Abstract
In animal models, devices such as indwelling catheters and intracranial cannulae are often fixed on the skull to allow sampling or injection in the freely moving animal. The most commonly used method to fixate these devices is by embedding them in a ‘helmet’ of cement which is fixed to the skull with screws. Methylmethacrylate cement is commonly used for this purpose. The disadvantages of this cement are the high polymerization temperature, poor bonding to the bone and long hardening time. We have evaluated the use of glass ionomer cement, carboxylat cement and cyanoacrylic glue as alternative for methylmethacrylate cement. Temperature increase during polymerization of methylmethacrylate cement and glass ionomer cement was measured in the cement on the skull and in the brain of 14 rats in an acute model. In a chronic model, 52 rats and 91 mice were equipped with a ‘helmet’ of one of the cements. The glass ionomer ‘helmets’ were applied without or with pretreatment of the skull. The attachment of the cement to the skull was checked every day. After four weeks the bonding strengths of the cements were measured. The glass ionomer cement had less temperature increase during polymerization and good bonding capabilities when compared with methylmethacrylate cement. Mechanical pretreatment of the skull resulted in a significant increase in bonding strength of glass ionomer cement in mice and rats as compared with chemical pretreatment. Furthermore, glass ionomer cement had a shorter hardening time than methylmethacrylate cement, and when the glass ionomer cement was used in prepacked capsules, it was possible to apply the cement sterilely and easily. Cyanoacrylic glue had good bonding capabilities to the skull of mice and is also a good substitute for methylmethacrylate cement.
Attachment of various measuring devices with screws and dental cement is a well-established technique for fixation and stabilization of these devices on the skull of laboratory animals. The technique allows undisturbed sampling and infusion in freely moving animals. The widely used application is the stabilization of intracranial cannulae such as microdialysis probes and intracerebroventricular (ICV) cannulae.1,2 Apart from stabilizing cannulae in specific brain areas, the head attachment is often directly connected to sampling tubing, requiring sufficient bonding strength to the skull. The technique is also used for the fixation of indwelling vascular blood catheters to the skull of mice 3 and rats.4,5 The catheter is inserted into a specific vein or artery and subcutaneously tunnelled to emerge at the top of the head.
In those cases where methylmethacrylate cement is used for the permanent head attachment in rats, screws are needed for the bonding strength. Methylmethacrylate cement has poor bonding capacities to bioactive surfaces such as the skull. 6 To accomplish bonding strength, two or three holes are drilled in the vicinity of the measuring device for the placement of anchoring screws. 7 Because of the less calcified and thinner skull of mice, screws are not commonly used in this species. 1 The bonding strength of the head attachment in mice thus solely depends on the bonding strength of the cement to the skull.
Although permanent head attachment with methylmethacrylate cement can be accomplished in chronic experiments in rats using additional anchoring screws, considerable dropouts can be expected when using methylmethacrylate cement in mice without the use of screws. Groseclose
We hypothesized that glass ionomer cement has a stronger bonding strength to the skull and results in a lower temperature during polymerization compared with methylmethacrylate cement. We evaluated the temperature increase and bonding strength of glass ionomer cement in comparison to methylmethacrylate cement. Additional bonding strengths of cyanoacrylic glue and carboxylat cement were evaluated. These materials are also commonly used for the fixation of devices to the skull of laboratory animals, and it has been reported that cyanoacrylic glue is a less time-consuming alternative to dental cements. 2 Temperature increase in carboxylat cement and cyanoacrylic glue was not investigated because in these materials temperature increases were suspected to be less. 8
Materials and Methods
Experimental design
Experiments were conducted in two institutions because of the local availability of equipment. The temperature increase during polymerization of the cement was measured in rats in an acute experiment conducted at the Rudolf Magnus Institute of Neuroscience. The temperature increase was measured in the ‘helmet’ of cement and in the brain. The success rate and the bonding strength were measured in rats and mice, in a chronic experiment conducted at Solvay Pharmaceuticals.
In the acute experiment two types of cement were tested. In the chronic experiment four types of cements and four different pretreatments were tested.
Measurements of temperature increase during polymerization in an acute experiment
The temperature increase was measured in 14 rats during the polymerization of methylmethacrylate cement (Polyfast®, Dental Union, Nieuwegein, The Netherlands) and glass ionomer cement (GC Fuji Plus®, Dental Union). The animals were anaesthetized with a mixture of fentanyl and fluanisone (0.03 mL intramuscularly; Hypnorm®, Janssen Pharmaceutica, Antwerp, Belgium) and midazolam hydrochloride (0.1 mL intraperitoneally; Dormicum®, Hoffman-La Roche, Amsterdam, The Netherlands). This mixture of anaesthetic agents provided sufficient anaesthetic depth for the entire surgical procedure and for the temperature measurements that took around 30 min. When pedal and corneal reflexes were absent, the heads of the animals were shaved and disinfected using 1% chlorhexidine (Sterilon®, Boots Healthcare BV, Naarden, The Netherlands). The corneas were covered with eye protecting oil (Caf®, Ceva Santé Animale BV, Maassluis, The Netherlands), and the animals were placed in a stereotaxic frame (David Kopf®, New York, NY, USA). Body temperature was rectally monitored and controlled by an automatic regulated heating pad (35 ± 1°C). An incision of approximately 3 cm was made medial over the skull and the periosteum was removed with a scalpel. Three stainless-steel screws (3.0 x 1.2 mm2) were inserted approximately 1 mm into the skull. An ICV cannula was stereotaxically placed in the lateral brain ventricle. The guide cannula consisted of an aluminium needle (outer diameter, 0.4 mm). 7 Stereotaxic coordinates (0.92 mm posterior to the bregma, 1.50 mm lateral to the mid-line saggital suture and 3.5 mm ventral to the dura) were adapted from Paxinos and Watson 9 ; a small hole was drilled using a dental drill with a hollow 2 mm bit (Dental Union) to place the ICV cannula. The first thermometer, a 80 TK thermocouple module (Fluke, Eindhoven, The Netherlands), was attached to a multimeter (Metex®, Testlab, Seoul, Korea) and placed near the cannula parallel to the surface of the skull. A second hole was drilled 2 mm lateral to the cannula. A second thermometer probe (Radionics Neurosurgical Instruments, Burlington, MA, USA) was placed at an angle of 45° towards the guide cannula using a second stereotaxic instrument (Figure 1). The thermometer was advanced until it made contact with the cannula, which was approximately at a depth of 2 mm in the brain. The cannula and first thermometer were fixed with 1 g of cement.
