Abstract
Objectives
The purpose of this study is to evaluate the effects of allogenic costal cartilage transplantation on preventing bony bridge formation and angular deformities for the treatment of partial growth plate injury using a rabbit model.
Methods
An experimental model of partial growth injury was created by resecting the medial part of the proximal tibial growth plate in male six-week-old New Zealand White rabbits. The rabbits were divided into four groups: no surgery; no transplantation; bone wax transplantation; and allogenic costal cartilage transplantation. The angular deformities of the tibia and bony bridge were analysed using radiographs and microcomputed tomography, and the repair of the injured growth plate cartilage and bony bridge formation rate were histologically evaluated.
Results
On radiographic evaluation, the varus deformities in the costal cartilage group were significantly improved compared with the no transplantation group at four and eight weeks after operation and with the bone wax group at eight weeks after operation. Micro-computed tomography showed that the bony bridge formation was prevented in the bone wax and costal cartilage groups. Histological findings showed that the bony bridge formation in the bone wax and costal cartilage group was decreased. In addition, the growth plate was continuous and stained with safranin O and immunohistochemically stained for type II collagen.
Conclusion
Transplantation of costal cartilage improved angular deformities and decreased bony bridge formation in the partial growth plate injury. Costal cartilage might be a suitable graft for the treatment of growth plate injury.
Introduction
Growth plates exist in the metaphysis of growing children. They consist of a hyaline cartilage layer that is involved in the longitudinal growth of long bones. In children, a bony bridge forms at the injury site when the growth plate is injured by fractures, tumours, infection or side effects after radiation therapy. Injured growth plate cartilage and the bony bridge can result in leg length discrepancy, angular deformities and rotational deformities.
In surgical treatment for partial growth injury, autogenous fat and non-biological grafts are clinically transplanted after bony bridge resection to prevent the bony bridge reforming and recurrence of angular deformities.1–5 Although these grafts may prevent the bony bridge reforming, the injured growth plate cartilage is not repaired. Using a growth plate injury model in animal experiments, chondrocytes and mesenchymal stem cells (MSCs) have been transplanted to repair the growth plate cartilage.6–12 Previously, Yoshida et al transplanted synovial MSCs in a rabbit model. 13 They showed that varus deformities were improved by the transplantation of autogenous chondrocytes and MSCs, and the growth plate cartilage was repaired. However, in a larger animal model, the injured growth plate cartilage was not completely repaired and fibrous tissue formed at the injury site. 10 Moreover, autogenous chondrocytes and MSCs must be collected before the operation and cultured to acquire a sufficient number of cells using ingredients such as foetal bovine serum (FBS).
To overcome these problems, we focused on costal cartilage. This has been used as a satisfactory graft for surgery in children, including in auricular reconstruction 14 and laryngotracheoplasty. 15 No major side effects have been reported over long observation periods. In this study, costal cartilage was transplanted for treatment of a partial growth plate injury in a rabbit model to evaluate its healing ability.
Materials and Methods
Experimental design
This experiment was approved by our institution's animal ethics committee. The 60 male six-week-old New Zealand White rabbits were used for the experiment, as the growth plate of these rabbits would be open for 16 to 24 weeks. The rabbits underwent operation for partial growth plate injury of the left tibia under general anaesthesia and were killed at four or eight weeks post-operatively. Rabbits were assigned to four groups: non-operated right tibia (non-operated control, n = 10); no transplantation for operated left tibia (operated control group, n = 10); bone wax transplantation for operated left tibia (bone wax group, n = 10); and costal cartilage transplantation for operated left tibia (costal cartilage group, n = 10). The costal cartilage grafts were not taken from the experimental rabbits, but were taken as allografts from rabbits in another study. Collected non-operated control group right tibia samples were analysed using radiography. The left tibia samples from the three operated groups were analysed using radiography, microcomputed tomography (μ-CT), histology and immunohistochemistry at four and eight weeks post-operatively.
Surgical procedure
The operation was performed as previously reported. 13 Briefly, a skin incision was made at the medial proximal left tibia. The medial half of the growth plate of the tibia was drilled from the inside using a high-speed steel bur and injured in the range of 3mm diameter and 5mm depth. The excavated space created by drilling was filled with either bone wax or costal cartilage graft, which was tailored to the shape of the space (Fig. 1). Nothing was transplanted in the operated control group. After the operation, the rabbits received 1.5 mg/kg of carprofen intravenously for post-operative analgesia, but did not have any load or mobility restrictions.
