Abstract
The single bile duct cannulated rat model was used for short-term intestinal absorption studies. Rats were randomly assigned to three groups: (A) bile duct cannulated, (B) control laparotomy and (C) no treatment. The body weight and health of the rats were monitored before and until day 5 after surgery, while bile flow was measured in group A on day 2. On the fifth day, tail vein blood was harvested, and alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase and total bilirubin were quantified. Analysis of variance showed no significant difference between the groups for these parameters. This study demonstrated the suitability of a surgical rat model feasible for evaluation of the impact of bile in pharmacokinetic studies.
Keywords
The use of animal models in biopharmacy and pharmacology can provide important knowledge about the preliminary pharmacokinetics and metabolism of new chemical entities, as well as detailed information about the influence of various biological aspects of absorption from different pharmaceutical formulations. Bile duct cannulated (BDC) rats may yield valuable information about the oral absorption of hydrophobic drugs, since endogenous bile salts and lipids facilitate their solubilization in mixed micelles. 1–3 Many different techniques have been reported in the literature for bile collection in rats. These include single cannulation, where the bile is drained out of the rats, 4,5 and double cannulation with intact enterohepatic circulation. 6 Variations include exteriorization of the catheter through the neck, 7 skull, 8 flank 9,10 or tail 11 of unrestrained conscious animals. 12,13 The present study describes a simplified and refined modification of the unrestrained freely moving single BDC rat model for short-term studies. 5,14 Blood sampling in this model makes it feasible for intestinal absorption studies.
Male Spraque-Dawley rats (Crl:CD, 225–250 g) were obtained from Charles River (Sulzfeld, Germany). Animals were acclimatized for at least five days in groups of 2–4 on wooden bedding (Tapvei, Kortteinen, Finland) in plastic cages, 595 × 380 × 200 mm, with a stainless-steel grid (Scanbur, Sollentuna, Sweden) in an air-conditioned building with controlled environmental parameters (relative humidity 50 ± 10%, temperature 20 ± 1°C, light 06:00–18:00 h). The animals were provided with enrichment including aspen wooden chew blocks, polycarbonate shelters and nesting materials all from Scanbur. Animals had ad libitum access to pellets (Altromin 1324; Altromin Spezialfutter, Lage, Germany) and fresh tap water.
The protocol was approved by the Animal Welfare Committee, appointed by the Danish Ministry of Justice, and all animal procedures were carried out in compliance with EC Directive 86/609/EEC, with Danish law-regulating experiments with animals and with the NIH guidelines on animal welfare. Animals were randomly assigned to one of the three groups (4 rats per group) and underwent either bile duct cannulation (group A), control laparotomy (group B) or no treatment (group C). SigmaStat for Windows version 3.5 (Systat Software Inc, Richmond, CA, USA) was used for the statistical calculations. Differences between groups were assessed using one-sided analysis of variance, where P ≤ 0.05 was statistically significant.
Prior to surgery, a cannula was fashioned from 35 cm BPE-T50 tubing (Instech Laboratories, Plymouth Meeting, PA, USA). Two beads, 2 mm 18G feeding tube (Instech Laboratories), were placed 5–7 mm and 50 mm from the bevelled end and sealed with cyanoacrylate (Dana lim, Køge, Denmark). The cannula was bent sharply in boiling water 3–4 cm from the bevelled end, and subsequently the cannula was flushed with iodine and afterwards isotonic sterile saline. Buprenorphine 0.01 mg/kg (Temgesic; Merck, Whitehouse Station, NJ, USA), carprofen 5 mg/kg (Rimadyl; Pfizer, New York, NY, USA) and enrofloxacin 10 mg/kg (Baytril; Bayer, Leverkusen, Germany) were administered subcutaneously preoperatively. Anaesthesia was induced with ∼4% isoflurane/oxygen via a nose cone and the skin shaved followed by disinfection with 70% ethanol. A 1:1 mixture