Drawing of the set-up that was used to measure the temperature rise in the cement and in the brain of rats during polymerization of the cement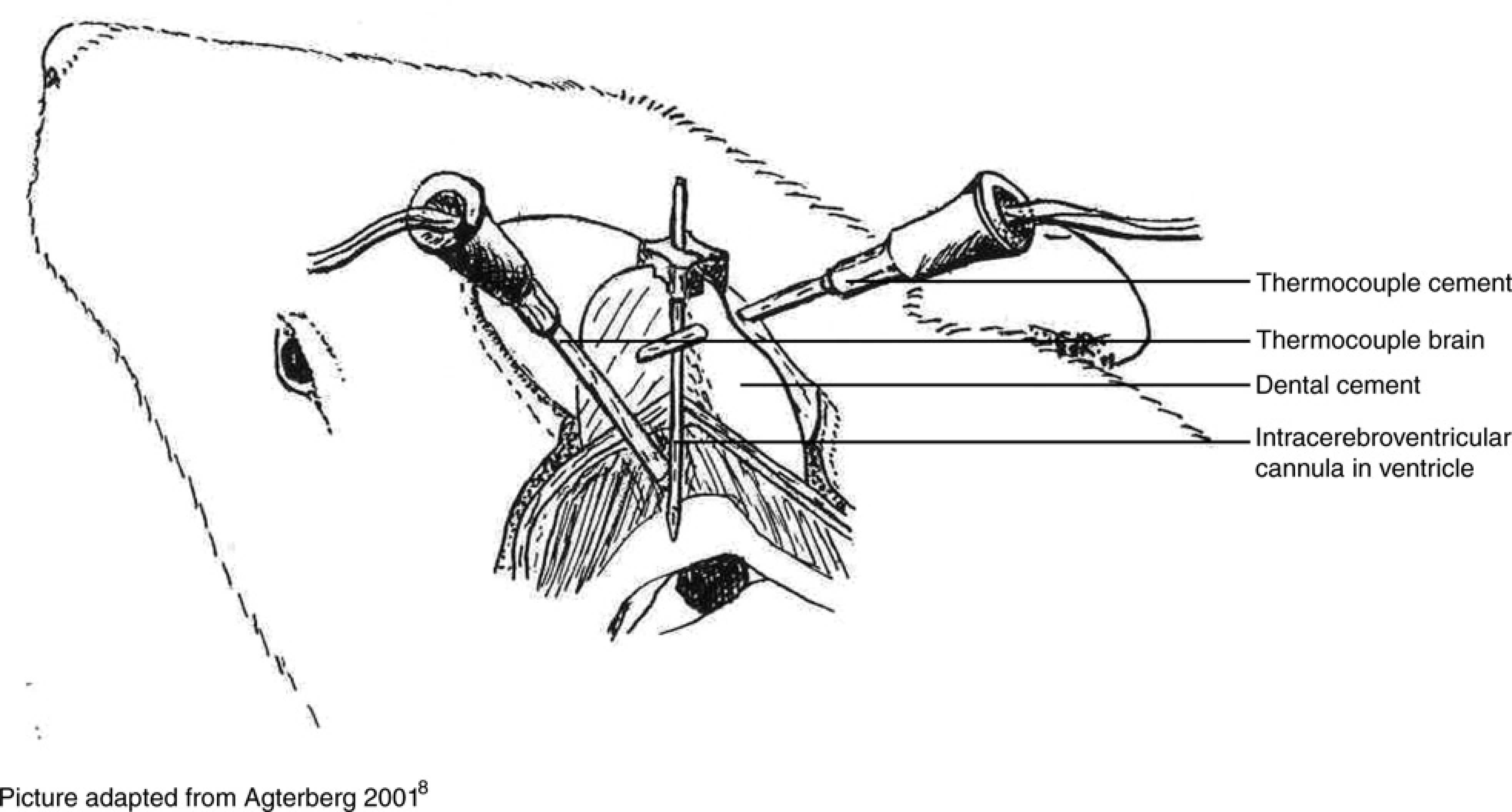
The acrylic cement powder was mixed with the methylmethacrylate solution (1:1 ratio) and applied on the skull using a spatula (rats,
Success rate and measurements of bonding strength in a chronic experiment
The success rate and bonding strength were measured in a total of 52 rats and 91 mice. In mice and in rats glass ionomer cement was tested with four different types of pretreatment of the skull. In addition three other cements were tested in mice. Methylmetacrylate cement and carboxylat cement were both tested with etching gel as pretreatment and cyanoacrylic glue was tested without pretreatment. The mice and rats were anaesthetized in an anaesthetic chamber with a mixture of isoflurane (4%) and O2. Loss of the pedal reflex was used as an index of the onset of surgical anaesthesia. The heads were shaved and disinfected with 1% Chlorhexidine solution. During the surgical procedure, general anaesthesia was maintained using an open inhalation circuit. A mixture of approximately 2% isoflurane (Forene, The Netherlands), oxygen and nitrous oxide (O2:N2O = 1:1) was directly delivered through a face mask to the animal. The corneae were moistened using eye drops (Lacriforte®, AST Farma, Oudewater, The Netherlands). Xylocaine 10% spray (Astra Pharmaceuticals, Amsterdam, The Netherlands) was applied on the skull approximately one minute before the periosteum was removed. Body temperature was rectally monitored and controlled by an automatic regulated heating pad (35 ± 1°C). An incision of approximately 2 cm was made medial over the skull. The periosteum in rats was removed using a scalpel, while cotton wool sticks were used in mice. After pretreatment of the skull, a bent 20-gauge sterile needle was cemented onto the skull for the measurement of the bonding strength. This bent needle was left
The animals were monitored for a period of four weeks. During the daily health checks, the head attachments were inspected by manually applying some force on the bent needle in the cement and visually inspecting the wound area around the cement. Animals with dislodged head attachments were taken out of the study (defined as dropouts) and, after closer inspection of the head attachment, euthanized. After four weeks the breaking force experiments were performed. Each animal was anaesthetized with CO2/O2 (0.5 L/min/1.0 L/min) and quickly decapitated. The head of the animal was fixed into the Kopf stereotaxic instrument. The 20-gauge bent needle embedded in the cement on the skull was connected to a purpose-built modified arm (plunger) of the breaking force apparatus (Schleuniger 6D, Dr Schleuniger Pharmatron AG, Solothurn, Switzerland) as shown in Figure 2. The plunger that connected the head attachment moved to the right, while the breaking force was measured by the counteracting the balance connected to the stereotaxic frame.
