Abstract
Nulliparous female Syrian hamsters were used to investigate the effect of two different breeding systems on the fertility of the female Syrian hamster. We hypothesized that females submitted to a harem system (HS) would deliver smaller and more female-biased litters than in a monogamic system. Ten female and 10 adult male hamsters housed individually (G1) were kept in a monogamic temporary breeding system, while 10 females and five males (G2) were submitted to HS with two females and a male permanently housed together since female weaning. Females from G1 and G2 delivered, respectively, 47 and 50 litters, and produced 364 (G1) and 383 (G2) weaned pups without any difference in litter size, mean weight of weaned pups and body condition of dams. Interparturition intervals were shorter and the percentage of male pups per litter was higher in the HS possibly as a result of different endocrine conditions provided by different breeding systems. Besides providing evidence that housing conditions can influence the sex of hamster offspring, our findings suggest a mechanism for the non-random distribution of male and female pups in hamster litters.
Despite their confirmed solitary social organization in the wild, 1 in animal facilities, Syrian hamsters (Mesocricetus auratus) are generally kept in isosexual groups from weaning until adult age. 2 Even in such conditions, grouping adult hamsters results in symptoms of stress. 3
At least two main breeding systems, namely the monogamic temporary system (MTS) and the harem system (HS), can be used to breed hamsters. In MTS, the animal's oestrous cycle is monitored daily and the female is placed in the male cage only during oestrus, when the female is sexually receptive. This system seems to mimic the species behaviour in the wild. 4 In HS one male and two or three females are kept together until pregnancy can be visually detected. Pregnant females are then individually housed until the weaning of the litter. By then the females are reintroduced in the male's cage. In most cases, these harems are formed at female weaning to avoid agonistic behaviour, but fights are not rare when a female is reintroduced in the group. HS results in repeated social defeat for some females, which may result in reduced fertility and biased offspring sex ratio. 5 However, HS is generally the first choice in most animal facilities since it is less labour consuming than MTS.
Herein, we investigated the effect of each breeding system on the fertility of the female Syrian hamster. The reproductive history of nulliparous females submitted either to HS or to MTS and mated repeatedly was monitored. We hypothesized that HS females would deliver smaller and female-biased litters.
Twenty non-isogenic, recently weaned female (age: 25 days; weight: 35–45 g) and 15 adult (age: 65 days; weight: 90–105 g) male Syrian hamsters were supplied by the Laboratory Animal Facility of the Faculty of Veterinary Medicine (University of São Paulo, Brazil), whose Ethics Committee approved the experimental design. The animals were housed under conventional conditions (12:12 h light:dark, lights on at 06:00 h; room temperature between 20 and 24°C; 20 changes of air per hour). Specific pelleted food (Nuvilab CR1, Nuvital, Curitiba, Brazil) and filtered bottled tap water were supplied ad libitum. The animals were free from any common pathogens according to the FELASA Health Monitoring Recommendations. 5
The animals were separated into two groups. In the first group (G1) 10 females and 10 males were individually housed and submitted to MTS. Five groups of two females and a male permanently housed together (HS) from female weaning formed the second group (G2). Females were individually housed as soon as pregnancy was visually detected.
All hamsters were mated repeatedly five times according to their respective breeding system. Weaning occurred 21 days after pups' birth. All reproductive events such as postovulatory discharge (POD), mating, parturition, number and sex of pups, and intervals between parturitions were recorded daily until the weaning of the fifth litter. Dams and pups were weighed at weaning. Dams were clinically examined at the end of the experiment.
The total and mean production was calculated for each parturition in each group, as well as the mean weight of dams and pups. These means were compared between groups by t-test. Sex ratio was determined for each group as the percentage of pups of each sex. Results are expressed as means ± SD.
The interval between parturitions was calculated only when a new pregnancy and full-term delivery followed the lactation and weaning of the precedent litter. Average interval was higher in G1 than in G2 (44.63 ± 3.86 and 40.91 ± 1.67 days, respectively, t-test with Welch correction, t = 5.19, P < 0.0001).
