Abstract
Foot ulceration annually affects millions of patients and accounts for billions of dollars in medical expenses in the US alone. Many previous studies have investigated co-morbidities associated with impaired healing, such as microbial infection, compromised circulation, and diabetes. By comparison, little is known about how wound healing proceeds in plantar skin, despite its many unique specializations related to its load-bearing function. One of the main challenges in modeling plantar wounds is the difficulty in maintaining wound dressings, as animals generally have a low tolerance to wearing bandages on their feet. With assistance from the MGH Center for Comparative Medicine, we developed a positive reinforcement-based behavioral training regimen that successfully induced tolerance for plantar dressings in swine, which is a critical first step towards enabling in vivo study of the wound healing process in this highly specialized skin area. This training program will be described in detail in this manuscript.
Introduction
Foot ulceration poses a large burden on the global healthcare system. The risk of developing foot ulcers is estimated at 15–25% for diabetic patients. 1 Non-healing ulceration is the leading cause of non-traumatic lower limb amputations 2 and costs billions of dollars in annual medical expenditures. 3 Even with treatment the recurrence rate for diabetic ulcers is approximately 40% in one year and 60% in three years. 4
Animal models are a critical tool in wound research, as the complex cellular and biochemical processes involved in wound healing are still impossible to fully replicate in in vitro culture systems. And while many models have been developed to simulate various pathologies associated with foot ulcers, such as vasculopathy, microbial infection, and diabetes, 5 the experimental wounds are predominantly placed on the animals’ trunks, and seldom produced on the foot, despite previously demonstrated body site-specific variations in skin histology, 6 growing evidence that wound healing proceeds differently in different parts of the anatomy, 7 and the many well-recognized specializations in plantar skin, which include substantial thickening of the epidermal layers, 8 reduced barrier function, 9 and a complete lack of hair (the density of which has been shown to correlate with faster healing 10 ). The general preference for using trunk skin in wound models is largely due to logistical reasons, as there is usually more skin area available for experimentation, and the paraspinal area in particular is relatively inaccessible to the animal’s mouth, which makes it easier to maintain dressings and minimize unplanned damage to the study wound sites.
Pigs are generally recognized as the best preclinical model for wound research, due to the many anatomic and physiologic similarities between human and pig skin, and high concordance between human and porcine wound data. 11 For modeling plantar wounds, the swine has the added benefit of similar body size to humans, which should lead to more comparable weight-bearing mechanics than rodent models, as it has been shown that peak plantar pressure during voluntary walking scales to the square root of body mass in a broad array of mammalian species. 12 To the best of our knowledge, there have only been a handful of reports in rodent models with wounds on the paws,13–15 but those studies did not address plantar-specific aspects of wound healing, and there are no published reports on plantar wound models in large animals. The aim of this study was to develop a behavioral training program to enable the study of porcine plantar wounds that is consistent with best practices in research animal welfare and would allow both adequate wound care and longitudinal monitoring of the healing process.
Materials and methods
Histology
Deidentified human skin tissue that would otherwise be discarded after surgery was obtained from the pathology department of the Massachusetts General Hospital (MGH), with approval by the MGH Institutional Review Board. Porcine plantar skin samples were obtained as part of the study described below. Tissue samples were fixed in formalin, embedded in paraffin, and processed for standard hematoxylin and eosin staining.
Animals
Three naïve female Yorkshire swine, aged about 8 weeks and approximately 30 kg at arrival in the housing facility, were used for this study. Neither the model nor the experimental procedures are expected to be gender specific. Animals were housed on a 12-h dark/light cycle. All procedures were conducted in accordance with the Public Health Service Policy on Humane Care and Use of Laboratory Animals, and with approval by the MGH Institutional Animal Care and Use Committee.
Behavioral training
In preliminary studies we found that naïve pigs had a low tolerance to wearing dressings on their feet and would remove/destroy the dressings by biting and tearing almost as soon as the animals recovered from anesthesia. A positive reinforcement-based behavioral training regimen was therefore designed, with guidance from MGH’s veterinary staff and behavioral specialist, to induce tolerance for wearing a protective boot (Figure 1). As an animal welfare measure to minimize potential negative impacts on mobility, plantar wounds would only be produced on one foot, therefore animals underwent training focused on one foot only. We elected to use a hind foot since it is less accessible by the animal’s mouth. The training program typically lasts four weeks, but adjustments may be made based on the animal’s behavioral endpoints, as discussed below.
