Abstract
Purpose
Based on previous investigations on the vascular blood supply to the femoral head, a technique for anatomical reduction after slipped capital femoral epiphysis was developed. This technique is a modification of the original technique by Dunn using a retinacular soft-tissue flap. This allows the visual control of the epiphyseal vascular blood supply. We report the experience at the inventor's institution with a critical discussion of the available literature.
Methods
Using a trochanteric osteotomy for surgical dislocation of the hip, a retinacular soft tissue flap is created containing the deep branch of the medial femoral circumflex artery, the external rotators and the capsule. The femoral epiphysis can be mobilised safely and reduced on the femoral neck after resection of the almost constantly present reactive metaphyseal callus.
Results
In our institution, the rate of avascular necrosis with 2% is comparably low to Dunn's original results. It is only present in cases where no bleeding was already evident before reduction of the epiphysis. The ten-year long-term results are favorable in these cases with a good functional result and only little progression of osteoarthritis. However, other authors have reported higher rates of avascular necrosis up to 24% in their initial experience.
Conclusions
In experienced hands using the correct meticulous surgical technique, the results are favorable regarding the rates of avascular necrosis, the functional outcome and the development of radiographic osteoarthritis – even in acute and severe cases. Avascular necrosis is rare but can be observed if there is no evidence of intra-operative femoral head perfusion before and after reduction of the epiphysis.
Keywords
Introduction
The treatment of slipped capital femoral epiphysis (SCFE) has been controversial for decades. It is in the nature of things that an anatomical reduction of the epiphysis provides the best condition for a good function of the hip joint in the long-term – given a surgical technique that can control the blood supply to the femoral head. The attempts to anatomically reduce the femoral epiphysis by a subcapital reorientation with an anterior approach in the mid of the last century have resulted in rates of avascular necrosis (AVN) up to 54%.1,2 In 1964, Dunn presented a promising technique of subcapital reorientation through a posterior approach with a trochanteric osteotomy with a risk of avascular necrosis of only 4%. 3 Similar to the observations in the last 15 to 20 years in our institution, Dunn pointed out that a callus formation at the posterior aspect of the femoral neck is present in most of the cases. 4 This callus formation can neither be seen nor addressed from anteriorly or through a closed reduction maneuver. This can explain the high AVN rate with these techniques, since the typically shortened retinacular vessels in SCFE are stretched and may be occluded when the epiphysis is reduced. He pointed out that a careful subperiosteal preparation of the retinacular vessels with a resection of the newly formed callus and some neck shortening is the key to preserve the blood supply to the femoral head (Fig. 1). Many authors have tried to reproduce Dunn's results. However, they report substantially higher rates of AVN up to 54%, 1 and the technique has not found a wide distribution.
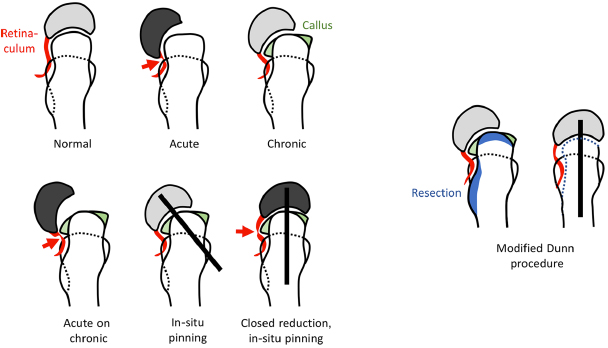
Overview of different conditions in slipped capital femoral epiphysis (SCFE). In the acute slip, kinking of the retinacular vessels (arrow) may occur leading to ischemia of the femoral epiphysis. In a chronic SCFE, callus formation has built reactively in particular on the posterior aspect of the metaphysis. In the acute-on-chronic situation, kinking may occur again (arrow). In-situ pinning of an acute or chronic slip usually preserves the femoral head blood supply. Closed reduction maneuvers stretch the retinaculum over the posterior callus leading to an increased risk of avascular necrosis. With the modified Dunn-procedure, the greater trochanteric massif is reduced and a soft tissue flap is developed containing the retinacular vessels. With additional shortening, the epiphysis can be reduced safely without tension to the retinaculum.