Apparatus (Schleuniger 6D) to measure the breaking force
Types of cement and pretreatment that were used in the chronic experiment
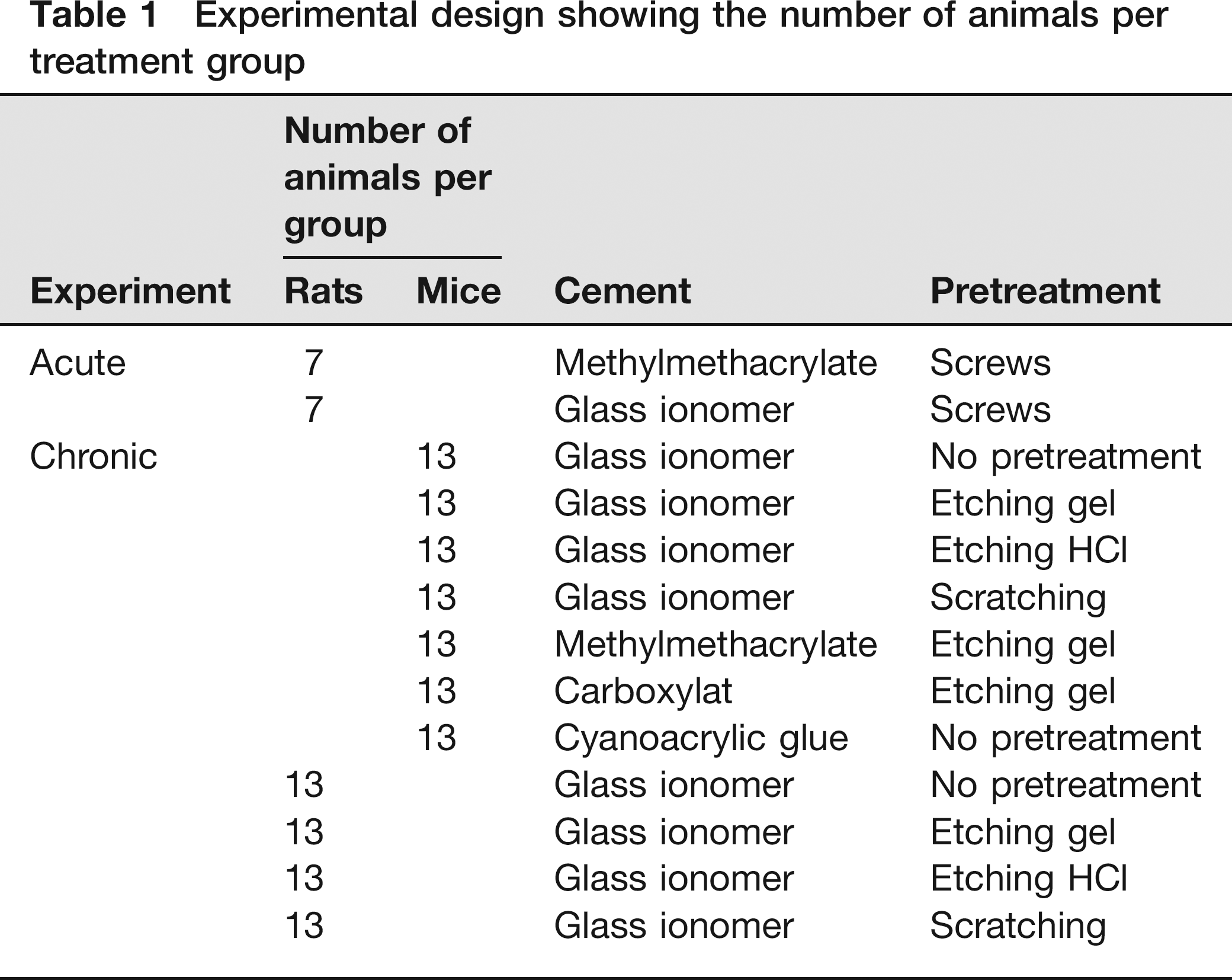
In the chronic experiment four types of cement were tested as follows: methylmethacrylate cement (Surgical Simplex®, Stryker, Newbury Berkshire, UK), glass ionomer cement (GC Fuji Plus®, Dental Union), carboxylat cement (Dentalon®, Henry Schein-Van den Braak BV, Utrecht, The Netherlands) and cyanoacrylic glue (Loctite® 545, Lochte Nederland BV, Nieuwegeim, The Netherlands). Subsequently, for glass ionomer cement four different pretreatments were tested as follows: no pretreatment, etching gel, etching with 1 mmol/L HCl and scratching.
In the condition without pretreatment, the skull was rinsed with saline after all bleeding had stopped. The etching gel contained 38% phosphoric acid (Dental Union). About 30 s after etching with either etching gel or 1 mmol/L HCl, the skull was rinsed with saline. In the condition with pretreatment scratching, scratches were made in the skull with a scalpel. Scratches had the shape of a chess-board. The different cements and pretreatments are listed in Table 1.
Experimental design showing the number of animals per treatment group
Although it is known that the bonding capacity of methylmethacrylate cement and carboxylat cement to bioactive surfaces such as the skull is low, 6 we tested these cements without the use of screws in mice. In rats, the success rate and bonding strength was only tested without the use of anchoring screws when glass ionomer cement was applied.