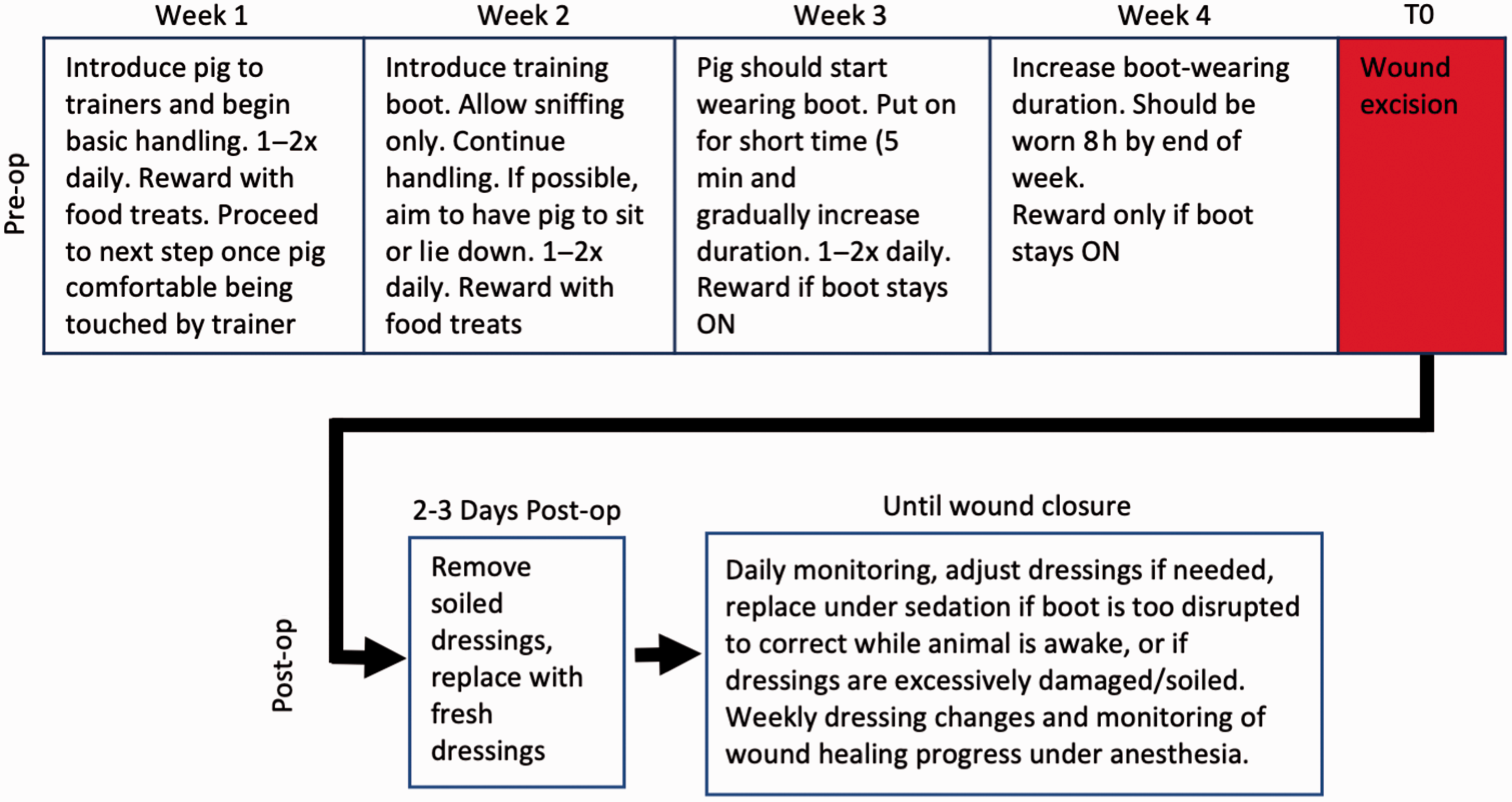
Flowchart for porcine plantar wound model. Protocol timeline for behavioral training and post-operation follow ups. The various phases of behavioral training should occur as depicted, but the timing should be adapted to each individual animal based on behavioral endpoints.
