Abstract
Intraperitoneal (i.p.) injection is the most frequently used method for implementing parenteral therapies in rats and mice. Whether the caecum is located in the right caudal quadrant or left caudal quadrant in the abdominal cavity is not clear. For that reason, we have developed a method for identifying the location of the caecum in rats and mice and thus revealed the most reliable location for i.p. injection in these animals. Two hundred Wistar albino rats and 100 BALB/c mice were used. The location of the caecum was determined by revealing the intra-abdominal organs immediately following euthanasia, photographing the organs, and archiving the images. Both digital photographic images and computed tomographic (CT) sections were analysed in terms of caecum morphology and location. In both rats and mice, the caecum was most commonly located on the animal’s left side. It was less frequently located on the right side or in the centre. The caecum was typically comma-shaped, but it was round or S-shaped in some animals. The direction of rotation of the caecum from the basis to the apex was mostly counterclockwise. Additionally, the apex showed a tendency to be evenly centred. This study demonstrated that the caecum was mostly located on the animal’s left side; and for that reason, the most suitable location for i.p. injection in these animals was understood to be the right caudal quadrant. Furthermore, when we compared the CT images and autopsy findings, the caecum did not change location in the abdominal cavity postmortem.
Rats and mice are the most frequently used experimental animals in biomedical research. Although the exact numbers are not known, according to the estimates of the British Union for the Abolition of Vivisection (BUAV), 100 million vertebrate animals are used worldwide each year, of which 95% are rats and mice. 1 During research, most experimental animals receive chemical compounds, drugs, antibodies, and other agents via enteral or parenteral routes. Intraperitoneal (i.p.) injection is the most common route for the parenteral administration of drugs in rats and mice, where substances are injected into the i.p. space. The absorption rate in i.p. application is typically much slower than for intravenous (i.v.) injection but faster than that observed after subcutaneous (s.c.) or intramuscular (i.m.) injection due to the intense blood circulation of the peritoneum. 2
The administration of relatively large amounts of fluids or various drugs in repetitive doses is possible via i.p. injection. 2 Drugs that are administered via this route are absorbed by the parietal and visceral peritoneums. Drugs such as atropine and progesterone are absorbed primarily by the visceral peritoneum and then transported to the liver via the portal venous circulation and metabolized by the liver. 3 Therefore, the bioavailability after i.p. administration compared with i.v. administration would be less for these drugs. 2 In contrast to the visceral peritoneum, the venous vessels of the parietal peritoneum empty into the systemic veins rather than the portal venous circulation. Thus, drugs and other compounds that are absorbed primarily by the parietal peritoneum bypass first-pass hepatic metabolism and may exert profound systemic effects when administered by i.p. injection. 3
