Abstract
In addressing the challenge of the low survival rates of rats with myocardial ischaemia, we developed a novel respiratory mask. We tested this mask on the rat model. We gave attention to several features of the mask: (1) shape, (2) size, (3) inlet, (4) outlet, (5) compatibility between rat head and the mask, (6) connection between mask and ventilator. We found certain features, especially to influence mask efficacy. These features include: mask shape, mask inlet and outlet, mask connection to the respiratory machine, mask mount on the rat head. We examined the rat mask in a model of chronic myocardial ischaemia; our model was the ligation of the coronary artery. The rats with the masks experienced an increase in survival by a factor of 50–90% compared with rats deprived of the masks. Towards the examination of myocardial ischaemia, our new mask may offer a platform replete with both efficiency and stability.
Coronary ischaemia, myocardial infarction and heart failure contribute to the high rates of cardiovascular morbidity and mortality. A plethora of studies have been conducted in order to elucidate the cellular mechanisms of myocardial ischaemia. Also pharmacotherapies that target acute myocardial ischaemia have been explored and designed. Nevertheless, the low survival rate in animal models has hindered the research on chronic myocardial ischaemia. 1,2 Until recently, the prominent myocardial ischaemic model has been the model from the ligation of coronary artery. This model is supported by the use of a respiratory machine and an endotracheal tube connected to a ventilator on one end and placed through the incised trachea on the other end, which serves as the conventional method for airway maintenance. With this technique, at the most, ≤50% of the rats are able to survive several weeks into surgery.
Data from anatomic and morphological studies that support this work have identified especially three lethal factors. The first factor is the respiratory tract obstruction that leads to respiratory failure. This is responsible for ∼90% of mortalities. The second factor is the lethal arrhythmia that sometimes follows myocardial ischaemia. This is responsible for ∼7% of mortalities. The third factor is progressive myocardial haemorrhage or pneumothorax. This is responsible for ∼3% of mortalities.
The data lead us to identify upper respiratory tract obstruction as the predominant factor for mortality in the context of the experimental model. Alleviating upper respiratory tract obstruction offers a great challenge. In this work we present an original respiratory mask for rats. In the current model of experimental chronic myocardial ischaemia, this mask provides an overt pro-survival efficacy of 50% to >90% in 10 days.
Materials and methods
Animals
Adult Sprague Dawley albino rats (n = 400) weighing 200–220 g were provided by the Laboratory Animal Center of the Fourth Military Medical University (FMMU). All animal procedures were reviewed and approved by the Institutional Ethics Committee of the FMMU.
Rat myocardial ischaemia model
A group of rats were anaesthetized with an intraperitoneal injection of 3% sodium pentobarbital (60 mg/kg). Then they were set in a supine position on a rodent operating table. Under sterile conditions, the skin was incised along the left edge of the sternum and the chest wall muscle was dissected in layers in order to expose the left fourth rib. Suture silk was placed around this rib. Two knots were tied. The rib was divided between the two knots. This division both opens the chest cavity and also exposes the heart. The pericardium was opened. The left anterior descending coronary artery was identified under microscopy. The left anterior descending coronary artery was then ligated ∼2–3 mm from its origin. Both rib stumps were then closed. A chest tube was placed. Then the chest wall was closed. The air in the chest was evacuated. The chest tube was pulled, and then the suture string was tied. The rats were connected to the ventilator. The skin incision was closed.
Rat groups
One hundred rats were divided into two groups. One group was named the ‘intubation group.’ The other group was named the ‘mask group.’ A neck midline skin incision was made in each rat belonging to the ‘intubation group.’ Each incised rat was intubated through an open tracheal incision. Following intubation, each rat in the ‘intubation group’ was connected to a ventilator. Then each rat in the ‘intubation group’ underwent thoracotomy. In the mask group, rats were ventilated through the respiratory mask connected to a ventilator prior to a similar thoracotomy procedure. Each rat in the ‘mask group’ was fitted with the new respiratory mask. Once the masks were positioned, each rat in the ‘mask group’ was connected via the mask to a ventilator. Then each rat in this group underwent thoracotomy identical to the rats in the ‘intubation group’.