During the first week, all trainers involved in the process were introduced to the animal to familiarize the animals with the trainers. The trainers began by staying at the opened door of the cage, offering food treats to entice the animal to approach the trainer. This would be repeated at least once daily, until the animal became familiar enough with the trainer to approach even without enticement, upon which the trainer would start entering the cage before offering treats. Once the animal became comfortable with the trainers’ presence in its cage, the next phase of the training program would commence to acclimatize the animals to basic handling, which was gradually increased based on the reaction of the individual pig. First, only the back was touched; once the pigs would not shy away, the trainer would then move to the belly, then head, and finally the legs and feet. Pigs were rewarded with food treats when they allowed the trainer to pet them. Training was paced at the comfort of the pig. For pigs that were more reluctant, additional time was spent on basic handling to encourage the pig to become more comfortable with the trainers. These interactions were performed once or twice a day depending on how well the pigs learned the desired behavior. Pigs that were slower to learn or more anxious were handled twice a day to increase their comfort level.
Once the animals became comfortable enough to approach and spend time with the trainers inside their pens, typically by the second week, training would progress to the next phase. The pigs were encouraged to sit or lie down by rubbing them on their bellies, which greatly facilitated the manipulation and/or changing of the boot. But if the pigs would not lie down, boot training still proceeded with donning of the boots while the pigs were standing. A training boot (Sheep Boot, Stock #: 220204, Davis Manufacturing, Brandon, WI, USA) was introduced to the pigs during the second week. This boot model is not durable enough for long-term wear in our animal model, but is advantageous for training purposes primarily due to the ease of donning and doffing. Pigs were allowed to sniff the boot; however, any forms of playing with, chewing or biting the boot were discouraged by removing the boot. This was to discourage the pig from chewing on or otherwise interacting too vigorously with the boot while wearing it. Training sessions continued at once or twice a day during the second week, with an emphasis on the trainers picking up the animals’ feet, to acclimatize the animals to having their feet handled by humans.
The animal will typically begin wearing the boot by the third week. The training boot is usually larger than the pigs’ feet and required gauze pads to improve fitting of the boot on the hoof (Figure 2(a)). At first the boot would only be fastened with the built-in strap, and only stayed on for a maximum of 30 s, with 2–3 repeats. This was generally the longest time that the pig would tolerate at the first training session. At the next session the boot was put on the pig and secured with self-adherent elastic wrapping (Coban Wrap, 3M, Saint Paul, MN, USA; Figure 2(b) to (f)). Typically, two rolls of wrapping were necessary to secure the boot. Pre-stretching of the elastic wrapping is not recommended, as the pre-stretched wraps often failed to secure the boot. Boot donning is greatly facilitated and could be performed by a single person if the animal would lie down (as it was trained in the previous week to do), but if necessary it could be done with the animal standing, in which case it is often advantageous to have two staff members present: one to apply the boot while the other distracts the animal with destructible enrichments, for example, fruit stuck to peanut butter inside a paper bag which would then be rolled up to conceal the treats – both sticking fruit to the peanut butter and rolling up the bag served to increase the time it took for the animals to access the food, which provided more time to apply the training boot and wrapping. The amount of time the boot remained on was incremental and increased based on how well the pigs tolerated the boot, starting at 5 min with the boot while supervised by the trainer. Pigs would be given food treats as reward only if the boot was kept on for the desired length of time. If the pig kept the boot on for the desired time, then the duration of the subsequent training session would be increased, with the goal of having the pig wear the boot continuously for at least 8 h, which generally occurred by the end of the fourth training week.

