Abstract
Diet-induced weight gain increases disease risk via disruption of the innate immune system. Flow cytometry is commonly used to assess the immune system; however, in mice such measurements traditionally require terminal procedures and tissue collection to generate sufficient sample. The present study refined an existing flow cytometry method to reduce the number of mice needed to longitudinally measure monocytes. CD-1 male mice were randomly assigned to one of the three groups: DS (diet-induced weight gain + sedentary), DE (diet-induced weight gain + forced treadmill running [total distance 35,755 ± 1832 m]) or NS (normal weight gain + sedentary). DS and DE consumed a 60% fat diet and NS consumed a 10% fat diet ad libitum. Saphenous vein blood samples were collected weekly for a period of six weeks and three-colour flow cytometry was used to measure changes in monocyte (CD11b+/14+) concentration and cell-surface toll-like receptor 4 (TLR4) expression. DS (18%) and DE (17%) gained more weight than NS (P < 0.001). On a group basis, DS expressed 17% more TLR4 than DE and NS (P = 0.005). The present study demonstrates that a longitudinal survival model can be used to reduce the number of animals needed to complete flow cytometry experiments. Exercise during diet-induced weight prevented some (decreased monocyte TLR4 expression) but not all aspects of innate immune system function.
Diet-induced weight gain is associated with innate immune system dysfunction and an increase in systemic, low-grade inflammation. 1,2 Such inflammation contributes to the onset and progression of type 2 diabetes mellitus and cardiovascular disease. 3–5 Many measures exist to assess innate immune system function, such as response to infection, monocyte concentration/activity and natural killer cell concentration/activity. The rationale for selecting monocytes for this study was that we had previously demonstrated that exercise-training had anti-inflammatory effects in humans that could be measured by reduced monocyte expression of toll-like receptor 4 (TLR4). 6–9 TLR4 is a cell-surface pathogen-associated molecular pattern receptor that responds to bacterial lipopolysaccharide and other endogenous substance elevated during diet-induced weight gain (i.e. leptin, fatty acids, etc.). 10,11
Weight gain is a complex phenomenon that can be difficult to measure. 12,13 Traditional flow cytometry require large sample volumes that can only be collected from mice using terminal procedures. Terminal models are not desirable because they increase the number of animals that must be used and limit the ability of the investigator to describe longitudinal changes in outcome measures. In the present study, we describe a small volume flow cytometry technique that has been adapted to work with very small blood volumes.
Anecdotal reports have speculated that exercise training may be able to offset the deleterious complications of diet-induced weight gain by preventing disturbances in monocytes. We hypothesize that aerobic exercise training during a period of diet-induced weight gain will prevent innate immune system dysfunction. This study aimed to determine whether forced treadmill running could prevent the effects of diet-induced weight gain on monocyte concentration and cell-surface TLR4 expression. Additionally, we have improved an existing flow cytometry method to reduce the number of animals needed to complete a longitudinal experiment.
Materials and methods
Animals
All procedures described in this investigation were reviewed and approved by the University of Houston Institutional Committee on the Care and Use of Animals. Young (6–8 weeks old), outbred CD-1 male mice were used for this study (Charles Rivers Laboratory; Wilmington, MA, USA). CD-1 mice were chosen because they are ideally suited for longitudinal studies. 14 Also, since CD-1 mice are an outbred strain, they tend to have more animal–animal variability than inbred strains. Such variability is similar to what is found in humans. Prior to group assignment, mice were allowed 14 days to acclimatize to the animal care facility and maintained on a reverse 12:12 h (lights off at 10:00) light–dark cycle for the duration of the study. During the acclimatization phase, mice were handled and routinely placed in the restraint used for blood collection (no blood was collected). These procedures were designed to allow the animals to become accustomed to routine handling that would be completed as part of the study. Mice were housed four per cage and identified via ID numbers, tattooed on the tail (AIMS Animal System; New York, NY, USA). Body weight and food consumption were measured on a weekly basis throughout the study using a digital scale. Food was presented to animals in wire cage lids. Twice a week food was changed, residual (food left in cage lid) and wasted (food in bottom of cage) were combined and weighed. Food consumed was determined by subtracting residual/wasted food from total food given.
Sample size calculation
An a priori sample size calculation was completed based on preliminary data from our laboratory. 15 In this preliminary study, six weeks of diet-induced weight gain resulted in a 21% increase in body weight (moderate effect size, 0.40). Given that the aims of our preliminary and present study differed slightly, we opted to use a small effect size (0.25) in order to take a conservative approach. We calculated (G-Power version 3.0; Bonn, Germany) that we would need approximately eight animals per group to detect at least a 13% increase in body weight after six weeks of diet-induced weight gain (80% statistical power). In order to account for potential death as a result of diet-induced weight gain, we increased our total to 12 mice per group (total n = 36). The smallest body weight difference in the present study was 17% (effect size = 0.30), thus our statistical power was >99%.