Indices
These parameters were used in order to evaluate ischaemic outcome. The ischaemic outcomes of both the ‘intubation group’ and the ‘mask group’ were evaluated.
Electrocardiography; Triphenyltetrazolium chloride (TTC) degree of myocardial ischaemia (evaluated by double staining with Evans blue and TTC); Survival frequencies at a predefined point (1, 3, 6, 12, 24 and 48 h after surgery, and 10 days after surgery); Postmortem determination of cause of death; Appearance of ischaemic heart and transmittance of the left ventricle 10 days after surgery.
Data analysis
All results were expressed as mean ± SEM. SPSS version 10.0 statistical software (IBM Corporation, Armonk, NY, USA) was used for the data analysis. Statistical significance was considered with P < 0.05.
Results
Development of the respiratory mask
According to a literature review, up to this point, no previous report for the use of a rat respiratory mask has been published. Also, no similar device is available. Key components define the mask, namely: shape and size of the mask, inlet and outlet of the mask, the compatibility between the rat head and the mask, and the connection between the mask and the ventilator.
Design of the respiratory mask
In our design of this mask, the first issue we confronted was whether to design the mask to cover both the maxilla and the submaxilla. A mask that covers both the maxilla and submaxilla would have a much larger inner volume; this mask would have a much larger size. We noted the inability of the rat to breathe through the mouth–tracheal airway while under anaesthesia. We addressed this matter and designed the mask in order to cover only the maxilla and apex nasi. Our mask exposes the submaxilla. This design offers a substantially reduced volume. Also, this design reduces manufacturing expenses.
The second issue that we addressed was the challenge presented by the rat teeth. Specifically, the upper incisors of the rat are particularly long and are prone to damage. Damaged and/or missing incisors dramatically affect the quality of life of the rat. Damaged and/or missing incisors contribute to both starvation and mortality because of inadequate food intake with broken/missing upper incisors. In response to this challenge, we designed a nick (incisor-protection nick) near the incisors (Figure 1a).
Schematic diagram of the respiratory mask. (a) Illustration of the respiratory mask: 1: Maxilla immobilizer, 2: Main body of the mask, 3: Teeth-protecting nick, 4: Mask-immobilizing tapes, 5: Inlet, 6: Outlet, 7: Resistance adjustor. (b) Immobilization of the mask. The head of the rat is first immobilized by the mandibular immobilizer before the mask is placed. (c) Schematic diagram of the respiratory mask connecting to a ventilator in the early phase of inspiration. Intra-mask pressure is greater than the outlet pressure, whereas it is lower than the intrapulmonary pressure. As a result, the remnant air of the last expiration is pushed out of the mask, leaving no dead space behind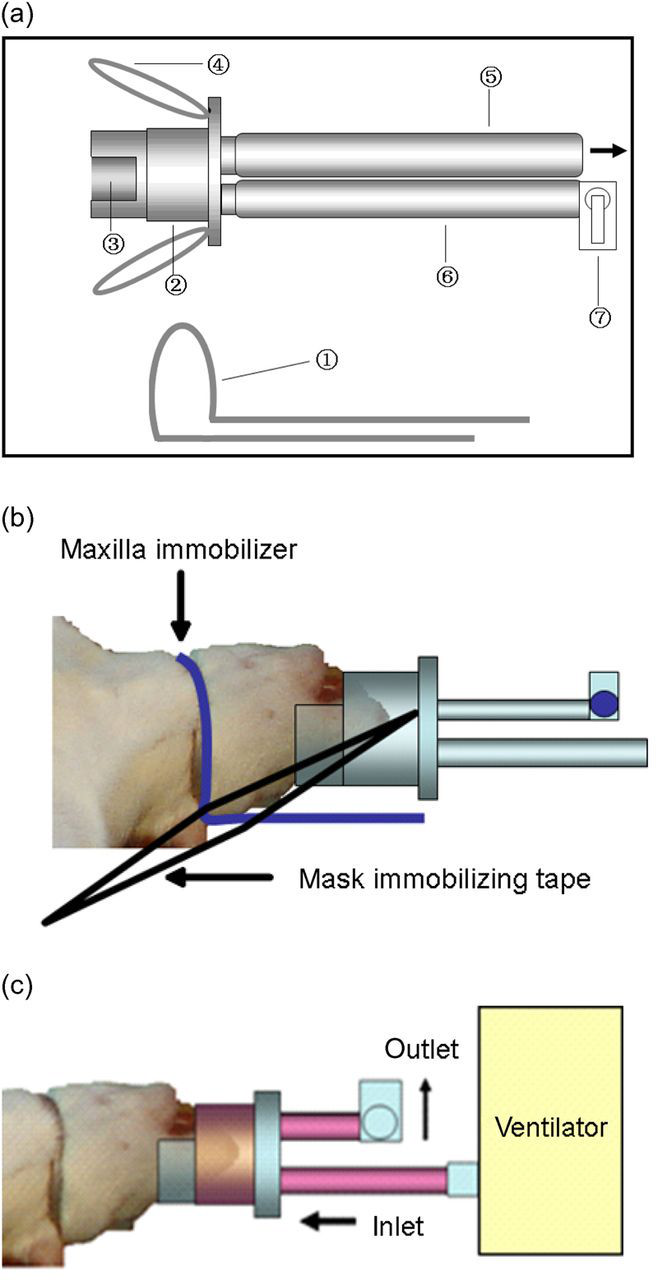