Behavioral training boot. Training boot for the acclimation/behavioral training period. (a) Inside of boot used for behavioral training, showing gauze stuffing to improve boot fit. (b) Boot fitted to a pig’s foot. (c) Training boot secured with elastic wrap (Coban). Note wrapping direction from hoof to hock. (d) First layer of wrapping completed to the hock. (e) A second roll of Coban, applied in the same direction and (f) Completed wrapping of the training boot.
Wounding procedure
Buprenorphine (0.3 mg/ml, IM) was given approximately 30 min prior to surgery for pre-emptive analgesia. Under general anesthesia, one leg on the animal was shaved from the hoof to the hock, washed, and disinfected by successive scrubbing with povidone-iodine and isopropanol. Skin biopsies of 10 mm diameter were then collected from the plantar surface of that foot (the weight-bearing and non-weight-bearing areas of the foot can accommodate up to two wounds each (Figure 3(a)). In weight-bearing areas the skin may be too thick for standard biopsy punches to penetrate completely, and we have found that removing parts of the plastic adjacent to the cutting blade (effectively increasing the cutting depth of the biopsy punch) is often necessary to produce full-thickness wounds in such areas.
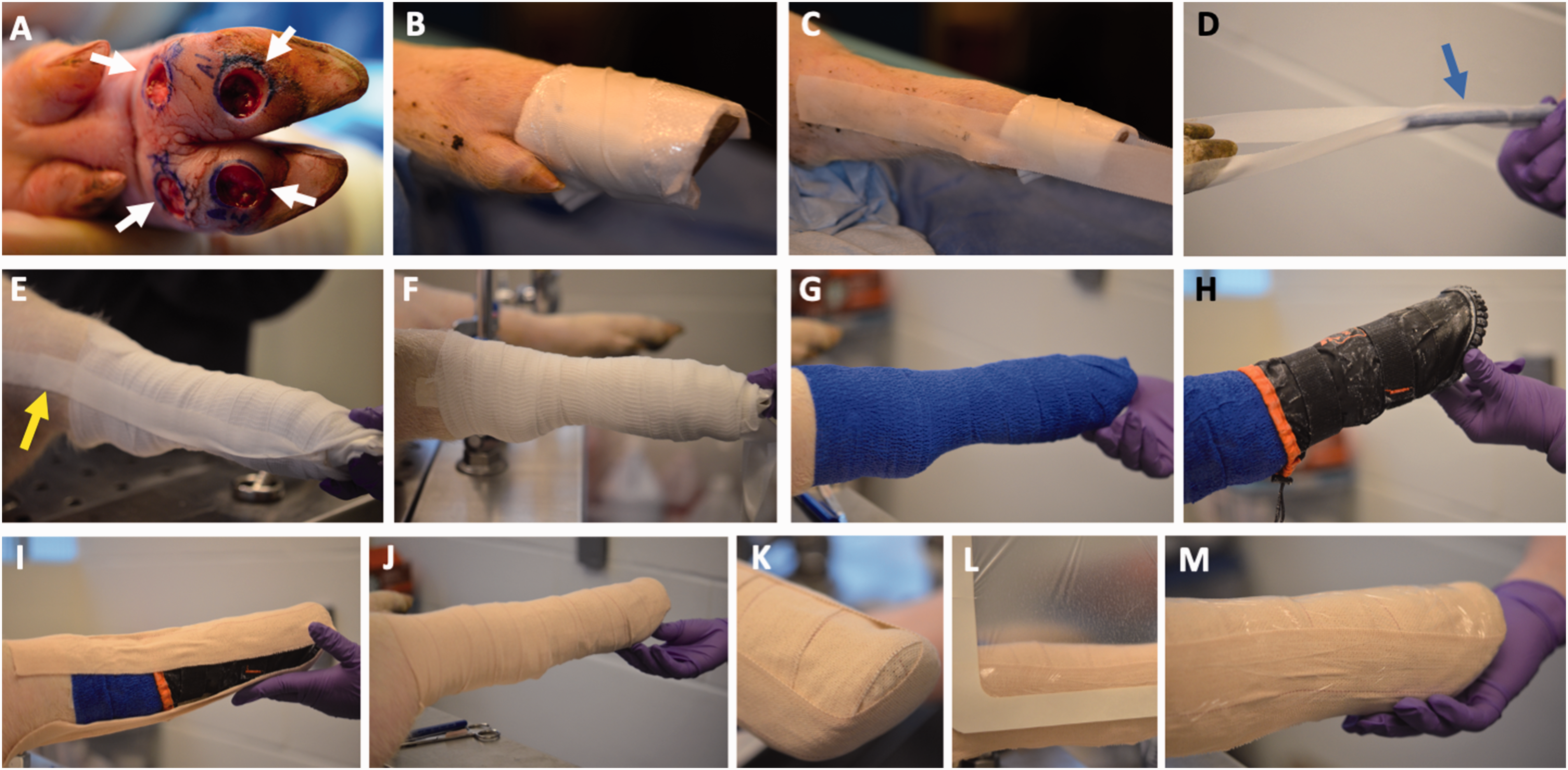
Post-injury wound wrapping. (a) Four 10mm-diameter, full-thickness skin wounds on the plantar surface of a pig’s foot, shown shortly after excision. (b) Telfa pads wrapped around the foot and held in place with tape. Freshly excised wounds would also be packed with Tegaderm gel and foam to facilitate hemostasis and blood/exudate removal (not shown). (c) Tape is applied to the medial and lateral aspects of the upper portions of the leg, over the Telfa pads, and with the ends extending beyond the foot. (d) The extended tape ends are prevented from sticking to each other by adhering them to a pen (arrow) temporarily. (e) Gauze is wrapped around the leg in an upwards direction stopping at the hock, then the tape ends are removed from the pen and twisted to form a stirrup then taped down to secure gauze (arrow). (f) A second layer of gauze is wrapped on top of the first. (g) Coban is then wrapped to cover the gauze, again in the hoof-to-hock direction. (h) The Medipaw boot is put on and adjusted to ensure proper alignment. (i) The first stirrup is made using Elastikon tape that goes from the front of the leg to the back. It is often necessary for another person to push against the bottom of the boot during this step, to ensure that the boot is fitted snugly against the bottom of the pig’s foot. (j) Elastikon is used to wrap around the entire leg. (k) A second stirrup is made that goes from the medial to the lateral aspect of the leg; (l) The outside of the dressing is covered with a water-impermeable film and (m) The completed dressing.
