Abstract
There is a general consensus that liver fibrosis in humans is potentially reversible, while scepticism prevails on the concept that cirrhosis can be truly reversed. The availability of suitable experimental models is fundamental for disease research. The experimental murine model of liver cirrhosis induced by carbon tetrachloride (CCl4) reproduces both the histological picture of the postnecrotic cirrhosis and its biochemical and clinical parameters. Normal hepatic structure is modified by formation of regeneration nodules. Fibrosis represents a morphological element of disease and an effect of hepatocyte necrosis. However, the relevance for research of this well-established model of liver cirrhosis is hampered by some spontaneous cirrhosis regression reported in mice and rats. It has been reported that CCl4 also induces experimental liver cirrhosis in rabbits, but it is not known whether the process is reversible in this species. The aim of our study was to investigate this question. Male New Zealand White rabbits were treated intragastrically with CCl4 or the vehicle only for 19 weeks and groups were sacrificed three and five months after treatment interruption. Cirrhotic and control livers were processed for routine light microscopy and for morphometric study of fibrosis by semiquantitative evaluation. The degree of fibrosis was based on the Knodell's scoring system.
Keywords
Histological observations and morphometric fibrosis evaluation demonstrated micronodular cirrhosis in CCl4-treated liver, and suggest that this may not be completely reversible.
Introduction
Hepatic fibrosis, a common pathological process of chronic hepatic disease, can lead to irreversible cirrhosis, characterized by morphological conversion from the normal hepatic architecture into structurally abnormal nodules. 1–3 Cirrhosis involves multiple cellular and molecular events that ultimately result in the accumulation of collagen and extracellular matrix protein in the space of Disse, 4 and is characterized morphologically by the loss of normal liver architecture, which is replaced by a new tridimensional structure based on a combination of variable degrees of liver cell regeneration and excessive connective tissue deposition. The reversibility of liver cirrhosis (tested in both the rat and mouse) is very controversial. 5–9 Until the end of the 1970s, it was universally accepted that in chronic liver disease, once fibrosis is established, the process becomes irreversible. 5 However, from experimental studies dealing with the specific issue of irreversibility of the process, Pérez-Tamayo 6 concluded that, in the rat, experimental cirrhosis of the liver progresses via two successive stages: a precirrhotic reversible stage, with histological features indistinguishable from actual cirrhosis, and a cirrhotic stage with finely or coarsely granular liver. Although increased reticular fibres are more easily and completely resorbed than thick collagen bundles, these experimental models are reversible when the inciting agent is removed and sufficient time for the return to normal liver structure is allowed. There is general consensus that liver fibrosis is potentially irreversible. 8,9 The pathogenic mechanism leading to liver fibrosis and cirrhosis results from the excessive secretion of matrix proteins by hepatic stellate cells (HSCs). 10–12 The mechanism of HSC activation has been established 13,14 and HSCs are considered to be the most important cell type involved in hepatic fibrogenesis. The alpha isotype of actin expressed by this cell type reflects their activation to myofibroblastic-like cells and represents a reliable marker of HSC differentiation. 15–17 Moreover, activation of alpha smooth muscle actin (SMA)-positive HSCs has been directly related to experimental liver fibrogenesis and indirectly to human fibrosis in chronic liver injury. The best characterized experimental model has been described by Issa and Zhou 18 who used a 12-week carbon tetrachloride (CCl4) injury murine model to establish the fibrotic appearances of macronodular and micronodular cirrhoses.
On the other hand, the murine animal models currently available do not accurately recapitulate the complex architectural changes observed in human liver disease. Mice and rats develop fibrosis over a relatively short period of time (weeks), while human liver disease develops over many years, and involves irreversible remodelling of matrix and of vascular structures that may not develop in murine models because of the limited time period. In addition, the process has been shown to be reversible in murine models, with the discontinuation of the cirrhogenic agent being followed by a period of liver repair, which produces a significant but relatively small reduction of fibrosis. 18,19 Studies by Di Vinicius et al., 20 Constandinou et al. 21 and Tasci et al. 22 suggest that, when the cause of cirrhosis is removed, the liver may adapt itself to a new and permanent structure, which is probably compatible with normal or near-normal function. This spontaneous regression of liver damage associated with the interruption of the toxic treatment reported in mice and rats 6,10,23 hampers the use of these models to test the value of therapeutics and innovative treatments.