Third, we noted how rat maxilla lips are fleshy and flexible. In response to this challenge, we designed a tight fit to the respiratory mask. The tightness maintains obturation (Figure 1b).
Inlet and outlet of the respiratory mask
The respiratory mask for humans has only one inlet with outgoing air being released through the valves on the mask. Therefore, the volume of the mask is relatively large for humans.
However, the volume of the respiratory mask for rats is relatively small, thus making it unrealistic for valves. Our system offers an individually designed inlet and outlet feature for airflow maintenance (Figure 1a).
Immobilization of the rat head with the fitted mask
What is the optimal positioning methodology for our new rat mask? We first fixed two elastic straps on both sides of the mask. First, we tested mask fit with two elastic straps, one on each side of the mask. We positioned the mask. Then, the elastic straps were tied to both sides of the operating table. The tie immobilized both the mask and the rat head. However, this immobilization technique failed to prevent rats from moving their heads, resulting in dismounting of the mask with movement of the rat's head.
In order to immobilize the mask, the head should be properly fixed. Due to the fact that the rat's jaw horn displayed relative protrusion, a mandibular fixator was designed. With immobilization of the head by this mandibular fixator, the mask was not easily dislodged with movement of the rat's head (Figure 1b).
Connection of the mask to a respiratory machine
Another major challenge was to reduce dead space in the mask. Increased pulmonary resistance following thoracotomy and the slender nasopharyngeal duct necessitate a high ventilation pressure to overcome the respiratory resistance and inflate the lung.
Our response to these challenges began with the placement of a resistance adjustor at the tip of the outlet. This resistance adjustor allowed the modification of the outflow resistance. This adjustor allowed us to make the outflow resistance slightly lower than the pulmonary resistance.
Because ventilatory pressure is between the outlet tube pressure and due to pulmonary resistance during the early phase of inspiration, residual air from the last expiration was ventilated out of the mask, resulting in increased ventilatory pressure.
Pulmonary resistance during the early phase of inspiration helps to contribute to ventilation pressure. Also ventilation pressure is localized between the outlet tube pressure. In addition, residual air from each expiration is ventilated out of the mask. As a result, the ventilation pressure is increased.