In 1991, Carney et al presented their large milestone data from Iowa on various treatments for SCFE. 5 They concluded that pinning in situ has the lowest risk of AVN, which eventually became the treatment of choice even for severe SCFEs. However, there is increasing evidence that the remaining deformities may imply a substantially higher risk of functional hip problems due to femoroacetabular cam-type impingement in the mid- and long-term with premature hip osteoarthritis.6,7
The profound studies on the vascular blood supply to the femoral head at our institution in the past 20 years have led to the development of a surgical dislocation of the femoral head with an almost negligible risk of AVN given a correct surgical technique.8–10 As an extension of this approach, the retinacular soft tissue flap can reproducibly be developed. 11 This allows full surgical access to the femoral neck and the epiphysis with visual control of the integrity of the retinacular vessels to the femoral epiphysis. Compared with a standard surgical hip dislocation, the dissection is even closer to the afferent vulnerable blood supply to the femoral epiphysis, which puts the retinacular vessels to a somewhat higher risk of iatrogenic injury. Finally, the technique of a subcapital reorientation through a surgical dislocation of the hip with development of a retinacular soft-tissue flap has been developed.12,13 In this article, we present the indications, the surgical technique, the currently available outcomes from the literature and a self-critical analysis of these results.
Indications
Basically, the modified Dunn procedure can be performed for any type of SCFE. At our institution, it is indicated for every moderate and severe (acute or chronic) SCFE, which covers more than 80% to 90% of our procedures. Mild slips with a Southwick angle of less than 15° are typically treated with in-situ pinning and concomitant arthroscopic or open osteochondroplasty. Here, the slight remaining malorientation of the femoral head cartilage and the mismatch between the acetabular fossa and the fovea of the femoral head seems to be acceptable.
It is crucial to recognise the maximum amount of slippage. This can typically not be seen on the conventional pre-operative radiographs. We therefore perform routinely pre-operative magnetic resonance imaging with acquisition of radial sequences.
Surgical technique
The patient is positioned in the lateral decubitus position with the operated leg positioned on a tunnel pillow (Fig. 2). The entire leg is prepped and draped in sterile fashion. A sterile bag is fixed on the opposite side of the operating table to allow the later dislocation of the hip joint in the figure of four position. A straight lateral incision centered over the greater trochanter is used. The fascia lata is incised along the anterior border of the gluteus maximus muscle and the Gibson interval between the gluteus medius and maximus muscles is developed (Fig. 3). Perforating epifascial vessels originating from the inferior gluteal artery can be used as landmarks to find the anterior border of the gluteus maximus muscle. The trochanteric bursa is incised and the piriformis and gluteus minimus muscles are identified by retracting the gluteus medius anteriorly. Care must be taken to avoid an iatrogenic injury to the anastomosis of the inferior gluteal artery, which runs at the inferior border of the piriformis muscle. This important and relatively constant anastomosis to the deep branch of the medial circumflex artery can potentially guarantee the blood supply to the femoral head in case of damage of the deep branch.
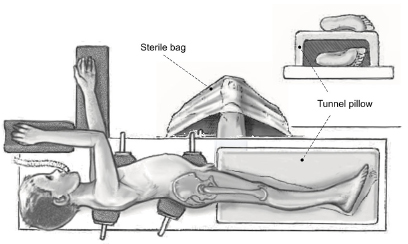
Patient positioning. Adapted with permission from Leunig et al. 13
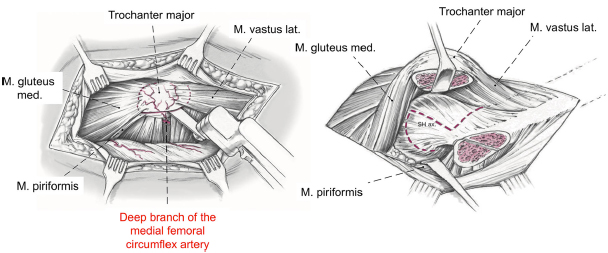
Greater trochanteric osteotomy and capsulotomy. Adapted with permission from Leunig et al. 13
A straight digastric trochanteric osteotomy is performed leaving the most posterior fibers (approximately 2 mm to 3 mm) of the gluteus medius tendon intact. This ensures that the osteotomy is not too medial potentially endangering the deep branch of the medial femoral circumflex artery. Using this technique most fibers of the piriformis tendon remain attached to the stable portion of the greater trochanter. The capsule is then exposed between the piriformis and gluteus minimus muscles by mobilisation of the trochanteric fragment together with the vastus, gluteus minimus and medius muscles ventrally (Fig. 3).
