Abstract
Severe immunodeficient mice are an essential tool for the examination of the efficacy and safety of new therapeutic technologies as a humanized model. Previously, non-obese diabetic (NOD)/Shi-scid IL2rγnull (NOG) mice were established as immunodeficient mice by combining interleukin-2 receptor-γ chain-knockout mice and NOD/Shi-scid mice. The NOG mice are used frequently in the research of therapeutic monoclonal antibodies and regenerative medicine for human diseases. Establishment of an efficient production system of NOG mice, using optimized reproductive techniques, is required to accelerate research. In this study, we investigated the efficacy of the superovulation technique using equine chorionic gonadotropin (eCG) and inhibin antiserum (IAS) in NOG mice of various ages (4, 8, 12, 24, or 54 weeks). Additionally, we examined the fertilizing and developmental ability of the oocytes through in-vitro fertilization using frozen–thawed sperm, embryo culture and embryo transfer. The results showed that NOG mice produced the highest number of oocytes at 12 weeks old following the co-administration of eCG and IAS (collectively IASe) (70 oocytes/female). IASe was more effective in increasing the number of oocytes v. eCG at all ages. The IASe-derived oocytes demonstrated the ability to fertilize and develop into blastocysts and pups. Finally, we demonstrated that three strains of genetically modified NOG mice were efficiently produced through the optimized reproductive techniques. In summary, we developed an efficient system for the production of immunodeficient mice using 12-week-old, IASe-treated female NOG mice.
Introduction
Severe immunodeficient mice are widely used as a host for human cells or tissues to examine the efficacy and safety of new therapeutic technologies in preclinical and translational research. Recently, several institutes and companies started to establish repositories of patient-derived tumour xenograft models for cancer research.1–4 The demand for immunodeficient mice in the scientific community – to systemically conduct large-scale studies using patient-derived tumour xenograft models – is rapidly increasing.
Thus far, various severe immunodeficient mice have been developed based on non-obese diabetic (NOD) 5 and severe combined immunodeficient (SCID) mice 6 backgrounds. In 2002, NOD/Shi-scid IL2rγnull (NOG) mice were established by combining interleukin-2 receptor-γ chain-knockout mice and NOD/Shi-scid mice in the Central Institute of Experimental Animals, Kawasaki, Japan.7,8 The NOG mice are characterized by lack of T-, B- and natural killer cells and poor function of macrophages and dendritic cells. Therefore, the NOG mice are widely used in cancer research and regenerative medicine using induced pluripotent stem cell-derived cell products. 9
In general, a colony of NOG mice is maintained through natural mating in a breeding facility. The average production efficiency of an NOG mouse is 5.3 pups/female mouse by natural mating. The high demand for NOG mice strongly requires an improvement of the productivity of the animal breeding system. Such an advancement may accelerate the research of cancer treatment using patient-derived tumour xenograft models and regenerative medicine using induced pluripotent stem cells. Previously, we developed an efficient production system using reproductive engineering techniques for the C57BL/6J mouse – the most widely used inbred strain to produce genetically modified mice.10–12 Additionally, the reproductive engineering techniques are applicable to major inbred strains of mice. 13 Especially, the ultrasuperovulation technique drastically enhanced the yield of ovulated oocytes and animal production through the co-administration of inhibin antiserum (IAS) and equine chorionic gonadotropin (eCG). 12 This superovulation technique involving the co-administration of IAS and eCG (collectively IASe) may be effective in increasing the productivity of NOG mice. However, the optimal conditions for this technique have not been determined.
In the present study, we examined the yield of ovulated oocytes in eCG- or IASe-treated NOG mice at various ages (4, 8, 12, 24, and 54 weeks). Additionally, the fertilization and developmental ability of the oocytes was evaluated through in-vitro fertilization (IVF) using frozen–thawed sperm, in-vitro embryo culture and embryo transfer. Finally, we demonstrated the productivity of genetically modified NOG mice using an optimized superovulation protocol.