Wound dressings
After wound excision, Tegaderm hydrogel (3M) was placed into each wound site, followed by Tegaderm foam dressing (3M) that was cut to the size of the wound and packed into the wound bed to provide compression and facilitate blood and exudate removal. These were overlaid with non-adherent dressing (Telfa, Covidien) then taped into place with surgical tape (Figure 3(b)). The next step was to begin creating the stirrup. Surgical tape was placed down each side of the leg from hock to hoof, with a “tail” of approximately 6 inches of tape to be used for the second part of the stirrup (Figure 3(c)). These “tails” were prevented from sticking to each other by placing a pen between the ends (Figure 3(d)). Two layers of gauze (Elastomull) were then wrapped around the leg starting at the hoof and moving upwards to the hock – the upwards wrapping direction is intended to prevent blood pooling in the hoof region. The tape ends were then twisted and taped back up the leg to form a stirrup to keep the gauze in place (Figure 3(e)). Pre-stretched elastic wrap (Coban, 3M) was then wrapped over the gauze, again starting at the hoof and moving upwards (Figure 3(g)). Depending on the size of the pig it may be necessary to add an extra layer of gauze and Coban to ensure better boot fit.
We were unable to find commercially available protective boots designed for pigs, so boots made for other animals were adapted for this study. After consulting with the MGH veterinary staff, the Cavallo Cute Little Boot (Cavallo Horse and Rider) and the Medipaw Rugged Dog and Cat Protective Boot (Medipaw) were identified as the most promising options, and both were tried in this study. After the elastic wrap, the boot was placed on the foot with adjustments to ensure that the toe of the boot aligns with the contralateral foot, which will provide a better fit and make walking easier (Figure 3(h)). For the Medipaw boot, the drawstring should be tightened in a way that does not rub against the leg or impede circulation, as having the drawstring too tight was found to cause chafing and skin irritation. The dressing was then secured with Elastikon tape (Johnson & Johnson). First, a stirrup was made going from the front of the leg to the back and a second stirrup was made from side to side. Then the entire dressing was wrapped with a layer of Elastikon, wrapping from hoof to hock (Figure 3(i) to (k)). The upper part of the Elastikon must be in tight contact with the skin (no folds in the tape) so that the dressing is secured to the leg. Lastly, the dressing was covered with adhesive film to help keep the bandage dry (Figure 3(l); we used expired Tegaderm film, but other similar film dressings should be able to serve the same purpose).
