Abstract
The vast majority of laboratory studies on animals have focused on ventricular fibrillation (VF) and not on cardiac arrest (CA) resulting from asphyxia. The aim of this study was to develop a clinically relevant animal model in Landrace/Large-White swine of asphyxial CA resuscitated using the European Resuscitation Council guidelines. Survival and 24 h neurological outcome in terms of functional deficit were also evaluated. Asphyxial arrest was induced by clamping the endotracheal tube (ETT) in 10 Landrace/Large-White piglets. After 4 min of untreated arrest, resuscitation was initiated by unclamping the ETT, 100% oxygen mechanical ventilation, 2 min chest compressions and epinephrine administration. Advanced Life Support algorithm was followed. In case of restoration of spontaneous circulation, the animals were supported for one hour and then observed for 23 h. Coronary perfusion pressure was significantly higher in surviving animals (P < 0.001) during cardiopulmonary resuscitation. End-tidal CO2 was significantly higher in the animals that survived than in non-surviving animals (P = 0.001). All of the animals were severely neurologically impaired 24 h after CA. This refined model of asphyxia CA is easily reproducible and may be used for pharmacological studies in CA.
The primary cause of a cardiac arrest (CA) in adults is of a cardiac origin. 1 Paediatric CA is usually asphyxial in origin and can be caused either by airway obstruction due to choking and drowning or by a depressed respiratory drive. 2–4 Clinical trials are scarce in asphyxial CA. 5 This is due to the retrospective nature of study designs and inconsistent definition of CA and cardiopulmonary resuscitation (CPR) in asphyxial CA. Furthermore, data regarding the pathophysiology of asphyxial CA are limited due to the fact that the vast majority of laboratory studies on animals have focused on primary cardiac events such as ventricular fibrillation (VF) and not on CA resulting from underlying asphyxial events. 6
The aim of this study was to develop a refined animal model of out-of-hospital asphyxial CA in Landrace/Large-White swine resuscitated using the European Resuscitation Council guidelines. 7 Survival and 24 h neurological outcome in terms of functional deficit were also evaluated.
Materials and methods
Animals
The experimental protocol was approved by the General Directorate of Veterinary Services (permit no. K/3037/22-4-2008), according to Greek legislation regarding ethical and experimental procedures (Presidential Decree 160/1991, in compliance with the EEC Directive 86/609 and Law 2015/1992 and in conformance with the European Convention ‘for the protection of vertebrate animals used for experimental or other scientific purposes 123/1986’). All experiments were performed on 10 healthy 12–15-week-old Landrace/Large-White male pigs with an average weight of 20 kg. They all came from the same breeder (Validakis, Athens, Greece) and were of conventional microbiological status. They were housed in single cages and the area of the cage was 1 m2. The conditions were climate-controlled with a 12 h light/dark cycle. The temperature was 22 ± 2°C, with a relative humidity of 55% and with 15 complete changes of filtered air per hour. The animals were fed a commercial food (Biozokat, Ekaterinis-Larissis, Greece) and were acclimatized to the laboratory conditions for a one-week period prior to the experiment. In order to ensure that the animals were healthy, all animals included in this study were clinically examined by a veterinarian. White blood cell count with differential counts, blood biochemistry, C-reactive protein and a chest X-ray were performed two days before experimentation. Animals with any suspicion of underlying pathologies were returned to the breeder.
Anaesthetic protocol
After one night's fasting and access only to water, the piglets underwent intramuscular sedation with 0.5 mg/kg midazolam (Dormicum® 5 mg/mL; Hoffman-La Roche, Athens, Greece), 10 mg/kg hydrochloride ketamine (Ketamin-Actavis 10 mg/mL; Actavis Group, Athens, Greece) and 0.05 mg/kg atropine (Atropine® 1 mg/mL; Demo, Athens, Greece). Two auricular veins were cannulated and connected to a 0.9% N/S infusion. Induction to anaesthesia followed with 2 mg/kg propofol 1% (Diprivan 1%; Abbott, Athens, Greece) and 2 μg/kg fentanyl (Fentanyl®; Janssen Pharmaceutical, Athens, Greece) intravenously. The pigs were intubated with an endotracheal tube (ETT) with a cuff (Portex, 5 mm ID, Smiths Medical, St Paul, MN, USA). The correct placement of the ETT was confirmed by auscultation of both lungs. Anaesthesia was maintained by continuous intravenous infusion of 5 mg/kg/h propofol, 20 μg/kg/h remifentanyl (Ultiva®; GlaxoSmithKline, Athens, Greece) and muscle relaxation with intravenous infusion of 0.3 mg/kg/h rocuronium (Esmeron®; Schering-Plough, Alimos, Greece). The pigs were mechanically ventilated by a volume-controlled ventilator (ventiPac Sims pneuPac, Luton, UK) with fractional inspired oxygen (FiO2) 21%. The tidal volume was initially set at 15 mL/kg. The respiratory frequency was regulated so that the end-tidal carbon dioxide (ETCO2) would be 35–40 mmHg. ETCO2 was monitored with a capnograph (Nihon Kohden Corp, Bergamo, Italy), which was connected at the end of the ETT. A pulse oximeter (Vet/Ox Plus 4700; Papapostolou, Athens, Greece) was also used for continuous recording of peripheral tissue oxygenation. The electrocardiogram was recorded continuously (Mennen Medical Envoy, Athens, Greece) using leads I, II, III, AVR, AVL, AVF and V5.