In vivo cirrhosis models remain fundamental for research 9 and identification of new suitable models is a constant aim for liver cirrhosis investigation. More recently, rabbits have been used as animal models for hepatic cirrhosis and sinusoidal fibrosis induced by endotoxins (association of CCl4 and phenobarbitone). 24,25 The aim of this work was to study, for the first time, a rabbit model of CCl4-induced cirrhosis and to investigate whether spontaneous regression occurs.
Materials and methods
Eighteen male New Zealand White (NZW) rabbits were purchased from Harlan Laboratories (San Pietro al Natisone, Udine, Italy). Upon arrival at the laboratory, animals weighing almost 2500 g were acclimatized for seven days to adapt to environmental conditions, food and water, and to allow daily evaluation of health status. All the rabbits were clinically healthy prior to the study and were kept in standard conditions during the experiment. The animals were housed singly in cages (floor area of 3500 cm2 with height 45 cm) in close visual contact. The cages were in rooms maintained at a temperature range of 22 ± 1°C, a relative humidity range of 45% and a 12 h light/12 h dark cycle; 10 ± 15 air changes per hour. Each animal was fed ad libidum with standard pelleted diet for rabbits (Altromin MSK®, Rieper, Bolzano, Italy) and provided with fresh water. The experimental protocol of this work was performed in accordance with the Italian ethics legislation (Ministry of Health, DL 116/92), and in accordance with the guidelines of the European Community Council for animal care and use. The rabbits were divided into four experimental groups: Group 1: control rabbits (n= 9); Group 2: rabbits treated with 99.9% pure reagent grade CCl4 (Sigma Aldrich, Milan, Italy) for 19 weeks (n= 3); Group 3 and Group 4: rabbits in which the CCl4 treatment for 19 weeks was followed by complete interruption of treatment for 12 (Group 3, n= 3) and 21 (Group 4, n= 3) weeks, respectively. At the end of each experimental period (end of CCl4 treatment, after 12 and after 21 weeks discontinuation), rabbits in the appropriate treated groups and three of nine control rabbits (Group 1) were sacrificed.
Kits for the determination of alanine aminotransferase (ALT), aspartate aminotransferase (AST) and albumin were purchased from BCP BioSed (Castelnuovo di Porto, Italy). Alkaline phosphatase (ALP) and urea kits were obtained from FAR Srl (Verona, Italy). ALT, AST, ALP, urea and albumin were measured using these enzymatic spectrophotometric kits according to the manufacturer's instructions. All other chemicals were purchased from Sigma Aldrich.
Experimental procedure
Liver cirrhosis was induced by CCl4 as described by Penteado and others 25,26 with slight modifications for the determination of the CCl4 dose. CCl4 was diluted daily at a concentration of 100 µg CCl4/mL with commercial corn oil. The treatment started with 20 µg (0.2 mL) CCl4. Subsequent doses were increased/decreased by 25% for each 100 U/L increment/decrement of ALT activity. The daily exact amount of CCl4 at the above concentration for each animal was calculated in advance and the correct amount was taken using a graduated 1 mL syringe. This volume was administered intragastrically with the aid of a mouth gag inserted behind the incisors. A rubber tube was then inserted through the hole in the gag as far as the stomach. Each week, the same operator administered the required dose of intragastric CCl4 after adjusting the volume. The weekly administration was always performed on the same day of the week and at the same time of the day (11:30 h). The day after each administration, blood samples were taken from the marginal ear vein and collected in heparinized tubes. Blood was centrifuged (3500 rpm, 15 min, at 6°C) to obtain the plasma.
ALT, AST, urea and albumin were measured using kits according to the manufacturer's instructions. The measurements were usually performed on the same day. Alternatively, aliquots of plasma were kept at −20°C till analysis.
The CCl4 treatment was continued for 19 weeks and then discontinued till sacrifice 12 (total experimental period 31 weeks) and 21 (total experimental period 40 weeks) weeks later. During the study, the animals were observed daily by a laboratory animal veterinarian and animal care staff and no adverse effects were observed. The rabbit body weight and relative food consumption were measured three times per week. Humane endpoints were established on the basis of the Morton and Griffith's parameters (1985). 27 No animals died during experimentation.
