Abstract
Blood sampling from awake non-human primates (NHPs) is classically performed under constraint in the cephalic or saphenous vein. It is a challenging, potentially harmful and stressful procedure which may lead to biased results and raises ethical concerns. Laboratory NHPs undergo a head-restrained procedure allowing for a safer procedure of collecting blood from their ears. Using regular capillary blood collection devices 500 µL of blood can be easily withdrawn per puncture point, which is sufficient for performing most of the usual modern biological assays. This procedure has been validated by measuring total proteins, cortisol and vasopressin concentrations from concomitant blood samples taken from the saphenous vein and the ear capillary vessels of macaques (n = 16). We observed strong correlations between the blood concentrations of total proteins, cortisol and vasopressin (r = 0.72, r = 0.63, r = 0.83, respectively; all P values <0.01) taken from the saphenous vein and from the ear capillary. There were no significant differences between blood concentrations taken from the saphenous vein and the ear capillary. Our alternative to the classical blood collection procedure is harmless and can be routinely performed, which can therefore improve scientific results while increasing animal welfare in accordance with the 3R (replacement, reduction and refinement) principles.
When it comes to non-human primates (NHPs), researchers face ethical concerns which sometimes require a shift of experimental strategy. 1 Classical methods of blood sampling in NHPs usually imply challenging and stressful procedures which increase the risk of injury for both the animals and the experimenters. In addition, extensive handling of the primates by the experimenter requires habituation. This trust relationship might be altered by this potentially harmful sampling procedure, making behavioural training harder and longer. In addition, the behavioural and physiological consequences of stress can induce potential scientific misinterpretation (as reviewed by Reinhardt 2 ).
Despite a recent growth of interest in microsampling methods in rodents, 3 no comparable alternative blood sampling method has been reported for NHPs. We propose here an original microsampling method from the ear capillary of head-restrained NHPs. Since blood contains biomarkers that provide insights into brain functioning, 4 the need for a convenient and ethical blood sampling procedure for NHPs used in neuroscience research is thus important. To validate our sampling method, we assessed the concentrations of plasma total proteins and two hormones (cortisol and vasopressin) in samples taken simultaneously from the ear capillary and the saphenous vein. Our hypothesis is that blood collected from the ear capillary will give similar information to blood collected from the saphenous vein.
Materials and methods
Animals
This study was approved by our local animal experimentation ethics committee (CELYNE) and used experimental procedures complying with the recommendations of the local authorities on animal care (Direction Départementale des Services Vétérinaires, Lyon, France) and the European Community standards for the care and use of laboratory animals. All animals were individually or socially housed at the Centre de Neuroscience Cognitive in Bron, France. Subjects were 16 macaques obtained from SILABE (Niederhausbergen, France) (14 males and 2 females, 9 mulatta and 7 fascicularis, mean age = 6.1 years, standard deviation [SD] = 2.9, mean weight = 7.4 kg, SD = 1.7).
Blood sampling procedures
We took advantage of a veterinary control procedure that required the animals to be anaesthetized (ketamine 10 mg/kg) in the morning to collect concomitant blood samples from the ear capillary and the saphenous vein.
Blood collection from the saphenous vein was performed using EDTA tubes and a 23 G needle.
The ear capillary blood sampling procedure on vigilant animals is described in Figure 1. Once the subjects had had their head fixed using a classic head restraint system, they were first familiarized with ear manipulation. Prior to the puncture, the ear was cleaned and shaved in order to avoid blood contaminations. The ideal locations for collecting blood are around the lobule vein and at the extremity of the ear (see Figure 1a). However, it is also possible to collect blood in other areas of the external ear. Using a micropuncture system (Safety-Lancet Super, blade of 1.5 mm; Sarstedt, Nümbrecht, Germany), a small puncture was made at the selected location which was adapted to each monkey (Figure 1b). Immediately after the puncture, a drop of blood appeared and started to ooze out (Figure 1c). The first drop of blood was systematically discarded. Blood was collected using a Microvette® 300 (Sarstedt), with the tip inclined at 45° downwards to the drop of blood to insure optimal collection (Figure 1d). If the blood flow stopped before the desired volume was collected, massaging around the puncture site was performed to stimulate blood flow. Depending on the puncture site, between 100 µL and more than 500 µL of blood can be withdrawn from each puncture point. Once enough blood was collected, a one-minute compression of the puncture site was performed to allow proper coagulation. The whole process required only one experimenter.
