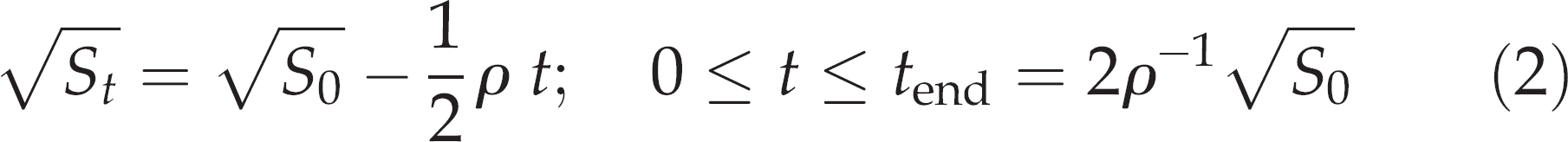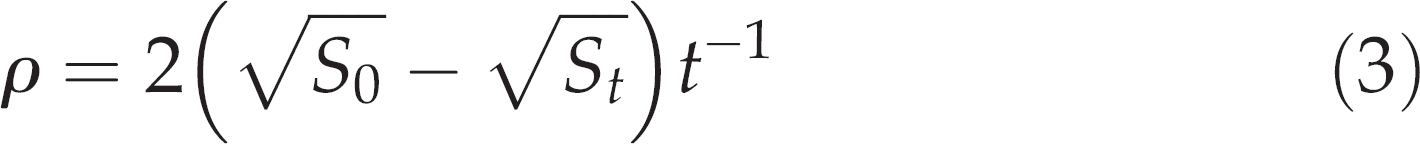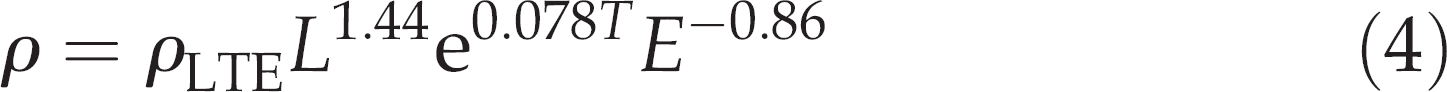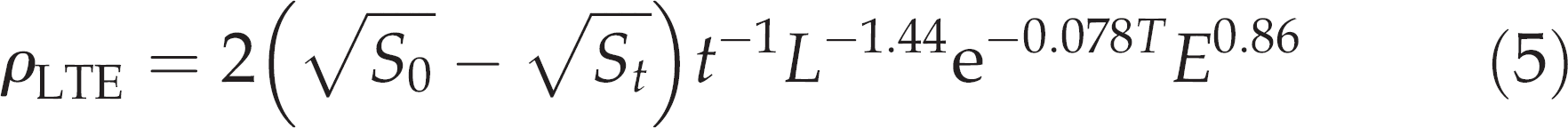Abstract
The current study investigated how the gastric evacuation rate (GER) was affected after surgically introducing dummies of a blood flow biotelemetry system into the abdominal cavity of Atlantic cod, Gadus morhua. Gastric evacuation experiments were performed two and 10 days postsurgery on surgically implanted and control G. morhua force-fed sandeel, Ammodytes tobianus. The results were compared with previously obtained estimates from unstressed conspecifics voluntarily feeding on a similar diet. After two days, GER was significantly lower in the group of fish with the dummy implants compared with the control group, but following 10 days of recovery no significant difference was seen between the two groups. The difference between implanted and control fish observed two days postsurgery may have resulted either from surgery, postsurgical stress and/or the presence of the implant. The conclusion is that 10 days of postsurgical recovery will stabilize GER in G. morhua, thus indicating that at this point the implant per se did not affect GER. Both the fish with surgical implants and controls in this study evacuated their stomachs much slower and with much higher interindividual variation compared with G. morhua feeding voluntarily on similar prey items. The lower GER and higher interindividual variation for force-fed fish indicate that handling, anaesthetization and force-feeding impair GER and that individual fish respond differently to the suppressing effects.
Invasive procedures are often necessary for the physiologist to understand the mechanisms behind various physiological parameters. In fish, measurements of cardiovascular parameters such as blood flow and blood pressure involve handling, anaesthesia and surgical implantation of catheters and flow probes in and around vessels and sutures to close the incisions. In traditional laboratory experiments, standard bench top blood pressure and blood flow measuring units are used, which means that the animals are ‘hardwired’ to the equipment during the experiments. Catheters and leads from flow probes penetrate the body wall and increase the risk of infection. Furthermore, to prevent tangling and ripping of wires the animals are most often confined in small containers, which severely limit the range over which they can move with the risk of introducing confinement stress. The animals are usually left for 18–48 h before experimentation is initiated, the main reason for such shorts periods being the high risk of infection after an extensive surgery. 1 In cases where catheters are used, limits on the time where these will remain open may also be decisive. However, unless appropriate recovery time is ensured following surgical procedures and instrumentation, major concerns are the validity of data and ethical considerations. In a study on rainbow trout Oncorhynchus mykiss using bioelectric potential recordings to measure heart rate, fH (i.e. a non-invasive method) the fish were handled and then left to recover for at least three days after which their basal heart rates were much lower than those presented in previous studies using traditional methods involving surgical procedures. 2 A lower heart rate is usually indicative of lower stress levels pointing to the fact that these animals were not as stressed due to confinement and the use of ‘hardwired’ techniques. Obviously, any misinterpretation of data may have important consequences for our understanding of the physiology of fishes and researchers should be encouraged to seek refinements of techniques to ensure more valid results as well as better animal welfare.