At the end of the experimental period, all animals were fasted overnight in order to reduce the stomach contents. After that the blood sample was taken, the rabbbits were fully anaesthetized with Ketavet® (ketamine hydrochloride, 35 mg/kg from Intervet Productions Srl, Latina, Italy) and Rompum® (xylazine hydrochloride, 5 mg/kg, Bayer SpA, Milan, Italy) and sacrificed with about 2 mL of Tanax® (embutramide, Intervet Italia Srl, Peschiera Borromeo, Milan, Italy) by intracardiac injection. Complete postmortem examinations (necropsies) were performed under the supervision of veterinary certified pathologists on all carcasses submitted for necropsy. The necropsy included an external examination and an examination of the internal organs. The abdomen was opened by a midline incision from the sternum to the pelvis. The abdominal and pelvic organs were examined in situ. After removal of the gastrointestinal tract, the liver was removed by cutting the hepatic ligaments and the hepatic tissues were examined. The excised livers were washed in sterile saline, dried on Whatman paper (Tecnochimica Moderna Srl, Rome, Italy) and weighed. Aliquots (400 mg each) of whole liver were used for histopathological and morphological examinations.
Histomorphological study
Liver samples (400 mg each) were excised, fixed in buffered formalin for 48 h and embedded in paraffin with a melting point of 55–57°C.
For histomorphological examination, sections were cut in 5 μm thickness and stained with haematoxylin and eosin and Masson's trichrome for collagen fibres. Observations were performed at magnifications of ×4, ×10 and ×20 with a light microscope (Leica Microsystems DM 4500B, Milan, Italy).
Morphometric study
Fibrosis semiquantitative evaluation was carried out as follows: images were captured with a light microscope (Leica Microsystems DM 4500B) connected to a videocam (ProgRes C10 plus) and provided with an Image Analysis System (Delta Sistemi, Rome, Italy) as described elsewhere. 15,28 The samples were evaluated independently and blindly by three investigators (Mammola C L, Franchitto A and Carpino G). To evaluate the degree of fibrosis, conventional Masson's trichrome stain and Knodell's scoring system were used. This classification system stratifies the degree of fibrosis as follows: 28,29
0 = no fibrosis; 1 = fibrous portal expansion; 2 = bridging fibrosis and 3 = cirrhosis
The modified Knodell's scoring system proposed by Ishak 30 was used to evaluate separate scores for the degree of necroinflammation (histological activity index [HAI]). 31–35
The necroinflammatory score (NI score) was assigned according to the severity of the inflammation and necrosis as follows:
0 = no lesions; 1 = mild focal necrosis; 2 = moderate to severe necrosis affecting less than 25% of liver tissue examined and 3 = severe necrosis affecting more than 50% of liver tissue examined
Statistical analyses
Data are reported as mean ± standard error of the mean (SEM). Two-way analysis of variance was used for analysis of rabbit weight and food consumption.
All other results were analysed by parametric Student's t-test. Changes were considered significant at P < 0.05.
Results
Doses of CCl4
To induce experimental cirrhosis in rabbits, Penteado et al. 25 started with a dose of 20 µg and then administered 40 μg when transaminases were <200 IU/L, 30 μg when their levels were between 200 and 400 IU/L and 10 μg when transaminases were ≥800 IU. 25 In a pilot experiment, however, using the CCl4 dosing protocol proposed by Penteado and others as above, 25,26 about 60% mortality were found in treated animals (5 of 9 rabbits), while the control rabbits were unaffected, and for this reason the pilot experiment was stopped and we adopted a modified method for the determination of the CCl4 dose. First, as AST and ALT levels do not always parallel each other, the CCl4 weekly dose was determined using ALT levels. 24 We started treatment with 20 µg CCl4 and subsequent doses were increased/ decreased by 25% for each 100 U/L increment/decrement of ALT activity as described in Materials and methods. Using this method the animals showed no adverse effects and there was no mortality during the experiment.