During i.p. application, the animal is restrained on its back and tilted so that the head is facing downward and its abdomen is exposed. An imaginary line is drawn across the abdomen just above the knees and the needle is inserted along this line on the animal’s left side. Thus, the injection is made into the left caudal quadrant of the abdominal cavity. Because of the risk of damage to the kidney and retroperitoneal large vessels (e.g. the abdominal aorta and caudal vena cava), the needle should enter the skin at an angle of 10–20° in mice and 20–45° in rats, but the most common mistake is to puncture the skin at too acute an angle, resulting in s.c. rather than i.p. administration.2,4,5
Few references exist for reports on incorrect i.p. injection. According to existing reports, the frequency of erroneous injections has been reported to be from 5.5% to 20%, and these i.p. injections were probably made into a site other than the peritoneal cavity.6–9 These other sites include the retroperitoneum, subcutis and the lumen of the stomach, caecum, or small intestine. Furthermore, misplaced injections have been made into the vascular lumen or the urinary bladder lumen.6,7,10 When the ventral abdominal wall of the rat and mice is portioned into four quadrants, the liver, stomach, and spleen are located in the cranial quadrants. Therefore, injection into these quadrants may lead to serious haemorrhages that can be fatal. 2 Because the caecum is located on the animal’s right side, the left caudal quadrant is recommended for i.p. injections. Otherwise, the caecum may be punctured with the tip of the needle, and the caecal content may leak into the i.p. cavity, leading to complications such as peritonitis, sepsis, shock, and even death. Because the caecum is a large organ and almost always contains some amount of dietary content, its wall tends to be distended, which makes it more susceptible to puncture with the tip of a needle, compared with the small intestine. 11
The caecum resembles a blind sac and is located at the junction of the small and large intestines. 12 Food contents are temporarily stored in the caecum while bacteria that are normally present here break down cellulose. 13 The caecum has three parts: the apex, base, and body. 14 Although the apex is rich with lymphoid tissue, it does not have a vermiform appendix in rodents, as is found in humans. 15 The base is where the ileum and ascending colon are connected to the caecum. The caecum has the macroscopic appearance of a comma, and it is located in the caudal abdominal cavity. 16 Whether the caecum is located in the right caudal or left caudal quadrant of the abdominal space has not been definitively established. This is due to discrepancies found in textbooks related to the anatomy of rats and mice, as well as to controversies among authors in terms of their experiences. According to two reference books for researchers, the caecum in a rat or mouse is located in the right caudal quadrant of the abdominal cavity; therefore, they recommend i.p. injections be made on the animal’s left side.2,4 In contrast to this classical understanding, Coria-Avila et al. found that the caecum was located in the left caudal quadrant in 74% of female rats and 59% of male rats in their study; therefore, they stated that the most appropriate location for i.p. injections is the right caudal quadrant. 11
During laparotomy to perform a caecal ligation and puncture (CLP) procedure to induce a sepsis model, researchers may encounter different locations of the caecum (right, left, or middle). Due to the variations in caecum location, some textbooks related to invasive techniques state that either the right or left side may be used for i.p. injections.17,18 Some authors even recommend that the injection be performed at the midline. 19 In our review of the literature, we did not encounter any other study investigating the location of caecum in rats apart from that by Coria-Avila et al. 11 Moreover, even though mice are the most frequently used experimental animals in scientific research, no study investigating the caecum location in these animals has been undertaken. For this reason, we aimed to determine the caecum location in rats and mice via both laparotomic examination and computed tomography (CT) in our study, and to establish the safest site for i.p. injections in these animals.