of long-acting bupivacaine (Marcaine; AstraZeneca, London, UK) and short-acting lidocaine (Xylocaine; AstraZeneca) was infiltrated at the surgical side (∼0.1 mL/cm). The prepared animals were placed on a CMA1500 temperature control heating mat (CMA Microdialysis, Solna, Sweden) under isoflurane (Schering-Plough, Uxbridge, UK) (∼2%)/oxygen anaesthesia and covered with sterile drape. Body temperature (thermometer probe in colon [CMA Microdialysis]) and the stage of general anaesthesia were monitored continuously during the procedure by the assessment of respiratory frequency, depth of respiration and signs of pain reaction after pinching the toes. Groups A and B underwent laparotomy, and group A was BDC (Figure 1b). The transition zone between the proximal bile duct and pancreas was ligated (6-0 silk; Ethicon, Sommerville, NJ, USA), a V-shaped hole cut above the distal ligature and the bevelled tip was gently placed in the bile duct. The suture ends were tightened around the beads, while a second proximal ligature was tied around the bile duct and the catheter. The distal and proximal suture ends were tied, and a microdrop of cyanoacrylate (Dana lim) was placed at the top of the catheter to prevent displacement. The intestine was placed in situ and the catheter ligated to muscle. Peritoneum and muscle layers were closed with a running suture (Vicryl 5-0; Ethicon) and skin using a running everted subcuticular technique (Prolene 5-0; Ethicon). A pair of pean forceps was tunnelled subcutaneously from a 3 cm incision in the neck towards the xyphoid process to guide the catheter out and through a Dacron button spring tether and a 20G swivel outlet on the lever arm (all from Instech Laboratories). The Dacron button was ligated to the neck muscles (Vicryl 5-0; Ethicon) and the skin closed by interrupted knots (Prolene 5-0; Ethicon). Another 40 cm length of polyethylene tube was connected to the swivel inlet by silicone adaptors and bile was sampled 5 cm below the cage. Operated rats were housed in individual round plastic bowls, with a diameter of 265 mm and a height of 375 mm (Scandidact, Kvistgård, Denmark), designed for free movement of the tethered rat on wooden bedding (Tapvei). Postsurgery, the wellbeing of the animals and the efficacy of subcutaneous caprofen (5 mg/kg) pain relief were monitored and additional analgesic was given if needed. The animals were controlled twice daily for clinical observations and possible infections in the wounds, without any major observations. During the recovery period, the rats had ad libitum access to pellets (Altromin 1324; Altromin Spezialfutter), apples, tap water, saltstone (Chrisco, Køge, Denamark) and isotonic NaCl with glucose (5%) (Fresenius Kabi, Bad Homburg, Germany).
Bile duct cannulation of rats. (a) Laparotomy was performed from the xyphoid and 5 cm caudally. The intestine was taken out, wrapped in sterile gaze and moistened with sterile isotonic saline. Liver lobes were displaced cranially using a gaze pack to help visualize the bile duct. (b) Schematic presentation of the abdominal section of the rat, where the laparotomy was performed. (c) Presentation showing the catheter (cannula) placed in the bile duct. (d) Schematic enlargement of the catheter placement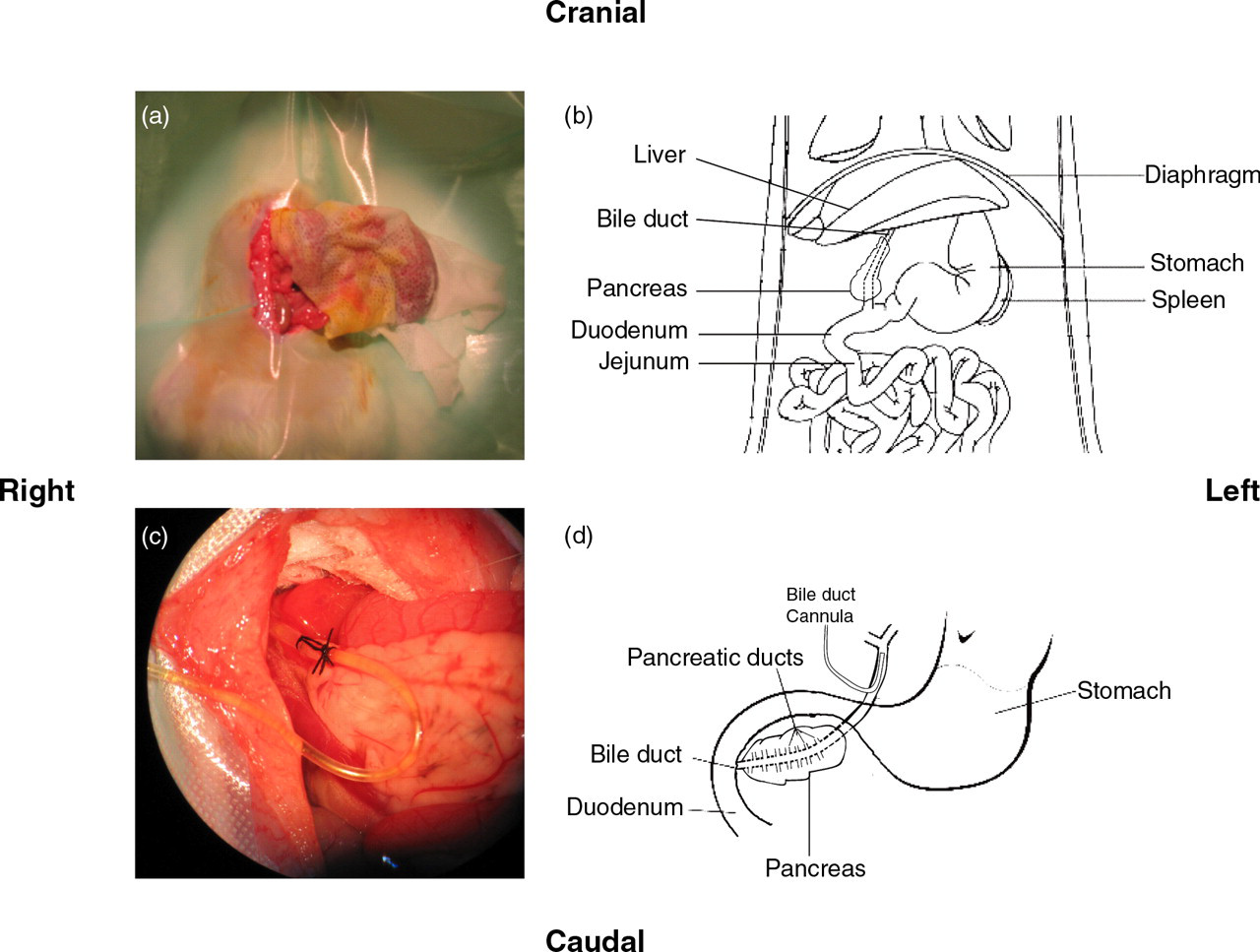
Serum biochemistry parameters for bile duct cannulated, sham-operated and intact rats five days after the surgery
Data are presented as means ± SEM (n = 4). No statistically significant differences were observed between the three groups for any of the four parameters
ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; Tbili: total bilirubin
Rats in groups A and B lost BW 1–2 days after surgery, but the rats had regained their initial BW 4–5 days after surgery. Clinical chemistry parameters are presented in Table 1. No statistically significant differences were observed between the three groups with regard to the liver parameters (ALT, AST, ALP) and total bilirubin (Table 1, P values: 0.13, 0.24, 0.81 and 0.32, respectively). 15 Bile flow in the BDC animals was continuous, with a flow rate of ∼4 μL/min/100 g, which is within the range of previously reported flow rates. 5,6