Experimental design
Diet-induced weight gain conditions were completed as detailed in a previous set of experiments. 16 In order to complete the aims of the study, mice were randomly divided into one of three groups (n = 12 per group): diet-induced weight gain, sedentary (DS), diet-induced weight gain, forced treadmill running (DE) or normal weight gain, sedentary (NS). The DS group consumed a commercially available, open-source high-fat diet (D12492; 60% calories from fat, 20% calories from protein, 20% calories from carbohydrate; 5.24 kcal/g) (Research Diets Inc; New Brunswick, NJ, USA) and remained sedentary. The DE group consumed a high-fat diet (D12492) and participated in a treadmill exercise intervention. Exercise sessions were completed five days per week (Monday–Friday) and each exercise session consisted of up to one hour of treadmill running (Columbus Instruments; Columbus, OH, USA) at ∼21–22 m/min and 1% grade. All exercise sessions were completed in the first 4 h of the dark cycle to correspond with the animals' active phase. The NS group consumed a commercially available, open-source low-fat diet (D12450B; 10% calories from fat, 20% calories from protein, 70% calories from carbohydrate; 3.85 kcal/g) (Research Diets Inc) and served as a control group. The fat source in both diets was soybean oil and lard (cholesterol content = 0.95 mg/g). All groups were provided ad libitum access to food and water for the duration of the study.
Refinement, reduction and replacement
The present study is part of our laboratory's long-term goal to reduce the number of animals needed to complete longitudinal experiments of diet-induced weight gain. Here within, we describe a novel, improved technique for completing microcapillary flow cytometry on small blood samples (<50 µL) collected from the saphenous vein. Alternative sites to saphenous vein (for example, retro-orbital, caudal) exist for the collection of blood when animal survival is desired; however, collection at these sites are either more invasive or require sedation. Using this analysis technique, we were able to track changes in monocytes of individual animals over time, adding considerable statistical power to our model. Such statistical power increased the effect size and decreased the number of animals required, compared with a cross-sectional sacrifice design. For example if the present study was completed using a cross-sectional sacrifice experimental design, >250 animals would have been needed.
Blood collection
Saphenous vein blood samples were collected according to the standard technique 17 with slight modification. On Monday morning, 2 h before the start of the dark cycle (07:00) mice were placed individually into a modified 50 mL centrifuge tube (Figure 1a). Hair was removed from the hindlimb using an electric clipper and a thin layer of petroleum jelly was applied to the skin. After which, the saphenous vein was punctured with a 21-gauge sterile lancet. Whole blood (∼40 µL) was collected into a plastic capillary tube treated with lithium heparin (Sarstedt; St Louis, MO, USA) (Figure 1b). All blood samples were collected 48 h after the last exercise session. Blood was processed for flow cytometry measurements within 2 h of collection.
Blood collection. (a) Mice were individually positioned into modified 50 mL centrifuge tubes and (b) ∼40 µL of whole blood was collected into capillary tubes treated with lithium heparin
Monocyte cell-surface TLR4 measurements
Total leukocyte concentration and viability were measured using a commercially available test kit (ViaCount; Millipore-Guava; Hayward, CA, USA). All reagents were purchased from e-Bioscience (San Diego, CA, USA). Three-colour flow cytometry was used to determine monocyte concentration (CD11b+/14+) and monocyte cell-surface TLR4 geometric mean fluorescence intensity (gMFI). Samples were prepared according to the standard technique. 6,7 Briefly, whole blood aliquots (10 µL) were incubated with an Fc receptor blocking cocktail (anti CD16/32) to reduce non-specific binding. Blocked cell pellets were labelled with either a three-colour positive (CD11b-PECy5, CD14-FITC and TLR4-PE) or a three-colour isotype control cocktail. Monoclonal antibodies (mAbs) were used for all experiments in the present study. After 30 min of labelling in the dark on ice, red blood cells were lysed using a commercially available buffer. Lysed cells were washed and fixed in 1% paraformaldehyde (Electron Microscopy Sciences; Hatfield, PA, USA) and acquired on a Millipore-Guava EasyCyte Mini flow cytometer (Hayward, CA, USA). Analysis was completed offline using FCS Expression (version 3; De Novo Software; Los Angeles, CA, USA).