Materials and methods
Animals
The study was approved by the Gaziosmanpasa University Local Ethics Committee for Animal Experiments (Tokat, Turkey). The study included 100 Wistar albino male rats, 100 Wistar albino female rats, 50 BALB/c male mice, and 50 BALB/c female mice. All animals were maintained under conventional housing conditions, with food and water available ad libitum in a temperature-controlled room (18–22℃) and in a 12 h:12 h light/dark cycle. These animals were obtained following euthanasia by decapitation under ketamine/xylazine (50/10 mg/kg i.m.) anaesthesia after they had been used in studies related to various topics (toxicology, behaviour, hypercholesterolaemia, testis torsion, etc.). Following euthanasia of the animal, the ventral abdominal wall was vertically cut from the symphysis pubis to the xyphoid process, and then the incision line was extended toward the sides with the guidance of the costal arcs so that the intra-abdominal organs were exposed, and the caecum location was determined. This view was photographed with a digital camera, and the image was archived.
Radiological imaging
Sixty of the rats (30 female and 30 male) were examined using CT prior to euthanasia to determine the premortem location of the caecum. Radiological examination was performed using a Siemens Biograph 2 PET/BT device (Siemens Healthcare GmbH, Erlangen, Germany). Organs and structures that were present in the abdominal area were scanned in 1 mm thick sections in the sagittal, coronal, and axial planes. Sections showing the caecum were determined and recorded in digital media. Images in a DICOM format were evaluated with the help of the Image J 1.48 program (National Institutes of Health, Bethesda, MD, USA) and using a 34 inch monitor.
Analysis of the images
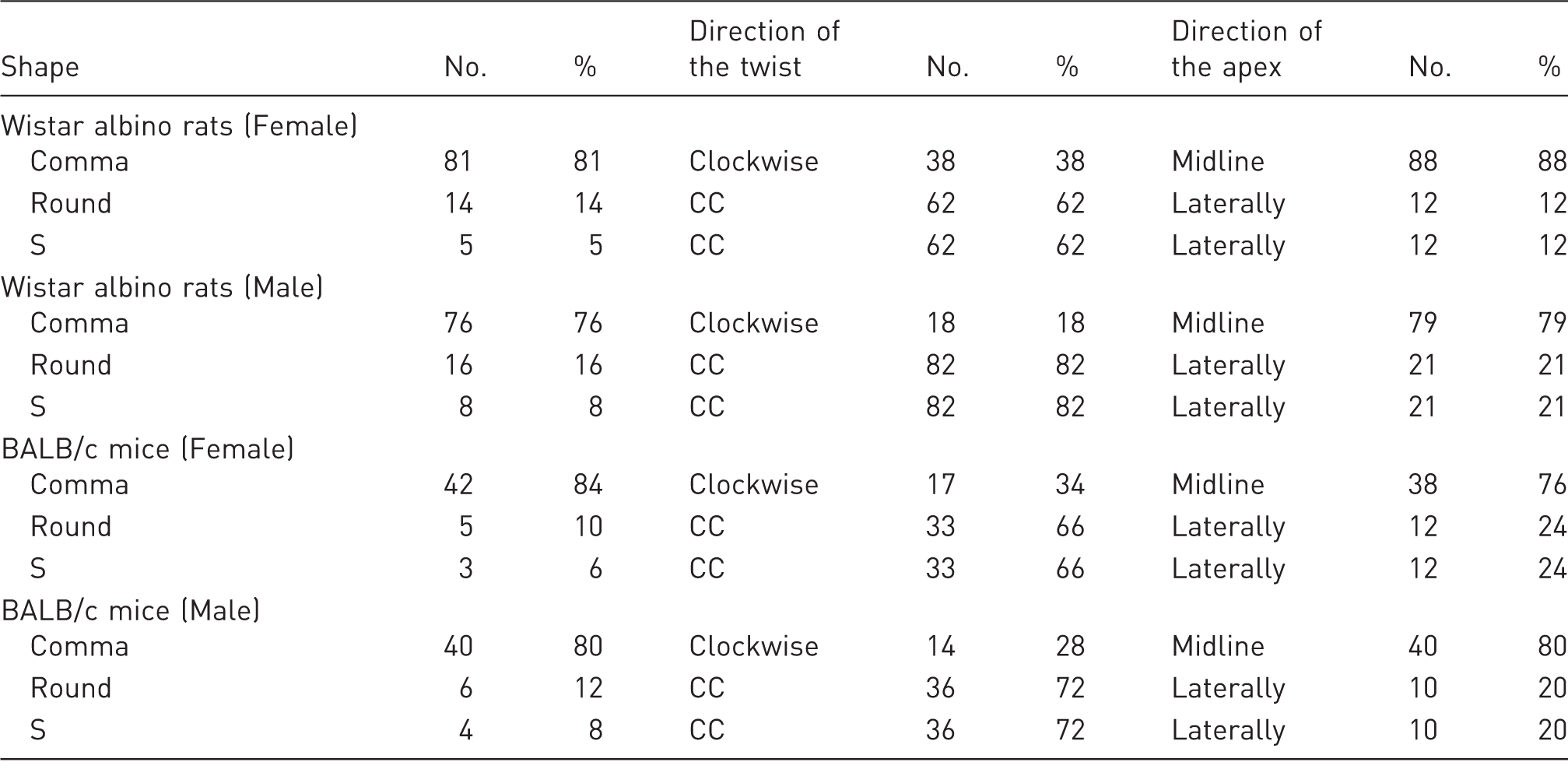
The digital photographic images and CT sections were evaluated in terms of the morphology and location of the caecum by two independent observers. After achieving an agreement on the images, the data were entered into an Excel program. In the majority of the animals, the caecum was clearly located on either the animal’s left or right side. However, in cases where its location was not clear, the decision was made according to whether three-fourths of the caecum was located on the left or right side, similar to the method defined by Coria-Avila et al. 11 When more than one-fourth of the caecum was present on the other side, the caecum was accepted to be in the middle. In addition to its location, the caecum was evaluated morphologically with regard to the following features: (i) whether the caecum appeared to be round, comma-like, or like the letter ‘S’; (ii) whether its twist from the base toward the apex was clockwise or counterclockwise; and (iii) whether the apex extended laterally or toward the midline.
Statistical analysis
Statistical analysis was performed using IBM–SPSS 20 software (IBM, Armonk, NY, USA). Comparisons of locations and morphological features of the caecum between the groups and sexes were made with a chi-square test. A P value of <0.05 was accepted as statistically significant.