The right internal jugular vein was surgically prepared and a Swan-Ganz catheter (Opticath 5.5F, 75 cm; Abbott Ladakis, Athens, Greece) was inserted via the vein into the right atrium for continuous recording of right atrial pressure. The internal carotid artery was dissected and a fluid-filled catheter (model 6523, USCI CR; Bart Inc, Athens, Greece) was inserted into the thoracic aorta for continuous recording of systolic and diastolic pressures. The pigs were administered 750 mg cefuroxime intravenously and received diclofenac (Voltaren®; Novartis Pharma, Athens, Greece) and paracetamol (Uni Pharma SA, Pharmaceutical Laboratories, Kifissia, Greece) every 12 h. Coronary perfusion pressure (CPP) was calculated as the difference between minimal aortic diastolic and right atrial pressure that was simultaneously recorded.
Following stabilization baseline data were collected and at the end of normal exhalation. Asphyxial CA was induced by ETT clamping. CA was defined as mean arterial pressure (MAP) of less than 10 mmHg and by the absence of aortic pulsations. 8 The piglets were left untreated for 4 min. The asphyxial time interval was defined as the period between the clamping of the ETT and the commencement of resuscitation efforts. Airway obstruction in animals triggered responses through the nervous system and activated the hypothalamic–pituitary–adrenal axis. Full muscle paralysis prevented any form of gasping that would be anticipated because of hypoxic injury. Animals exhibited cyanosis of the skin on the extremities within minutes. They developed purple discoloration of the skin on the ventral abdomen and extremities. No animal had posthypoxic seizure.
Four minutes following untreated CA, the ETT was unclamped and resuscitation efforts started with mechanical ventilation, 2 min chest compressions and administration of 0.02 mg/kg epinephrine. Mechanical ventilation was provided with 100% oxygen and adjusted to obtain ETCO2 of 35–40 mmHg. Chest compressions were delivered by a mechanical chest compressor (LUCAS Chest Compression System; Jolife AB, Mantzaris, Greece) providing 100 compressions per minute. Compression depth was equivalent to 30% of chest diameter. Following 2 min of chest compression, advanced life support was provided according to resuscitation guidelines. 7 When VF occurred, defibrillation was attempted with a 4 J/kg monophasic waveform shock (Porta Pac 190, Medical Research Laboratories, Inc, Athens, Greece) and with 12 cm adhesive electrodes.
Endpoint of the experiment was return of spontaneous circulation (ROSC) and asystole or persisting VF after the third defibrillation attempt. ROSC was defined as restoration of spontaneous rhythm with a MAP of 50 mmHg for a minimum of 10 min. After ROSC, mechanical ventilation was maintained with 100% oxygen and adjusted to maintain ETCO2 of 35–40 mmHg. The pigs that restored spontaneous circulation were monitored for 60 min, while anaesthesia was maintained.
Afterwards the blood vessels were ligated to prevent bleeding and the wounds were sutured. The ventilator was switched off and the animals were ventilated by manual squeezing of the reservoir bag with a 100% oxygen (FiO2 100%). After the first spontaneous swallowing reflex was detected, 0.2 mg/kg atropine was administered, followed by 0.05 mg/kg neostigmine (Neostigmine®; Cooper, Athens, Greece) intravenously. The tracheal tube was connected to a T-tube for oxygen administration when spontaneous breathings began. The animals were extubated when adequate inspiration depth was ascertained, the cranial and upper way reflexes were active and the SpO2 measurement was more than 97%. Thereafter oxygen was given by face mask to keep the SpO2 above 97% and vital signs were monitored through recovery for one hour. Monitoring included leads II AVL electrocardiogram recordings, heart rate (HR) and non-invasive arterial blood pressure. Also peripheral tissue oxygenation was recorded through a pulse oximeter. At that point each successful resuscitated animal was returned to its cage whereas pigs that failed to wean off mechanical ventilation were euthanized and autopsy was performed. Surviving animals were kept in their cage for 23 h. Animals had free access to water and food.