The resistance adjustor allowed both for the resistance to be overcome and also for the inflation of the lungs. Also, fresh air or fixed oxygen instead of residual air from the last expiration was ventilated into the lungs. As a result, respiratory dead space was minimal. The inlet tube was connected to the ventilation machine. The resistance adjustor was connected to the outlet in order to modulate the outlet resistance (Figure 1c). Excessive air in each ventilation escaped from the mask through the outlet tube to minimize overventilation-induced alveolar damage.
The rat model of chronic myocardial ischaemia and respiratory mask application
The application potential of the mask was assessed. The respiratory mask was examined in the rat chronic myocardial ischaemic model. Data from the current study revealed that the survival of the ‘mask group’ reached >90%. These data demonstrate the high value of the mask in the experimental rat model of chronic myocardial ischaemia.
Electrocardiogram fluctuations before and after myocardial ischaemia
Ligation of the left anterior descending coronary artery resulted in a significant rise in the ST-T wave (P < 0.01) (Table 1). Eighty percent of the rats displayed various patterns of arrhythmia including: premature ventricular contraction, paroxysmal ventricular tachycardia, reversible ventricular fibrillation and irreversible ventricular fibrillation (Figure 2).
Ischaemic arrhythmia. (a) Before ischaemia. (b) Prominent elevated ST-segment caused by ischaemia. (c) Sporadic ischaemic premature ventricular systole. (d) Frequent premature ventricular systole. (e) Episodic premature ventricular systole. (f) Ventricular fibrillation Comparison of ST-T wave changes before and after ligation of coronary artery *P < 0.01 versus before ischaemia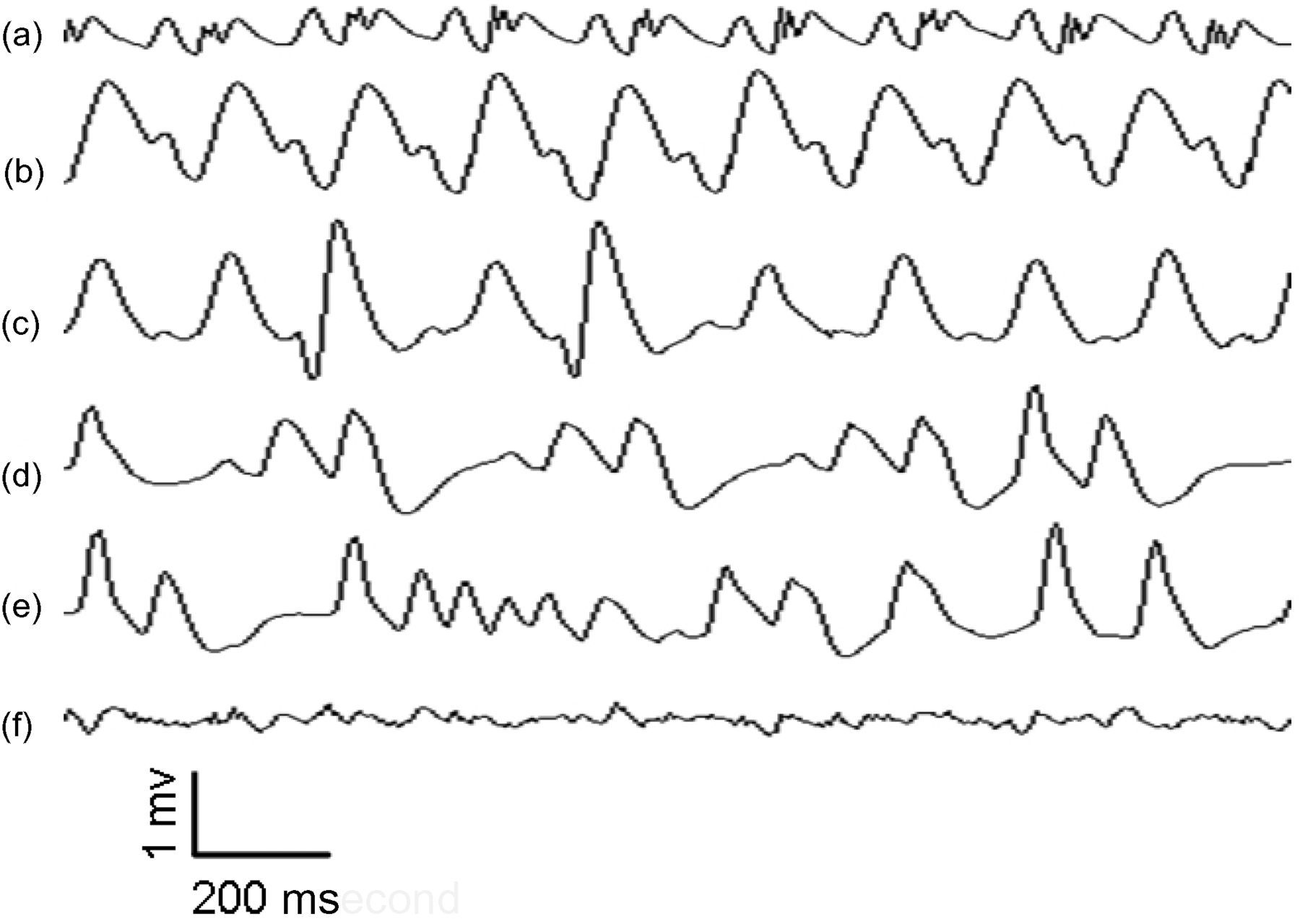