Materials and methods
Animals
Female (aged 4, 8, 12, 24, 54 weeks) or male (aged 12 weeks) mice of the NOD/ShiJic/scid/IL-2Rγnull (NOG) strain (CLEA Japan Inc., Tokyo, Japan) were used as oocyte or sperm donors, respectively. Three strains of genetically engineered mice with a NOG background (aged 12 weeks) were similarly used as sperm donors. Jcl:MCH(ICR) mice (aged 8–16 weeks) (CLEA Japan Inc, Tokyo, Japan) were used as recipients of two-cell embryos. The mice were reared under the following conditions: room temperature, 22 ± 0.5°C; humidity, 55 ± 5%; lights on 07:00–19:00. Food (CA-1; CLEA Japan Inc, Tokyo, Japan) and water were provided ad libitum. All mice were maintained in the Central Institute for Experimental Animals under specific pathogen-free conditions. All animal experiments were approved by the Institutional Animal Care and Use Committee (certification number: 16031) and were conducted according to the institutional guidelines. This study is described in accordance with the ARRIVE guidelines. 14
Media
A mixed solution produced from 18% raffinose pentahydrate and 3% skim milk (R18S3) was used for sperm cryopreservation. 15 Calcium-enhanced human tubal fluid (mHTF) containing 1.0 mM reduced glutathione was used as fertilization medium.11,16,17 Sperm preincubation was performed in mHTF containing 1.0 mM methyl-β-cyclodextrin.10,18 Modified Whitten’s medium (mWM) was used to wash the oocytes and culture the two-cell embryos. 19 Potassium simplex optimized medium (KSOM) was used to culture two-cell embryos to the blastocyst stage. 20 Embryo cryopreservation was performed using a pre-treatment solution (P10: 10% propylene glycol in phosphate-buffered saline (PB1)) and vitrification solution (PEPeS: 10% propylene glycol, 30% ethylene glycol, 0.3 M sucrose and 20% Percoll in PB1). 21 SPB1 (0.3 M sucrose in PB1) was used to warm the vitrified embryos. Percoll was obtained from GE Healthcare (Uppsala, Sweden). Other reagents used to prepare the solutions were purchased from Sigma–Aldrich Co.
Sperm freezing and thawing
The procedure for sperm freezing and thawing was performed as described previously with slight modification.15,22 An aliquot of R18S3 solution (120 µl) was placed in a 35-mm culture dish and covered with paraffin oil. After euthanasia of male NOG or genetically engineered NOG mice through cervical dislocation, their cauda epididymides were removed and completely cleansed of all fat and blood under a microscope before transfer to a drop of R18S3. Subsequently, they were cut into five or six pieces using micro-spring scissors. Freezing straws (0.25 ml plastic straw; IMV) were loaded with 100 µl mHTF and 10 µl sperm suspension, and both sides of the straw were heat sealed. The sealed straws were cooled in the liquid nitrogen gas layer for 10 min. The straws were plunged directly into liquid nitrogen and stored until use.
Superovulation and collection of oocytes
Female NOG mice were intra-peritoneally injected with 3.75 IU of eCG (ASKA Pharmaceutical Co. Ltd, Tokyo, Japan) or 0.2 ml IASe (a mixed solution of 0.1 ml of inhibin antiserum and 3.75 IU eCG, CARD HyperOva, Kyudo Co Ltd, Saga, Japan) at 17:00. At 48 h after the administration of these reagents, 7.5 IU human chorionic gonadotropin (hCG, ASKA Pharmaceutical Co. Ltd., Tokyo, Japan) were intra-peritoneally administered to the mice. At 17 h after injection of hCG, the mice were sacrificed through cervical dislocation and their oviducts were promptly collected and transferred to a fertilization dish covered with paraffin oil. Under microscopic observation, cumulus–oocytes complexes were collected from the oviducts and transferred to 90 µl of fertilization medium. The number of ovulated oocytes and the fertilization ability of oocytes in each group were examined. Genetically modified NOG mice received IASe and hCG to induce superovulation.