The rapid development of telemetric devices opens new research areas that have not been possible with hardwired animals and during the last few decades there have been great advancements made in applications of biotelemetry in fish physiology. Biotelemetry allows data collection from specimens that move around freely and can behave in a more natural way. In the telemetrically instrumented animal, the risk of infections is lower since there are no skin-penetrating wires. Furthermore, long-term recovery and recording periods are possible in reducing stress from handling, making this an attractive alternative. Altogether, this provides a number of welfare advantages and the opportunity to obtain a higher quality of the measured variables and more reliable routine values. A recent study on white sturgeon (Acipenser transmontanus) showed that routine heart rate of freely moving biotelemetric instrumented fish was lower compared with hardwired, confined conspecifics as well as with previous values reported for white sturgeon.3,4 The downside is that the animals are carrying the entire recording/transmitting unit, increasing their body mass and possibly impairing the capacity to generate thrust by altering the ability to bend the body. It may furthermore interfere with the internal organs if placed in the body cavity. Nevertheless, a fully implantable system maximizes the likelihood that the studied fish will be treated normally by surrounding fish 5 and minimizes the risk of infection and expulsion. 6 Even the presence of a small, protruding antenna has been shown to cause adverse tissue reaction at the antenna exit7,8 and to elicit aggressive attacks from other individuals of Atlantic salmon smolts. 5
We are looking to use a fully implantable biotelemetry system4,9 to study blood flow distribution in Atlantic cod, Gadus morhua L. voluntarily feeding and moving between different habitats. Before employment of such a system it is, however, imperative to evaluate appropriate postsurgery recovery time and possible adverse effect of the telemetric implant. Using a dummy version of the dual blood flow biotelemetric system used by Gräns et al.,4,9 the aim of the present study was to examine the effects on the gastric evacuation rate (GER) of handling, surgery and instrumentation of G. morhua with a dual channel blood flow telemetric system following short-term (2 days postsurgery) as well as prolonged (10 days postsurgery) recovery. Both the surgically implanted and control fish were force-fed to standardize the amount of food and the time of feeding. The ability to evacuate the force-fed meal was determined and compared with estimates obtained from unstressed conspecifics that fed voluntarily.
Materials and Methods
Fish
Wild G. morhua ranging in total body mass from 290 to 815 g were caught in late 2009 by fyke net in the vicinity of the Marine Biological Laboratory in the northern part of Øresund, Denmark. The fish were transferred to the laboratory where they were kept in 9–10°C re-circulated, aerated seawater. The tank was covered by tarpaulin to avoid unnecessary disturbance of the fish. Light/dark conditions were 16:8 h. All fish were acclimatized to laboratory conditions for a minimum of 12 weeks before experiments were initiated and fed ad libitum three times a week with chopped herring (Clupea harengus L.) or Raitt's sandeel (Ammodytes marinus Raitt). Before the experiment the fish were starved for five days to ensure that their stomachs were empty.
Preoperative care
Individual G. morhua were anaesthetized in 15 L of water containing 0.15 g/L 3-aminobenzoicacid ethyl ester (MS-222) until gill ventilation ceased. Each fish was weighed and its length measured before it was positioned with the ventral side facing up on an operating table covered with wet sponges. Anaesthesia was maintained during the surgery by pumping oxygenated sea water containing MS-222 (0.075 g/L) over the gills. All surgical instruments, the dummy implant and associated leads were sterilized using a cold steriliant (Cidex, Johnson & Johnson Company, New Brunswick, NJ, USA).
Dummy implant
Our long-term goal is to implement the fully implantable biotelemetry system used by Gräns et al.4,9 This system weighs 35 g (total mass of implant and battery) in air and we aim for a transmitter:fish mass ratio of 3–4%. Consequently, the dummy implant was constructed so that its total mass equalled 19 g in air, thus, corresponding to an average of 3.7 (0.3)% of the mean body mass of the present experimental G morhua. It was made of silicon-coated stainless steel (L = 35 mm, H = 5 mm, W = 16 mm) with an attached silicon-coated dummy battery made of plastic tubes (L = 18 mm, D = 11 mm) and two silicon leads with a probe cuff each, and the implant fitted easily within the abdominal cavity of the fish.