Degree of myocardial ischaemia 2 h after ligation of coronary artery
Double staining with Evans blue and TTC was used to identify the area of myocardial ischaemia. The area coloured blue indicated normal myocardial tissue. The area coloured red indicated tissue that experienced myocardial ischaemia. The white area was considered to be the infarcted region (Figure 3A, see online version for colour references).
Morphological alteration of chronic myocardial ischaemia and comparison of survival rate between the tracheal intubation and respiratory mask groups (A) Staining of ischaemic myocardium for 2 h and a, b, c, d are arranged from the tip to the bottom of the heart. The white part symbolizes infarcted myocardium. Red and dark parts represent ischaemic and normal myocardium, respectively. Upper panel: mask group. Lower panel: control group. (Ba) Appearance of rat heart 30 days following coronary artery ligation in mask group. (Bb) Extent of the transparency of ventricular wall. (C) Comparison of survival rate between the tracheal intubation and respiratory mask groups (see online version for colour references)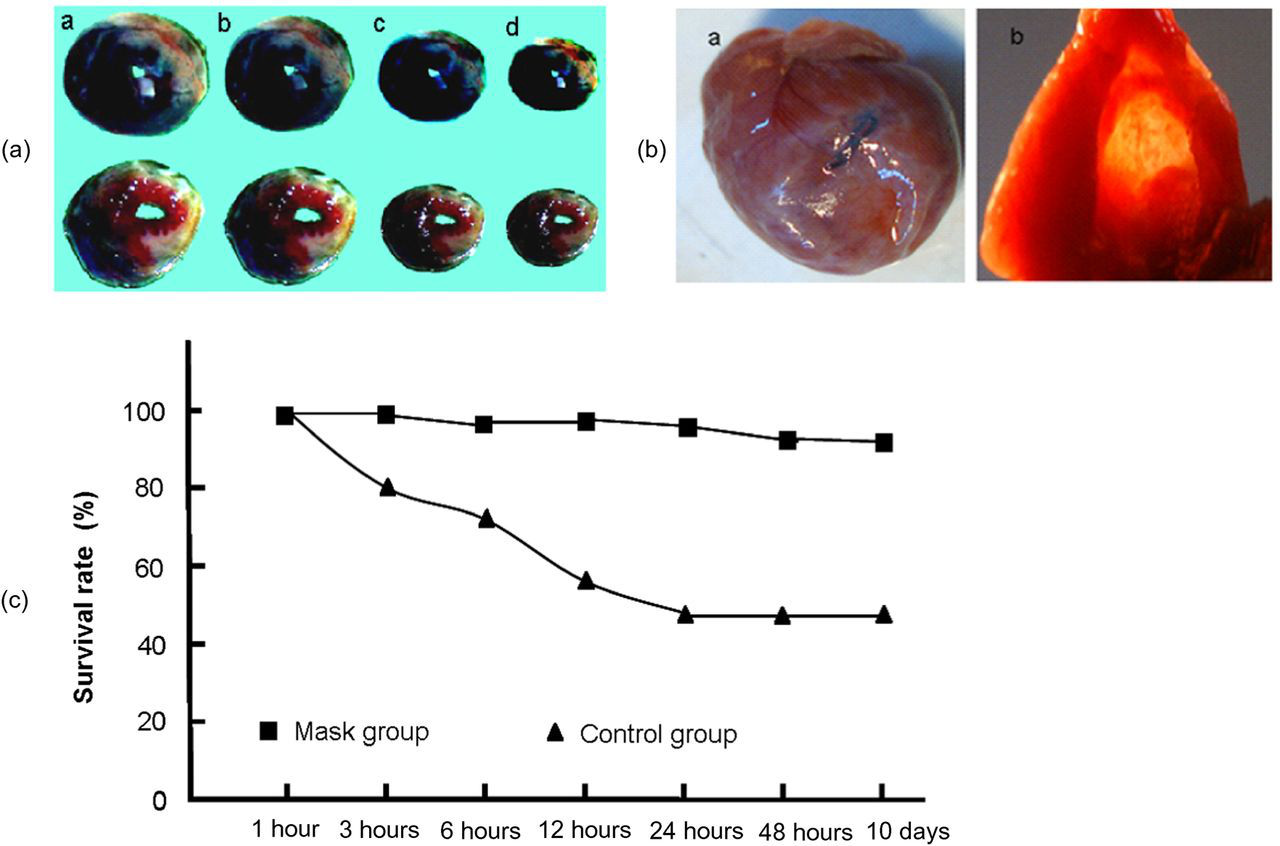
The novel respiratory mask increased the survival rate in the chronic myocardial ischaemic model
Comparison of the survival rate between the two methods (%)
*P < 0.01 versus the control group
Postmortem analysis of cause of death
Analysis of the causes of death (%)
URT: upper respiratory tract
*P < 0.01 versus the control group
Heart appearance and left ventricle wall transmittance 30 days after ligation