A z-shaped capsulotomy is performed, which allows a direct visualisation of the epiphyseal slip including the prominent metaphysis. In case of an unstable slippage, the femoral epiphysis is temporarily pinned with 2 mm-Kirschner wires (Fig. 4). The viability of the femoral epiphysis is ensured by drilling of the femoral head. In most cases, the epiphysis is bleeding since the posterior slippage basically relaxes the retinacular vessels. However, in severe slips, there is a potential that the retinacular vessels may be kinked leading to a relevant temporary ischemia of the epiphysis.
Dislocation of the femoral head after temporary in situ pinning of the displaced epiphysis. Adapted with permission from Leunig et al. 13
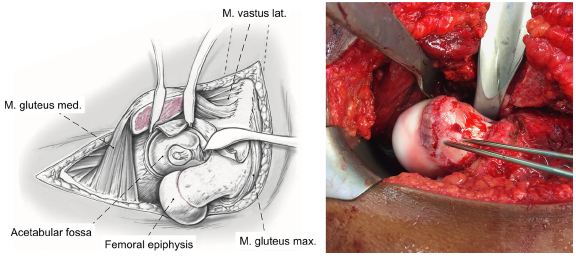
After cutting the capitis femoris ligament, the femoral head is then dislocated by placing the lower leg in the opposite sterile bag in flexion and external rotation. This allows a full inspection of the corresponding damage to the acetabulum (Fig. 4). Interestingly, there is already considerable damage even in acute slips indicating that there has been some previous ongoing pathology. The chondrolabral damage is often severe independent from the degree of slippage. In mild cases, the asphericity can even enter deeper into the socket than in severe slips.
After relocation of the femoral head, the retinacular soft tissue flap is developed (Fig. 5). This flap contains the deep branch of the medial femoral circumflex artery, the periosteum, the piriformis muscle, part of the posterior capsule, and the short external rotators. The flap is developed by stepwise resection of the most posterior portion of the greater trochanter. The reduction of the greater trochanter starts at the level of the trochanteric apophysis which indicates the base of the femoral neck. The periosteum of the femoral neck is incised along its axis, and the retinacular flap is then carefully mobilised proximally with the periosteal elevator. Distally, the flap should be extended to the level of the lesser trochanter, which significantly reduces the tension from the retinacular vessels (Fig. 6). Anteriorly, the typically partially torn periosteum is peeled off the bone with a periosteal elevator with the head in dislocated position.
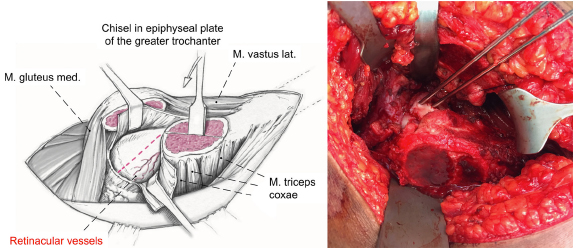
Periosteal incision and development of the retinacular soft-tissue flap. Adapted with permission from Leunig et al. 13
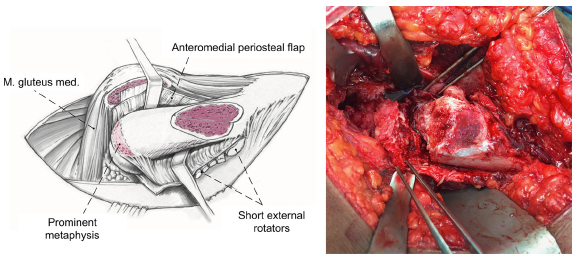
Final appearance after complete development of the retinacular soft-tissue flap. Adapted with permission from Leunig et al. 13
The femoral head is then stepwise mobilised using chisels starting from anterior (Fig. 7). This gradual separation of the femoral epiphysis from the metaphysis is facilitated with external rotation. Eventually, the epiphysis becomes completely mobile but still attached to the posteroinferior retinacular soft tissue flap. The metaphyseal stump can be best exposed when the epiphysis is relocated in the acetabular socket. The callus formation of the metaphysis is basically always present except in traumatic SCFE and must be resected until a spherical surface of the neck becomes apparent. Further shortening of the neck may be necessary in case of difficulties with the reduction of the epiphysis to prevent tension to the retinacular vessels (Fig. 8).