Ear capillary blood sampling procedure. (a) Optimal puncture site is delimited by a dashed line. (b) Puncture performed using a Safety-Lancet. (c) A successful puncture. (d) Blood collection using a Microvette®.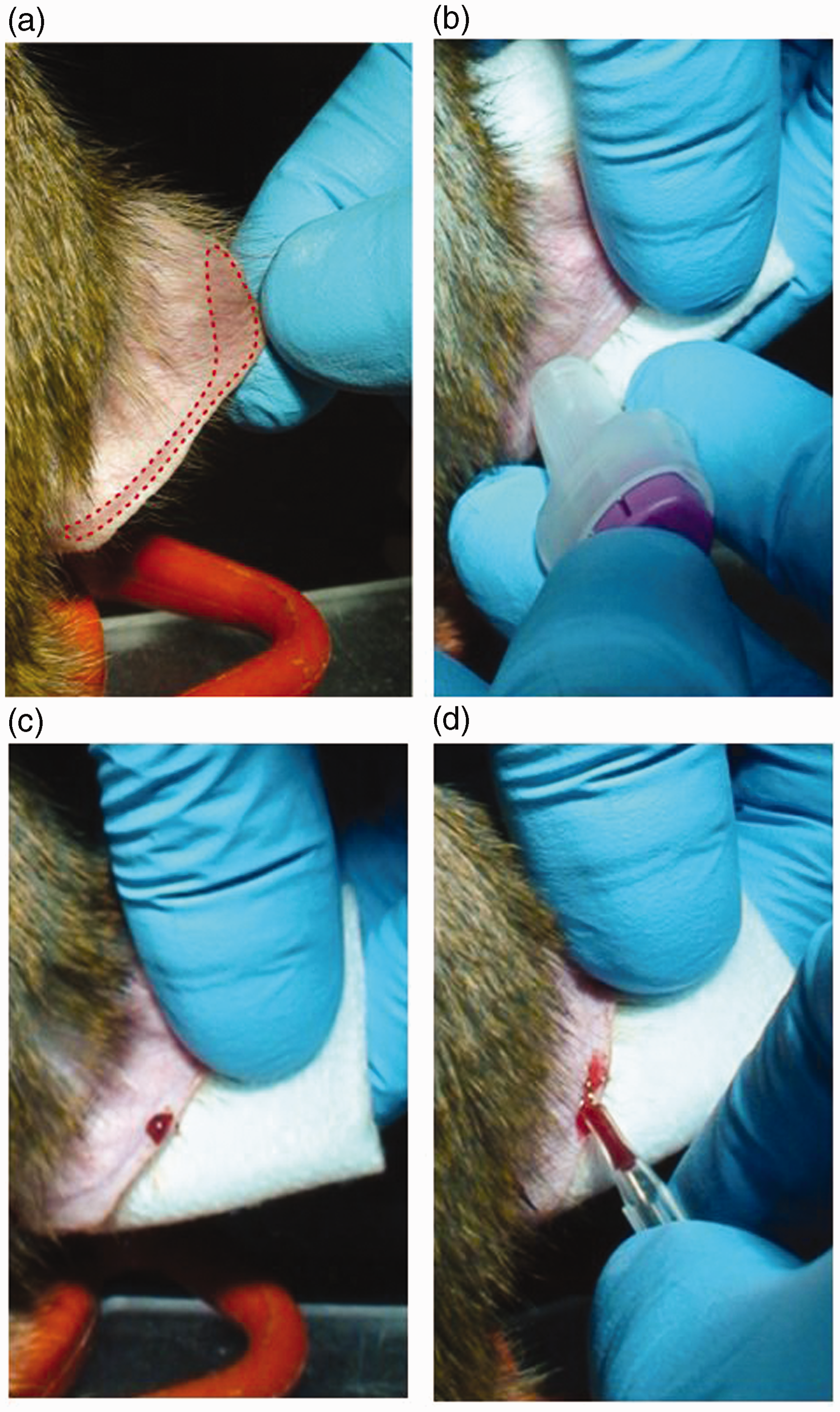
Assays
All blood samples were immediately centrifuged at 4℃ for 10 min at 2000
Results
All P values are corrected for multiple comparisons (Bonferroni). No significant differences were found between venous and capillary concentrations of protein, vasopressin and cortisol (Wilcoxon signed rank test, all P values >0.1).
Concentrations of capillary proteins were correlated with concentrations of venous proteins (r = 0.73, P = 0.008; Figure 2a). To correct for non-Gaussian distribution, cortisol and vasopressin concentration values were log-transformed, and correlations were found between venous concentrations and capillary concentrations (r = 0.84, P < 0.01 and r = 0.65, P = 0.021, respectively; Figures 2b and c).