Surgery
The splanchnic circulation in G. morhua is derived from the celiacomesenteric artery (CoMe). The CoMe is the largest single branch of the dorsal aorta and branches to form the coeliac artery and mesenteric artery. The coeliac artery supplies parts of the cardiac stomach, the pyloric stomach, the proximal intestine and parts of the distal intestine, while the mesenteric artery supplies the remainder of the gastrointestinal system, the gallbladder and the spleen. 10 The surgical procedures were performed on 10 fish (body mass 550 [144] g and length 38.7 [3.2] cm) and done in accordance with the guidelines described in permission 2010/561–1812 from the Danish Ministry of Justice. For placement of the first dummy probe, a 2 cm long incision was made alongside the basibranchial bone left of the midline, and the liver carefully retracted, after which the CoMe artery was exposed and cleared. The dummy implant and battery were then carefully placed in the abdominal cavity and the retracted organs restored to their places of origin. Furthermore, an individual passive integrated transponder (PIT) tag (12 mm × 2 mm, ISO standard 11784/11785, FDX-B) was placed in the body cavity for later identification of the fish. To access the ventral aorta for placing the second dummy probe, a 0.5 cm ventral incision was made posterior to the gill juncture and carefully, without disrupting the pericardium or damaging any vessels, the dermal and subdermal musculature and connective tissue were separated using blunt dissection tools to expose the ventral aorta. The probe was tunnelled under the skin from the abdominal cavity to the incision above the ventral aorta (see Figure 1 for schematic overview of positioning of the system). The wires from the probes were anchored with a single stitch of 3/0 silk suture in the subdermal muscle tissue and the two incisions were closed using sterile monofilament prolene 3–0 suture. Subsequently, the cod was given a subcutaneous injection of 0.4 mg/kg [1 mmol/L] butorphanol (Torbugesic, Fort Dodge, IA, USA) for postoperative pain relief in addition to an injection with 10 mg/kg [1 mmol/L] enrofloxacin (Baytrilo, Bayer, Shawnee Mission, KS, USA) antibiotics, to minimize the risk of infection. Finally, to reduce incidence of oomycete infections, povidone iodine powder was applied to the closed incision before returning the fish to recovery water. The surgical procedure including anaesthesia and awakening took approximately 20 min for each fish and when ventilation and locomotion had been re-established, the fish were returned to their home tank. All but one fish survived the surgery.
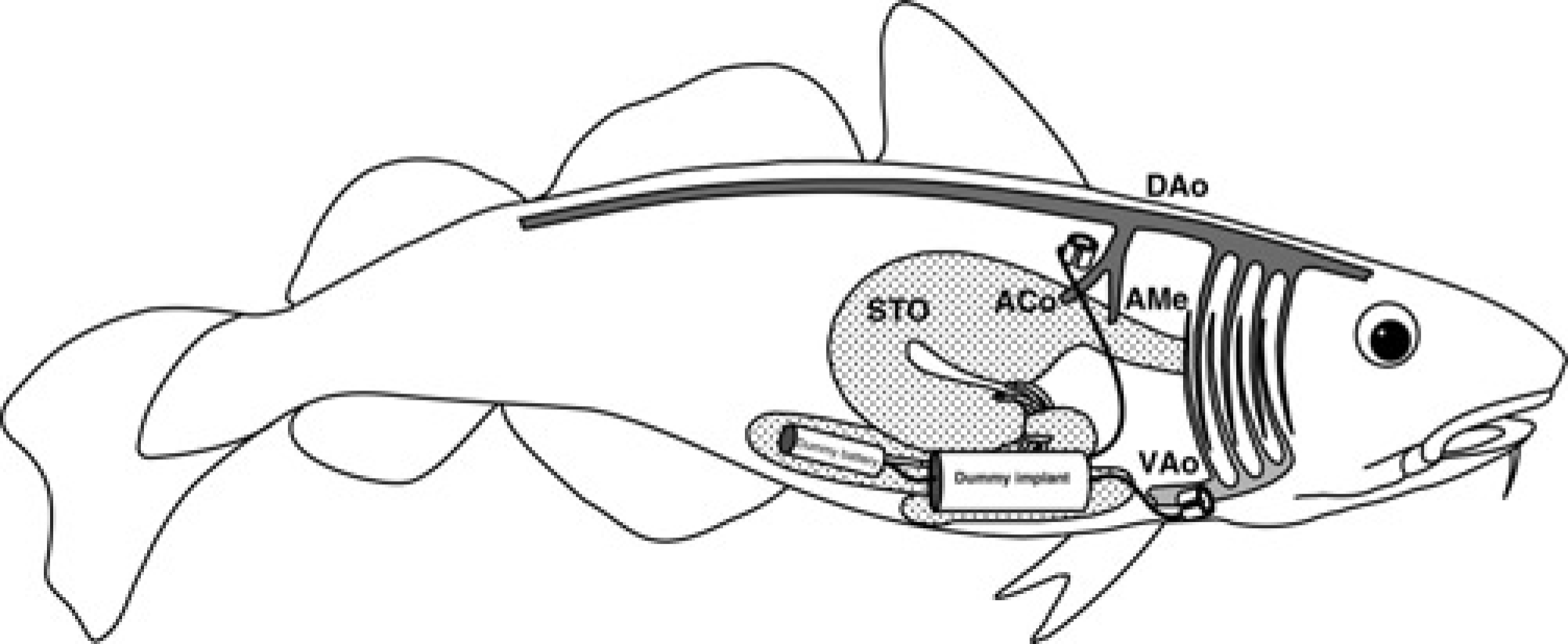
Schematic drawing of a Gadus morhua showing the placement of the dummy implant and battery (both drawn to scale) in the abdominal cavity and the flow probes belonging to it. The celiacomesenteric artery branches off the dorsal aorta (DAo) and subsequently divides into the coeliac (ACo) and the mesenteric artery (AMe). VAo: ventral aorta; STO: stomach
The control fish (n = 8, body mass 518 [155] g and length 37.1 [2.7] cm) were anaesthetized as described above, and subsequently weighed, length measured and individually PIT tagged. The tags were placed in the abdominal cavity, using a syringe implanter, taking care not to harm any organs. The control fish were likewise left to recover before being returned to their home tank.