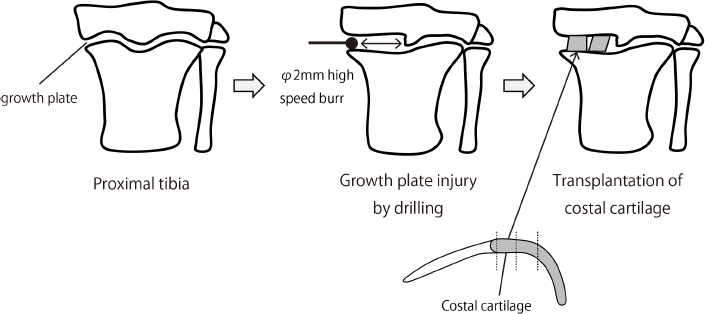
Schematic showing the surgical procedure. The medial half of the growth plate of the left tibia was drilled from the inside using a high-speed steel bur, and injured in the range of 3mm diameter and 5mm depth. Tailored costal cartilage was transplanted to the excavated space.
Radiographic analysis
Collected non-operated right and operated left tibias were radiographed using an MX-20 Cabinet X-ray System under specific conditions (35 kV, 300 μA and 240 s). For the radiographic evaluation, the medial proximal tibial angle (MPTA), defined as the angle between the mechanical axis of the tibia and proximal tibial articular surface, was measured at four and eight weeks post-operatively. The proximal tibial articular surface was defined as the line from the medial corner of the articular surface to the lateral corner of the articular surface. The mechanical axis was defined as the line from the centre of the articular of the knee to the centre of the articular of the foot.
μ-CT analysis
Representative left tibia samples were scanned by μ-CT (R_mCT; Rigaku Mechatronics, Tokyo, Japan) for animals at 90 kV and 200 μA. Image data were reconstructed and visualised using image analysis software (TRI/3D-BON: RATOC System Engineering, Tokyo, Japan).
Histological evaluation
All left tibia samples were fixed with 10% formalin and demineralised with ethylenediaminetetraacetic acid. Paraffin-embedded sections (3-μm thick) were stained with haematoxylin and eosin (H&E) and safranin O. The bony bridge formation rate, defined as the ratio of the total width of the bony bridge formation to the width of the growth plate injury, was measured quantitatively on H&E sections, with samples cut at the mid-coronal plane of the tibia.
Immunohistochemical evaluation
Serial 3-μm sections from the costal cartilage group were deparaffinised and pre-treated with proteinase K (DAKO, California) for five minutes. Next, all sections were incubated in 3% hydrogen peroxidase in PBS for five min at room temperature to block endogenous peroxidase activity. After blocking in normal horse serum, the sections were stained with anti-human type II collagen monoclonal antibody (COL2; 1:100 dilutions, Kyowa Pharma Chemical Co., Toyama, Japan). Detection was performed using a VECTASTAIN ABC KIT (Vector Laboratories, California). A 3,3’-diaminobenzidine (DAB) HRP substrate (DAKO, Glostrup, Denmark) was used to change bound antibodies to a visible brown colour. Finally, the sections were counterstained with H&E.
Statistical analysis
JMP 11 (SAS Institute Inc., Cary, North Carolina) was used for data analysis. Comparisons of MPTA and bony bridge formation rates among the three operated groups were performed using Tukey's honest significant differences method. A p-value of < 0.05 was considered statistically significant.
Results
Radiographic assessments
The mean MPTA in the non-operated right tibia control group was 88.2° (86° to 90°) at four weeks, and 87.3° (83° to 90°) at eight weeks post-operatively. In the operated control group, the mean MPTA was 69.1° (62° to 78°) at four weeks and 48.8° (37° to 65°) at eight weeks post-operatively. There were significantly more varus deformities in the operated control, bone wax and costal cartilage groups than in the non-operated control group at four and eight weeks post-operatively. In the operated control group, the proximal tibia had severe varus deformities at four weeks post-operatively, which had proceeded further at eight weeks after the operation.
In the bone wax group, the mean MPTA was 77.7° (70° to 88°) at four weeks and 63.8° (56° to 71°) at eight weeks post-operatively. The radiographic results showed that the bone wax group had significantly fewer varus deformities than the operated control group at both four and eight weeks post-operatively (Figs 2a and 3b).