Because one female from G1 died three days after the birth of her second litter, G1 females delivered only 47 litters through five pregnancies and parturitions. In this group 43 litters (364 pups) survived until weaning. G2 females gave birth to 50 litters and 44 of these litters (383 pups) reached weaning. The mean number of pups per litter (G1: 7.68 ± 4.11, G2: 7.66 ± 4.27) did not differ significantly between the groups (t-test, t = 0.02, P = 0.98). Table 1 shows the mean number of pups weaned per litter in both groups after each delivery. Mean body weight of pups at weaning (calculated as the mean body weight of pups in each litter divided by the number of litters in each group) did not differ significantly between the two groups (G1: 41.46 ± 6.70 g, G2: 40.14 ± 5.90 g, t-test, t = 0.47, P = 0.65).
Number of pups (mean ± SD) weaned for litter in monogamic (G1) and harem (G2) groups
Female body condition was measured by weighing dams after the weaning of the first litter (G1: 119.59 ± 10.04 g, G2: 115.57 ± 8.25 g, t-test, t = 0.86, P = 0.40) and the fifth one (G1: 156.78 ± 10.04 g, G2: 158.93 ± 11.94 g, t-test, t = 0.44, P = 0.67). All females were also clinically examined at the end of the trial. Mean body weight did not differ significantly between groups, and the clinical exam did not detect any indicator of morbidity except for some mild bite scars in G2 animals.
Whereas in G1 (kept in MTS) more female pups survived until weaning (n = 207, 56.87%) than male pups (n = 157, 43.13%), a significantly different (t-test, t = 2.516, P = 0.02) situation occurred in G2 (HS group: male pups n = 204, 53.26%; female pups n = 179, 46.74%). Although the difference was only significant for the last litter (t-test, t = 2.914, P = 0.01), possibly due to the small sample size, the percentage of male pups was consistently numerically higher in G2 than in G1 even when more females were weaned in both groups as occurred after the third delivery (Figure 1).
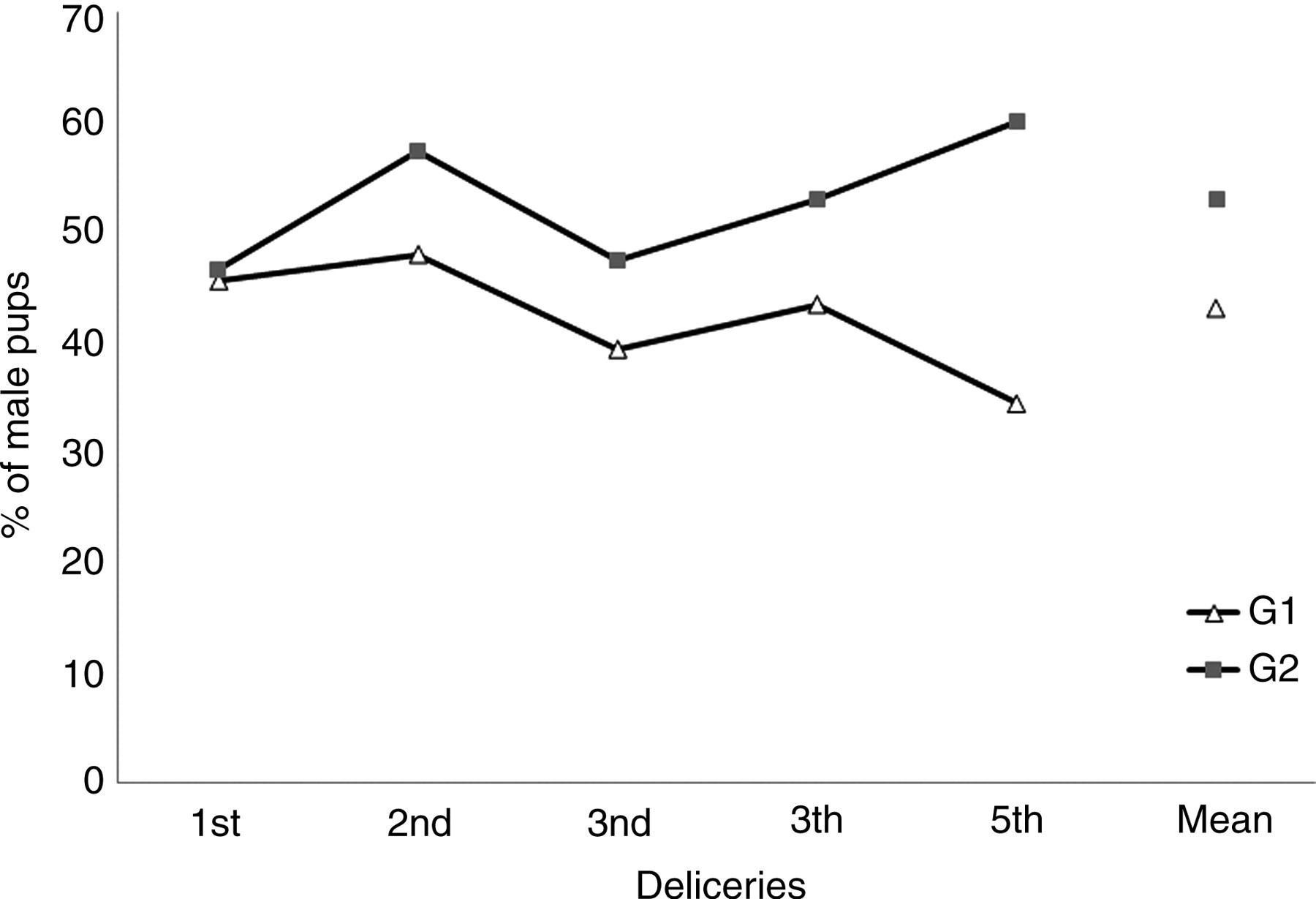
Percentage of male pups delivered in each group through five successive litters. G1: monogamic temporary system; G2: harem system
The four-day (an oestrous cycle) difference between the mean interparturition intervals can be easily explained by the characteristics of each breeding system: in HS (G2) the male is able to detect the first oestrus after weaning and copulation occurs during this first cycle. In MTS (G1), the pairs are only allowed to mate after the first oestrus has been detected, a posteriori, by the observation of POD. Thus, in this system, mating only occurs during the second cycle after weaning, four days later.