Animals
Animals used at the Rudolf Magnus Institute for Neuroscience were male Wistar/Crl:WU outbred rats (Charles River, Sulzfeld, Germany) weighing between 300 and 350 g. Before the experiment animals were housed socially in Makrolon cages measuring 425 x 266 x 180 mm (1 x w x h) at a controlled temperature between 21 and 23°C, relative humidity between 47% and 63% and a light/dark cycle of 12/12 h. Standard food pellets (RM1 (E), Special Diet Services, Witham, Essex, UK) and tap water were provided
Animals used at Solvay Pharmaceuticals were male C57BL6/J01aHsd inbred mice (Harlan, The Netherlands) weighing between 25 and 30 g, and male HsdHan™. WIST outbred rats (Harlan, Horst, The Netherlands) weighing between 300 and 350 grams (Harlan, The Netherlands). Mice were housed individually in Makrolon cages measuring 267 x 207 x 140 mm (1 x w x h) (Type II cages, Tecniplast, Exton, PA, USA) with wood chips bedding (Harlan Teklad, Horst, The Netherlands). Rats were housed individually in Makrolon cages measuring 425 x 266 x 155 mm (1 x w x h) (Type III cages, Tecniplast) with wood chips (Harlan Teklad). All animals were subjected to a 12 h dark/12 h light cycle, with water and food (Harlan Teklad) given
Statistics