Prior to analysis, samples were labelled with each mAb individually and electronic colour compensation was used to eliminate overlapping emission spectra. Primary gates were established to identify monocytes (CD11b+/14+) and secondary histograms were used to identify TLR4 expression (gMFI). Routine quality control analysis was completed using standard sized polystyrene beads and daily fluorescence variation was <3%.
Statistical analysis
All statistical analyses were completed using PC-based SPSS (version 17.0; Chicago, IL, USA). Prior to formal statistical testing, the data were examined to ensure that the assumptions of normality and constant variance were met using quantile–quantile and residual–predicted plots, respectively. Outlier samples were identified as those that were more than 2 SD from the mean. All dependent variables were analysed using separate 3 (Group: DS, DE or NS) × 7 (Time: 0, 1, 2, 3, 4, 5 and 6 weeks of study) analysis of variance (ANOVA) with repeated measures on the second factor. Food intake was analysed using a 3 (Group) × 6 (Time) ANOVA with repeated measures on the second factor. Significance was set at P < 0.05. Significant P values were adjusted using the Huynh-Feldt correction to account for the repeated measures design. Location of significant effects was determined using Tukey's post hoc test.
Results
Exercise training
Over the course of the six-week forced treadmill running protocol, the DE group averaged a total exercise duration of 1571 ± 82 min (mean ± SD) per mouse. Considering the treadmill running speed, this translates to an average total distance run of 35,755 ± 1832 m (mean ± SD) per mouse.
