Abstract
The need for using immunodeficient mice for xenoimplantation of tumours is increasing in translational research in radiation oncology. However, adverse effects of radiation and infectious diseases may ruin the experimental work, in particular when appropriate facilities are not available. In this report, we describe a procedure to deliver fractionated radiotherapy to xenoimplanted tumours in immunodeficient mice using a medical linear accelerator, a method that was devised as an alternative to the lack of facilities devoted to radiation research. The mice were irradiated under anaesthesia and aseptic conditions. Thirty Gray in 10 days using a 6 MV photon beam were delivered only to the right thigh of the mice where tumours were implanted. The mice were evaluated twice a week up to planned euthanasia. The follow-up of mice was completed without premature interruption due to toxicities or infectious diseases, an observation which demonstrates the feasibility of the method.
Keywords
Every year, millions of cancer patients around the world undergo radiotherapy, alone or in combination with drugs. The rise in radiotherapy use, together with an avenue of novel drugs that modulate radiotherapy action, has prompted an increased interest in radiation oncology research. An important part of this research is based on the irradiation of human tumours grown in mice. 1,2 However, such experimental models are associated with a number of difficulties that can ruin the experimental outcomes. First, it is difficult to shield radiosensitive healthy tissues from the radiation field and avoid its adverse effects in the absence of appropriate equipment to irradiate mice, such as an orthovoltage X-ray treatment machine. 3 Second, to prevent the tumour rejection mediated by a normal immune system, human tumour cells have to be implanted in immunodeficient mice which are prone to lethal infectious diseases. Third, there is a general lack of specific pathogen free (SPF) facilities to irradiate mice. Since these have to be transferred from the SPF colony to medical radiotherapy units, the risk of infectious outbreaks is greatly increased. Finally, to make matters even more complex, often experimental design requires multiple fractions of radiotherapy, which exposes mice to pathogens more frequently.
In this short report, we describe the experimental procedure and the refinements we applied to successfully deliver high doses of fractionated radiotherapy to xenoimplanted human tumours, protect radiosensitive tissues and decrease the microbiological risk when irradiating immunodeficient mice using a medical linear accelerator. Importantly, we devised this procedure as an alternative to the lack of experimental facilities devoted to radiation research in immunodeficient mice.
All experimental methods were approved in accordance with our own institutional IDIBELL guidelines for animal care and ethics. Six to eight-week-old female athymic mice (Athymic Nude-Foxn1nu, Harlan, Gannat, France) were used. Complete health reports, especially the microbiological status of the animals based on the Federation of European Laboratory Animal Science Associations (FELASA) recommendations, were certified by the vendor. Tumours were generated by injecting one million human cancer cells delivered in 100 μL into the subcutaneous tissue. The mice were intraperitoneally anaesthetized (1 mL/kg of each solution: 50 mg/mL ketamine and 1 mg/mL medetomidine with 5 mg/mL atipamezole for the reversal of the anaesthesia effects) in order to precisely control the area in which the cells were to be injected and to obtain homogenous growth of tumours. We decided to inject the cells in the right thigh of the mice, which allowed us to irradiate exclusively a limited part of the body. This decision served to protect the rest of the animal body from radiation.
At a suitable tumour size, and two days before irradiation, the mice were moved from the SPF area to a quarantine room so they can adapt to the new housing conditions, and where the animals were to be permanently housed in closed autoclaved plastic cages (5 mice per cage). In this room, the mice were manipulated under a laminar flow hood using aseptic conditions. Before each radiotherapy session, they were anaesthetized, as mentioned above, and transported to the radiotherapy unit in clean autoclaved closed cages covered with a drape to conceal the animals from plain view.
Upon arrival at the radiotherapy room, the treatment table was disinfected with alcohol and covered with sterile drapes. Surgical caps, masks and sterile gloves were worn. Figure 1 shows the scheme of how an appropriate setting for selectively irradiating tumours and reducing infections in the mice was achieved. Of note is the placement of the thigh on the edge of the radiation beam to allow sufficient coverage of the tumour, while protecting the body of the mice as much as possible.