After surgery, carprofen (2–4 mg/kg PO, q12–24 h) was given for 72 h for analgesia. Animals were monitored for standard humane endpoints, including excessive weight loss, abnormal breathing and cyanosis, vomiting/diarrhea resulting in severe dehydration, and signs of pain or distress not alleviated by analgesics. Some limping or otherwise favoring of the wounded foot is expected, therefore mild abnormalities in ambulation were not taken to be a humane endpoint. None of the three animals in this study exhibited humane endpoints.
Dressing changes
It is advisable to replace the dressings 2–3 days after the initial wounding procedure, otherwise the hardened clot may be difficult to remove from the deeper plantar wounds. Thereafter the dressings were changed weekly, until the wounds visibly closed. These scheduled dressing changes were performed under anesthesia, as applying the dressings in awake animals was difficult even with the pre-surgery training. The Elastikon and Coban dressings were cut with bandage scissors to facilitate removal. The soiled dressings were removed, the wounds were evaluated and photographed, then dressed again with fresh dressings. At each dressing change, a best effort was made to change the location where the Elastikon tape adhered to the skin, in order to minimize irritation and damage to the underlying skin.
Between bandage changes the animals were monitored by study staff at least daily, usually in the morning, for signs of damage or dislodgement of the dressings. For minor peeling of the Elastikon tape or small slips in the boot (roughly 5 cm or less), the boot could be adjusted without anesthesia. This can be facilitated by having one person distract the pig with treats while a second person adjusts and secures the boot with more Elastikon. More extensive displacements of the boot, or any adjustments that required adjusting the layers of Coban and gauze, would be performed under anesthesia.
Results
Histology
As has been previously described,8,16 human plantar skin exhibits distinctive structural characteristics primarily in the epidermis, which is substantially thickened due to both increased number of cell layers and enlargement of keratinocytes and corneocytes, with prominent rete ridges interdigitating deeply with the underlying dermis (Figure 4(a)). Porcine plantar skin is thicker overall, and lacks friction ridges (an evolutionary adaptation to arboreal habitats that is found only in primates and koalas), but otherwise shares many similarities with its human counterpart, including thickening of both the viable and cornified epidermis, pronounced rete ridges, and a compact stratum corneum composed of densely packed, enlarged corneocytes (Figure 4(b)). Non-plantar skin samples from both human and swine are shown in Figure 4(c) to (f) to further underscore the remarkable structural differences between plantar and non-plantar skin in both species.