Caloric intake
After six weeks, the DS group had a total caloric intake of 207.5 ± 22.0 kcal (mean ± SD) per mouse, the DE group had 223.6 ± 25.0 kcal (mean ± SD) per mouse and the NS group had 170.1 ± 17.7 kcal (mean ± SD) per mouse. Caloric intake did not differ statistically between groups.
Weight gain
A significant group × time interaction was found for body weight gain (Figure 2, F = 6.38, P = 0.005). Significant group differences were apparent beginning at week 3. The greatest difference occurred at week 6, where both the DS (18%) and DE (17%) groups were significantly heavier than the NS group. Compared with the respective baseline values, body weight significantly increased for the DS (45%), DE (43%) and NS (23%) groups. No significant differences were observed between the DS and DE groups (1.5% difference based on final weight gain) at any time point.
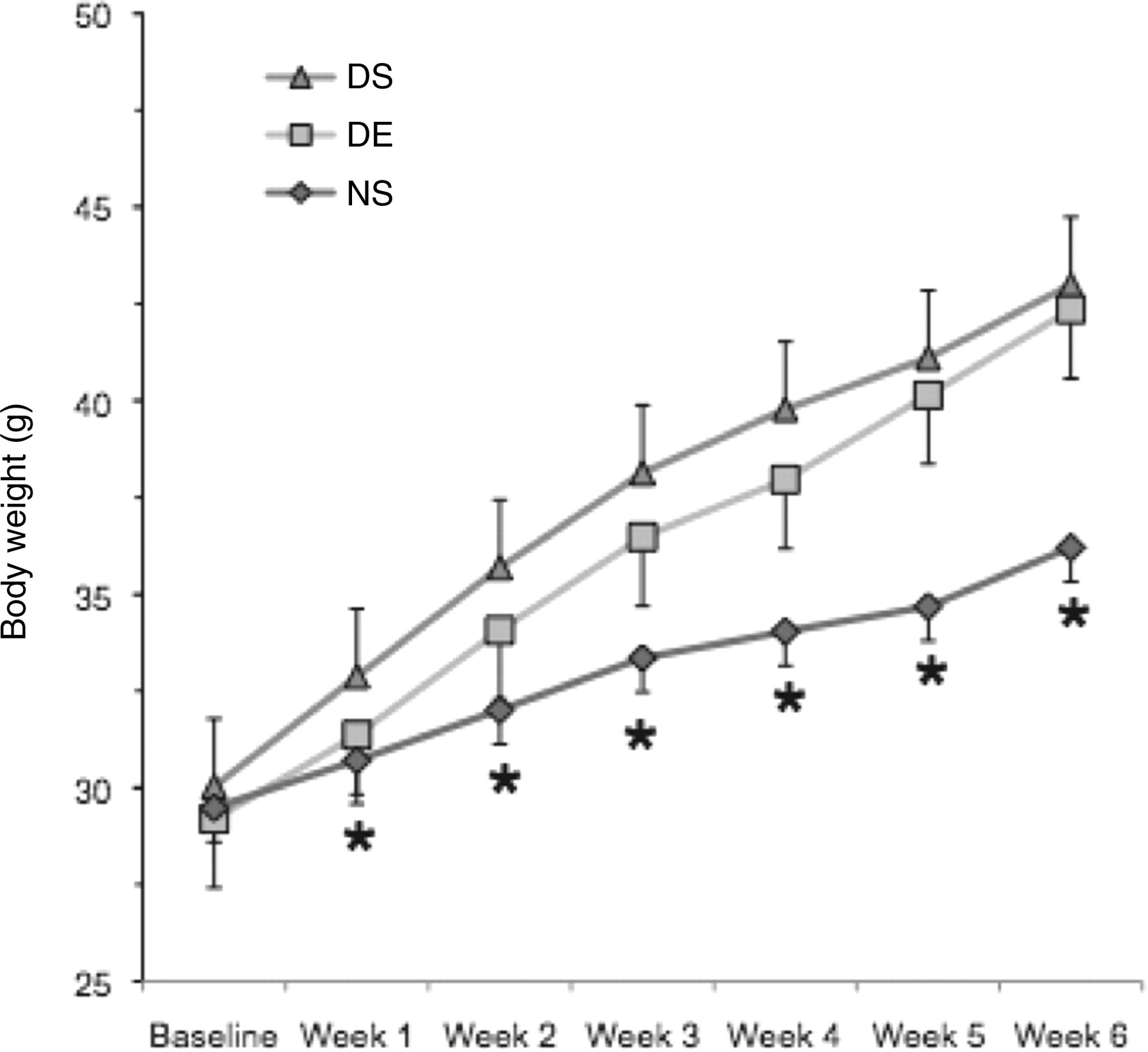
Weekly group weights. Body weight was measured on a weekly basis using a digital scale. The DS group remained sedentary and consumed a 60% fat diet (n = 12), the DE group ran a total distance of 35,755 m and ate a 60% fat diet (n = 12), and the NS group remained sedentary and consumed a 10% fat diet (n = 11). Values are mean + SE. *Indicates significantly less than the DS and DE groups (group × time interaction; P < 0.0001). DS: diet-induced weight gain, sedentary; DE: diet-induced weight gain, forced treadmill running; NS: normal weight gain, sedentary
Monocyte concentration