Results
Location
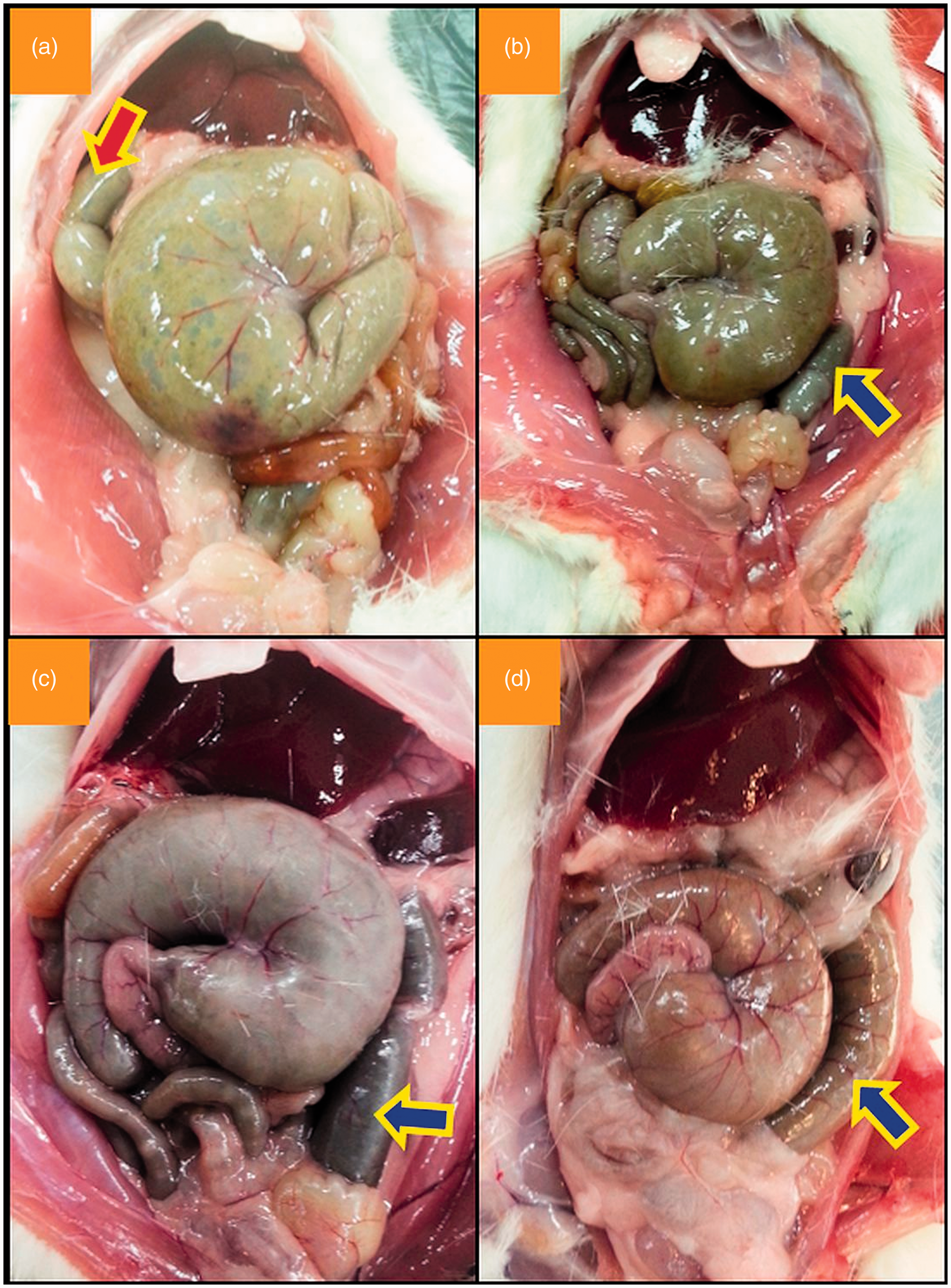
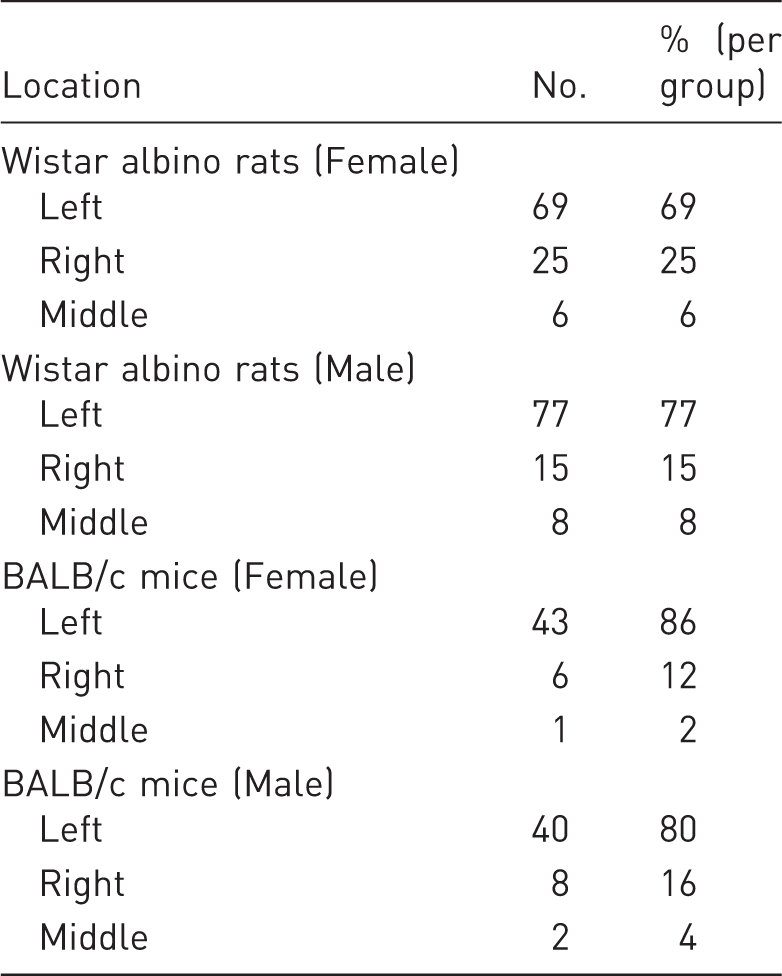
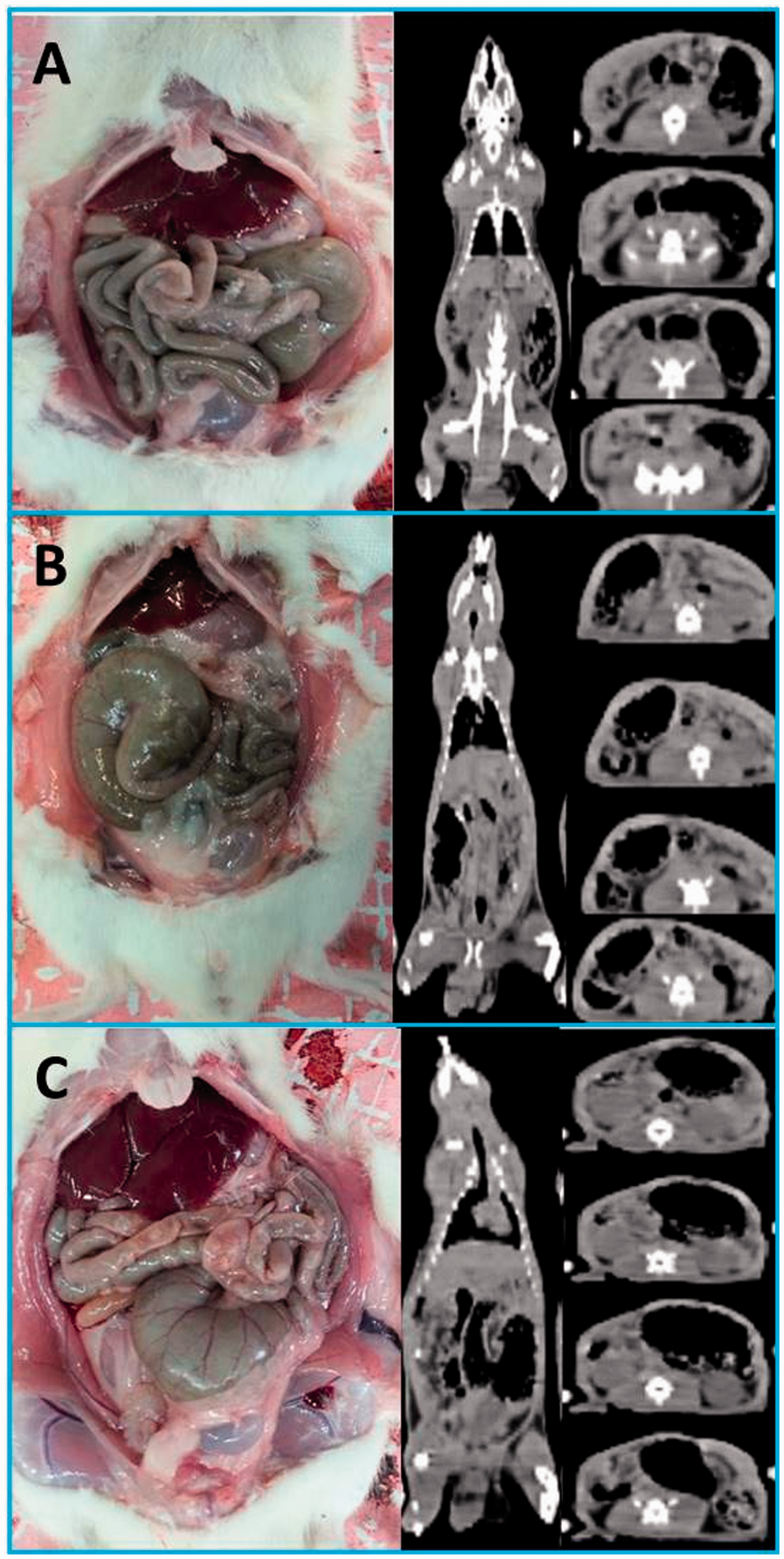
The caecum was located in three main regions in rats and mice – the right, left, and middle of the caudal abdominal cavity (Figure 1). In both rats and mice, the caecum was mostly located on the animal’s left side (Table 1). Among female Wistar albino rats, the caecum was located on the left in 69%, on the right in 25%, and in the middle in 6%. Among male Wistar albino rats, these rates were 77%, 15%, and 8%, respectively. In both female and male rats, the greater incidence of the caecum on the left was statistically significant (P < 0.001). No significant differences were observed between male and female rats with respect to their caecum location (P = 0.073). Among female BALB/c mouse strains, the caecum was located on the left in 86%, on the right in 12%, and in the middle in 2%. Among male BALB/c strain mice, these rates were 80%, 16%, and 4%, respectively. The differences between these rates were statistically significant in both sexes (P < 0.001). In 10 out of 14 rats (71%) and in all mice (100%) that had their caecum in the middle, the proximal or distal colon was filled with faecal content (Figure 2).