Food
Ammodytes marinus constitutes a natural and large part of the diet of wild G. morhua and one specimen per cod was used as the experimental meal. To minimize variation in size and condition, specimens of A. marinus from a single commercial batch (TripleNine, Hvide Sande, Denmark) were used for force-feeding. The energy density of these prey fish was determined by bomb calorimetry (IKA C-7000 bomb calorimeter [www.ika.net]) on a representative sample after drying it at 60°C until constant mass according to the procedure described in Pedersen & Hislop. 11
Gastric evacuation experiments
The first experiment was initiated 48 h postsurgery. On the day of experiment, A. marinus were quickly thawed in running water, and those of similar total lengths were selected. These were subsequently dabbed dry with paper towel and weighed, and individuals within a mass range of 9.4–15.7 g were chosen (corresponding to 2–3% of the body mass of the G morhua). This food ration was chosen from the largest daily feeding rates observed in wild G. morhua. 12 In random order individual cod were anaesthetized by placing them in 15 L of water containing 0.07 g/L MS-222. They were then force-fed by gently pushing a whole A. marinus, bent in the middle, through the oesophagus and into the stomach. Immediately thereafter, the fish were returned first to recovery water and subsequently to their home tank. The exact time of force-feeding was noted for each fish, as was the length and mass of the prey.
Stomach contents were recovered 22–26 h post-force-feeding by stomach flushing following anaesthetization (0.07 g/L MS-222). The stomach content from each fish was collected on a filter with a mesh size of 200 μm, and gently patted with a moist paper towel and weighed. The exact time for recovery of stomach content was noted for each cod.
The fish were subsequently left to recover for seven days in their home tank, after which the above described protocol was repeated in a second trial.
Calculations and statistical analyses
In accordance with the cylinder model of Andersen and Beyer,
13
the GER of stomach contents in G. morhua (as well as in a variety of other piscivorous fishes) can be described independently of meal size by the current mass of stomach contents St (g) together with the rate parameter ρ:
Integrated from time 0 of force-feeding to time t (h) of recovery of stomach contents, the solution to Equation (1) is G. morhua evacuating its stomach contents according to the relationship
where tend is the time of complete evacuation of the prey.
The ability to evacuate the stomach contents is then described by ρ, which can be calculated for each individual G. morhua by the reorganization of Equation (2):
The rate parameter ρ depends on predator (total) length L (cm), temperature T (°C) and prey energy density E (kJ/g). The effects of these variables were estimated by Andersen 14 for different gadoids fed fish prey, and the following relationship was obtained:
The basic rate parameter ρLTE specifies the general ability of the predator to digest and evacuate a specific prey type from the stomach (or reversed: it indicates the resistance of the specific prey to the digestive processes in the stomach). The value 0.00142 ± 0.00011 (estimate ± SD) of ρLTE was obtained from an experiment by Andersen 14 on G. morhua that fed voluntarily on lesser sandeel Ammodytes tobianus L. The temperature (10.3°C) and mean body size of G. morhua (41 cm; 664 g) were similar to those of the present study. This value of ρLTE was used to decide the evacuation time t of c. 24 h employed in this study, which was shorter than tend and yet long enough to ensure that a significant part of the meal was evacuated from the stomach prior to recovery of the remains.