Rabbit body and liver weight
Body and liver weights in control and CCl4-treated rabbits
Body weight and liver weight, and the liver weight as a percentage of the body weight in rabbits at the end of CCl4 treatment (19 weeks) (Group 2), and 12 (Group 3) and 21 (Group 4) weeks after discontinuation of the treatment. Values for control rabbits (Group 1) were obtained after sacrifice of three of nine animals at the end of each experimental period (end of CCl4 treatment, and 12 and 21 weeks after discontinuation of CCl4 treatment)
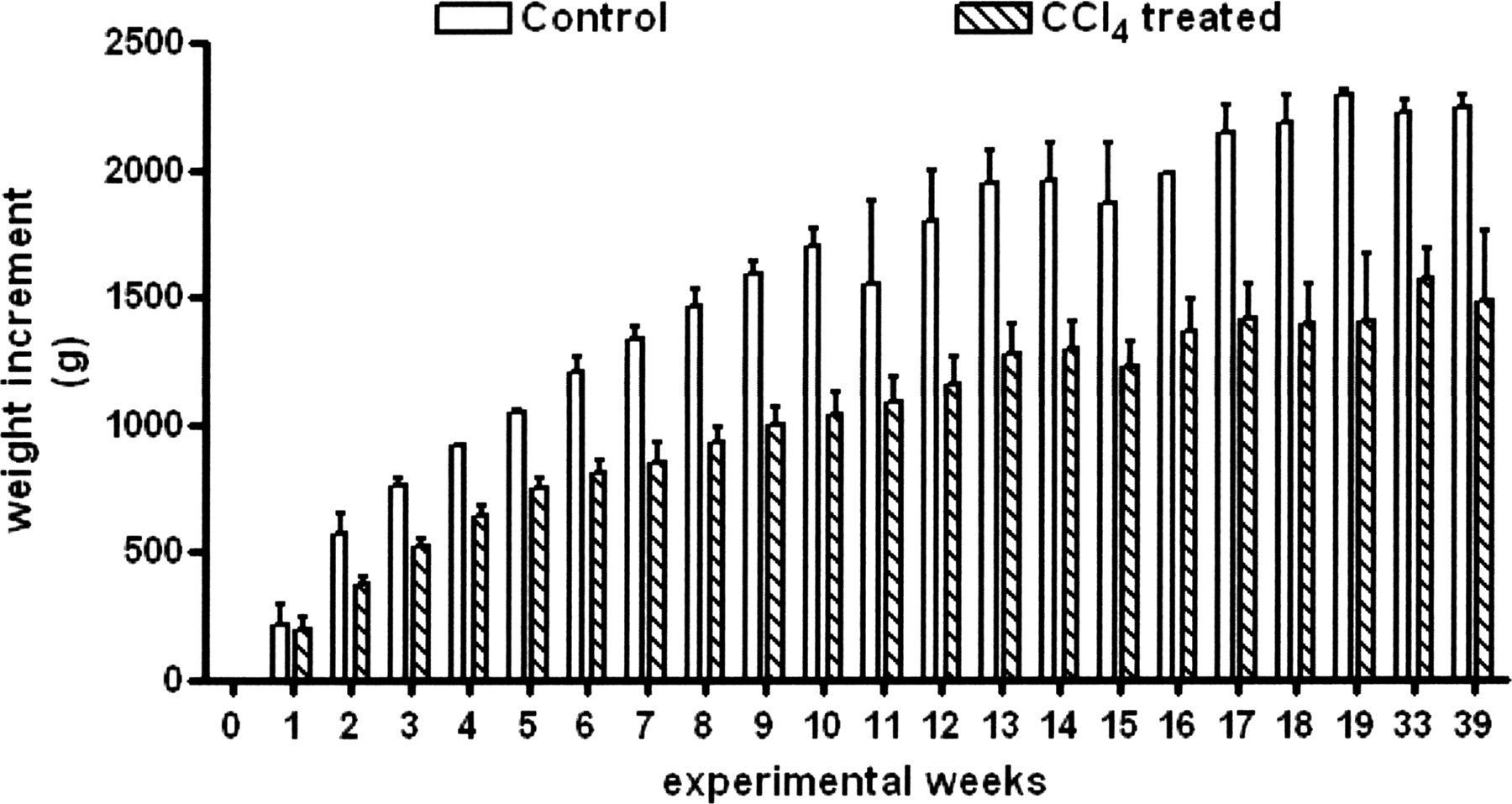
Rabbit body weight increment during the experiment. Time course of the body weight increment in control and CCl4-treated rabbits during the experiment. For both groups: 0–19 weeks: n= 9; 31 weeks: n= 6; 40 weeks: n= 3
Light microscopy
Liver cirrhosis was induced by CCl4 and the treatment of Group 2 was continued for 19 weeks. The histological features of experimental cirrhosis were apparent after this time (Figure 2).