Ten days following their operations, the surviving rats in each group received a normal diet. The wounds healed well and without notable infections or scars. These rats were physically inactive but otherwise appeared normal. Thirty days after the surgeries, the (surviving) rats were anaesthetized, and these rats underwent thoracotomy. Opposite motion of the ischaemia region was observed during systole and diastole. In addition, the heart wall was thin and folded (ventricular aneurysm) in regions below the ligation. A cut was then made between the anterior wall of the left ventricle and interventricular septum. The left ventricle transmittance was subsequently observed with a 30 W cold-light illuminator. Figure 3B indicates both the thinness of the central portion of the ischaemic region and also the relative increase in thickness of the peripheral regions.
Discussion
Due to intubation methods, the development of the traditional chronic myocardial ischaemic model witnessed the constant incidence of airflow occlusion in the upper respiratory tract. During experimentation with the chronic myocardial ischaemic model, secreta would deposit in the slender nasopharyngeal duct. The secreta would dry and harden, and this dry secreta would further occlude the nasopharyngeal duct.
The extra attention given to the traditional experimental model for myocardial ischaemia led to an observation: anaesthetized rats do not breathe through the mouth. This observation may explain many cases of respiratory failure and anhelation. These facts help us to arrive at the statement that the prevention of airway narrowing and airway occlusion from dried secreta stems from the preservation of normal airflow through the nasopharyngeal duct.