Left (a), right (b), and middle (c) localization of the caecum in Wistar albino rats. In some rats that had their caecum at the midline, the red arrow (a) points to the proximal colon filled with faecal content, and blue arrows (b–d) point to the distal colon filled with faecal content. Caecum location in Wistar albino rats and BALB/c mice.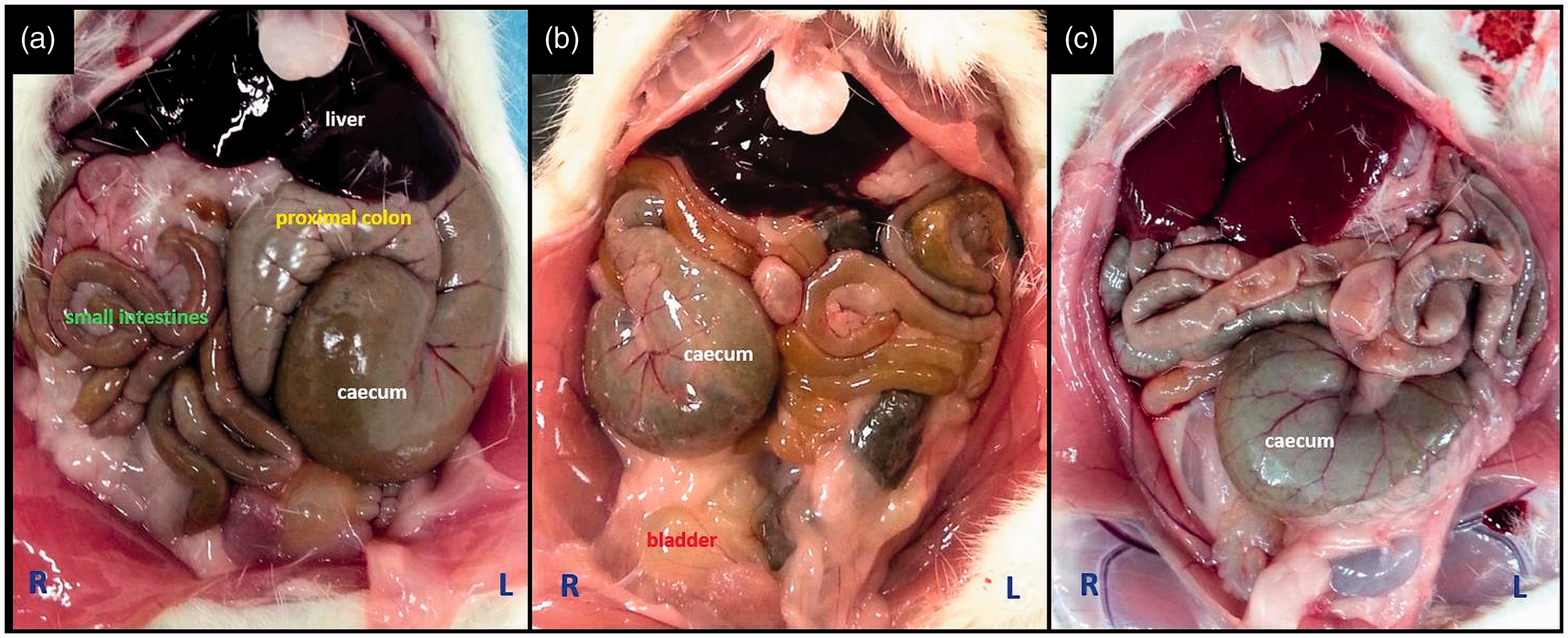
Morphology
The caecum was morphologically categorized according to three groups – comma-shaped, round, and S-shaped (Figure 3). In both rats and mice, caeca were mostly observed to be in the shape of a comma (Table 2). In some of the animals, the caeca had a round appearance due to the apex making a curve toward the ileocaecal junction. Furthermore, a few animals showed their apexes diverging away from the ileocaecal junction, and therefore, these caeca had the appearance of the letter ‘S’. The higher frequency of a comma-like caecum compared with the other types was statistically significant in both species of animals (P < 0.001). No significant differences existed between male and female animals with respect to caecum morphology (P = 0.271 for rats, and P = 0.367 for mice). None of the animals that were examined with CT showed a displaced caecum postmortem (Figure 4).