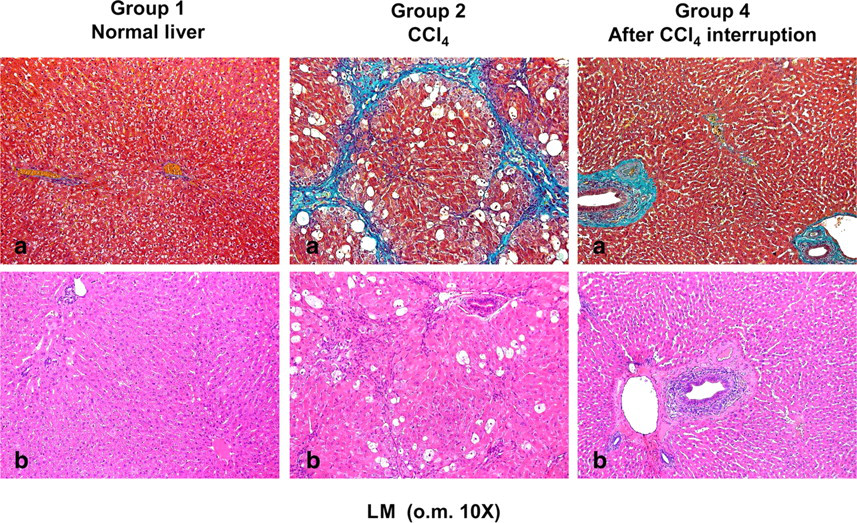
Histomorphological observations of rabbit liver (light microscopy [LM] o.m. ×10). (a) Masson stain. (b) Haematoxylin–eosin stain. In control rabbits (Group 1) light microscopy showed a normal parenchyma. Hepatic laminae spread out regularly from central veins. Border of the hepatic lobules was not lined by any connective tissue. Network sinusoids appeared continuous and interconnected. Necrosis and fibrosis were absent. In CCl4-treated rabbits (Group 2), the liver appeared diffusely nodular. Fibrosis was present in uniform septa of connective tissue and the nodules were defined by a thin fibrous capsule. Necrosis areas were present. In rabbits 21 weeks after CCl4 treatment interruption (Group 4), livers showed recovery to normal parenchymal architecture with the disappearance of nodular shapes. Connective tissue was organized into fibrotic bundles. Necrosis was almost completely absent. Each experimental group included three rabbits
In control rabbits (Group 1), light microscopy examination showed a normal hepatocytic structure with regular hepatic laminae radiating from the central veins. The borders of the hepatic lobules were not lined by any connective tissue. The network sinusoids appeared normal as a continuous and interconnected structure. Necrosis and fibrosis were absent: the NI score and Knodell's scoring system were 0 and the morphometric analysis assigned a percentage of connective tissue as 0.8 (Figure 2).
In cirrhotic rabbits of Group 2 after 19 weeks of CCl4 treatment, at light microscopy, the livers appeared diffusely nodular with micronodular shapes. Fibrosis was present in uniform septa of connective tissue and the nodules were defined by a thin fibrous capsule. This, in some cases, extended through the parenchyma across the liver nodules (Figure 3). The NI score was 2, corresponding to moderate necrosis affecting less than 25% of liver tissue examined. The degree of fibrosis evaluated by morphometric analysis was 14% and Knodell's scoring system was 3, corresponding to cirrhosis (Figure 3).