In-vitro fertilization
The procedure for preincubation and IVF using frozen–thawed sperm was performed as described previously with slight modification. 11 A straw containing cryopreserved sperm was removed from liquid nitrogen and thawed in a 37°C water bath for 2 min. The thawed sperm suspension (10 µl) was added to 90 µl of sperm preincubation medium covered with paraffin oil. The thawed sperm were preincubated for 30 min at 37°C in an atmosphere containing 5% CO2 to induce capacitation. Subsequently, the sperm suspension (10 µl) was carefully collected, transferred to 90 µl of IVF medium containing cumulus–oocytes complexes (female mouse/drop), and cultured for 3 h at 37°C in an atmosphere containing 5% CO2. The final concentration of motile sperm in the fertilization medium was 400–800 sperm/µl. At 3 h after insemination, the oocytes were washed using three drops of mWM (50 µl) and the number of ovulated oocytes was counted. At 24 h after insemination, the rate of fertilization was calculated as the total number of two-cell embryos divided by the total number of inseminated oocytes and multiplied by 100. Two-cell embryos of NOG mice were divided into two groups to be used in embryo culture with KSOM and cryopreservation. In embryo culture, a number of the two-cell embryos of NOG mice were transferred to 50 µl of KSOM and incubated for 72 h. The developmental rate of blastocysts was calculated as the total number of blastocysts divided by the total number of two-cell embryos and multiplied by 100.
Embryo cryopreservation
Embryo cryopreservation was performed as described previously. 21 After IVF, two-cell embryos of NOG mice were added to approximately 100 µl of P10 solution and subsequently transferred into other drops of 100 µl of P10 solution for 5 min at room temperature. An aliquot of P10 solution (5 µl) containing the embryos was placed and equilibrated in a cryotube for 1 min at 0°C. An aliquot of PEPeS (95 µl) was added to the cryotube and incubated for 1 min at 0°C. After equilibration for 1 min, the cryotube containing the embryos was immersed in liquid nitrogen and stored. The vitrified embryos were warmed by adding 0.9 ml of sucrose solution pre-warmed to 37°C. The vitrified-warmed embryos were collected from the sucrose solution and transferred to 50 µl of mWM. After 10 min, the survival rate was calculated by counting the number of morphologically normal embryos. Embryo transfer was then performed using the morphologically normal embryos.
Embryo transfer
Embryo transfer was performed as described previously. 23 Vitrified-warmed or freshly harvested two-cell embryos of NOG or genetically engineered NOG mice were transferred into the oviducts of Jcl:MCH(ICR) females (8–12 embryos/oviduct) on the day a vaginal plug was present (day 0.5 of pseudopregnancy). Embryo transfer was carried out under three mixed anaesthetics (0.75 mg/kg medetomidine, 4.00 mg/kg midazolam and 5.00 mg/kg butorphanol). 24 After embryo transfer, medetomidine antagonists as painkillers (0.75 mg/kg atipamezole) was administered. The embryos were transferred through the wall of the fallopian tube. The number of offspring was recorded after 19 days.
Statistics
Results are expressed as the mean ± SD. Group results were compared using analysis of variance after arcsine transformation of the percentages; p < 0.05 denoted statistical significance.
Results
Administration of IASe in mice aged 12 weeks produced the highest number of oocytes
IASe-treated NOG mice produced more oocytes than eCG-treated NOG mice at all ages (Figure 1). The highest number of ovulated oocytes was produced by 12-week-old IASe-treated mice. The responsiveness to IASe differed among female mice of varying ages (54 weeks <4 weeks <24 weeks <8 weeks <12 weeks).