Twenty-four hours following CA the animals were sedated intravenously with 10 mg/kg ketamine and 0.5 mg/kg midazolam and euthanized by intravenous solution of 2 g pentobarbital (Nembutal® Sodium Solution, Lundbeck Inc, Deerfield, IL, USA). Then, necropsy was performed on surviving animals in order to detect internal organ damage and ribcage fractures. Survival and neurological status were evaluated using a previously described neurological alertness score 9 24 h following CA. The experimental protocol of the study is shown in Figure 1.
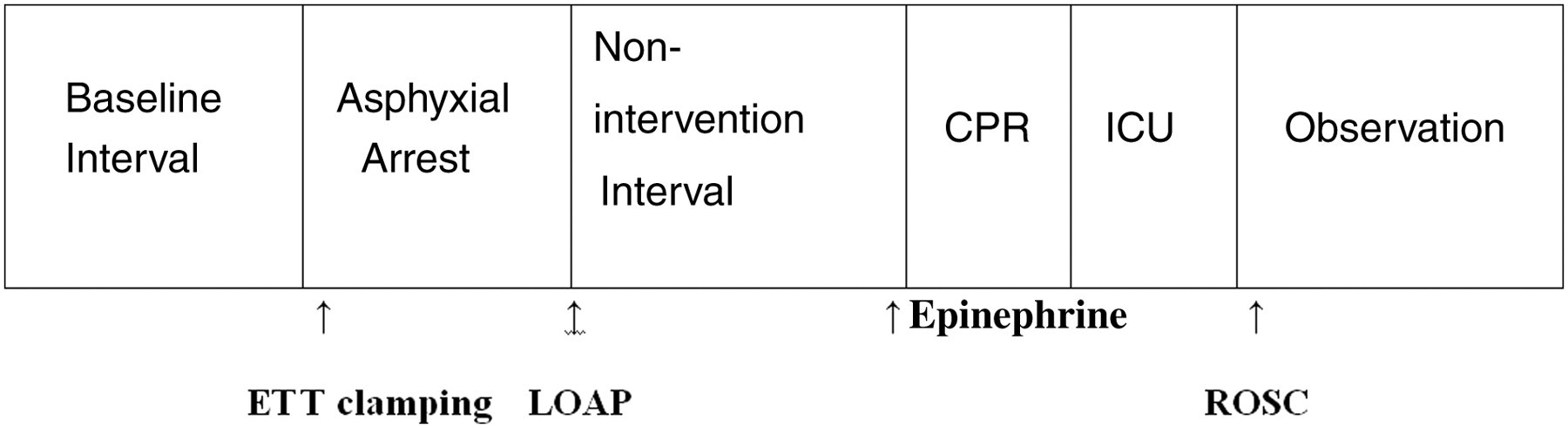
Experimental protocol timeline. ETT: endotracheal tube; LOAP: loss of aortic pulsation; CPR: cardiopulmonary resuscitation; ROSC: return of spontaneous circulation; ICU: intensive care unit
Data are expressed as mean ± standard deviation (SD) for continuous variables unless otherwise noted. Comparability of data was tested using t-test for continuous variables. The Kolmogorov–Smirnov non-parametric test was used for normality analysis of the parameters. Comparisons between time-based measurements on the same animals and differences between surviving and non-surviving animals were analysed with analysis of variance test. In the case where homogeneity of variances of the variables was not met, the Welch test was used. On all tests, P values less than 0.05 were considered to be statistically significant. The SPSS computer software program was used for all analyses (SPSS 17.0; SPSS Inc, Chicago, IL, USA).
Results
All animals were in normal sinus rhythm prior to ETT clamping. Baseline variables did not differ between resuscitated and non-resuscitated animals (Table 1). The mean time from ETT clamping until loss of aortic pulsation was not statistically significant between the two groups (9.67 ± 1.36 versus 9.25 ± 1.50 min, P = 0.661). The mean asphyxia time interval was 13.50 min. The rhythms encountered at loss of aortic pulsation and at 4 min of untreated CA are shown in Table 2.
Baseline variables
NS: not significant; ETCO2: end-tidal CO2
Encountered rhythms in animals at LOAP and at 4 min of untreated CA
LOAP: loss of aortic pulsation, CA: cardiac arrest
In the interval between tracheal clamping and loss of aortic pulsations, the HR in surviving animals was longer than that in the non-surviving animals. The HR in the surviving animals peaked at the 6th min following ETT clamping while in non-surviving animals it peaked at the 5th min. HR then steadily decreased in both groups until loss of aortic pulsation (Figure 2).