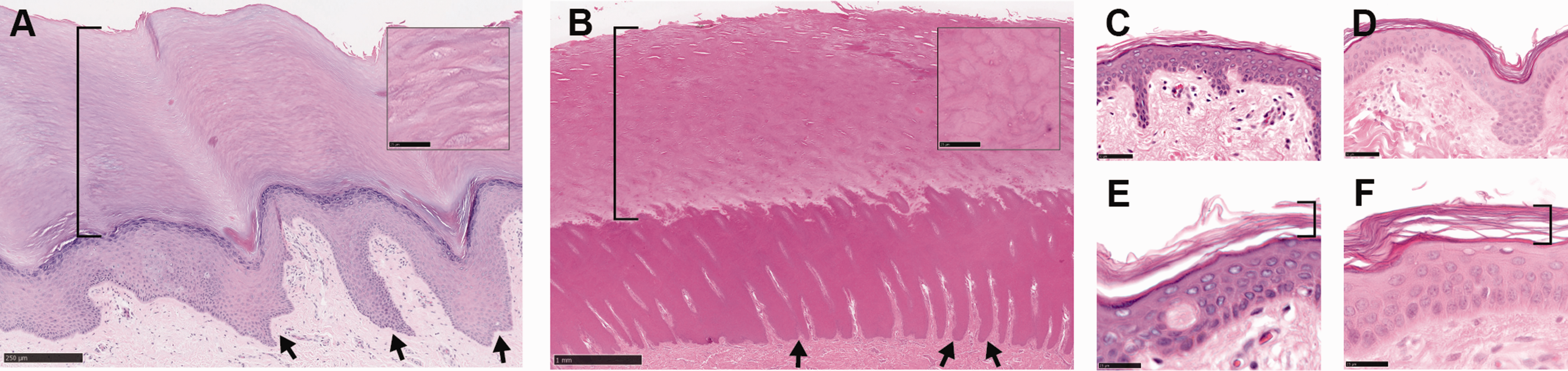
Plantar versus non-plantar skin histology in humans and swine. (a) Human plantar skin, showing distinctive features including prominent rete ridges (arrows) and a compact stratum corneum (bracket) that is thicker than the underlying viable epidermis. (b) Porcine plantar skin shows similar characteristics, with rete ridges and stratum corneum marked by arrows and bracket, respectively. Insets in (a) and (b) show the stratum corneum at higher magnification to better visualize the enlarged and tightly packed corneocytes. (c) Human abdominal skin. (d) Porcine skin overlaying the chest wall. (e) and (f) are high-powered views of the same skin types as shown in (c) and (d), respectively, with the stratum corneum marked by brackets. Note in non-plantar skin the much thinner viable and cornified epidermal layers, reduced interdigitation between dermis and epidermis, and the loosely layered corneocytes in the stratum corneum. Scale bars: (a): 250 µm; (b): 1 mm; (a) and (b) insets: 25 µm; (c) and (d): 50 µm; (e) and (f): 25 µm.
Behavioral training
One of the most difficult aspects of the behavioral training was the association pigs made between the trainers and treats. Once the pigs began to associate the trainers with treats, the animals would often search the trainers for treats instead of performing the desired behaviors, such as lying down. A few different adjustments were employed to disassociate the trainers from the treats, including leaving treats outside the pen, leaving treats in a corner of the pen, or putting the treats in a hanging bucket out of reach from the pig. Using a bucket or leaving treats outside the pen were the most successful methods of getting pigs to perform the intended training behaviors. This method was utilized until approximately week 3, when boot training began. At the start of week 3, destructible enrichment was used so that the pig remained distracted while the boot was put in place.
Plantar wounds
Similar to humans, in pigs the plantar skin (particularly in the weight-bearing regions) is substantially thicker than skin in other parts of the anatomy, which necessitated some technical adjustments. As described above, full-thickness plantar wounds were often difficult to produce with standard biopsy punches, unless modifications were made to increase the cutting depth. The depth of the wounds also created a “well” that blood and exudates could pool in, and if not removed in a timely manner could lead to the formation of thick clots that are difficult to remove and can interfere with wound closure. Porcine plantar skin is also stiffer than most other skin areas, which makes it challenging to clamp hemostats around bleeding vessels to achieve post-surgical hemostasis, and primary closure is virtually impossible even when it might otherwise be desirable (e.g. after taking biopsies at designated study timepoints). With proper wound care and dressing (detailed below), it is possible to maintain and monitor these plantar wounds longitudinally until wound closure, typically by the fourth or fifth post-wounding week. A more detailed study of the healing process in this plantar wound model is currently underway, and will be described in a later report.
Dressing regimen
All three pigs were able to remain on the study until their respective designated endpoints (four weeks for one, eight for the others), the wounds were well-protected by the regimen and did not develop clinical signs of wound infection, or any other complications that would require early termination of the study. The animals often favored the wounded foot especially at early time points, but otherwise did not display other signs of pain or distress. The boot-wearing foot was more prone to slipping if the cage floor was too smooth, and those cases were remedied by placing rubber mats on the floor. The Cavallo Cute Little Boot was very sturdy, but even the smallest size available was substantially larger than the pig’s foot, which made the fitting difficult even after stuffing much of the boot’s cavity with gauze pads. The thick soles on these boots also caused the animals’ hips to become uneven while standing, and the animals often placed more weight on the heels to compensate, which in turn led to a propensity for the boots to become dislodged. Consequently, these boots needed frequent adjustments (often several times per week) and despite our best efforts still became substantially displaced, as to require sedation to replace the dressings, three times for each animal over the four- and six-week course of boot wearing.