Correlations between venous and capillary concentrations. (a) Total proteins. (b) Cortisol. (c) Vasopressin. AVP = vasopressin.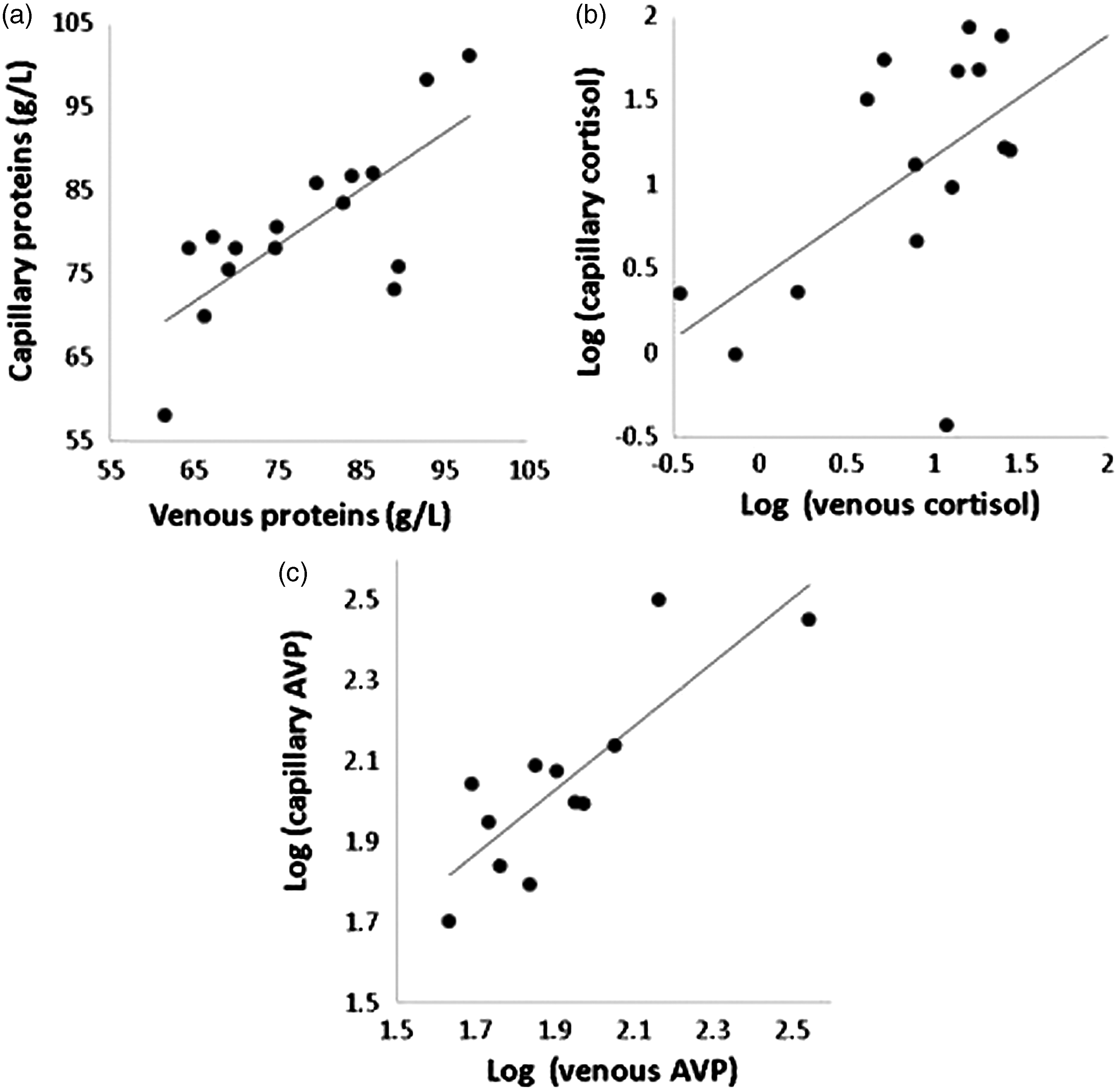
Discussion
The present study proposes ear capillary blood sampling as a suitable and harmless way of collecting blood from a head-restrained NHP.
Our physiological analysis validates this method as a reliable technique for measuring blood concentrations of meaningful biomarkers such as cortisol or vasopressin. Indeed, all measured biomarkers were strongly correlated between ear capillary samples and venous samples. Moreover, there were no significant differences between blood concentrations taken from the saphenous vein and the ear capillary.
This blood sampling technique requires minimal animal handling and is unlikely to produce any significant discomfort. Indeed, even though no controlled objective measures of pain were performed, we did not notice any changes in facial mimicry or general activity of the macaques while being sampled. Importantly, the ear lobe puncture has been shown to be less painful than classical arterial puncture in humans. 5 In addition, our sampling method was performed in the animal facility for more than one year and we did not notice any infections of the puncture site. Additionally, as neuroscience experiments may require water restriction, we propose that such sampling methods should also be routinely used to assess the hydration states of the animals. 6
The main limit of our procedure is the lower amount of blood that can be collected compared with classical venepuncture. However, recent assay procedures require smaller volumes (usually around 200 µL of plasma), therefore, depending on the number of molecules measured, one puncture site should generally provide enough blood for further biological analysis.
In conclusion, ear capillary blood sampling is a good alternative when behavioural training is precluded. It can represent a routine procedure in neuroscience research as it provides unaltered data, increases the well-being and safety of both animals and experimenters and performing it requires only one experimenter.
Footnotes
Funding
This work was supported by the LABEX (ANR-11-IDEX-0007) of University de Lyon within the program “Investissement d'Avenir” and by grants from the Rhône-Alpes Region and from the Agence Nationale de la Recherche (ANR-2010-BLAN- 1431-01, ANR-13-BSV4-0010-01) to JRD. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest statement
The authors have no conflicts of interest to declare.