The male-biased sex ratio in HS litter is more intriguing. In our experiment, all the animals were kept in equal environmental conditions and the body condition of dams and pups was similar in both groups. Therefore, such variables cannot be viewed as responsible for skewing the offspring sex ratio. Actually, the only differences between the two groups were housing and breeding system. Social stress can reduce the proportion of male pups born to defeated female hamsters. 6 The MTS seems at first glance to offer more natural, and then less stressful, conditions. However, dams in this system had equal body condition and produced less male pups than the harem kept females. Because our results do not show differences in the number of pups per litter between groups, it is likely that the difference in sex ratios might not be associated with sex selective spontaneous fetal loss. Therefore, sex ratio adjustment probably occurs around conception.
Although the mechanism of such a bias remains unclear, mammalian sex ratios at birth are partially controlled by the hormone levels of both parents at the time of conception. 7,8 Higher levels of oestrogens and testosterone in the blood of the parents would result in male-biased sex ratio while higher concentrations of progesterone would have an inverse effect. No hormone measurements were performed in our study. However, because the animals were raised and kept in conventional laboratory conditions, it seems reasonable to assume that their endocrine profile followed the pattern described in the literature. 4,9 The two different breeding systems resulted in different mating schedule, which in turn, because of the very quick succession of hormonal events in this species, likely resulted in different endocrine conditions at mating for both males and females.
As far as attested by the good physical condition of dams at the end of the study, both tested breeding systems warranted female welfare. Moreover, we did not observe any significant difference neither in the total number nor in the weight of the pups born in HS and in MTS. On the other hand, the shorter interval between two subsequent parturitions in HS may be an advantage when a very intensive production is necessary, and so does the male-biased sex ratio provided by the HS, since males are generally preferred in biomedical experiments. Besides providing evidence that housing conditions can influence the sex of hamster offspring, our findings suggest a mechanism for such an effect. Further studies including hormone measurements will be essential to better delineate the endocrine linkage responsible for the non-random distribution of male and female pups in hamster litters.
Footnotes
Acknowledgement
This work was partially supported by a grant awarded by Fundação de Amparo à Pesquisa do Estado de São Paulo to Marie Odile Monier Chelini (00/00282-6).