G. morhua used in the present study varied to some extent in size (Table 1). ρLTE was therefore used to represent the ability of individual G. morhua to evacuate their force-fed meals of A, marinus. By using this parameter, the results could further be compared directly with the above estimate obtained from unstressed G. morhua that fed voluntarily. Combining Equations (3) and (4), the value of ρLTE for each individual G. morhua was calculated by
Basic data on gastric evacuation experiments (mean [SD]) on Gadus morhua
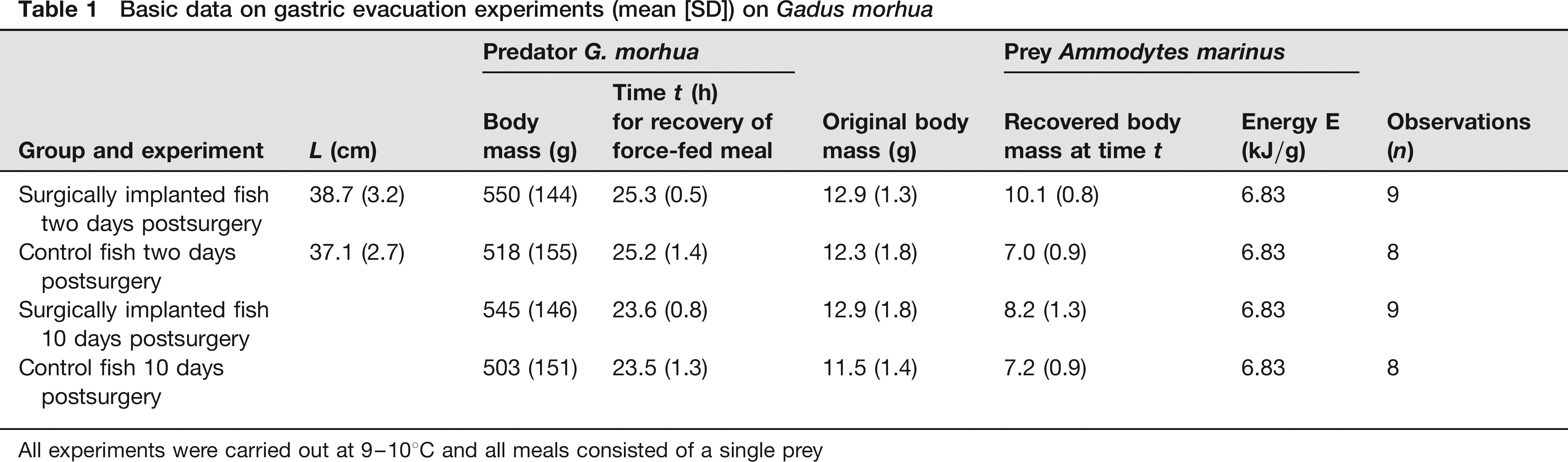
All experiments were carried out at 9–10°C and all meals consisted of a single prey
Assuming a G. morhua to operate with its individual value, the basic rate parameter ρLTE can be considered the outcome of a normally distributed stochastic variable. 13 Then, the performance of two groups of G. morhua with regard to GER can be compared statistically using a t-test to the values of this parameter calculated for each G. morhua according to Equation (5). Before t-tests were performed (SigmaStat version 3.5) the assumptions of normality (Kolmogorov-Smirnov test) and homogeneous variance of data (Bartlett's test) were tested. Significance was accepted at P < 0.05.
Coefficient of variation (CV) of ρLTE is constant and does not depend on the value of this parameter. 15 According to Sokal and Rohlf, 16 this implies that the variances of two values of In ρLTE are identical, and that a test of equality of two values of CV is equivalent to H-test of equality of the variances of the logarithmic transforms of ρLTE. The latter was therefore used here to test for equality of CV obtained from the different treatments of fish. Significance was accepted at P < 0.05.
Results
Short-term recovery (2 days)
Two days postsurgery, evacuation of force-fed sandeel was considerable slower in fish with surgical implants compared with control fish. The calculated GER parameter ρLTE was thus significantly lower (P < 0.001) in implanted G. morhua compared with the control, being respectively 4.2 × 10-4 (1.2 × 10-4) and 9.3 × 10-4 (1.9 × 10-4) (mean [SD]) (Figure 2). Because the effects of fish length were accounted for by ρLTE (Equation (5)), while temperature and energy density of the prey were similar for all animals (Table 1), the observed difference in GERs can be attributed to pure physiological causes. When comparing our data with the value of ρLTE = 14.2 × 10-4 (1.1 × 10-4) obtained in the study by Andersen 14 on G. morhua feeding voluntarily on A, tobianus (i.e. involving no handling or anaesthesia priory to recovery of the stomach contents), the present GERs only constituted approximately one-third (fish with implants) and two-thirds (control) of this rate (Figure 2), i.e. both being significantly lower (P < 0.001). This difference was also reflected in that on average only 21% and 42% (instrumented and control fish, respectively) of the meals had been evacuated 25 h post-force-feeding, which is inefficient compared with the prediction 14 that 62% of the meal should be evacuated.