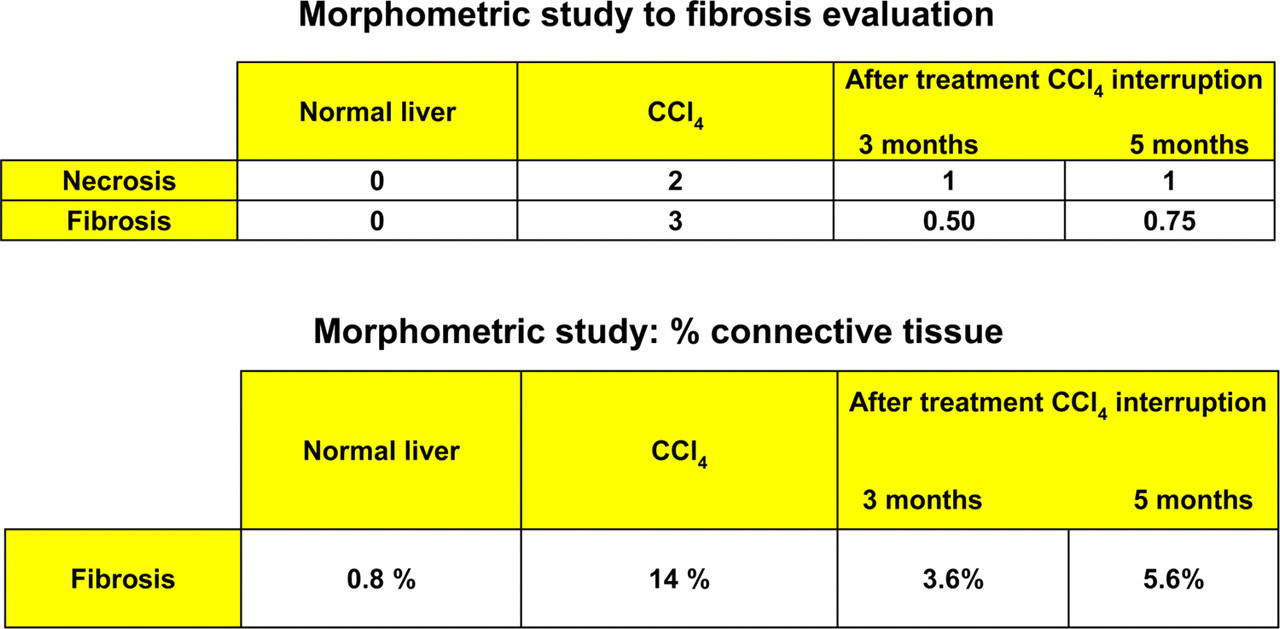
Morphometric study of the fibrosis and percent connective tissue in rabbit liver
The animals from Group 3 killed three months after interruption of the treatment showed progressive disappearance of connective tissue bands and returned to normal liver architecture: the NI score was 1, corresponding to mild focal necrosis. However, thin fibrous septa were present, and the morphometric evaluation assigned a percentage of connective tissue equal to 3.6. By Knodell's scoring system the degree of fibrosis was 0–1 (Figure 3).
The animals from Group 4, killed 21 weeks after interruption of the treatment, showed recovery of the normal hepatic parenchymal architecture with the disappearance of nodular shapes, as seen in Group 3 animals (Figure 2). The NI score was 1 as in the previous group but, in contrast to Group 3 livers, connective tissue appeared to be organized into thicker fibrotic bundles. The morphometric analysis assigned a percentage of connective tissue equal to 5.6, while by Knodell's scoring system the degree of fibrosis was 1.5 (Figure 3).
Biochemical parameters
Transaminase levels the day after CCl4 treatment
Average values of ALT and AST activities in the plasma of control and CCl4-treated rabbits at the start of the experiments (week 0) and at each two-week interval during the treatment (total treatment time = 19 weeks)
ALT: alanine aminotransferase; AST: aspartate aminotransferase
Plasma biochemical parameters in control and CCl4-treated rabbits
Biochemical parameters in rat plasma determined at the end of CCl4 treatment (19 weeks) (Group2), and 12 (Group 3) and 21 (Group 4) weeks after discontinuation of the treatment. Values for control rabbits (Group 1) were obtained after sacrifice of three of nine animals at the end of each experimental period (end of CCl4 treatment, and 12 and 21 weeks discontinuation of CCl4 treatment). Each group included three rabbits
*P< 0.05 versus the relative control
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase
Discussion
The murine CCl4 liver cirrhosis model is well studied, but little information is available about this model in rabbits. In the original description of the model, Penteado et al. 25 determined the CCl4 dose on the basis of transaminase levels. However, AST and ALT levels do not always parallel each other (Table 2). Furthermore, in a pilot experiment, we found that the dose of CCl4 used in previous reports 24,25 resulted in a high mortality rate. Since the circulating levels of AST are mainly derived from the liver, while the ALT plasma activity may also be derived from other organs (i.e. heart), 26 in this study the dose of CCl4 was determined using plasma levels of the more liver-specific ALT. We adjusted the weekly CCl4 dose by increasing/decreasing the CCL4 dose by 25% for each 100 U/L increment/decrement of ALT activity in the precedent assay. With these adjustments, mortality was abolished and all the rabbits were included in the study.