Based on this rationale, we developed a respiratory mask for the first time to ensure normal ventilation during surgery. As we designed our respiratory mask, we focused on certain critical areas. These areas included: appearance, inlet and outlet of air, connection to the ventilator, immobilization of rat head to mask. The newly developed mask was then applied in a rat model of chronic myocardial ischaemia. Our results suggest that the fitting of the mask on to the rat contributes to a significant increase in survival rate. The mask has proven its value in the context of the study of chronic myocardial ischaemia.
What we observed in this experimentation with the current model for chronic myocardial ischaemia supports our observations from the previous study. 3 Specifically, we noted various forms of cardiac arrhythmia within 2 h of the coronary artery ligation procedure. Also, we noted that a massive infarction could be observed within 2 h of the coronary artery ligation procedure.
Our results demonstrate that necrotic myocardial cells are absorbed and replaced by fibrous tissue. We noted that this occurs at least 10 days after surgery. As a result, the ventricular wall became thinner, whereas opposite motion of the ischaemic zone was observed during systole and diastole. Furthermore, the ventricular wall was expanded during systole and collapsed during diastole. All of these observed phenomena demonstrated that the mask was tested on the optimum model of chronic myocardial ischaemia for the rat. As shown in our present study, the respiratory mask reduced the mortality rate from 50% or higher to less than 10%. Further study revealed up to 90% of mortalities in the ‘intubation group’. The ‘mask group’ had no mortalities. This lack of mortalities indicates that the respiratory mask is capable of rectifying the narrowing and occlusion of the respiratory tract that results from secreta deposition. Because of the mask, normal ventilation was maintained both during and after thoracotomy. The mask supports normal ventilation, a critical factor for survival in the chronic myocardial ischaemic experimentation model.
Our new respiratory mask confers specific advantages. One advantage is the simplification of the operational procedure. Another advantage is the reduction of surgical risk. Still another advantage is the reduction of risk from tracheal incision. Lastly, the most salient advantage is the increase in survival rate.
An advantage that the mask confers is the simplification of the operational procedure. With our mask, the processes of tracheal incision, tracheal intubation, clearance of secreta and wound repair are simplified significantly. As a result, the time of surgery with the mask decreases from more than one hour to about 15 min.
Another advantage that the mask confers is the reduction of surgical risk. A plethora of blood vessels surround the rat neck. Surgical trauma to any one of these vessels may easily lead to any range of disorders, including subcutaneous emphysema and haematoma. Trauma to any one of these vessels may contribute to the depression of animal respiration. Depression of animal respiration may distort experimental results. The use of our new mask may circumvent all of this, at least in part.
Our mask also confers a reduction of risk of infection. In the myocardial ischaemic model of experimentation, tracheal incision and intubation may easily result in infection. The use of our mask simplifies the process of the establishment of ventilation and avoids the risks of infection that accompany many intubation procedures.
Finally our mask confers an increased survival rate. The use of our mask significantly increases the survival rate from less than 50% to greater than 90%. Our mask prevents death from upper respiratory tract obstruction.
We have designed a completely original respiratory mask for rats. This mask supports comprehensive ventilation. Also, this mask prevents airway occlusion during thoracotomy. The application of our mask in the chronic model of myocardial ischaemia dramatically improved survival rate from less than 50% to greater than 90%. This improved survival rate indicates the potential for use of the mask in the experimental model of chronic myocardial ischaemia. Our respiratory mask offers many distinct advantages. Some of these advantages include stability, efficiency, convenience, reduced surgery time and low cost. This respiratory mask has resolved the inherent problem that for many years limited the experimental model of chronic myocardial ischaemia. This new mask provides a stable and effective platform for the investigation of ischaemic heart disease. The new respiratory mask offers new options for many surgical procedures that require thoracotomy.
Footnotes
ACKNOWLEDGEMENTS
This work was supported by grants (Nos 30900535, 30971060 and 31100827) from the National Natural Science Foundation of China (NSFC), grants from Shaanxi Province (Nos 2010K01–195 and 2011JM4001) and grants from the National New Medicine Research Foundation of China (Nos 2009ZX09103–671 and 2009ZX09301–009-BD11). We thank Professor Sharon Morey for English editing.