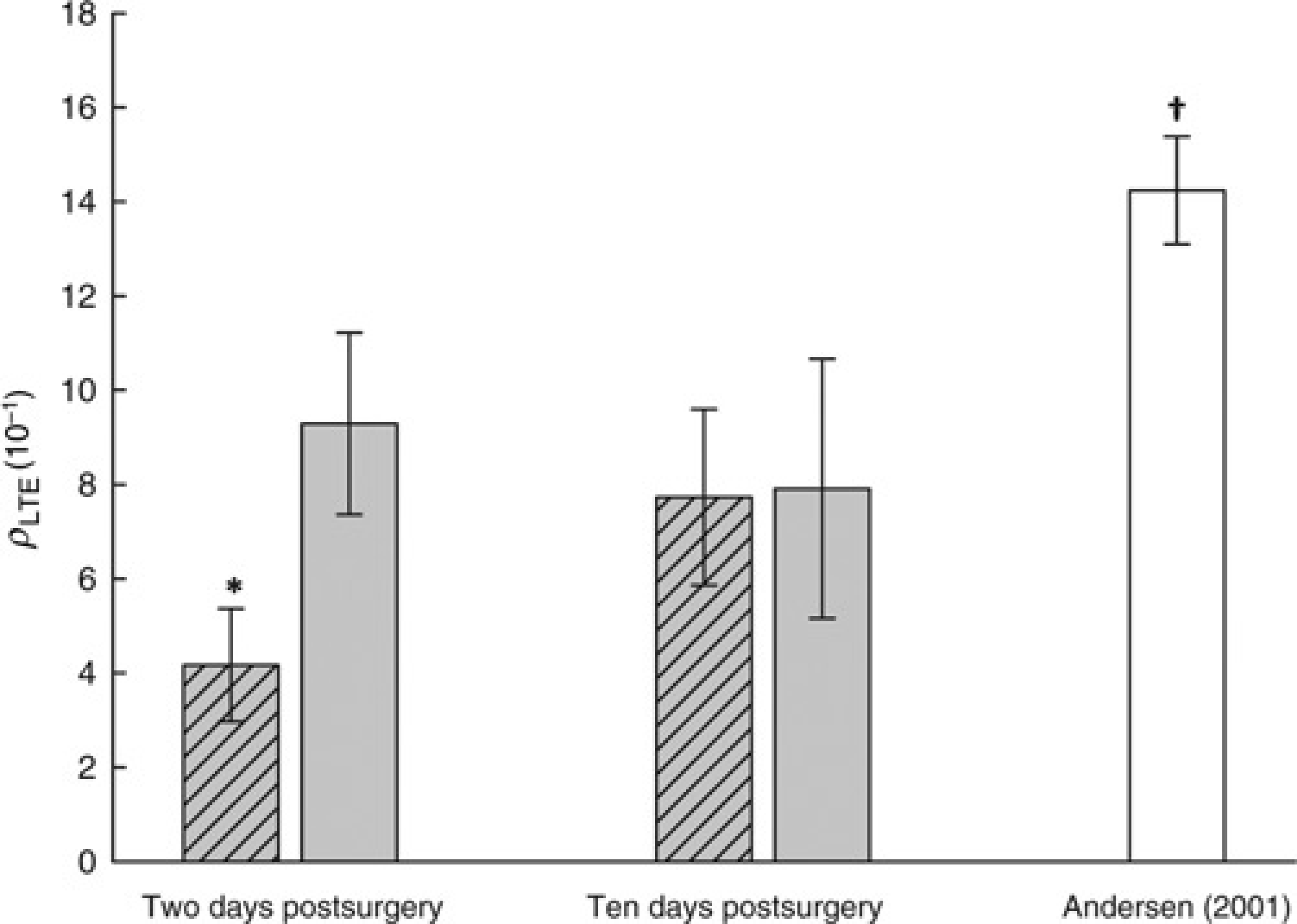
Comparison of the basic rate parameter rho (ρLTE) two and 10 days postsurgery for force-fed surgically implanted (hatched, n = 9) and control (grey, n = 8) Gadus morhua and with estimates from non-operated voluntarily feeding conspecifics (white). 14 Data are presented as mean (SD), *indicates a significant difference (P < 0.05) between the force-fed groups two days postsurgery, †indicates significant difference (P < 0.05) between non-operated voluntarily feeding fish and all force-feeding trials (i.e. both 2 and 10 days postsurgery)
According to Andersen and Beyer 15 the CV of the basic rate parameter ρLTE is constant (i.e. independent of the estimated value of this rate parameter). CV should therefore be used when comparing the variability of ρLTE in the different groups of G. morhua. For the two groups of force-fed G. morhua, CVs of 0.21 and 0.29 (controls and operated, respectively) were significantly higher (P < 0.002 and P < 0.001, respectively) than the value of 0.08 obtained from G. morhua that fed voluntarily 14 (Table 2).
Coefficient of variation (CV) of the basic rate parameter ρLTE obtained from G. morhua fed an A. marinus
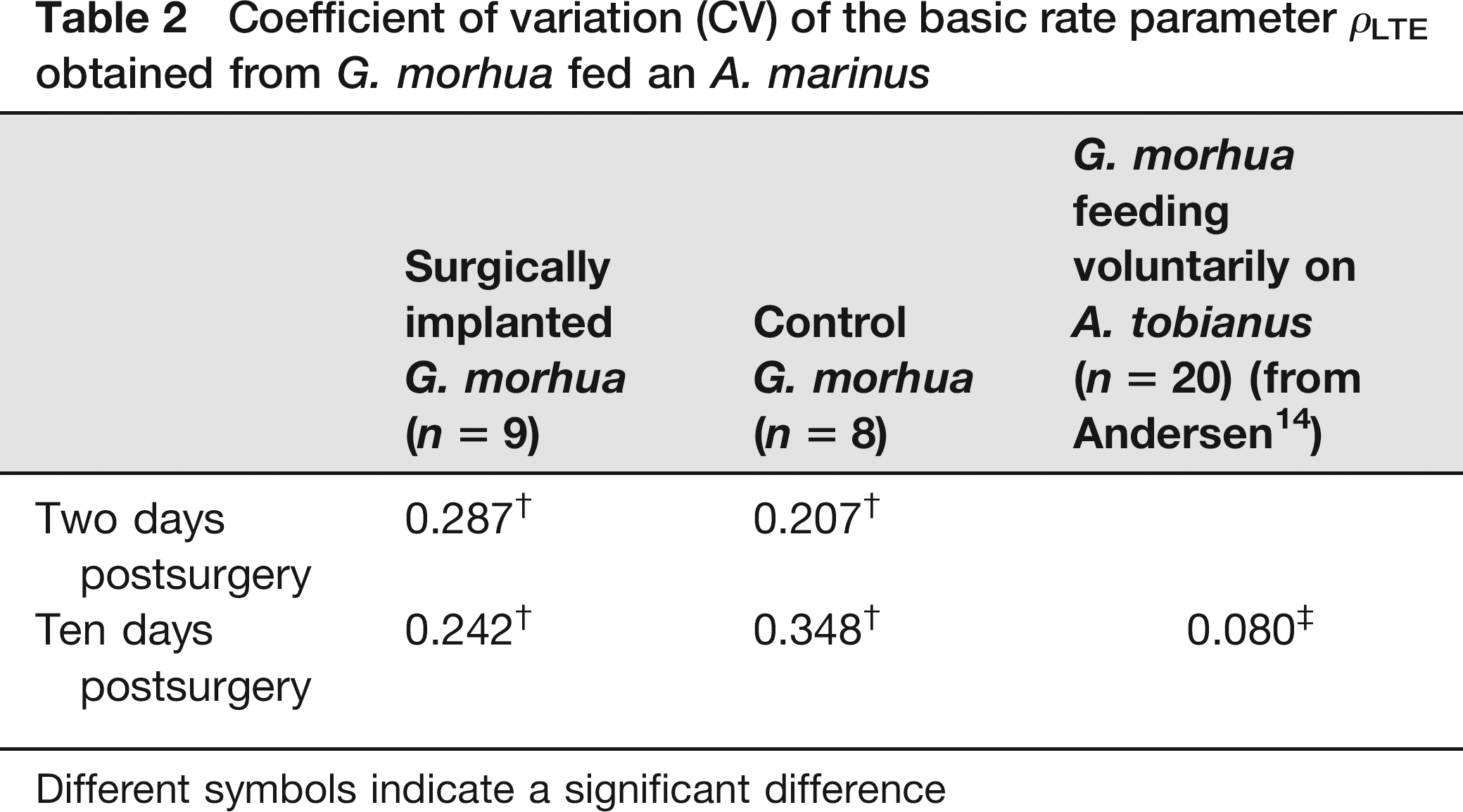
Different symbols indicate a significant difference
Prolonged recovery (10 days)
Ten days postsurgery, there was no significant difference (P = 0.878) between the rate at which animals with implants evacuated their meal compared with control fish (Figure 2). These estimates of ρLTE were however still significantly lower (7.7 × 10-4 [1.9 × 10-4] for implanted and 7.9 × 10-4 [2.8 × 10-4] for controls) than the value of 14.2 × 10-4 (1.1 × 10-4) obtained from G. morhua feeding voluntarily on Ammodytes spp. (P < 0.001). 