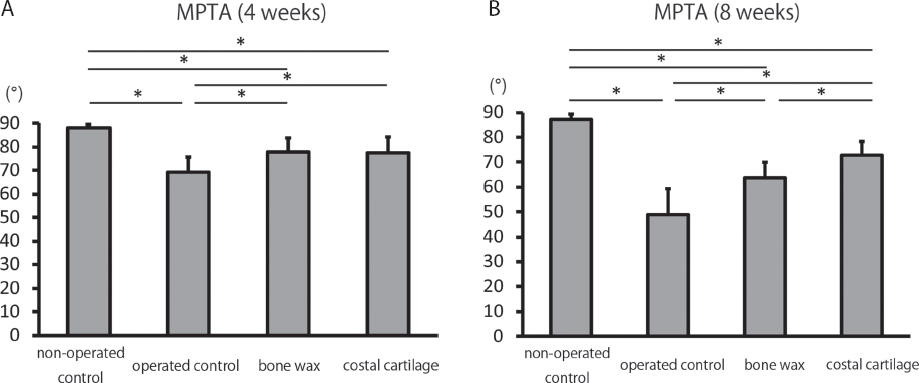
Bar graphs showing the mean medial proximal tibial angle (MPTA) on radiographs – (
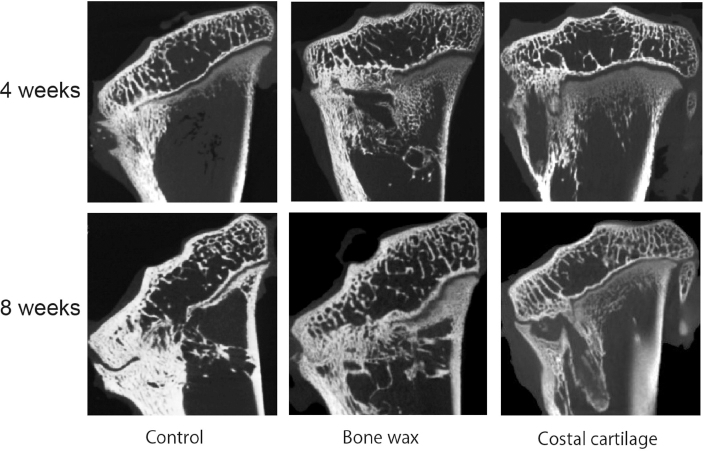
Representative microcomputed tomography (μ-CT) images of the mid-coronal proximal left tibia at four and eight weeks after operation for each group.
In the costal cartilage group, the mean MPTA was 77.5° (69° to 85°) at four weeks and 72.9° (67° to 82°) at eight weeks post-operatively. The costal cartilage group showed significantly fewer varus deformities than the operated control group at both four and eight weeks. There was no difference in MPTA between the costal cartilage and bone wax groups (p = 0.9974) at four weeks post-operatively. However, the costal cartilage group showed significantly fewer varus deformities than the bone wax group at eight weeks after operation (p = 0.0348).
μ-CT assessments
In the operated control group, the bony bridge was developed at the site of growth plate injury and the proximal tibia had severe varus deformities at four and eight weeks post-operatively (Fig. 3). In the bone wax and costal cartilage groups, bony bridge formation was partially prevented at the injured growth plate. Part of the transplanted costal cartilage was placed at the same level of the normal growth plate. In contrast, the bone wax was placed at the distal level of the normal growth plate.
Histological findings
Histological findings in the operated control group showed that the bony bridge was formed between the epiphysis and metaphysis at four and eight weeks post-operatively with H&E staining (Fig. 4). The mean bony bridge formation rate was 78.5% (50% to 100%) at four weeks, and 86% (60% to 100%) at eight weeks, post-operatively. Safranin O staining showed the growth plate was discontinuous at the operated site.
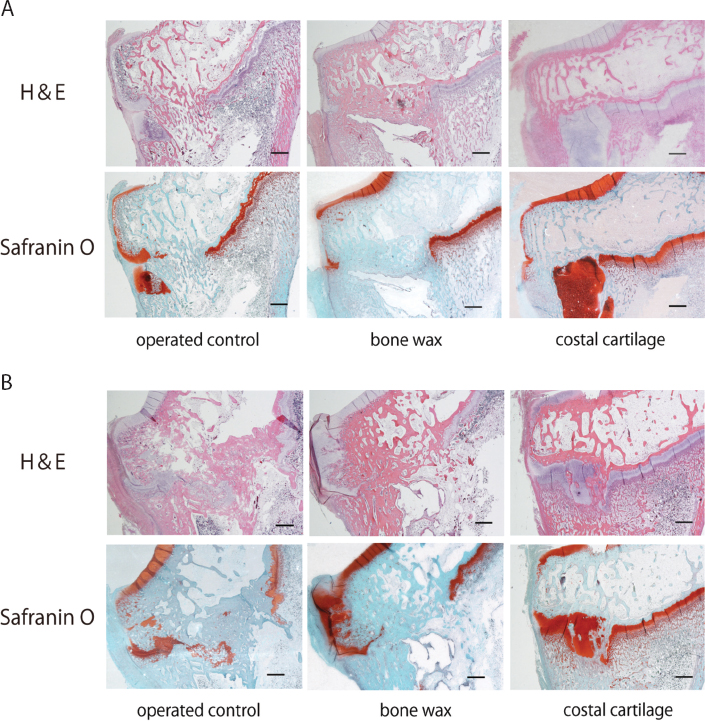
Representative histological appearance of the mid-coronal proximal left tibia. H&E staining (magnification ×2, scale bar = 1 mm) and safranin O staining (magnification ×2, scale bar = 1 mm) – (
In the bone wax group, the transplanted bone wax remained and the bony bridge was partially formed. The mean bony bridge formation rate was 26% (0% to 40%) at four weeks and 31% (10% to 60%) at eight weeks post-operatively. The growth plate was discontinuous, similar to that in the operated control group.