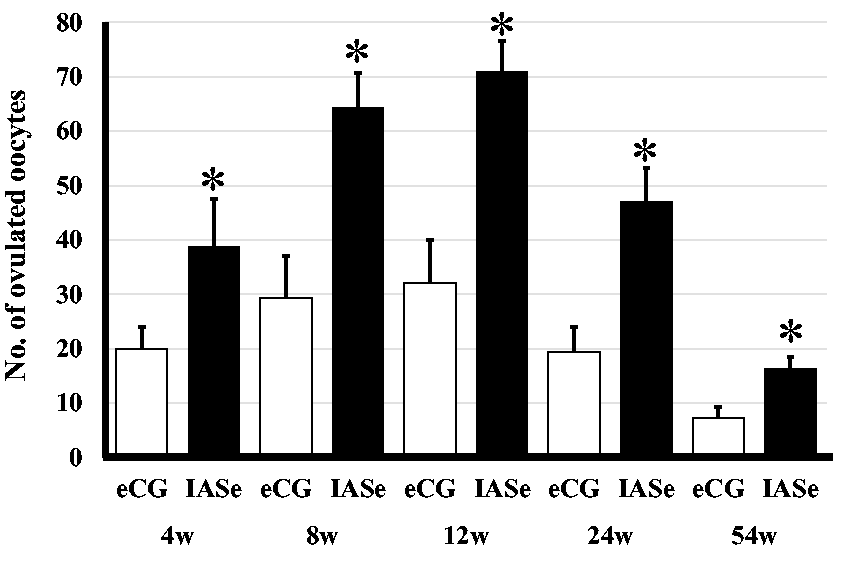
Effect of eCG or IASe on the numbers of ovulated oocytes in NOG mice aged 4–54 weeks. At 17 h after injection of hCG, oocytes were collected from eCG- or IASe-treated female mice. The number of morphologically normal oocytes was counted at 3 h after insemination.
IASe-derived oocytes had fertilizing ability at various ages of NOG mice
Fertilized oocytes were obtained from eCG- or IASe-treated female NOG mice through IVF using frozen–thawed sperm (Table 1). The fertilization rates of IASe-derived oocytes were decreased at 12 and 24 weeks of age.
Effect of IASe or eCG on the fertilization ability of oocytes derived from NOG mice aged 4–54 weeks.
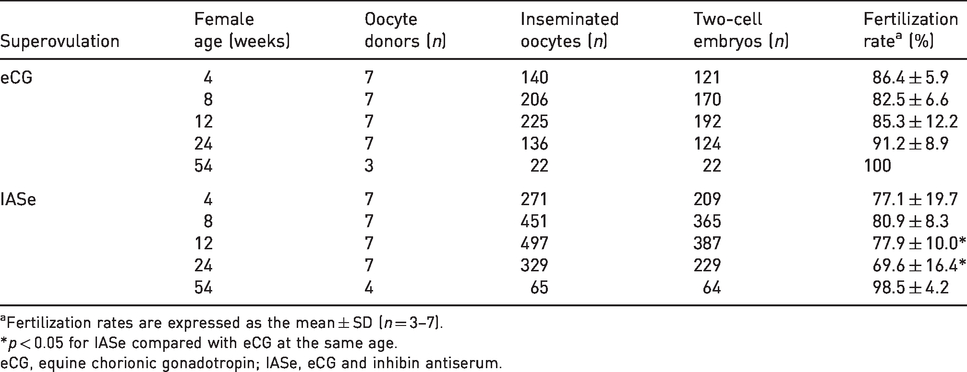
aFertilization rates are expressed as the mean ± SD (n = 3–7).
*p < 0.05 for IASe compared with eCG at the same age.
eCG, equine chorionic gonadotropin; IASe, eCG and inhibin antiserum.
IASe-derived embryos developed to blastocysts in vitro
Two-cell embryos derived from IASe-treated NOG mice developed normally into blastocysts at all ages (Table 2). There was no significant difference between eCG- and IASe-treated female mice at same age in the developmental rates from two-cell embryos to blastocysts in vitro.
Effect of IASe or eCG on the developmental ability of two-cell embryos derived from NOG mice aged 4–24 weeks.