Comma-like (a–c), round (d–f), and ‘S’-like (g–i) appearances of the caecum in Wistar albino rats (a, b, d, e, g and h) and in BALB/c mice (c, f and i). Premortem computed tomographic section images of the caecum in Wistar albino rats and the postmortem photographic images of the same animal. Caecum morphology in Wistar albino rats and BALB/c mice. CC: counterclockwise.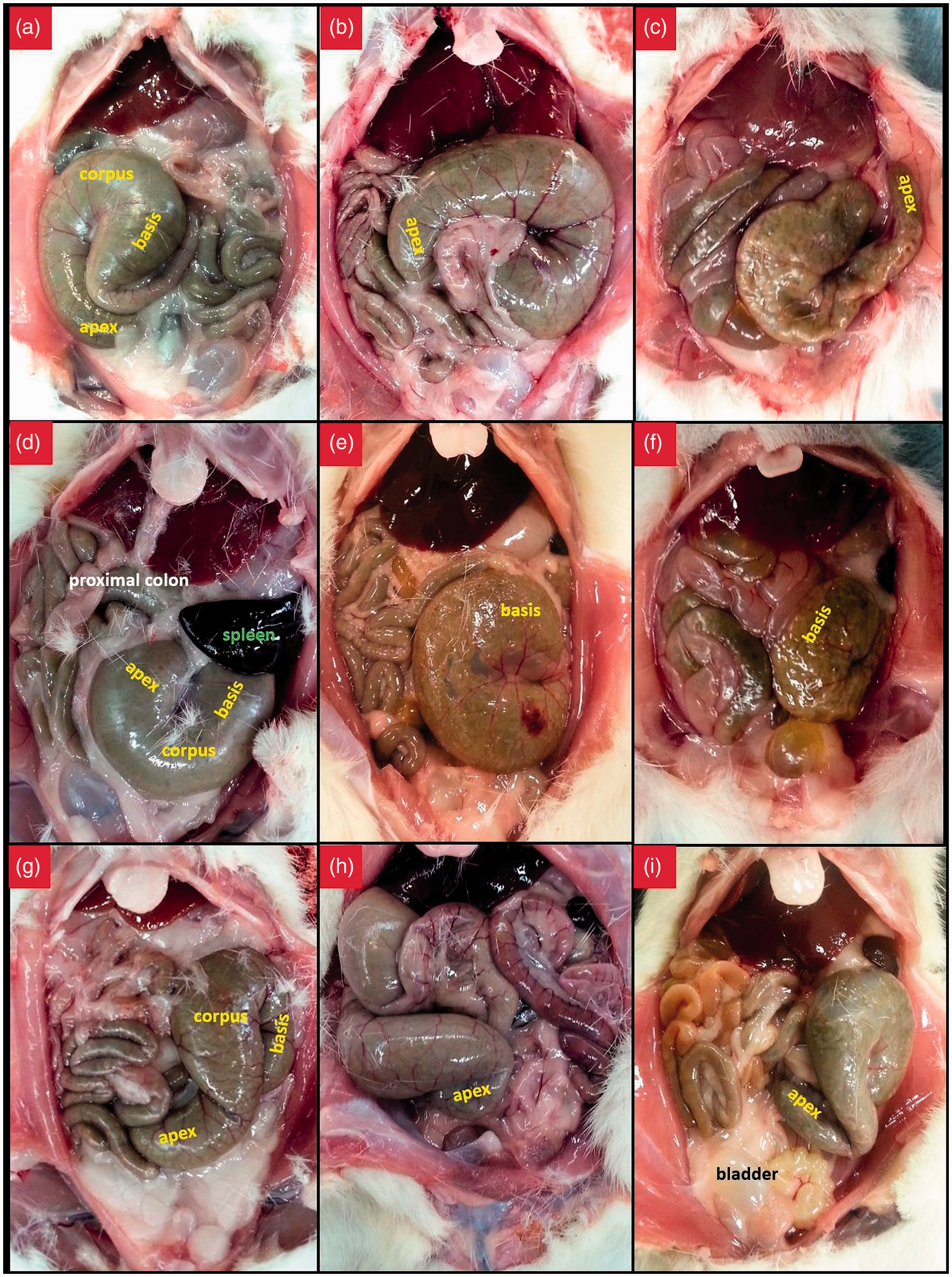
In all animals, the caecum twisted either clockwise or counterclockwise from its base to the apex (Figure 5, Table 2). This twist was counterclockwise in 72% of the rats and 69% of the mice. Although no significant differences existed between male and female mice with respect to the direction of the twist (P = 0.260), male rats had more caeca with a counterclockwise twist compared with female rats. However, this difference was not statistically significant (P = 0.096).