Representative plots were generated to demonstrate negative and positive staining for monocytes and TLR4 (Figure 3). A significant time effect was found for monocyte concentration (CD11b + /14 +) (Figure 4, F = 4.59, P < 0.001). Starting at week 2, there were more monocytes in all three groups than the respective baseline samples. Over the course of the six-week study, the average response across all groups was a 41% increase in monocyte concentration.
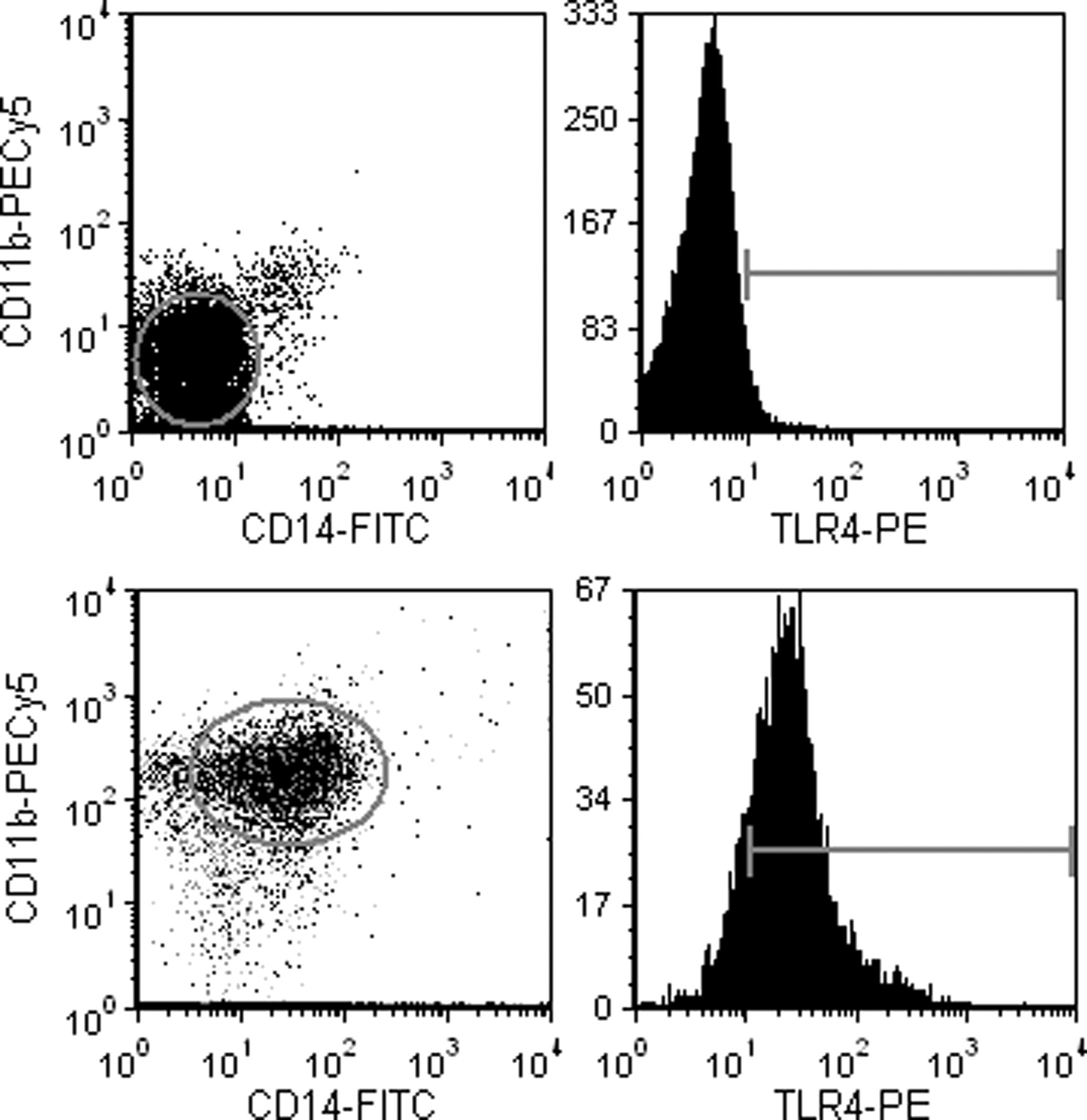
Representative flow plots. The upper two plots represent data collected for a representative negative control (note that negative populations are within the 1st log decade on each plot). The two lower plots represent a positively stained cell population. Monocytes expressed CD11b (between 2nd and 3rd log decade) and CD14 (between 1st and 2nd log decade). TLR4 cell-surface expression for positively stained cells was between the 1st and 3rd log decade. The presented plots had greater than 90,000 events. TLR4: toll-like receptor 4
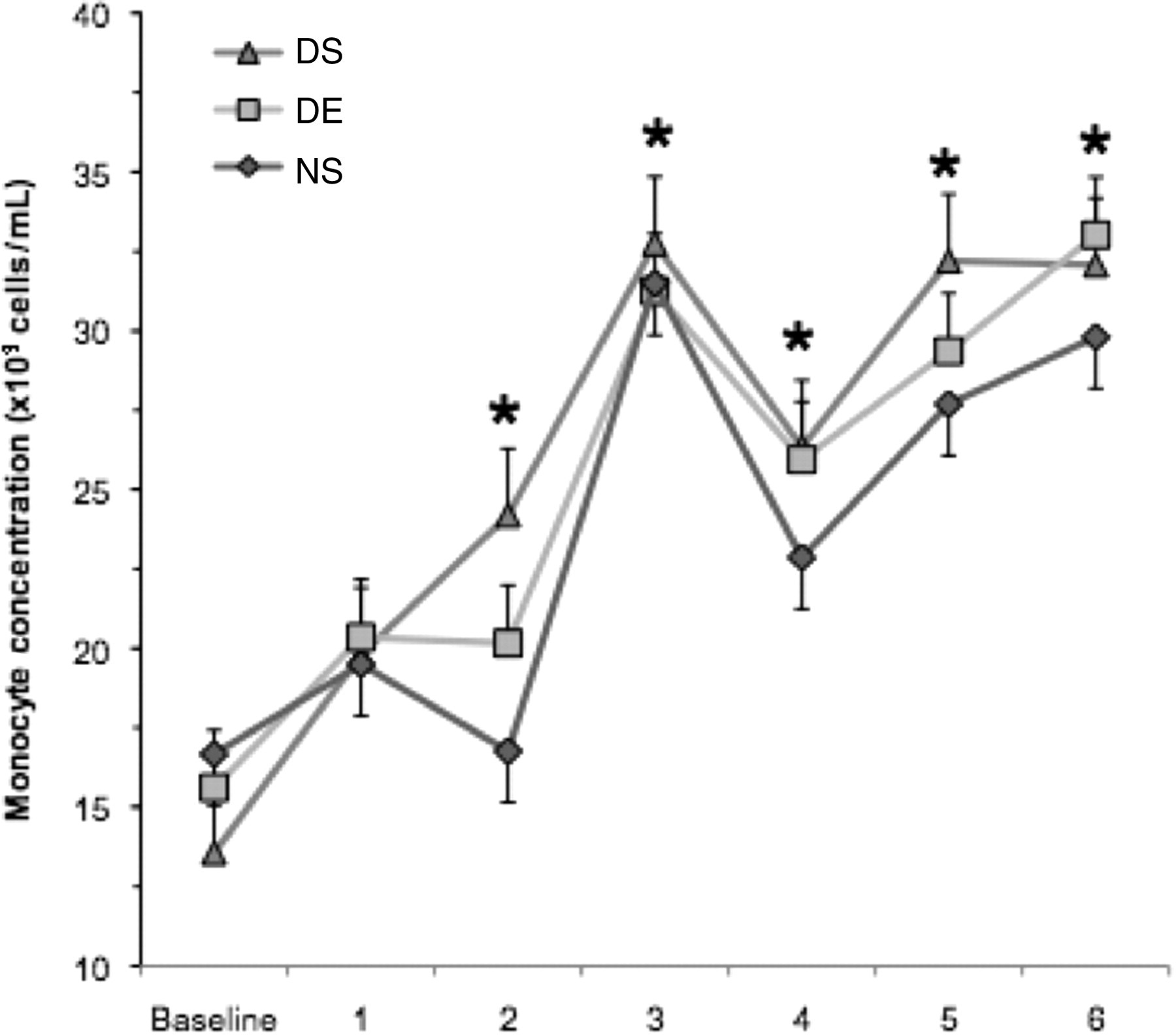
Monocyte (CD11b + /CD14 +) concentration. Measurements were made on weekly blood samples collected from the saphenous vein. Monocyte concentration was determined by two-colour flow cytometry. The DS group remained sedentary and consumed a 60% fat diet (n = 12), the DE group ran a total distance of 35,755 m and ate a 60% fat diet (n = 12), and the NS group remained sedentary and consumed a 10% fat diet (n = 11). Values represent the mean ± SE. *Indicates significantly greater than baseline (time effect; P < 0.001). DS: diet-induced weight gain, sedentary; DE: diet-induced weight gain, forced treadmill running; NS: normal weight gain, sedentary
Monocyte TLR4 expression