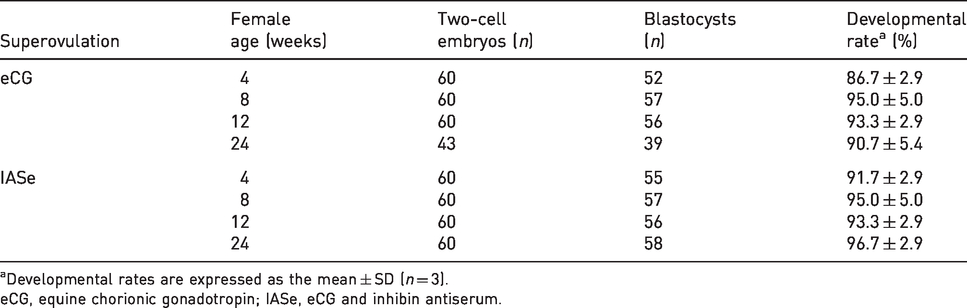
aDevelopmental rates are expressed as the mean ± SD (n = 3).
eCG, equine chorionic gonadotropin; IASe, eCG and inhibin antiserum.
IASe-derived embryos developed to pups
After vitrification and warming of two-cell embryos derived from IASe-treated NOG mice, the embryos developed normally into pups (Table 3). The birth rate of IASe-derived two-cell embryos was lower than that of eCG-derived two-cell embryos at 4- or 8-weeks old.
Effect of IASe or eCG on the birth rates of cryopreserved two-cell embryos.
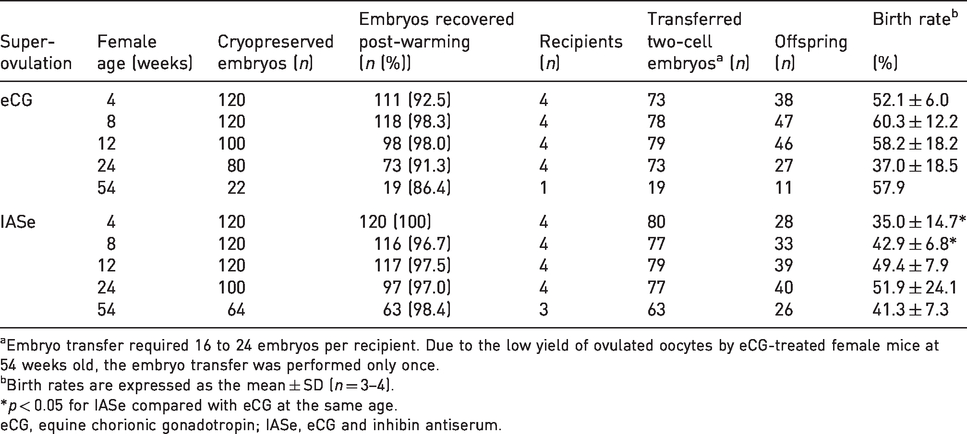
aEmbryo transfer required 16 to 24 embryos per recipient. Due to the low yield of ovulated oocytes by eCG-treated female mice at 54 weeks old, the embryo transfer was performed only once.
bBirth rates are expressed as the mean ± SD (n = 3–4).
*p < 0.05 for IASe compared with eCG at the same age.
eCG, equine chorionic gonadotropin; IASe, eCG and inhibin antiserum.
Efficient production of genetically engineered NOG mice through IVF using IASe-treated female mice aged 10–15 weeks
The production efficiency of genetically engineered NOG mice was evaluated by superovulation using IASe, IVF and embryo transfer. Oocyte and sperm donors were from three strains (strain A, B and C) of homozygous female and male mice. On average, 59 oocytes were obtained from each female mouse via treatment with IASe (Table 4). After the IVF, 83% of the ovulated oocytes were fertilized and 56% of the two-cell embryos developed to pups by embryo transfer. Theoretically, 27.4 pups of NOG mice with a genetic modification can be produced from an oocyte donor by IASe treatment.
Efficient production of genetically engineered NOG mice through IVF: IAS-derived oocytes and frozen–thawed sperm.