14 Again, as described above for G. morhua two days postrecovery, the CVs in both of our groups of fish were significantly higher (P < 0.001), being 0.24 in surgically implanted fish and 0.35 in the control group (Table 2) compared with the value of 0.08 obtained from G. morhua that fed voluntarily. In contrast to the improved evacuation ability of the surgically implanted G. morhua following prolonged recovery, the percentage of food that had been evacuated by control G. morhua at c. 23 h post-force-feeding (36%) was comparable both to the week earlier (43% c. 25 h post-force-feeding after 2 days of recovery) and to the value for the surgically instrumented fish (36% c. 23 h post-force-feeding).
Discussion
The present study demonstrates that following two days of postsurgical recovery after introduction of a dummy implant into the body cavity, G. morhua is not capable of evacuating a meal at a rate similar to non-operated fish. By using ρLTE (Equation (5)) to compare gastric performance between groups of fish we have accounted for the effects of fish length, while temperature and energy density of the prey were similar for all animals (Table 1). Our results thus suggest that the observed difference in GER can be attributed to pure physiological causes derived either from surgery, postsurgical stress and/or the presence of the implant. But following 10 days of recovery the suppressing effects on gastric performance caused by surgical implantation was gone. Thus at this stage the presence of neither the implant per se nor the surgery was limiting the GER. Reduced postsurgical gastric performance has been reported previously. Twenty-four to 36 h subsequent to invasive surgery, sea bass (Dicentrarchus labrax) L. showed a significantly higher gastric evacuation time (GET) (the relationship between GET and GER can be deduced from Equation (3)) compared with controls, with stomach contents still above 60% of the initial 24 h post-force-feeding, whereas control animals at this time had less than one-third of the meal remaining in the stomach. 17 This is thus in accordance with our results which show that GER had not stabilized in G. morhua two days postsurgery.