In our rabbit model after 19 weeks of CCl4 treatment the histological picture and clinical aspects are consistent with experimental cirrhosis and the findings of other authors. 24,25 The present results show that experimental hepatic damage induced by prolonged treatment with CCl4 presents an anatomopathological picture of postnecrotic cirrhosis according to aetiopathogenic classification of the disease. 2,3
Parenchymal normal architecture was largely changed, with destruction of sublobular or – sometimes – lobular areas. Fibrosis appeared to be evident from various fibrous thickening extending through the parenchyma-lined nodules of different sizes.
In established cirrhosis, necrotic shapes are marked by the presence of scarring tissue, while the presence of inflammatory infiltrates is an expression of the active chronic process. Fibrous tissue which is formed in response to the inducing agent is found near the portal tract, around the lobular vein, or directly in the spaces of Disse.
The action of sclerosing scar tissues on blood vessels is mainly responsible for the dramatic haemodynamic changes, which may lead to portal hypertension.
Vascular reorganization is important for maintaining the residual parenchyma functions and for allowing regeneration in cirrhosis.
Recovery from comparatively advanced cirrhosis is possible, and results in remodelling from a micronodular cirrhosis to a macronodular cirrhosis. 18 Issa et al. 18 and Kumar 35 suggest that cirrhosis resolution is characterized by apoptosis of HSCs 18 and that a capacity for recovery from any degree of fibrosis, including those associated with cirrhosis, is possible. 35 The term ‘reversal’ is often used to indicate a complete restoration of normal architecture, whereas ‘regression’ indicates that ‘the fibrosis content is less than earlier, without quantifying the extent of regression’. 35 Then, the factors involved in fibrosis are dynamic and are a balance between fibrogenic and fibrolytic forces.
CCl4 treatment reduced the natural body weight increase of the rabbits, and the body weight difference between the untreated and treated groups remained similar after the discontinuation of the treatment. Liver weight was also reduced by CCl4 treatment, and its percentage with respect to rabbit whole body weight was lower in the cirrhotic groups than that in the control group. Interestingly, the liver weight as a percentage of body weight continued to decrease even three and five months after the termination of the CCl4 treatment, suggesting that the general recovery of functional hepatic activity is not accompanied by a recovery in liver mass. It is possible that this finding may be due to the partial recovery of body weight observed after discontinuation of CCL4 treatment, but this seems unlikely, as the absolute liver weight in the treated animals is very low. It has been suggested that the organ-to-brain weight ratio may be more useful than that of the organ-to-body weight ratio in toxicological studies. 36 However, when Bailey et al. 36 compared the use of the two ratios they concluded that, although the organ-to-brain weight ratio is appropriate for some organs, such as the adrenal gland and ovary, for the liver, organ-to-body weight is the better measure. In contrast to the findings on liver weight, the levels of transaminases were elevated during CCl4 treatment, but normalized after discontinuation of the treatment. The levels of the other hepatic indexes such as ALP, cholinesterase and albumin showed no consistent alteration during the whole study.
Our findings show that discontinuation of the toxin for 12 weeks produced a reduction of fibrosis (although collagen was still 4.5 times greater than control). Moreover, 21 weeks after treatment interruption, the connective tissue percentage in Group 4 was seven times greater than that in the control rabbits.
Our findings indicate that, when the inducing agent of cirrhosis is removed, the liver may adapt itself to a new and permanent structure, which is probably compatible with near-normal function. Ultimately, however, it seems there is a partial regression of fibrosis, but not a true reversal, in our rabbit model. In fact, the percentage of fibrosis was less when the inciting factor was interrupted both at 12 and 21 weeks compared with the CCl4-treated animals. Both the structure of the hepatic parenchyma and the levels of hepatic functionalities (indices) seem to stabilize after the suspension of the treatment in the time periods considered. Furthermore, because of this spontaneous tendency to recover from hepatic alterations, caution should be taken in the use of rodent and rabbit models in which experimental cirrhosis is induced by CCl4 for the evaluation of hepatic, histomorphological and functional alterations in potential long-term therapeutic approaches.
Footnotes
ACKNOWLEDGEMENTS
The authors are grateful to Mr Antonio Di Virgilio and Mr Agostino Eusepi for their skilled help with animal work and to Dr Ghada M Safwat for her help. The authors are also grateful to Dr Kathleen M Botham for her useful comments and revision of the English language.
