TNF-α blockade with a single 3 mg/kg infliximab infusion given in 2 hours gave promising results in an open trial,27 but even 5 mg/kg was not more effective in pain control (reduction in leg pain from baseline to 12 weeks) than placebo in a controlled clinical trial.28 Local overexpression of interleukin-1 receptor antagonist (by gene transfer) might offer a more effective means of delivery for therapeutic agents.30 The present work confirms that degenerated intervertebral disks contain TNF-α positive macrophages and fibroblasts, which were not found in healthy disks. The effects of local disk pathology were, in addition to changes of TNF-α in the disk itself, also reflected in TNF-α expression in some fibroblasts of the spinal ligaments, which may have been subjected to abnormal strain. In many respects, the local role of TNF-α in juxtacrine, autocrine, and paracrine pro-inflammatory processes seems to be quite well established in intervertebral disk degeneration.47
IL-6 takes an intermediate place between the pro-inflammatory and anti-inflammatory cytokines.48 IL-6 down-regulates many chemokines,19 TNF-α37 and IL-137, and up-regulates IL-1Ra43 and soluble TNF-R type I (p55),42 shifting the pro-inflammatory and anti-inflammatory balance toward recovery and repair. Differential labeling for IL-6 was not demonstrated in this study at 3 months. Although the reason for this is not clear, it might be due to differences in reaction kinetics and long-term regulation of the target cytokines studied in the present work.
The most consistent changes of all were seen in IL-10. In spite of the degenerative and inflammatory nature of the intervertebral disk disease, paradoxically, IL-10 was most consistently up-regulated in the disk itself or in the paradiskal structures studied, namely the bone marrow, vertebral bone, vertebral end plates, and spinal ligaments. This certainly reflects to some extent the relatively late phase of the experimental accelerated intervertebral disk disease studied here, which at 3 months in pig has progressed quite far in the direction of repair, although loss of the nucleus pulposus does not allow complete recovery. Another apparent reason for the predominance of IL-10 is probably its high responsiveness to pro-inflammatory stimuli, like lipopolysaccharide, which has been shown using cultured murine disk-derived cells and enzyme-linked immunosorbent assay for the measurement of IL-10 secreted into the cell culture medium.35 The anti-inflammatory IL-10 in intervertebral disk disease is probably increased as a result of the tight coupling of the pro-inflammatory arm of the local disk disease process with an anti-inflammatory regulatory arm of the response, which is necessary to prevent excessive stimulation and tissue destruction.
Cytokine balance contributes to the onset, progression, and resolution of inflammation. TNF-induced receptor activator of nuclear factor κB ligand synthesis by bone marrow stromal cells is a fundamental component of inflammatory osteolysis, whereas IL-10 in vivo suppresses infection-induced bone resorption36 and cartilage destruction.21 This is seemingly at odds with an earlier piece of work in which IL-10 messenger RNA (mRNA) could be found in only 2 of 23 (9%) human herniated disk samples when a sensitive real-time reverse transcription-polymerase chain reaction (RT-PCR) method was used.1 Because of technical problems in isolating mRNA from intervertebral disks, this work1 may have underestimated IL-10. Isolation of IL-10 mRNA from such tissue can be quite tricky, in particular from disk tissue, as the long glycosaminoglycan (GAG) chains apparently behave like RNA. TRIzol has been found to work for all soft tissues, but not for cartilage and in particular not for the disk tissue. Furthermore, contaminating proteoglycan GAG also seems to inhibit reverse transcriptase46 (E. Vuorio, personal communication), and isolation may require pulverization, extraction in 4 M guanidine hydrochloride, 10 mM Tris-HCl, and 140 mM NaCl containing proteinase inhibitors followed by clarification by centrifugation at 15,000 × g for 40 minutes at +4°C.24, 46 Second, because IL-10 expression is regulated differently in different areas, sampling bias might explain the differences between the present observations and the RT-PCR results published earlier.1 Increases similar to those reported in this article for IL-10 were described earlier in disk degeneration for other molecules and were associated with repair and healing rather than inflammation and tissue destruction.24
In conclusion, immunohistochemical analysis indicates that there are slight increases in TNF-α and IL-10 in late intervertebral disk disease compared with controls, which suggests that they participate in local inflammation, repair, and tissue remodeling. The present work suggests that pure disk trauma and the resulting degeneration affect not only the injured unit but also contiguous spinal structures, which undergo secondary pro-inflammatory and anti-inflammatory remodeling processes, possibly because of altered load transfer caused by biomechanical derangements. Finally, this porcine model seems useful in the study of pathomechanisms involved in intervertebral disk disease.
Footnotes
Acknowledgements
This study was supported by Clinical-evo grants, Finska Läkaresällskapet, ORTON Foundation, Sigrid Jusélius Foundation, EU COST Biotribology Action 533, TBGS National PhD Graduate School, Danish Council for Strategic Research, Wilhelm and Else Stockmann Foundation.