Bonding strength data and temperature data are presented as mean ± standard deviation (SD). Statistical analysis included a multivariate analysis of variance, with Scheffe subgroup testing and a
Results
Figure 3 clearly shows the difference in temperature change on the skull of rats during polymerization of glass ionomer cement and methylmethacrylate cement. Seventy seconds after starting the temperature measurements, a significant difference in temperature between glass ionomer cement and methylmethacrylate cement was evident (
Temperatures (°C) on the skull of rats during polymerization: glass ionomer cement (▪) versus methylmethacrylate cement (▴). Data are expressed as mean ± standard deviation. *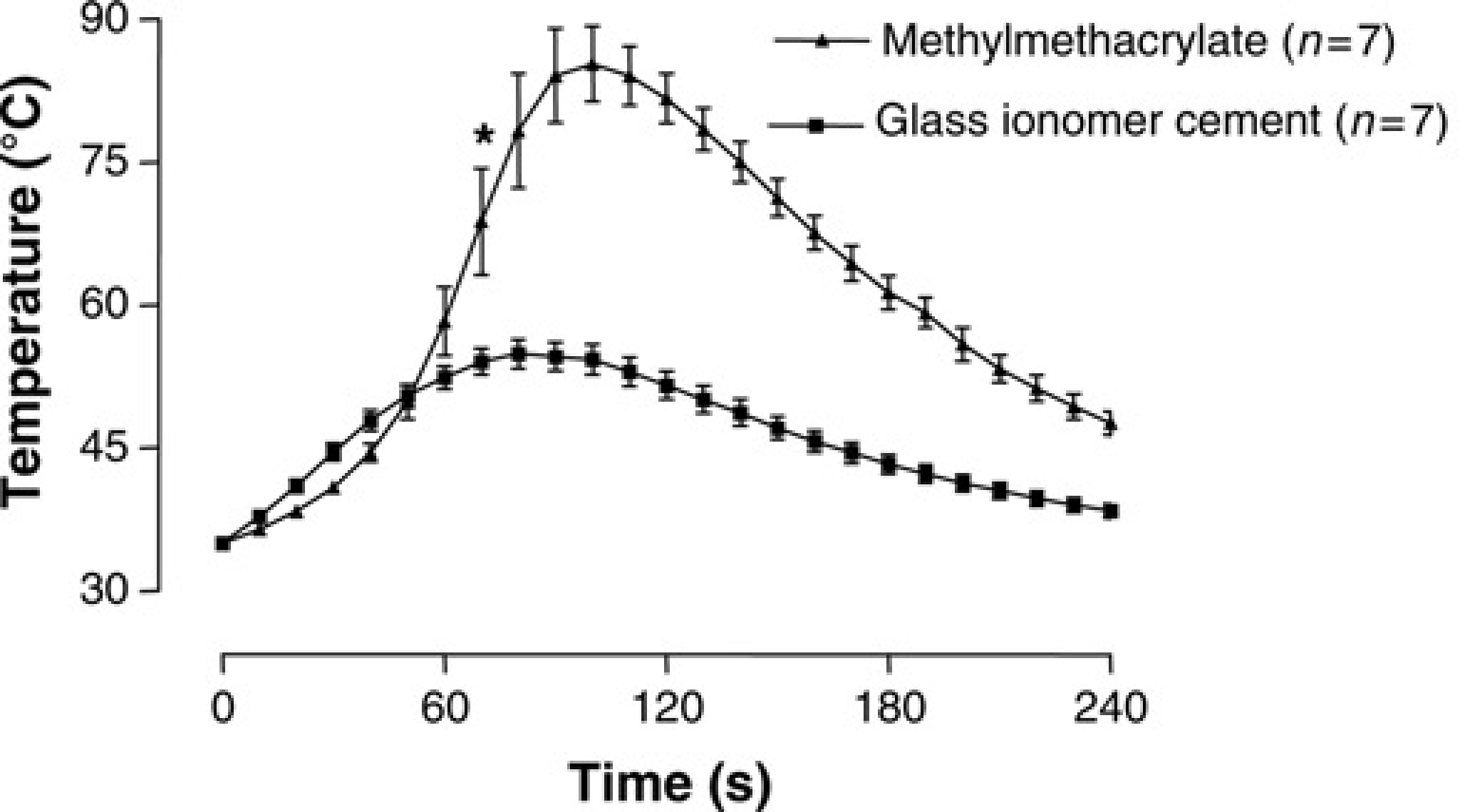
In the brain, the temperature increased with approximately 5°C during polymerization of the cements, as is shown in Figure 4. Although a significant temperature difference developed after 160 s, the maximum temperatures reached in the brain were not significantly different (
Temperatures (°C) in the brain of rats during polymerization: glass ionomer cement (▪) versus methylmethacrylate cement (▴). Data are expressed as mean ± standard deviation. *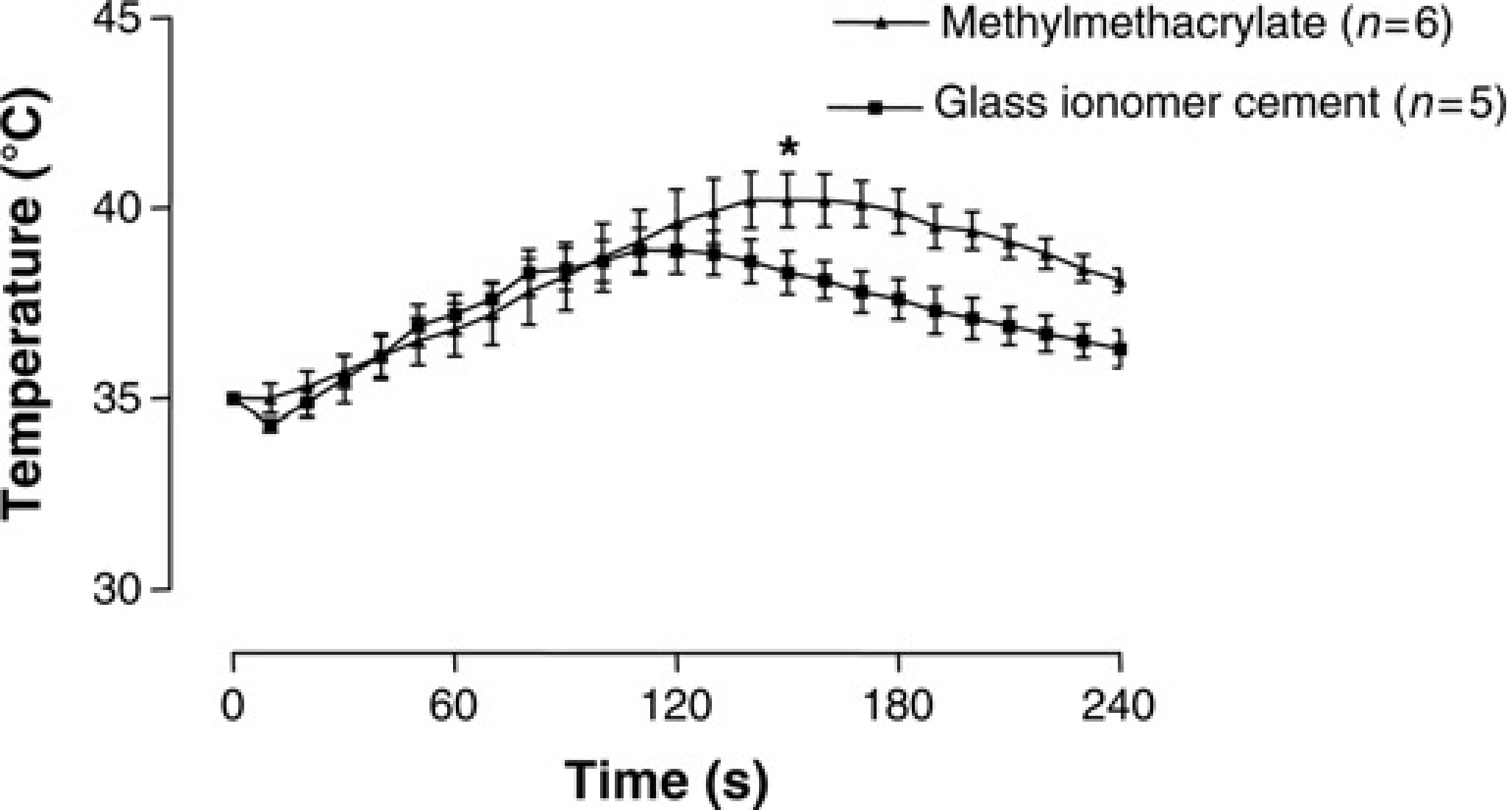