A factor that may affect the recovery and even long-term function is the effect on buoyancy. A fish with an implant has to counteract the downward force exerted by the added mass which can be done by secretion of gases into the swimbladder.18,19 With the present implant in the body cavity, a fish, irrespective of its size, would have to increase its swimbladder volume (5% of the total volume in G. morhua 18 ) with 14.2 cm3 of air to become neutrally buoyant. In the smallest G. morhua, this implies an almost doubling of swimbladder volume, which may approach the limit for their capabilities. 20 Swimbladder adjustment is a low-cost solution but is slow (up to a day or 2),21,22 meanwhile compensation must occur through active swimming which is energetically expensive.19,23 Such excess energy use will reduce the aerobic scope available for other processes 24 and this may be part of the explanation for the lower gastric performance observed two days postsurgery. Furthermore, active swimming may result in blood being shunted away from the stomach region to prioritize oxygen delivery to the working muscle.25,26
The use of biotelemetry devices allows experimenters to provide their animals with long recovery periods to facilitate complete recovery from instrumentation procedures. Nonetheless, for biotelemetry systems to hold their true/full potential it is imperative that the fish is capable of dealing with not only the effects arising from anaesthesia, handling and surgical intervention but also any potential disadvantages related to carrying the entire recording/transmitting unit. Taken together, our results show that a potential for returning to presurgical levels indeed exists, as GER was stabilized 10 days postsurgery in fish carrying the implant in the body cavity. Thus the presence of the implant per se did not seem to affect the average gastric performance at this point. The fish in the present study weighed between 290 and 815 g resulting in transmitter:fish mass ratios from 2.3% to 6.5%. It has been suggested that the mass of telemetry tags should not exceed 2% of body mass.24,27 Several experiments (including experiments on cod) using tags larger than this have however found no significant effect on the swimming performance.28,29 We found no correlation between GER and body mass indicating that cod can recover from carrying tags that represent up to 6.5% of their body mass. Along the same lines results from salmonids on recovery of swimming performance following surgical implantation of telemetry devices have shown that Juvenile Chinook salmon Oncorhynchus tshawytscha (Walbaum) had a significantly lower critical swimming speed one day postsurgery compared with controls, whereas full recovery was accomplished after 21 days (in-between evaluations are lacking). 21 Although full recovery was not established, the swimming performance of tagged juvenile Salmo salar L. had improved seven days post-tagging at termination of the experiment. 29 Suggestively, the fish may either have recovered in the long term, or the tags, representing on average 8.5% of the fish mass, may have been too heavy or have reduced the mobility of the fish.
When comparing the variability in gastric performance between different groups of G. morhua, the CV of the basic rate parameter ρLTE should be used (for further details on this see Materials and methods). The CV value of 0.080 estimated from the voluntarily feeding fish by Andersen 14 (Table 2) compared well with the estimate of 0.098 obtained from a variety of predatory gadoids and their fish prey. 15 This variation probably reflects the interindividual variation in gastric performance. Substantially higher values of CV were obtained from both groups of force-fed G. morhua, which may then be explained by an additional variability due to the suppressing effects of handling, anaesthetization and force-feeding on GER to which the individual fish responds differently. An alike large variability has been observed in O. mykiss; the lag phase from force feeding until the stomach started to empty varied between zero and 5 h.30 As anticipated, for the present G. morhua this furthermore resulted in on average 30% lower values of ρLTE from control fish compared with the voluntarily feeding G. morhua in the study by Andersen. 14 Studies on cod using intragastric transmitters have shown that if the fish voluntarily ingest the baited transmitter (in this case a transmitter wrapped in a fillet of herring) high food consumption rates are maintained in the days subsequent to tagging, in contrast to fish tagged by forced insertion (involving handling and anaesthesia) where food intake was notably lower for up to 15 days post-tagging. 31 This indicates that appetite prevails despite the presence of a transmitter as long as handling and anaesthesia are avoided.
To omit interference from anaesthesia and handling in future studies, one important unanswered question to investigate is how long it takes for instrumented fish to commence voluntarily feeding following the surgical procedure.
Although the primary intention of this study was not to focus on wound healing, this is an important matter, not only for ethical considerations but also because biotelemetric methods enable long-term measurements where open incisions may facilitate internal infections and/or cause changed behaviour and performance thus inflicting data invalidity. Two days postsurgery the surgically implanted fish had no or only slight signs of inflammatory reaction around the incisions. The exception was two fish, one in which the larger wound had opened, and the other where it gapped at one end. These animals were instantly euthanized and data omitted from the analysis. Ten days postsurgery an inflammatory response (redness and slight swelling) was noted around all the larger incision made alongside the basibranchial bone. These observations are in accordance with a previous study in which G. morhua (kept at comparable temperatures of 9.5–14°C) were surgically implanted with dummy transmitters into the body cavity via incision along the linea alba. 32 In these animals inflammatory responses begun 5–7 days postsurgery and subsided for 4–8 days with complete wound healing after a total of 24–34 days. Obviously, the time course of, and how to secure proper wound healing following surgical implantation demand further attention.
In summary, two days of postsurgical recovery will not stabilize GER in G. morhua, but 10 days will, when using a standard force-feeding protocol. The results indicate that at 10 days postsurgery, the presence of a dual channel dummy telemetric implant per se did not affect GER but that the effects observed two days after instrumentation are due to surgery, postsurgical stress and/or the presence of the implant. Biotelemetry has the welfare advantages of allowing long recovery periods and avoiding the unnecessary stress arising from handling and confinement, altogether improving quality of data. We believe our results have left a promising future for implementation of fully implantable biotelemetry systems in fish.