In the costal cartilage group, the mean bony bridge formation rate was 17.5% (0% to 40%) at four weeks and 23% (0% to 60%) at eight weeks post-operatively. The growth plate was thickened, continuous and stained with safranin O in some specimens at eight weeks after operation.
The mean bone bridge formation rates in the bone wax and costal cartilage groups were significantly less than in the operated control group at both four and eight weeks post-operatively (Fig. 5). No specimens showed inflammatory or immune reactions.
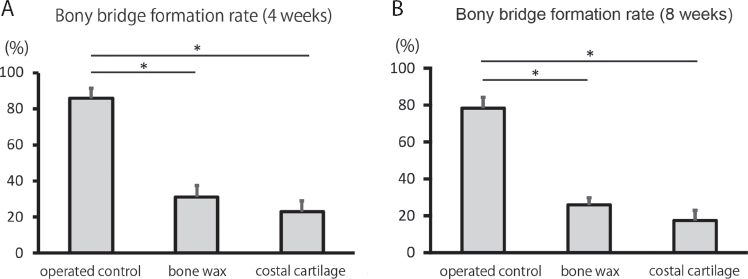
Mean bony bridge formation rate on histological sections of the mid-coronal proximal left tibia from each group – (
Immunohistochemical findings
In the costal cartilage group, transplanted costal cartilage and growth plate cartilage were deeply immunohistochemically stained with COL2, a chondrogenic extracellular matrix gene at eight weeks after operation, suggesting that the transplanted costal cartilage had the property of cartilage. (Fig. 6). The transplanted costal cartilage was continuous with the normal growth plate on one side.

Representative immunohistochemical staining of type II collagen (COL2) in the growth plate of the mid-coronal proximal left tibia at eight weeks after operation (magnification ×2, scale bar = 1 mm).
Discussion
Growth plate injuries are common in childhood (15% to 30% incidence in all paediatric fractures).16–18 Growth arrest and angular deformities occur in 1% to 10% of these injuries.16,19 For surgery in partial growth arrest, Langenskiöld first reported surgical resection of the bony bridge and transplantation of autogenous fat. 20 That report showed that transplanted fat prevented reformation of the bony bridge and angular deformities were improved. After Langenskiöld's report, surgical treatments for humans using various grafts to prevent the bony bridge reforming were reported, such as polymeric silicone, 1 bone wax,2,5 bone cement 4 and artificial dura. 3 However, there are no available reports on the regeneration of injured growth plate cartilage in humans.
Cell transplantation therapy has been performed in many animal experiments to repair injured growth plate cartilage. Previously, cultured chondrocytes or MSCs were transplanted to the growth plate defects in a rabbit model and growth plate cartilage was repaired.7–9,12 However, there are some problems with cell transplantation therapy. First, in large animal models (e.g. miniature pigs and sheep), some reports indicated that the growth plate cartilage was not repaired completely.6,10,11 In particular, new fibrous tissue was formed at the site of transplantation in the sheep model. 10 Second, cell transplantation therapy requires previous cell collection, time and money to acquire sufficient numbers of cells for transplantation. Finally, ingredients such as FBS are needed for the cell culture medium. 21 For these reasons, the clinical application of cell transplantation therapy is not easy in children.
To overcome these problems, we focused on costal cartilage as a new graft for the treatment of growth plate injury. It is not necessary to collect tissue in advance and culture cells for costal cartilage transplantation. Costal cartilage or costal osteochondral grafts are often used in paediatric surgery, including auricular reconstruction surgery, 14 laryngotracheoplasty 15 and osteochondritis dissecans. 22 No long-term side effects have been reported in previous studies. Moreover, our results showed that costal cartilage has a rich cartilaginous extracellular matrix. Therefore, costal cartilage could be a long-term, safe and adequate graft for the treatment of growth plate injuries.