Figure 5 shows the success rate during the four-week postoperative period of four different cements in mice with etching as pretreatment. In the glass ionomer cement group four animals lost their head attachment. In the cyanoacrylic group two animals lost their head attachment. In the methylmethacrylate group 10 animals lost their head attachment and in the carboxylat group all animals eventually lost their head attachment during the four-week period. The glass ionomer cement was also applied without pretreatment, after etching with HCl and after scratching the skull (data not shown). In these groups of animals all head attachments remained attached to the skull for the entire four weeks.
Success rate in mice during postoperative period of four weeks: comparison of cyanoacrylic glue (▪), carboxylat cement (•), methylmethacrylate cement (▴) and glass ionomer cement (▾)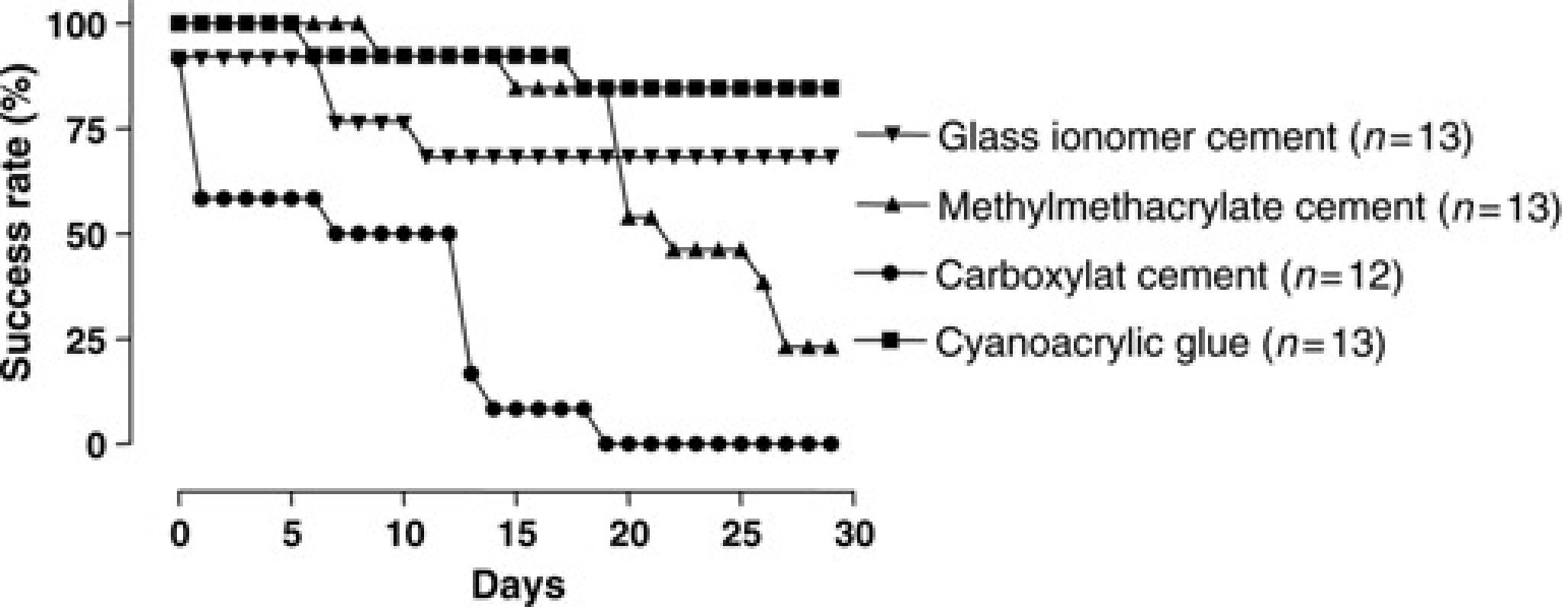
Figure 6 shows the breaking force of glass ionomer cement after different pretreatments in mice. Compared with the group that received no pretreatment, etching the skull with 1 mmol/L HCl and scratching significantly increased the breaking force (
Breaking force measurement (N = Newton) to dislodge the head cap in mice, comparing different pretreatments using glass ionomer cement as cement: no treatment, etching gel, etching (1 mmol/L HCl) and scratching. Data are expressed as mean ± standard deviation. *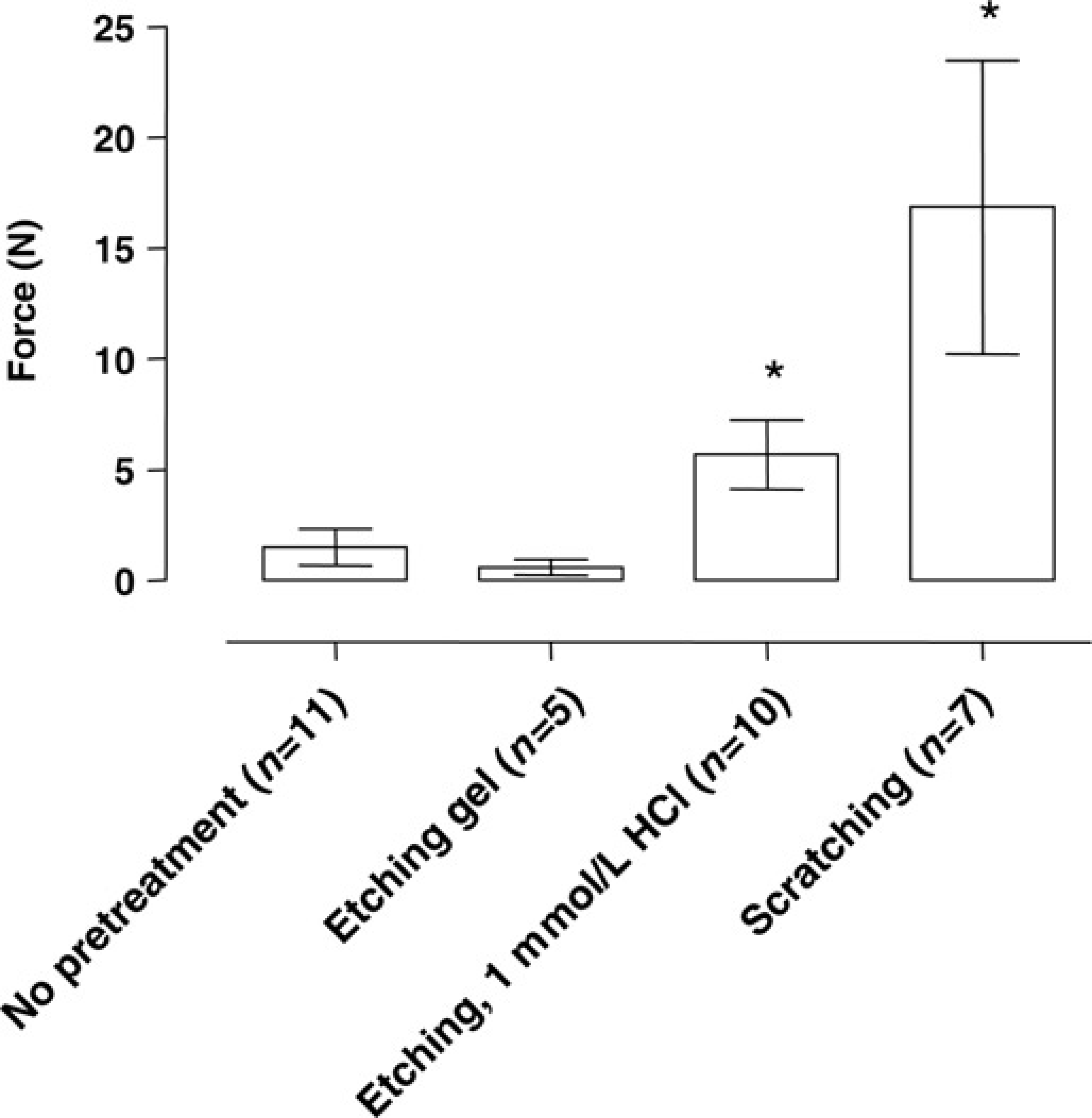