These challenges led us to transition to the Medipaw boot. The Medipaw boot was easier to fit as it was closer in size to the pigs’ feet, and had a long sleeve that facilitated taping and securing to the animals’ legs, although the same sleeve was prone to slipping down the leg if it was not properly secured. When applying the boot, it should be aligned with the contralateral foot to help the boot stay in place. We also found that adding stirrups with the Elastikon helped secure the boot. We tried three different configurations with the stirrups: one that went from the lateral side to the medial side, one from the front of the leg to the back, and a third configuration that used both. We found that the configuration using both stirrups was most successful at keeping the boot in place. We also found that taping of the central stirrup should go from front to back, while pulling slightly towards the back at the toes before taping up the back of the leg. This was because when the Medipaw boot did slide down the leg, the toe of the boot would often slide forward off the hoof, so adding pressure while taping back around the toes helped to keep the boot in place. There were initial concerns that the Medipaw boot may be more prone to damage as it is made of more flexible materials, but it remained largely intact over eight weeks, required fewer adjustments between the weekly designated dressing changes, and only once needed replacement under sedation outside of the scheduled dressing changes. Overall, we found the Medipaw boot to be better suited for this experimental model.
Conclusion
We have developed a behavioral training regimen that allows for the application and maintenance of protective dressings over experimental plantar wounds in pigs. In addition to enabling investigations into plantar-specific aspects of wound healing, this model could potentially be combined with other previously developed approaches to model various diabetes-related wound complications, such as compromising the circulation, 17 introducing microbial infection, 18 and/or inducing diabetes, 19 to more accurately simulate the pathologies commonly associated with foot ulcers in humans.
Inflammation is a critical component of the body’s response to injury, and dysregulation of the inflammatory response is thought to be a key contributing factor to the etiology of chronic ulcers. 20 In this study we used the non-steroidal anti-inflammatory drug (NSAID) carprofen for analgesia, since in our previous experiments we have not found it to impact healing in our porcine wound models, at the relatively low dosage (2–4 mg/kg) and brief duration (three days) used in this study. Long-term, high-dose treatment with NSAIDs has been reported to affect healing outcomes in rodent wound models.21,22 If the anti-inflammatory effects of NSAIDs are of concern, alternative analgesia medications, such as opioids, could be used instead.
Limits of this model include higher cost, labor-intensive procedures, requirement for more specialized staff skills, and incompatibility with genetic manipulations, compared with common rodent models. There are also likely to be inter-species differences in plantar skin biology between pigs and humans, which may be intrinsic (previous studies comparing non-plantar skin between humans and pigs, while noting the overall similarity, have reported disparities such as seasonal shedding of hair, lack of eccrine sweat glands, and biochemical differences in pig skin,23–25 or extrinsic (e.g. different weight-bearing and locomotion biomechanics in bipeds versus quadrupeds). As with any animal model, such inter-species differences should be considered when attempting to extrapolate animal data to make inferences on human physiology.
Footnotes
Acknowledgements
The authors thank staff members of the MGH Center for Comparative Medicine and Knight Surgical Research for their outstanding technical assistance and advice.
Data availability
Data generated in the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Military Medicine Technology Transformation Collaborative, Award Number HU0001-17-2-0009. The Uniformed Services University of the Health Sciences (USU), 4301 Jones Bridge Rd., Bethesda, MD 20814-4799 is the awarding office and is administered through the Henry M. Jackson Foundation for the Advancement of Military Medicine, Inc. (HJF). The information or content and conclusions presented here do not necessarily represent the official position or policy of, nor should any official endorsement be inferred on the part of, USU, HJF, the Department of Defense, or the U.S. Government. RRA was partially supported by the Lancer Endowed Chair in Dermatology.