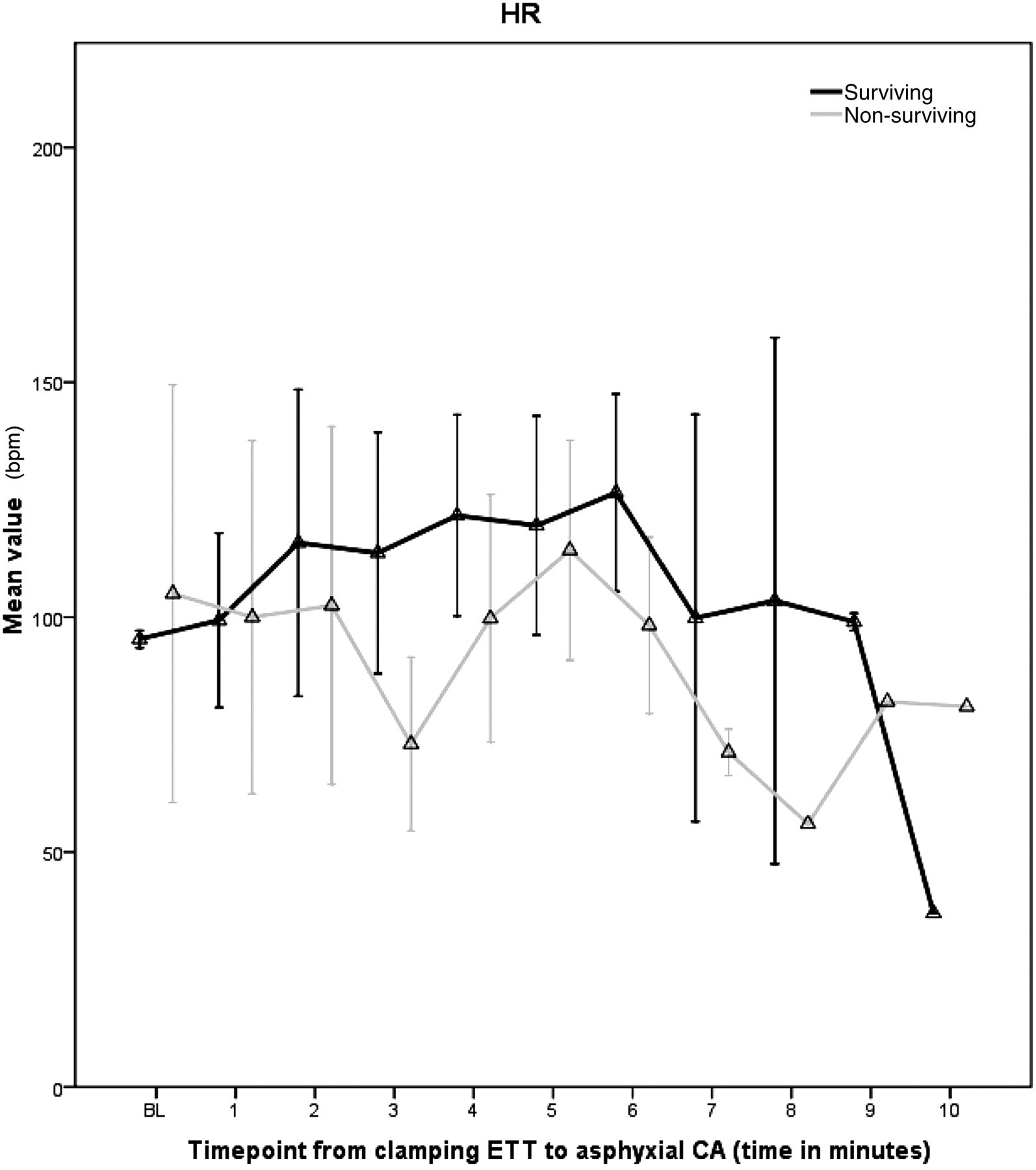
Heart rate (HR) in the time interval between clamping of ETT until loss of aortic pulsation for both groups. CA: cardiac arrest; ETT: endotracheal tube
Three minutes after ETT clamping, MAP in surviving animals was 100 ± 6.48 (P = 0.001), while in non-surviving animals it was 115 ± 4.40 (P = 0.032). By the end of 4 min of untreated CA, MAP decreased from 87.33 ± 5.57 to 10 ± 7.97 mmHg in surviving animals and from 86.75 ± 13.15 to 7 ± 8.08 mmHg in non-surviving animals (P = NS). By the end of the first cycle of CPR the surviving animals achieved MAP of 69.83 ± 7.78 mmHg, while the non-surviving animals achieved 31 ± 8.04 mmHg (P < 0.001). CPP was significantly higher in surviving animals (P < 0.001) at all time points of CPR. CPP fluctuations in time can be seen in Figure 3.