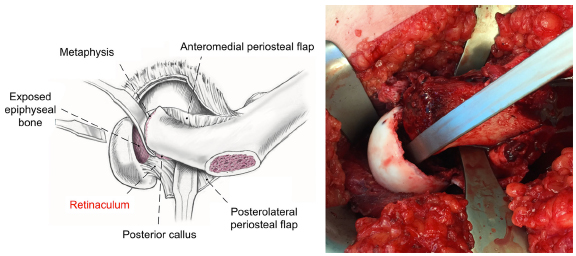
Mobilisation of the epiphysis from the metaphysis. Adapted with permission from Leunig et al. 13
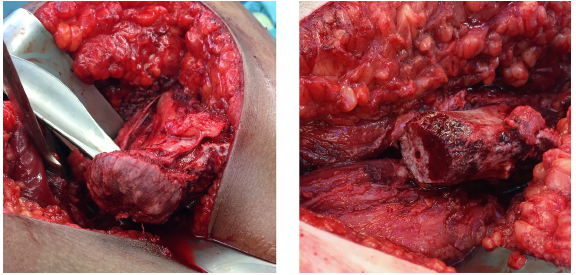
The metaphyseal stump with the prominent reactive callus formation can be inspected with the epiphysis relocated in the socket (left). The metaphysis is then cleaned from any excessive callus bone.
The epiphyseal scar of the head is then cleaned from growth plate tissue with a curette or a high-speed burr (Fig. 9). Under visual control of the retinaculum, the epiphysis is then reduced manually on the metaphyseal stump. A 3.0 mm fully threaded Kirschner-wire is placed anterograde starting at the fovea of the femoral head (Fig. 10). One or two more Kirschner-wires are then placed divergently from retrograde in the epiphysis. The correct alignment of the epiphysis is checked by fluoroscope. Epiphyseal perfusion after the capital realignment is checked routinely using a 2 mm-drill hole with observation of subsequent bleeding.
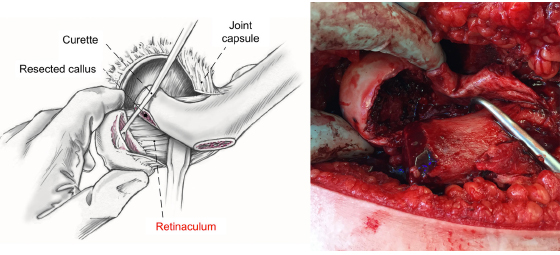
Curettage of the epiphyseal growth plate. Adapted with permission from Leunig et al. 13
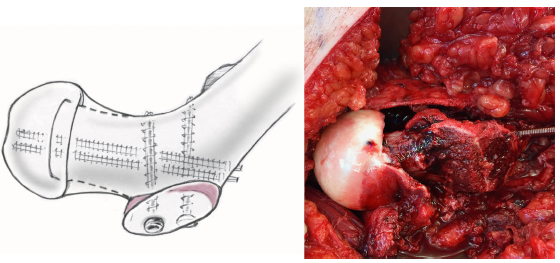
Fixation of the epiphysis with two to three 3.0 mm fully threaded Kirschner wires. Adapted with permission from Leunig et al. 13
The anterior periosteum and the posterior retinacular flap are then reattached loosely with tension-free sutures. Similarly, the capsule is closed without tension. The greater trochanter is readapted with two 3.5 mm cortical screws optionally with advancement, if needed.