Scheme used for localized irradiation of xenografted tumours in multiple fractions. Tumours (T) were placed into the gap created by two polymethyl methacrylate (PMMA) plates (b), each were 35 cm × 35 cm × 1 cm in size, separated by a smaller plate (30 cm × 30 cm × 1 cm). The edge of the beam was adjusted to cover the tumour with 1 cm margin, assuring that PMMA plates were included into the beam. The thighs were kept in place using an adhesive tape, while the body of animals was maintained out of the beam. Sterile drape that cover the treatment table (a) and wrapped PMMA plates are represented by the grey lines. Dotted line illustrates the sterile transparent film put on to the mice, protecting them from the non-sterile top PMMA plate, as it can also be observed in the pictures on the right. The source-plate distance was set to 100 cm on the centre of the top plate. Note that dimensions in the diagram are not to scale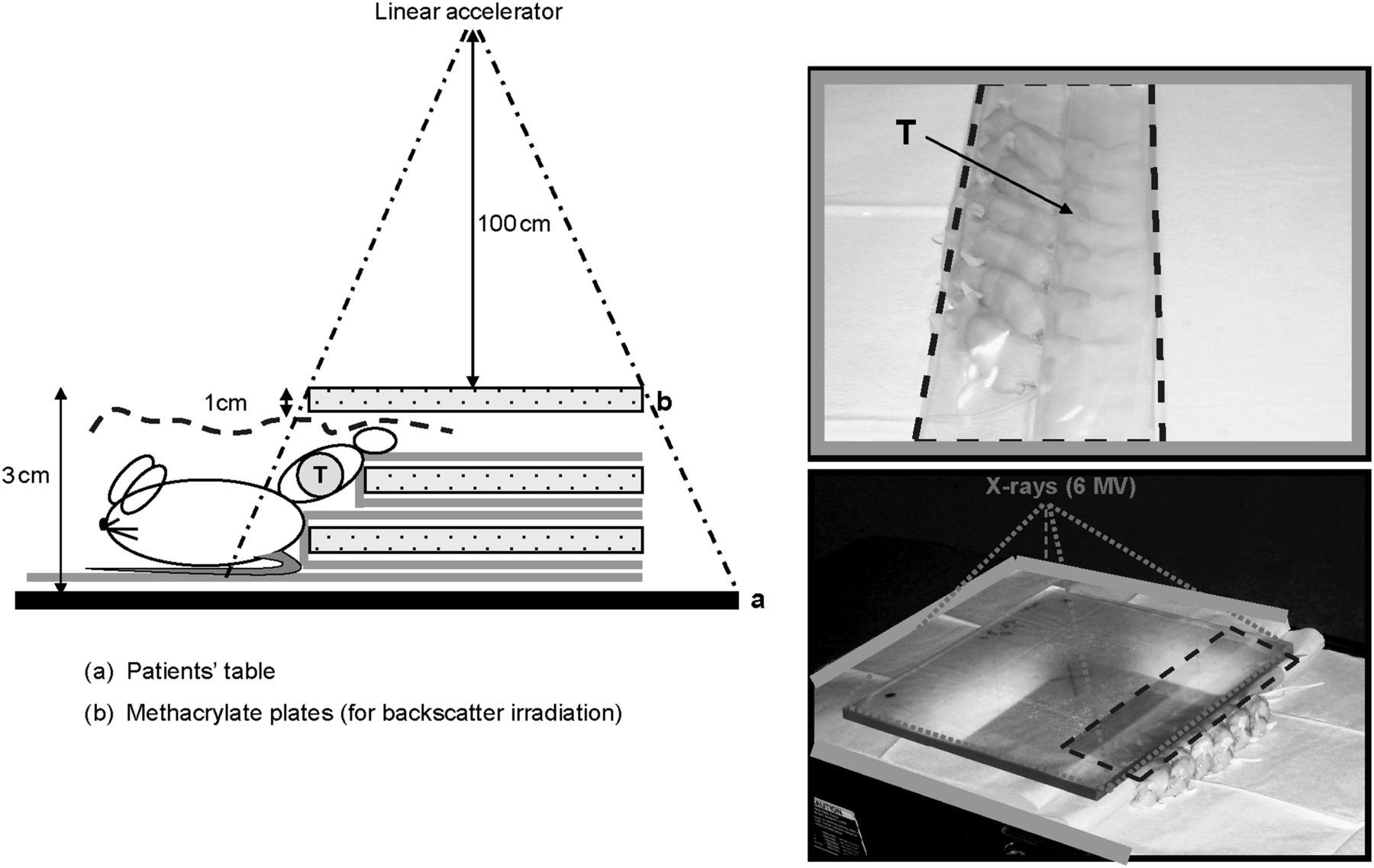
To irradiate the animals, as shown in Figure 1, a 6 MV photon beam from a Varian Clinac 2100 linear accelerator was used. A total dose of 30 Gray (Gy) in fractions of 3 Gy, separated by 24 h, excluding Saturdays and Sundays was administrated. The dose rate was 2.7 Gy per minute. In order to verify the dose that the tumours received, as well as the precision of the set-up used, an in vivo dosimetry was performed by means of radiochromic films (Gafchromic EBT, International Specialty Products, Wayne, NJ, USA). In total, 40 dose measurements were carried out during four separate days. Films were placed in contact with the tumour at the beam entrance. We found less than 3% variation in the doses received by different mice on the same sessions and less than 1.5% variation between different days. The mean deviation in absorbed dose in the tumours was 2.3% of the prescribed dose. The aforementioned results confirmed that our experimental set-up was homogeneous and reproducible.
Quality of life of the mice during and after localized irradiation in multiple fractions
Values are the proportion of an observation expressed in grades related to the total number of animals (n) within the period of follow-up
† Weight loss. Grades: 0 no weight loss; 1 less than 10%; 2 between 10 and 20%; 3 more than 20%
‡ Physical appearance. Grades: 0 normal; 1 changes in skin colour; 2 paleness and cyanosis; 3 hunching and loss of muscular mass
¶ Clinical signals. Grades: 0 no presence; 1 hypothermia; 2 bleeding or mucosal secretion in any orifice; 3 abdominal strain and cachexia