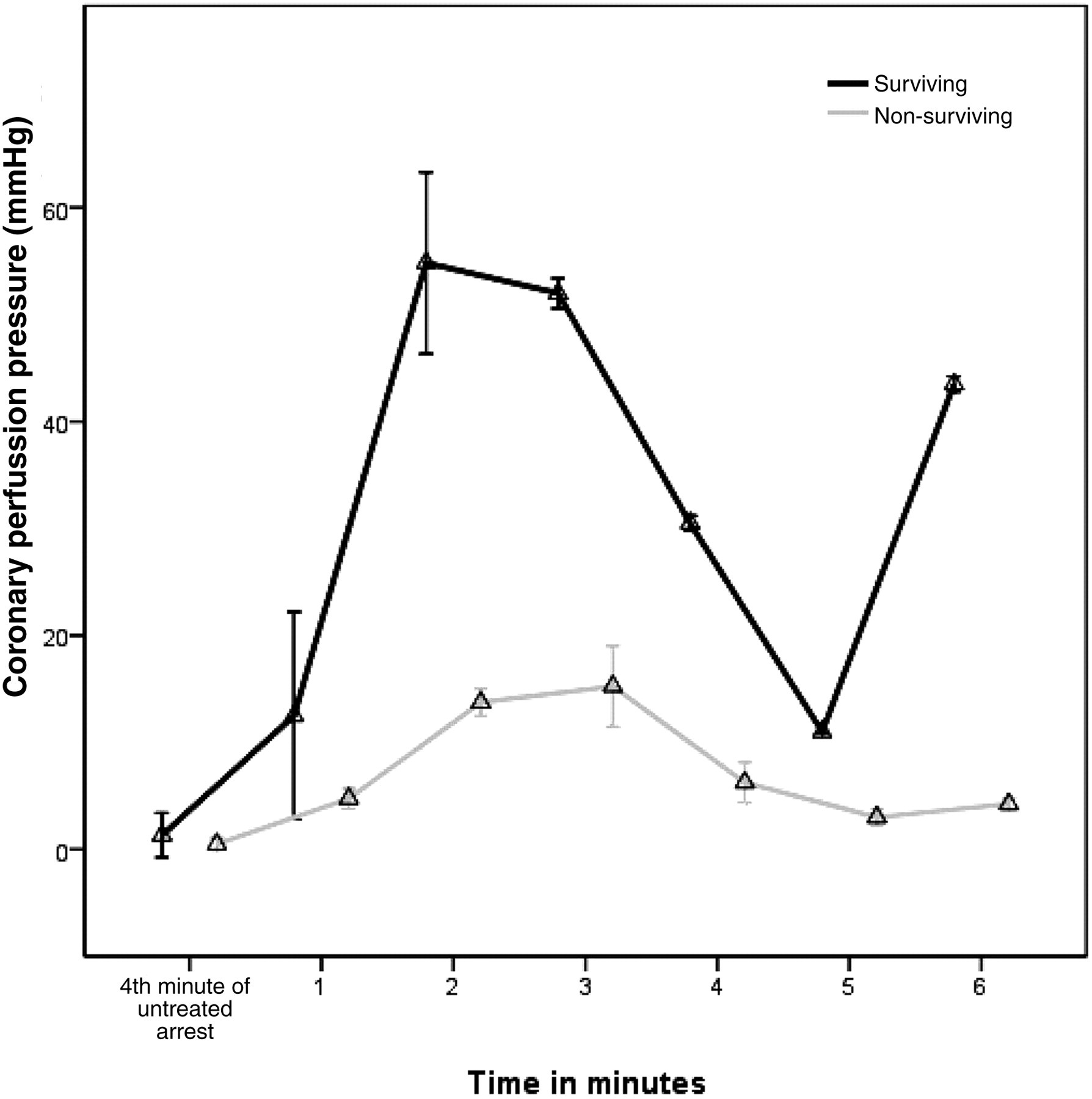
Coronary perfusion pressure fluctuation during asphyxia arrest and CPR. CPR: cardiopulmonary resuscitation (P < 0.001 surviving group versus non-surviving group)
In the first minute of CPR, ETCO2 was 26.17 ± 4.46 mmHg in the surviving animals and 34.00 ± 8.67 mmHg in non-surviving animals (P = 0.17). By the end of the second CPR cycle, ETCO2 changed to 23.50 ± 0.71 mmHg in surviving animals and to 14.25 ± 4.43 mmHg in non-surviving animals (P = 0.022). During the third cycle of CPR in surviving animals it changed to 22.50 ± 0.71 mmHg but only to 14.25 ± 1.26 mmHg in non-surviving animals (P < 0.001). At the end of the third cycle of CPR, there was a statistically significant difference between the two groups (P = 0.001, Figure 4).