The number of intact head attachments in the methylmethacrylate group (
Figure 7 shows the breaking force of cyanoacrylic glue (24.8 ± 5.0 N,
Breaking force measurements (N = Newton) to dislodge the head cap in mice after a period of four weeks postoperative when cyanoacrylic glue was used after scratching as pretreatment of the skull. Data are expressed as mean ± standard deviation. The dotted line indicates the breaking force of glass ionomer cement after scratching as pretreatment. Breaking force measurements were not performed in the carboxylat group because all animals in this group lost their cement ‘helmets’. *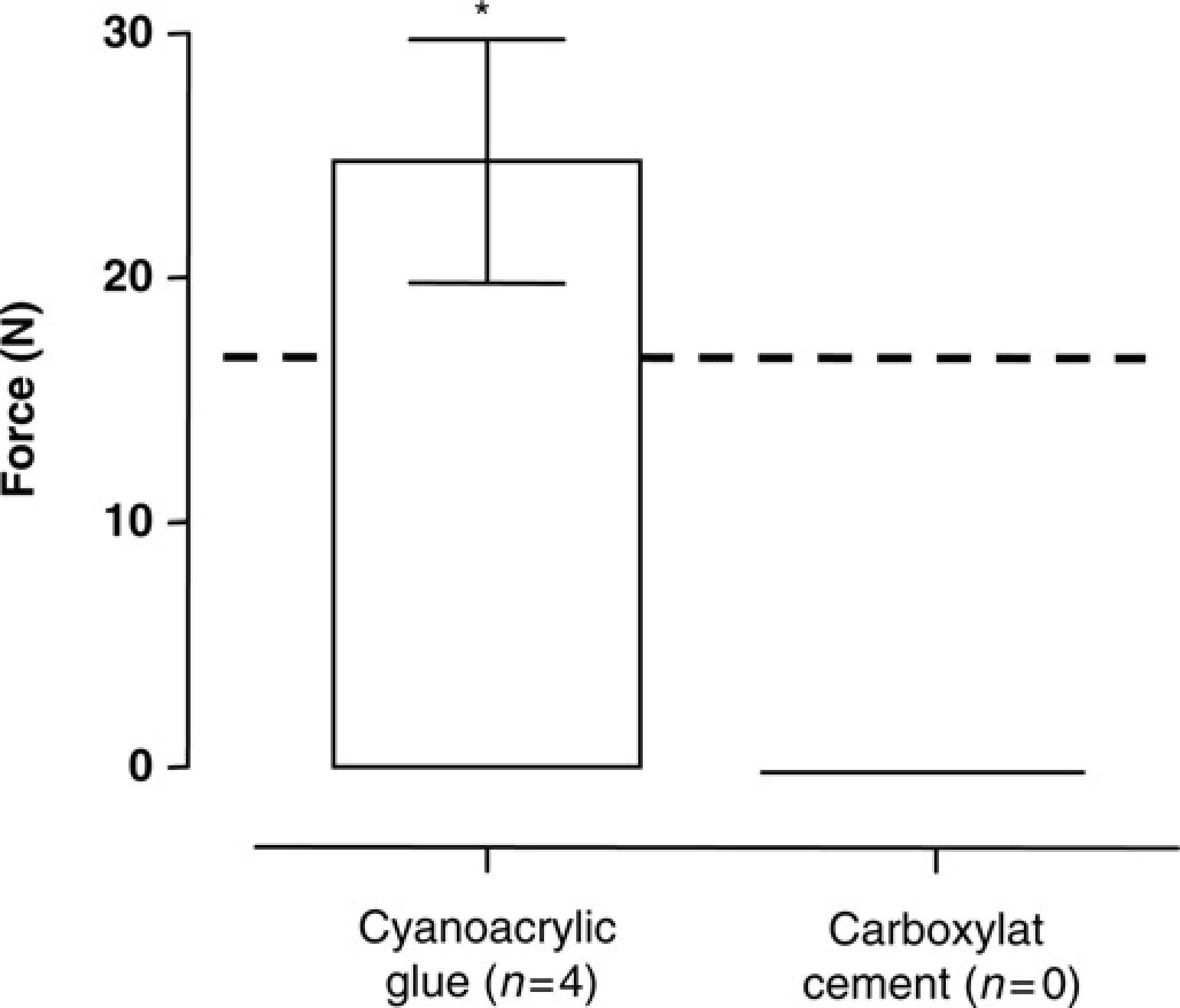
In rats, the differences in success rate between the pretreatments of the skull were not as evident as in mice. In the last two days of the four-week postoperative period, one rat in the ‘no pretreatment’ group lost its head attachment. In the ‘etching group’ three rats lost their head attachment on the last day of the four-week period. The head attachments of the ‘1 mmol/L HCl’ group and the ‘scratching group’ remained intact during the entire four-week period. We have also tested three group of rats (data not shown) that were pretreated by the placement of three anchoring screws. In these groups none of the animals lost their head attachment during the four-week period.