Our radiographic results showed that the MPTA was higher in the costal cartilage and bone wax groups than in the operated control group at both four and eight weeks post-operatively. There was no difference in MPTA at four weeks after operation between the bone wax and costal cartilage groups. However, the MPTA was higher in the costal cartilage group than in bone wax group at eight weeks post-operatively. Histological findings showed the bony bridge formation rate was significantly decreased in the bone wax and costal cartilage groups compared with the operated control group. The bony bridge formation rate in the costal cartilage group tended to decrease compared with the bone wax group. However, this difference was not significant. The transplanted costal cartilage was deeply stained with safranin O and COL2. These findings suggest that the transplanted costal cartilage retained the property of cartilage. A previous report showed that when a costal osteochondral graft was transplanted to injured knee articular cartilage in a rabbit model, the transplanted cartilage was viable and stained with safranin O up to 48 weeks. 23 We therefore assumed that the chondrocytes had potential to be alive and active. Moreover, in the operated control and bone wax groups, the growth plate was discontinuous, whereas the growth plate in the costal cartilage group was continuous. In summary, there were two differences between the transplantation of bone wax and costal cartilage. First, there were fewer varus deformities in the costal cartilage group than in the bone wax group. Second, the cartilage was continuous in the costal cartilage group, whereas it was discontinuous in the bone wax group. Varus deformities were improved at eight weeks post-operatively, despite no difference in the bony bridge rate between the bone wax and costal cartilage groups. The costal cartilage was at the same level as the normal growth plate for eight weeks after operation. In contrast, the bone wax moved to the distal side as the tibia grew longitudinally. This might have been because costal cartilage is biological material whereas bone wax is non-biological material. Therefore, the costal cartilage may be more suitable than bone wax as an interpositional material, supporting our assumption that the growth plate cartilage was partially repaired by costal cartilage transplantation. The thickened growth plate which we found in the costal cartilage group at eight weeks post-operatively was consistent with past findings of repaired cartilage. 13 However, a long-term functional analysis using larger animal models is required to prove the repair of cartilage functionally.
Bone wax is a non-absorbable and non-biological material suitable for interposition at the injured site to prevent bony bridge formation. Costal cartilage is also a good interpositional material, particularly as it remains at the transplanted site for longer than MSCs. In our previous study, transplanted synovial MSCs were not seen at eight weeks post-operatively. However, in this study, the transplanted costal cartilage was seen at eight weeks post-operatively and maintained its cartilaginous properties as shown by deep safranin O staining and immunohistochemical COL2 staining. Chung et al reported that undesirable bony bridge formation increased with the reduction of cartilage-related gene expression such as Sox9 and COL2. 24 Costal cartilage may help prevent formation of a new bony bridge by maintaining the property of cartilage. Previously, Yoshida et al showed that varus deformities in a synovial MSCs group were not significantly lower than in a bone wax group at eight weeks post-operatively, despite the repair of growth plate cartilage. 13 In our study, there were significantly fewer varus deformities in the costal cartilage group than in the bone wax group at eight weeks post-operatively. This result might have been obtained because costal cartilage is a superior interpositional material to synovial MSCs.
There are two limitations of this study. First, we did not use autograft. The six-week-old New Zealand White rabbits were small and it was difficult to exfoliate pleura from the costal cartilage. We were concerned about the occurrence of pneumothorax and undesired death by surgical procedure. Therefore, we selected allogenic costal cartilage as graft material to reduce the number of rabbits used in the experiment. Second, evaluating the function of repaired growth plate cartilage requires long-term functional analysis. Therefore, a study using a larger animal model is required before clinical application in humans.
In conclusion, our study showed a decrease in angular deformities and bony bridge formation and suggests the growth plate has potential to be partially repaired by the transplantation of costal cartilage graft. The costal cartilage graft is cartilage extracellular-matrix-rich and safe, and is a good interpositional biological material. Costal cartilage may therefore be a suitable graft for the treatment of growth plate injury. Further study is required using autograft and evaluating the long-term function of repaired growth plate in larger animal models.
Footnotes
This study was funded by JSPS KAKENHI (Grant Number JP26861188). No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
All applicable international, national, and institutional guidelines for the care and use of animals were followed.
K.Yoshida received research grants from JSPS KAKENHI Grant Number JP26861188.