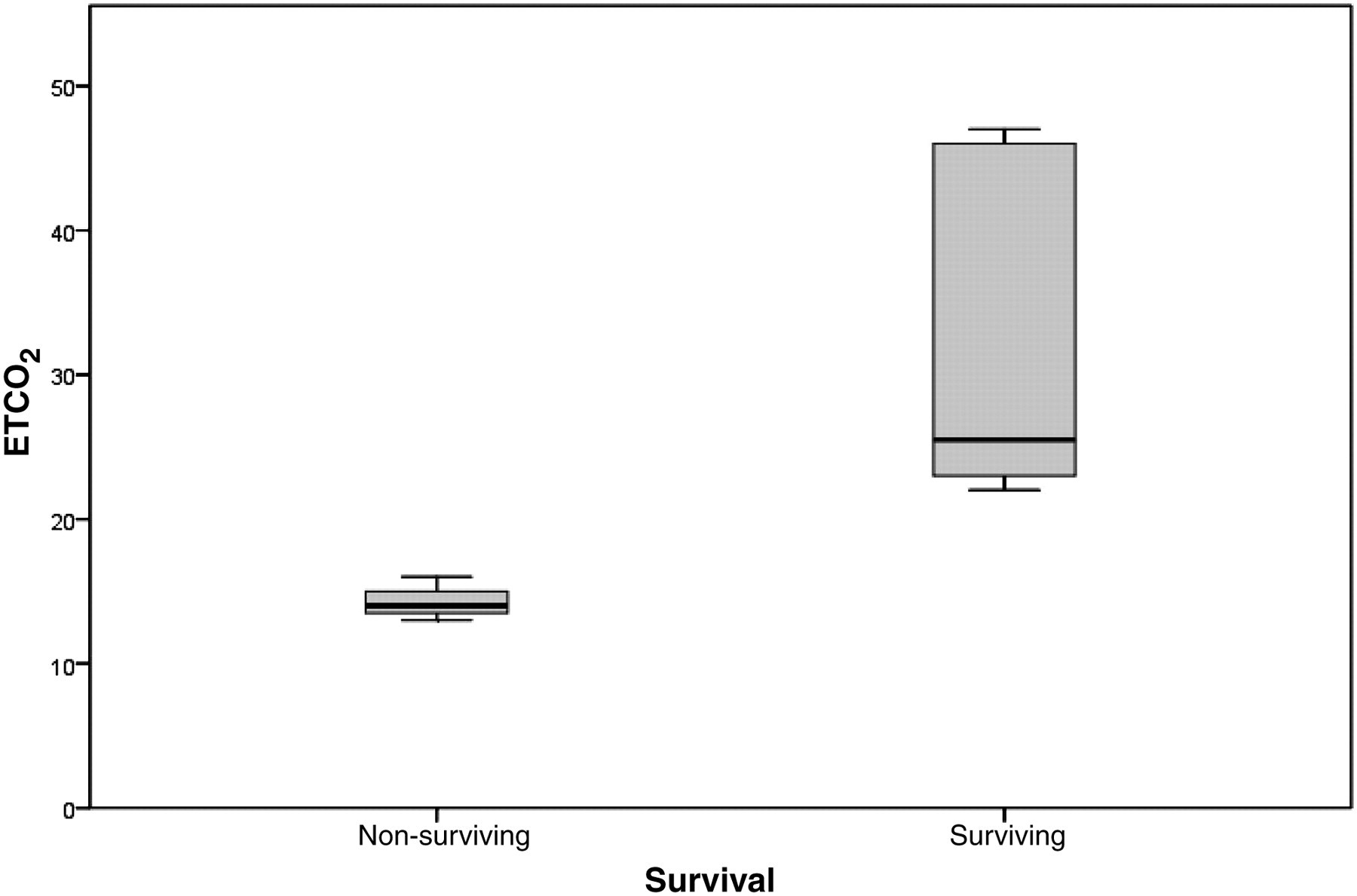
Boxplots of end tidal CO2 (ETCO2) in surviving and non-surviving animals. The shaded area indicates the standard deviation (SD), and the horizontal lines above and below the shaded area represent the maximal and minimal values, respectively
Six out of 10 animals were successfully resuscitated (60%). Two animals were resuscitated after one CPR cycle, two after two cycles, while two animals were resuscitated after three cycles of CPR. All animals that were successfully resuscitated exhibited supraventricular tachycardia, which progressively changed to sinus rhythm in the immediate postresuscitation phase. There was no incidence of recurrent VF. All surviving animals were successfully weaned off the ventilator. Neurological outcome was evaluated as functional deficit. All of them were severely neurologically impaired 24 h after their CA. They were all lying on each side and responded only to painful stimuli. Four animals had a neurological alertness score of 40 and two animals had a score of 50. Necropsy in all of the resuscitated and non-resuscitated animals revealed no injuries of the rib cage or of the intrathoracic organs. In addition, lungs were examined macroscopically and no signs of infection were observed. No haemorrhage in the pericardium and the pleural space were seen. No injuries were detected to the upper airway, heart and thoracic aorta. Finally no haematomas were present in the liver and spleen.