A significant group effect was found for monocyte TLR4 gMFI (Figure 5, F = 7.83, P = 0.005). The DS group expressed 17% more TLR4 than both the DE and NS groups. The DE and NS groups were not significantly different from each other. No other significant differences were observed.
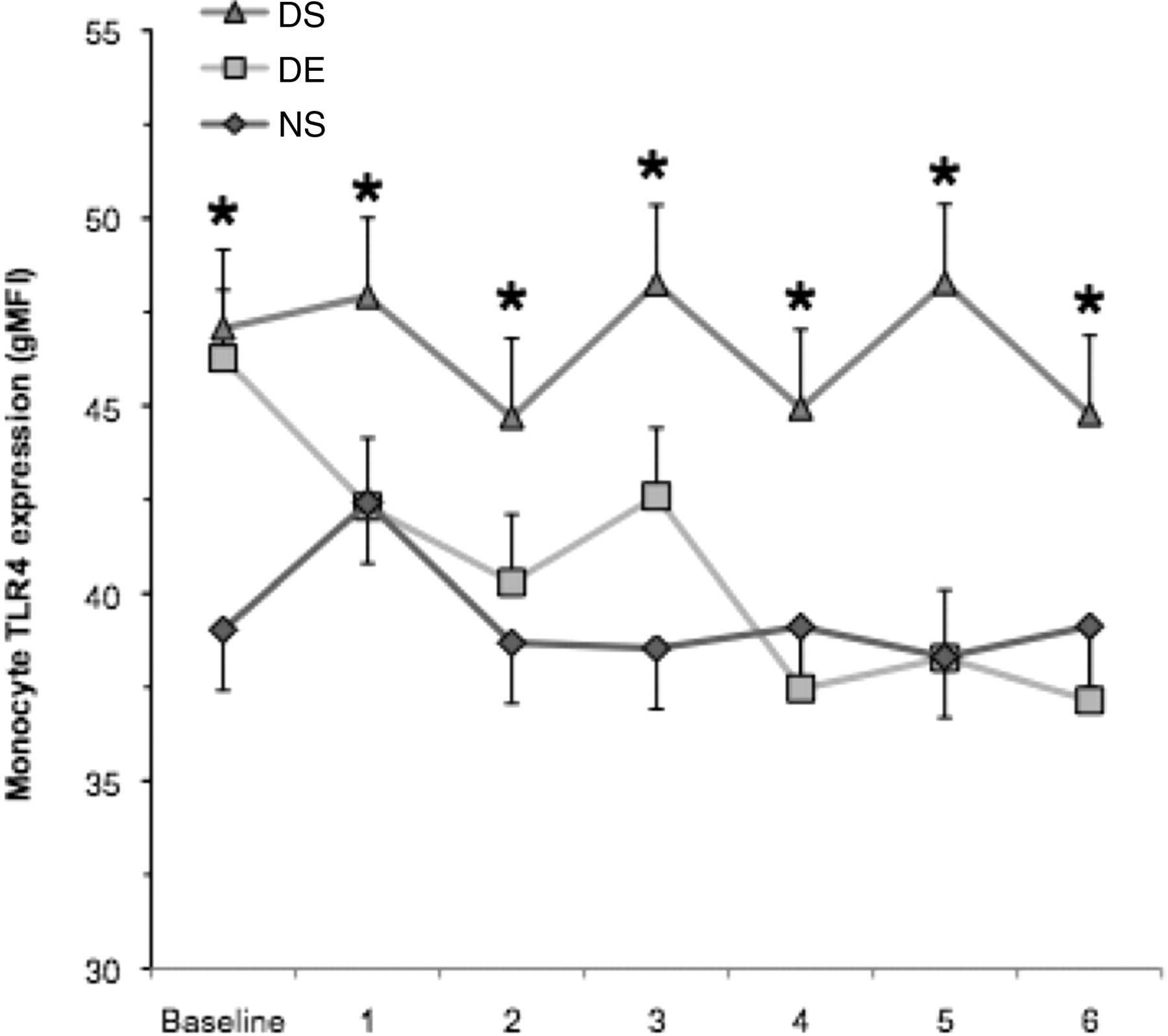
Monocyte cell-surface TLR4 expression. Measurements were made on weekly blood samples collected from the saphenous vein. Monocyte TLR4 expression was determined by three-colour flow cytometry. The DS group remained sedentary and consumed a 60% fat diet (n = 12), the DE group ran a total distance of 35,755 m and ate a 60% fat diet (n = 12), and the NS group remained sedentary and consumed a 10% fat diet (n = 11). Values are mean ± SE. *Indicates significantly greater than the DE and NS groups (group effect P = 0.005). DS: diet-induced weight gain, sedentary; DE: diet-induced weight gain, forced treadmill running; NS: normal weight gain, sedentary; TLR4: toll-like receptor 4
Discussion
The key finding of the present study was that forced treadmill running during a period of diet-induced weight gain did not mitigate weight gain or an increased monocyte concentration, but was associated with reduced monocyte TLR4 cell-surface expression. Reduction in monocyte TLR4 expression may be indicative of improved immune system function. To our knowledge, this was the first study to evaluate forced treadmill running as a countermeasure to innate immune dysfunction caused by diet-induced weight gain. While we acknowledge that more work is needed to further describe the longitudinal, survival model used in the present study, we believe that our approach may change the way that other investigators use mouse diet-induced weight gain models. A major advantage of our model is that it requires substantially fewer animals than a comparable cross-sectional sacrifice model.
In our study forced treadmill running did not prevent diet-induced weight gain. The literature in this area is somewhat inconsistent due to the use of many different exercise models. Bhattacharya et al. 18 reported that BALB/c mice that consumed a 35% fat diet and completed 14 weeks of forced treadmill running (total distance: 40,360 m) gained significantly less body weight than sedentary mice (5.8% compared with 9.1%). While the total distance exercised between our study and that of Bhattacharya et al. was similar, the duration of the study and dietary fat content differed. In a related study, Shimotoyodome et al. 19 reported that in C57BL/6J mice, 15 weeks of forced treadmill running (total distance: 39,375 m) prevented weight gain when consuming a 30% fat diet compared with a sedentary group (∼4% difference). In comparison, the present study included a treatment that was half as long and a dietary fat content that was twice as high. Of these design differences, it is likely that the high dietary fat content used in the present study created an energy imbalance that simply could not be overcome by exercise, thus translating to an increase in body weight. Typically diet-induced weight gain models use a diet, which is at least 45% fat and thus it is possible that previous studies were not completed under the same auspices as the present study.