Figure 8 shows the breaking force of glass ionomer cement after different pretreatments in rats. Breaking force measurements were not performed in rats carrying anchoring screws, since the screws had a leverage effect in our set-up (set-up is shown in Figure 2), thereby making representative measurements impossible. The pretreatments had no significant effect on the breaking force (
Breaking force measurement (N = Newton) to dislodge the head cap in rats with different pretreatments and using glass ionomer cement as cement after a postoperative period of four weeks. Data are expressed as mean ± standard deviation. *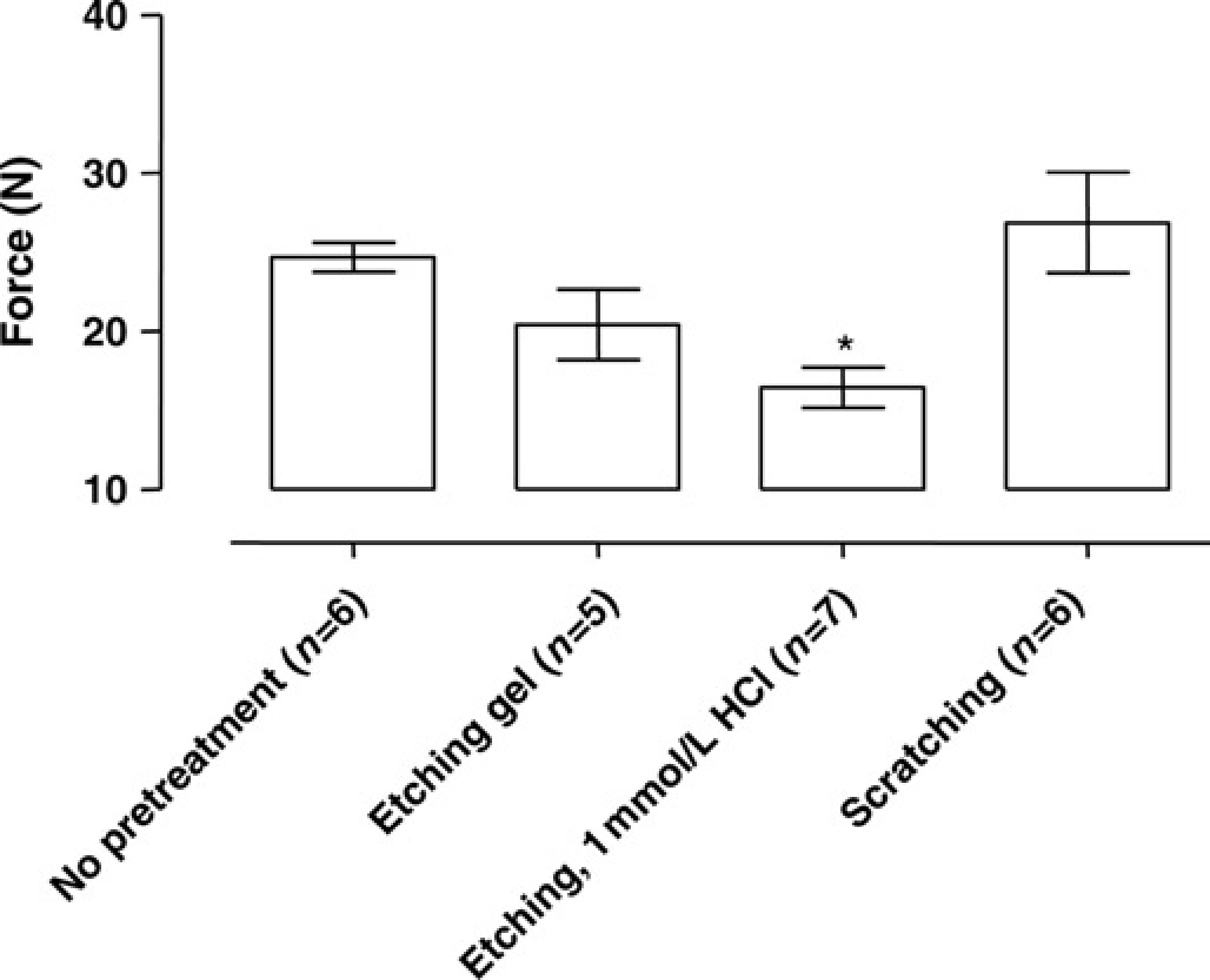
Discussion
The present study showed that the temperature during polymerization of cement on the skull of rats remains significantly lower when glass ionomer cement was used instead of methylmethacrylate cement. Notably, the temperature in ‘helmets’ built with glass ionomer cement was 31.6°C lower than in ‘helmets’ built with methylmethacrylate cement. In this study postoperative wound inspection did not show any signs of necrosis. However, histological evaluation of surrounding tissues, especially in the methylmethacrylate group in which the temperature during polymerization of the cement was more that 85°C, is indicated to investigate the effects of this extreme temperature rise. In the brain the temperature increased during polymerization of both methylmethacrylate and glass ionomer cement. Although we recorded a significant difference in temperature 160 s after we started to measure the temperature (Figure 4), we found no significant difference in maximum temperature. We did not perform temperature measurements in the brains of mice. Because the skull of mice is thinner, it is likely that the temperature increase in the brain of mice is more pronounced. On the other hand, the temperature rise in the brain might be less because in mice less cement is used for the fixation and stabilization of the devices to the skull. Also in mice histological evaluation is indicated to investigate a possible infection or inflammatory response due to the materials used. 1
Because the skull of mice is too thin to support anchoring screws, the breaking force is merely dependent on the bonding of the cement to the bone. 1 In mice the best fixation of a ‘helmet’ onto the skull without use of anchoring screws was achieved with cyanoacrylic glue and glass ionomer cement (Figure 5). In mice pretreatment with 1 mmol/L HCl or scratching of the skull increased the breaking force (Figure 6). In the ‘etching gel’ group (strong acid), we found no significant difference in breaking force compared with the ‘no pretreatment’ group. The reason for this unexpected result is probably the capacity of strong acids to decalcify the bone matrix. Calcium is an important salt in the chemical bonding of glass ionomer cements. 10
In rats the best attachment to the skull is achieved with anchoring screws. To investigate the possibility to achieve attachment to the skull without anchoring screws, rats were equipped with a ‘helmet’ of glass ionomer cement. In the groups with 1 mmol/L HCl and scratching as pretreatment, none of the animals lost the head attachment. However, breaking force measurements did not confirm a better bonding strength in these groups than in the group without pretreatment. This is in contrast with the results in mice. In conclusion, glass ionomer cement can be used in rats without the use of screws for a period of four weeks because of the chemical bonding of the cement to the skull, 10 and the most important part of the pretreatment in rats is that the skull is clean and dry before the glass ionomer cement is applied.
Apart from the advantages of less temperature increases of the glass ionomer cement and good bonding capacities to the skull, the use of the glass ionomer cement had other advantages. Glass ionomer cement is available in prepacked capsules (GC Fuji Plus® system). These capsules are sterile and it is possible to sterilize the cement applicator. The advantage is that, when applied with the GC Fuji Plus® system, the glass ionomer cement can be used for aseptic surgical techniques. The last advantage of the glass ionomer cement was the shorter polymerization time of about 4 min (approximately 13% of the overall surgical time) as compared with the polymerization time of methylmethacrylate cement. This reduction in surgical time is previously reported in rats 8 and is also reported when cyanoacrylic glue was used as a substitute for cement in mice. 2
Glass ionomer cement applied with the GC Fuji Plus® system is more expensive than the use of methylmethacrylate cement or cyanoacrylic glue. In our opinion the benefits of GC Fuji Plus® system outweigh the increased costs. We think that additional histological evaluation is important to demonstrate the possible negative effects of the extreme temperature rise in methylmethacrylate cement. When methylmethacrylate cement is used it is important to apply the cement in layers. Although time consuming this limits the temperature increase during the polymerization of the cement.