Post-operative care includes the use of a motorised continuous passive motion device during the hospital stay. Partial weight-bearing is needed for eight weeks. After radiographic evidence of consolidation, stepwise transition to full weight-bearing is permitted with normal use of the hip after three months.
Results from the inventor's institution
We have presented the mean 5 and 10-year follow-up of the first 30 hips in 30 patients undergoing the modified Dunn procedure at our institution starting in 1998.14,15 In this series, there were no cases of avascular necrosis (Fig. 11). The epiphyseal anatomy could be restored with residual slip angles ranging from 4° to 8°. Considerable chondrolabral damage of the acetabulum was observed consistently, regardless of the stability of the slip. 16 These promising results could be reproduced by the Bostonian group. 15 Although there were initial cases of hardware failure using screws instead of threaded Kirschner-wires, the functional long term outcome of the patients improved significantly. 14 Over the years, 14% of the patients required a secondary procedure to address bone spurs at the femoral head-neck junction and/or the acetabulum that had developed over time leading to a secondary femoroacetabular impingement. Overall, the cumulative ten year survival rate was 93%, which was defined as no progression of OA, no conversion to THA and no poor clinical outcome. 14
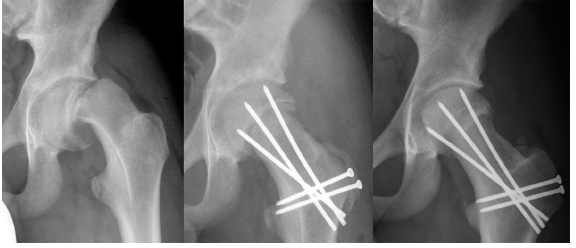
Example of a 14-year-old male patient with an acute-on-chronic SCFE treated with a modified Dunn procedure using a surgical hip dislocation approach with development of a retinacular soft tissue flap: pre-operatively (left), post-operatively (mid), and at four years follow up.
In a second series from our institution including 23 patients between 2004 and 2007, we found one case of osteonecrosis. 17 In this patient with symptoms for more than three weeks, no perfusion of the femoral head was evident after drilling although the retinacular vessels appeared to be macroscopically intact. Unfortunately, unlike in another published case, 18 there was no evidence of reperfusion in a bone scan two weeks post-operatively. Eventually, the patient developed avascular necrosis of the femoral head and required arthrodesis. Based on these two publications, the overall incidence of AVN in our institution is 2%.
Results from other institutions
Given the very promising results from our institution, the modified Dunn procedure has been used in several other centers for moderate and severe SCFEs (Table 1). In a comparative study in severe stable SCFEs, the modified Dunn procedure was found to have a better radiographic correction, a better clinical outcome and a lower reoperation rate compared to in situ pinning. 19