The loss of BW in groups A and B did not reach statistical significance at any time point and was regained 4–5 days after surgery, which is consistent with BW loss previously reported in BDC rats. 6,11 Surgery has a debilitating effect 11,13 and weight loss was most likely due to surgery-related stress rather than the specific surgical procedure, as demonstrated by the similarity between groups A and B. This means that the simplification of the BDC method did not compromise the wellbeing of the animals more than a simple laparotomy. Weight loss in operated rats is probably multifactorial. Surgical trauma overloads the organism and disturbs homeostasis, leading to a pathophysiological response. 16,17 These immunological and neuroendocrine responses can affect appetite following surgery, but may depend upon the time and extent of the surgery, which consequently could affect the degree of inflammatory response, postoperative pain and surgical complications. 11,17
The overall clinical appearance and BW recovery were supported by the serum measurements, where no statistically significant differences were observed between ALP, AST, ALT and total bilirubin five days after surgery, when comparing the three groups. Though not statistically different, ALP and subsidiary AST values were slightly lower in operated rats when compared with untreated animals, despite a fairly good agreement with control data intervals on fasted rats reported in the literature. 18 Postoperative variations in clinical biochemistry parameters were described by van Wijk et al. 11 who noted them to be outside normal limits in rats following surgery. Also, Faure et al. 13 reported enzyme and total bilirubin parameters outside the normal range on day two in 15–65% of rats following surgery, but approximately half of these animals regained normal values eight days postsurgery. Furthermore, deviations in liver enzymes have been reported by West et al. 19 in BDC rabbits. A major portion of serum ALP in rats originates from intestinal ALP, 20,21 which varies with food intake, particularly lipid, i.e. high food intake leads to high ALP and low intake to low ALP levels. 20,22 Lower food intake in the operated rats, and notably the bile-deficient BDC rats would have decreased intestinal absorption of lipids, which may affect intestinal and bile ALP activity and thus the total serum ALP level. Loss of ALP in bile has been reported 21,23,24 and also investigated in the rat. 25,26 The single bile duct cannulation is often used for short-term studies as continuous bile loss may affect the homeostasis. Single BDC rats have been kept for 14 days with saline replacement. 27,28 For long-term studies double cannulation of bile duct–duodenum or bile duct–bile duct may be more advantageous, since the enterohepatic circulation and bile salt pool is intact. 6,11
Sampled blood was obtained in the BDC rats by tail vein puncture. BDC rats used in pharmacokinetic studies have previously been subjected to a more invasive vascular catheter to withdraw blood. 14 Tail vein puncture was used in the present study to reduce the surgical trauma and pathophysiological response. Thus, the present BDC method has updated previously described procedures 5,14 on several points. Ether was previously used to anaesthetize the animals, whereas we used isoflurane. The present work introduced pain relief and updated the surgical method, and while Balabaud et al. 5 fixed the swivel using metal clips in the skull, we fixed it to muscle. Based upon monitoring of BW and liver parameters, these changes were shown to impact the animals at the same level as a sham operation, and hence demonstrated that the BDC model was suitable for drug absorption and pharmacokinetic studies. In comparison with previous methods, the advantages of the present method are (1) less stress for the animal, (2) optional blood sampling and (3) a higher throughput.
Footnotes
Acknowledgements
Christian J Jensen, Niels Coley and Marianne Kaarde are acknowledged for skilful technical support and assistance with the animals, respectively.