Excessive weight gain is purported to cause innate immune system dysfunction, which can be tracked using a variety of measures. For the present study we chose to examine peripheral blood monocyte concentration and TLR4 expression because it has been a previous focus of our laboratory and necessary samples could be collected such that the animals survived. Similar increases in monocyte concentration were observed in all groups over the course of the study. Numerically the DS and DE groups appeared to be greater than the NS group at weeks 4–6; however, this effect did not reach statistical significance. The response we observed is not consistent with what others have reported regarding diet-induced weight gain. 20,21 It is plausible that the individual components of the high-fat diet may have exerted an effect independent of the energy content of the food. Unfortunately, the present study was not designed to evaluate the effect of individual dietary components. One limitation of our monocyte measurement technique is that we did not examine changes in classic and non-classic monocyte subsets, which should be a focus of future investigations.
While we did not observe a group difference for monocyte concentration, we did for monocyte cell-surface TLR4 expression. This finding is particularly interesting because diet-induced weight gain in the absence of exercise appears to maintain the TLR4 expression at a high level compared with the group that exercised while undergoing diet-induced weight gain. This protective effect occurred even in the presence of similar amounts of diet-induced weight gain. Also, the response in TLR4 expression for the DE group mirrored the response for the NS group. Thus, despite no group difference in monocyte concentration, a reduction in TLR4 expression as a result of exercise could be viewed as a protective effect that preserved innate immune function. The TLR4 exercise response observed in the present study is consistent with what we have previously reported in exercising humans. 6,7,22 To date the effect of exercise on mouse monocytes has not been extensively studied. Interestingly, the exercise effect was observed early in the training programme (at week 1), which leads us to speculate that forced treadmill running had a strong immune boosting effect demonstrated by altered monocyte TLR4 cell-surface expression. Also, it is possible that the exercise effect may have been even more pronounced if we had limited caloric intake.
There has been some debate in the scientific literature regarding how exercise-training programmes should be administered in mice. Specifically, it has been argued that voluntary exercise is more effective at reducing body weight than forced exercise. When allowed free access to a running wheel, C57BL/6 mice will exercise an average of 10,000 m/day. 23 Compared with humans, this volume of exercise would be considered extreme; however, the activity patterns of humans and rodents differ significantly. Bradley et al. 24 reported that C57BL/6 mice that consumed a 45% fat diet and completed six weeks of voluntary wheel running (total distance: 143,460 m) gained 9% less body weight than sedentary controls. In addition to the voluntary running, a key design difference between the study of Bradley et al. and the present study is that they included a four-week sedentary high-fat feeding period prior to the six weeks of exercise. This ‘prefeeding’ period essentially created an increased body weight that the mice had to attempt to overcome by voluntary wheel running. The present study only used 27% of the running distance that Bradley et al. documented. By calculation, if our mice had exercised a distance of 143,460 m, it is possible that we would have observed a 6% difference in body weight between the DS and DE groups, which may have been in agreement with the findings of Bradley et al. 24 Single exercise sessions do not disrupt monocyte concentration or TLR expression in mice. 25 More research is needed to compare voluntary and forced exercise in mice to determine if one is more effective at minimizing weight gain and immune system dysfunction.
In summary, forced treadmill running during six weeks of diet-induced weight gain decreased blood monocyte TLR4 cell-surface expression, but did not alter monocyte concentration or body weight gain. To our knowledge, we are the first to use a repeated-measures longitudinal model to examine changes in monocyte concentration and TLR4 cell-surface expression in response to diet-induced weight gain with and without forced treadmill running. In traditional experimental designs, it is common to sacrifice the animal to collect tissues and while this may be useful for certain scientific aims, it fails to provide survival analysis in longitudinal designs. Given the short duration of the present study, additional studies are needed to determine the long-term validity of our survival approach. One of the most compelling reasons to utilize a repeated measured model was that it significantly reduces the number of animals that must be used, while significantly expanding the volume of data that can be collected. More research is needed to fully understand the time course of change in monocyte functional capacity in response to diet-induced weight gain and/or exercise training.
Footnotes
Acknowledgements
This study was funded in part by a grant from the UH College of Education Faculty Research Grant Award Program (McFarlin) and a Texas ACSM Student Development Grant Award (Esposito). The authors did not have any conflict of interest associated with the present study. Esposito was the lead author and organizer of this study. Simpson was a senior author who assisted in study design and data analysis. Strohacker and Carpenter assisted in data collection/analysis and manuscript preparation. McFarlin was the senior/corresponding author and oversaw all aspects of the study.