Results of the modified Dunn procedure.
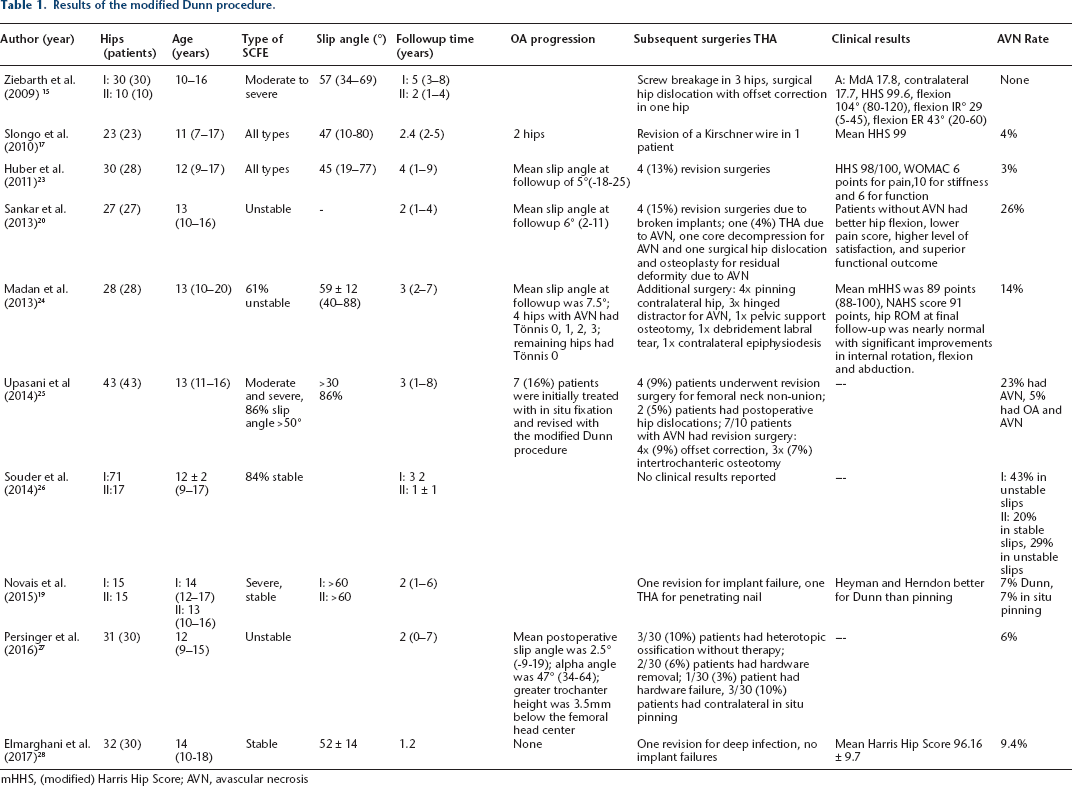
mHHS, (modified) Harris Hip Score; AVN, avascular necrosis
Analogously to the historical experience with Dunn's original technique, the results from the inventor's institution could only be partially reproduced. The reported rates of AVN varied between 3% and 24% (Table 1, Fig. 12). However, when analysing these results critically, there might be several reasons for this. In particular, the technique was mostly used in acutesevere cases with an inherently higher risk of avascular necrosis in the natural course of the disease. Another limitation might be the relatively high number of surgeons involved compared to our setup resulting in less routine with this demanding surgical technique. The proposed minimum number of ten surgical hip dislocations 20 per year does not seem to be the appropriate number to justify a modified Dunn procedure in our experience. In addition, the exact amount of the individual resection of the callus formation at the posterior level of the femoral neck is crucial. Finding the balance between a too short femoral neck (resulting in instability) 21 and an appropriate neck length to ensure a tension-free reduction of the femoral epiphysis is hard and requires years of surgical experience. 22
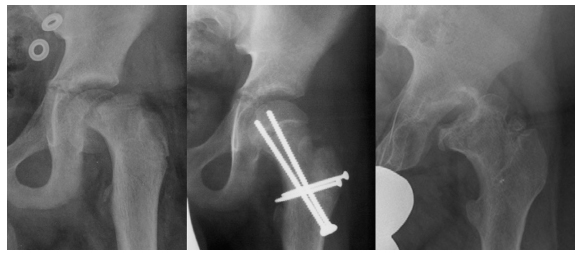
Example of a 12 year old boy with an unstable left SCFE treated with a modified Dunn procedure (with permission from Sankar et al) 20 pre-operatively (left), post-operatively (mid) and after hardware removal (right) revealing evidence of avascular osteonecrosis. Adapted with the permission of Sankar et al. 20
The modified Dunn procedure using the retinacular soft-tissue flap offers the opportunity to anatomically reduce a displaced femoral epiphysis while visually controlling the retinacular perfusion of the femoral head. In experienced hands using the correct meticulous surgical technique, the results are favorable regarding the rates of AVN, the functional outcome and the development of radiographic osteoarthritis – even in acute and severe cases. Avascular necrosis can be observed if there is no evidence of intra-operative femoral head perfusion before and after relocation of the epiphysis.
Footnotes
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
Each author certifies that his or her institution has approved the human protocol for this investigation and that all investigations were conducted in conformity with ethical principles of research.
One author (MT) received funding from the Swiss National Science Foundation (SNSF; Nr. PP00P3_144856). None of the remaining authors (LJ, TL, FS, KZ, KS) received funding.