Discussion
The aim of the present study was to develop a refined Landrace/Large-White swine model of out-of-hospital asphyxial arrest, to describe the evolution of haemodynamic and respiratory parameters during and after the asphyxial CA and to evaluate the 24 h neurological outcome.
Out-of-hospital CA in children results in dismal outcomes. ROSC is achieved in approximately 30% of children who suffer out-of-hospital CA, but only 12% of these children survive to hospital discharge. Moreover only 4% of children who sustain out-of-hospital CA have intact long-term neurological outcomes. 10–12 The majority of out-of-hospital paediatric arrests is unwitnessed and has low rates of bystander CPR. 13
This protocol simulates the event of loss of aortic pulsation by clamping the ETT followed by a 4 min interval of asphyxial CA. Resuscitation was accomplished by unclamping the ETT, 100% oxygen mechanical ventilation, chest compressions for 2 min and administration of epinephrine. The Advanced Life Support (ALS) algorithm was followed afterwards according to the guidelines published by the International Consensus on Cardiopulmonary Resuscitation. 7
To simulate an unwitnessed out-of-hospital asphyxial arrest we used a 4 min non-intervention interval before initiation of resuscitation. Although the non-intervention interval is an important experimental variable and affects the outcome, 14 it is not defined and varies in out-of-hospital asphyxial CA. In the present study the asphyxia time interval was 13.50 min representing the average time for the arrival of specialized personnel. Many animal models used in resuscitation research on asphyxia out-of-hospital CA use asphyxia time intervals for less than 10 min of CA prior to arrival of a specialized paramedic team. These models may not address the clinical situation faced by the majority of paediatric CA victims who may have a longer period of collapse. 15–18 Asphyxial arrest was induced by clamping the ETT in the presence of room air ventilation, while the animal had full muscle relaxation. In this way we prevented any form of gasping that would be a severe confounding variable in the asphyxial model.
CPR research depends mostly on the use of animal models that are designed to simulate CA in humans. 19–21 The animal models that have been used in studies are swine, rodents, 22,23 canines, 24,25 primates, 26 rabbits, sheep 27 and cats. 28 Among others, asphyxial arrest in animals such as rhesus monkeys, rabbits and sheep were extensively studied in the 1960s by Dawes and colleagues. 27 The animals that have been used most frequently for asphyxial CA research are rats. The rat asphyxial CA model is an established model for evaluating neurological injury after CA. 29 Canines have also been used as asphyxial CA models, but they are not as close to the thoracic and coronary anatomy of humans as swine are. 30–32
Recently, swine have gained popularity in biomedical research because of certain advantages compared with other animal models. The swine breed has been used extensively for experiments modelling infants and children. The anatomical and physiological parameters of the cardiovascular system of the swine have been defined during experimental studies 33–35 and are comparable with those of humans. 36–39 Swine cardiac electrophysiology and the coronary artery anatomy are more similar to humans. 40 Compared with rodents and canines, swine allow for better preclinical studies on techniques and interventions that take place during the CPR period. The oval chest wall shape of the swine closely resembles that of humans. 6,41,42 Pig's lungs are physiologically and anatomically comparable to those of humans though the right lung has four lobes while the left has two. 43
Landrace/Large-White swine, a farm pig breed, has already been used successfully as a research model for VF 44 and its physiological parameters and haemodynamic measurements are close to humans. 45,46 By contrast, Yucatan miniature and micropig haemodynamics are not as close to humans as Landrace/Large-White swine. 35
The real circumstances of asphyxial CA in humans are difficult to simulate. Many types of asphyxial arrest models have been developed. The most common are those of airway obstruction and of progressive anoxia. Obstruction can be caused by the ETT clamping and the tracheostomy tube disconnection from the mechanical ventilation. 21,22 In a study by Albaghdadi et al., 47 asphyxia was induced by insertion of a rubber stopper into the end of the ETT. A description of an injection of neuromuscular blocking agent for paralysis and apnoea has also been reported. 15 Progressive anoxia can be produced by manipulating the concentrations of inspired gas mixtures. In a study by Solevag et al., progressive asphyxia was induced by adding CO2 in order to achieve a PCO2 > 70 ka. In addition, oxygen was reduced to 8% and the ventilator rate was reduced to 10/min. 48
Following ETT clamping, there was a characteristic increase in HR and MAP in both surviving and non-surviving animals. Then, both HR and MAP steadily decreased until loss of aortic pulsation. This characteristic pattern of mild tachycardia-hypertension–bradycardia-hypotension has already been described in other asphyxial experimental studies. 32,49,50 In the present study we found that surviving animals had a higher HR compared with non-surviving animals. Until now no other study has reported similar findings. It is well known that the autonomic nervous system influences the haemodynamic response to asphyxiation in many ways. 28 Non-surviving animals are more sensitive to the asphyxia insult and therefore unable to increase their HR as much as surviving animals can. Further studies are needed to investigate the HR and ROSC correlation in asphyxial CA and to confirm whether HR could be used as a prognostic factor.