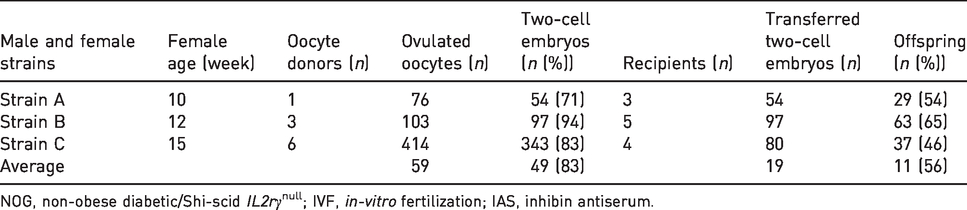
NOG, non-obese diabetic/Shi-scid IL2rγnull; IVF, in-vitro fertilization; IAS, inhibin antiserum.
Discussion
In this study, we firstly reported the effect of IASe on superovulation in immunodeficient mice. The highest number of oocytes was obtained from 12-week-old IASe-treated NOG mice. The oocytes were fertilized via IVF using frozen–thawed sperm. The IASe-derived two-cell embryos were subjected to cryopreservation and embryo transfer. Using the IASe treatment, genetically engineered NOG mice were efficiently produced from a minimal number of oocyte donors.
Previous studies have investigated superovulation in immunodeficient mice using treatment with eCG and hCG. In NOD mice, 22 or 29.3 oocytes were ovulated in mice aged 3–4 or 8–16 weeks.21,25 NOD/SCID mice aged 8–16 weeks ovulated 17.4 oocytes. 21 NOD-Rag1−/−IL2rγcnull (NRG) mice aged 4 or 8 weeks ovulated 15 or 26.7 oocytes 26 . In this study, NOG mice – aged 4 or 8 weeks and treated with eCG and hCG – ovulated 18.3 or 29 oocytes We demonstrated for the first time that IASe possesses the ability to increase the number of ovulated oocytes in NOG mice at various ages (i.e. 4 weeks old, 34.8; 8 weeks old, 63.8; and 12 weeks old, 71.1). This result suggests the treatment with IASe may be effective in increasing the yield of oocytes in different strains of immunodeficient mice.
Our previous study of the ultrasuperovulation technique using IASe showed that >100 oocytes were produced in 4-week-old C57BL/6J female mice. 12 Recently, we demonstrated that the effect of IASe strongly depends on the age of female C57BL/6J mice, with the highest number of oocytes produced at 4 weeks of age.27 Similarly, after treatment with eCG, female C57BL/6N mice aged 25 days produced a higher number of oocytes compared with mice aged 50 or 90 days. 28 However, the present study revealed that 12 weeks was a suitable age in NOG mice. The optimal age to obtain the highest number of oocytes differed between C57BL/6 and NOG mice. Previously, we observed that IASe was more effective in producing large numbers of oocytes v. eCG in major inbred (i.e. A/J, BALB/cByJ, C3HeJ, DBA/2J and FVB/NJ) and outbred (i.e. CD1) 4-week-old mice. Based on the results of this study, optimizing the female age for the injection of IASe may be effective in increasing the number of oocytes obtained from individual strains.
Treatment with IASe to increase the number of ovulated oocytes is used for the efficient production and archiving of genetically engineered mice in mouse repositories and transgenic facilities.12,13,29,30 Oocytes derived from juvenile female mice treated with IASe were used in IVF to produce offspring through embryo transfer or store cryopreserved embryos in liquid nitrogen as bioresources. Additionally, IASe-derived fertilized oocytes at the pronuclear stage are used in gene modification through microinjection or electroporation.31–33 In this study, we demonstrated that our optimized superovulation protocol for NOG mice was effective in efficiently and promptly expanding a colony of genetically engineered NOG mice. Additionally, an efficient production and storage system of NOG mice was developed through embryo and sperm cryopreservation. This technique is relevant to the strong demand for immunodeficient mice by the academic research community and pharmaceutical industry.
In summary, we developed an efficient production system of immunodeficient NOG mice using reproductive technology. Using an optimized protocol of superovulation, treatment with IASe yielded the most efficient production of NOG mice, while minimizing the number of oocyte donors. We strongly suggest that this improved system for the supply of immunodeficient mice may reduce the number of animals used and accelerate innovation to address currently unmet medical needs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Central Institute for Experimental Animals (CIEA).