# Behaviour alterations. Grades: 0 no alterations; 1 unable to move normally; 2 impossible to arrive to food/drink; 3 unconsciousness and no response to stimuli
†† Percentage of animals with infectious disease. Grades: 0 no infections; 1 animals infected that survived; 2 animals infected that died. Grade 3 not evaluated (NA)
‡‡ Stools appearance. Grades: 0 normal; 1 soft stools; 2 visible blood. Grade 3 NA
¶¶ Percentage of tumour necrosis. Grades: 0 no tumour necrosis; 1 tumour necrosis. Grades 2 and 3 NA
## Local radiation-induced toxicity. Grades: 0 no presence; 1 erythema; 2 exudative lesion; 3 necrosis
In this report, four objectives were considered during radiotherapy design. First, minimize irradiation of healthy tissues around the tumour. Second, fulfil the standard irradiation principles for human treatment. 5,6 Third, reduce time and increase efficiency by irradiating several mice at a time. Finally, be easy to reproduce and repeat on a daily basis by only two people. We demonstrated the feasibility of fractionated irradiation using immunodeficient mice to evaluate the role of radiotherapy on experimental tumours simulating a clinical setting. The major contribution of this study, however, was to demonstrate that general or local effects of irradiation did not jeopardize the tumour radiation–response observation over a long follow-up. In addition, the absence of infect–contagious diseases was particularly decisive for the success of experiments. This provided evidence that the barrier protections used accomplished the goal of preventing microbiological diseases in these animals. Since cancer patients are often immunosuppressed, concerns about zoonoses should not be ignored. Nevertheless, it should be emphasized that the potential risk of zoonoses is extremely low when good laboratory practices and healthy athymic mice are used. To our knowledge, there is no report in the medical literature about the incidence of opportunistic zoonoses in patients treated in such radiation oncology departments.
In conclusion, this study describes an effective method to irradiate human tumours implanted in immunodeficient mice which allows monitoring of tumour responses without interference from the adverse effects of radiation and infection. We demonstrate the feasibility of delivering fractionated radiotherapy using a medical accelerator when a specific experimental facility is not available for radiation research.
Footnotes
ACKNOWLEDGEMENT
The authors would like to acknowledge the financial support of the Spanish Association Against Cancer, Barcelona Committee.