CPP is the difference between minimal aortic diastolic and right atrial pressure that was simultaneously recorded and calculated as such. In this laboratory model of CA, we observed that there is a strong correlation between ROSC and higher levels of CPP. Although CPP has already been studied in the Landrace/Large swine model of VF, 44 there are no data about the evolution of CPP in an asphyxial CA model. In our study, CPP increased during the first CPR cycle and then decreased and increased again during the 6th CPR min. In the VF model, CPP rose for the basic life support (BLS) period and rose further after implementation of the ALS protocol. In previous studies, CPP has been found to be the key determinant for successful defibrillation in humans 50 and various animal models. 51
In the present study, ETCO2 levels decreased from baseline values to lower values during CPR. The very first reading taken after unclamping the ETT was noted to be much higher than subsequent readings taken during CPR, probably reflecting CO2 that had accumulated in the alveoli during asphyxia but before CA. 52 This has already been observed and verified in other studies as well. 53–55 In the experimental asphyxial study by Mananda et al., 39 ETCO2 levels were initially high during CPR, decreased to low levels and increased again at ROSC. This pattern is different from the one that was observed in animal CA that was caused by VF during which ETCO2 decreased to almost zero after the onset of the arrest, then began to increase at the onset of effective CPR, and increased to normal levels at ROSC. 56,57 This was also confirmed by another study where prior to CPR, ETCO2 was higher in an asphyxial arrest model than in a VF/pulseless ventricular tachycardia (VT) CA model. 37
By the end of the third CPR cycle, ETCO2 in the present study was significantly higher in the animals that survived than in non-surviving animals. Our results were consistent with previous studies in animals 58,59 which have demonstrated a strong correlation between ROSC and higher levels of ETCO2. Studies have confirmed that the measurement of ETCO2 is predictive of the outcomes of CPR. 56,60 Human studies have suggested that ETCO2 correlates closely with successful resuscitation and predicts the outcome of CA reliably 61,62 The initial ETCO2 immediately after unclamping the ETT did not correlate with ROSC in the present study. This has already been described in previous animal studies. 63
The cardiac rhythms were evaluated at the time of loss of aortic pulsations, followed by a 4 min interval of untreated arrest. In this model of asphyxial CA we established that by the end of the 4 min of untreated arrest, VF occurred in six out of 10 piglets. Even though, the rhythms in paediatric CA were thought to be asystole and bradydysrhythmias, 64,65 Walsh and Krongrad 66 first noted that VF could occur as a terminal rhythm in all paediatric age groups.
Like any other life-saving effort, the optimal goal of CPR is the neurologically intact survival. Neurological outcome was evaluated as functional deficit. All of the animals were severely impaired 24 h after their CA. Four animals had a neurological alertness score of 40, were lying on each side, had no gait, responded only to painful stimuli, had no convulsions and they had anisocoria. Two animals that had a score of 50 were also lying on each side, had no gait and no convulsions. They also responded to painful stimuli but their pupils were normal. Posture and gait were mostly impaired in all animals. Our results agree with other studies that show that asphyxia results in early cerebral ischaemic injury. 67
We have developed a model of out-of-hospital asphyxial arrest with clinical relevance to asphyxia and resuscitation that closely mimics guidelines published by ILCOR used in humans 4 and also has the capacity for assessment of 24 h neurological outcome assessment. This model could be used to test the efficacy of new pharmacological interventions during asphyxia CA and CPR. Moreover, it will prove useful in evaluating potential neuroprotective agents for victims who have suffered from asphyxia. Future paediatric resuscitation research must focus not only on how to increase survival from out-of-hospital CA but also how to maximize the probability that these survivors remain neurologically intact.
This study has some limitations. The interactions of the anaesthetic agents were not assessed, the number of animals was relatively small and that the experiment was performed on healthy pigs, whereas most of the victims of CA have underlying pathologies.
In this laboratory model of asphyxia CA, a successful basic Landrace/Large-White swine model of resuscitation in asphyxial arrest was for the first time developed that may prove useful in future studies. There was a strong correlation between ROSC and higher CPP levels. Among the different ways of inducing asphyxia arrest, clamping of the ETT is extremely easy. Neurological evaluation 24 h after ROSC revealed a moderate neurological deficit, as expected in asphyxia CA.