Examples of the caecum twisting counterclockwise (a) and clockwise (b).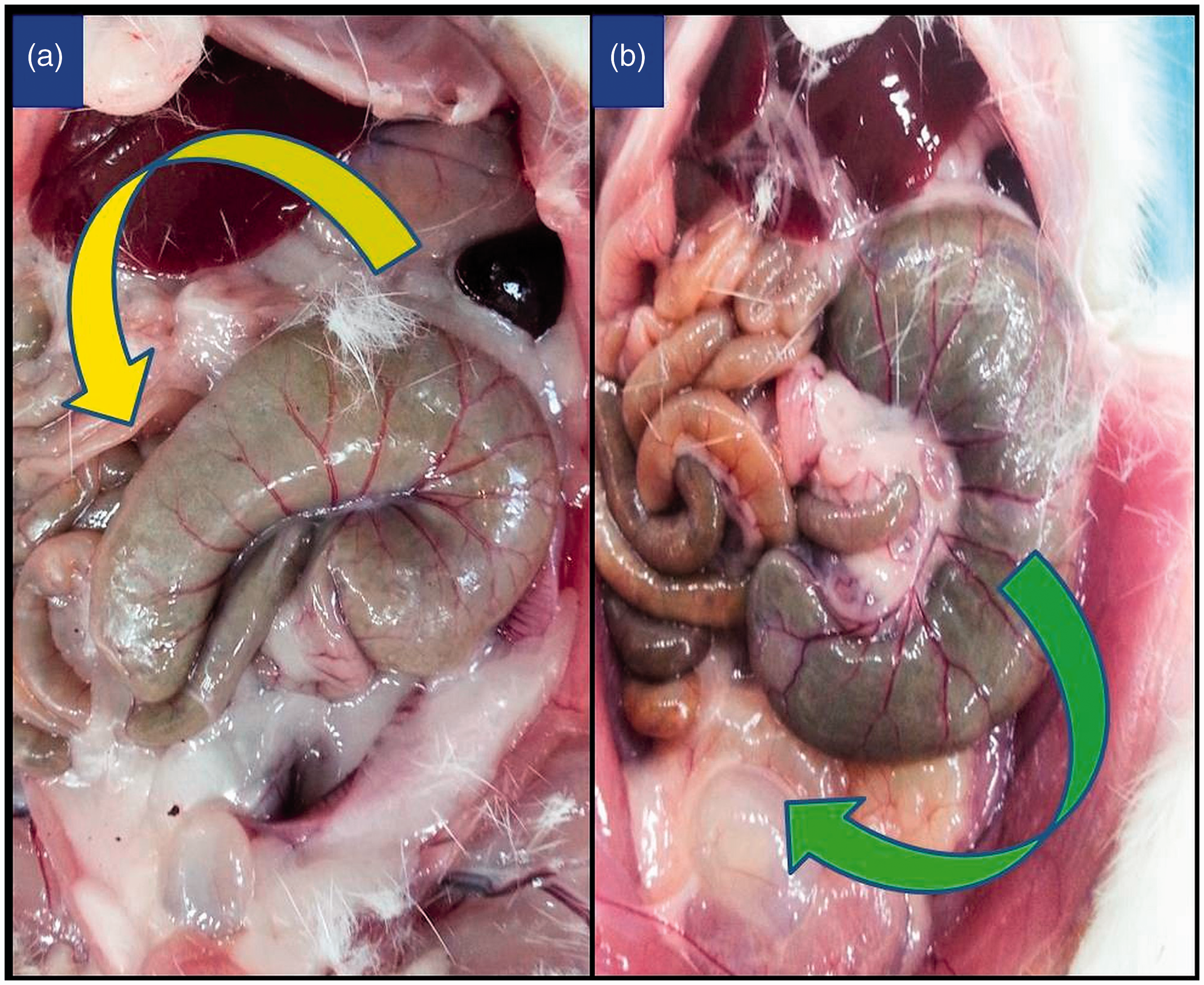
Due to the twist of the caecum from its base to the apex, the apex projected toward either the midline or laterally (Figure 6, Table 2). In 89% of rats and 80% of mice, the apex was close to the midline. This difference in favour of projection toward the midline was statistically significant in rats and mice (P < 0.001). On the other hand, the location of the apex relative to the midline did not differ between male and female animals (P = 0.086 for rats, and P = 0.128 for mice).
Apex of the caecum projecting toward the midline (a) and laterally (b).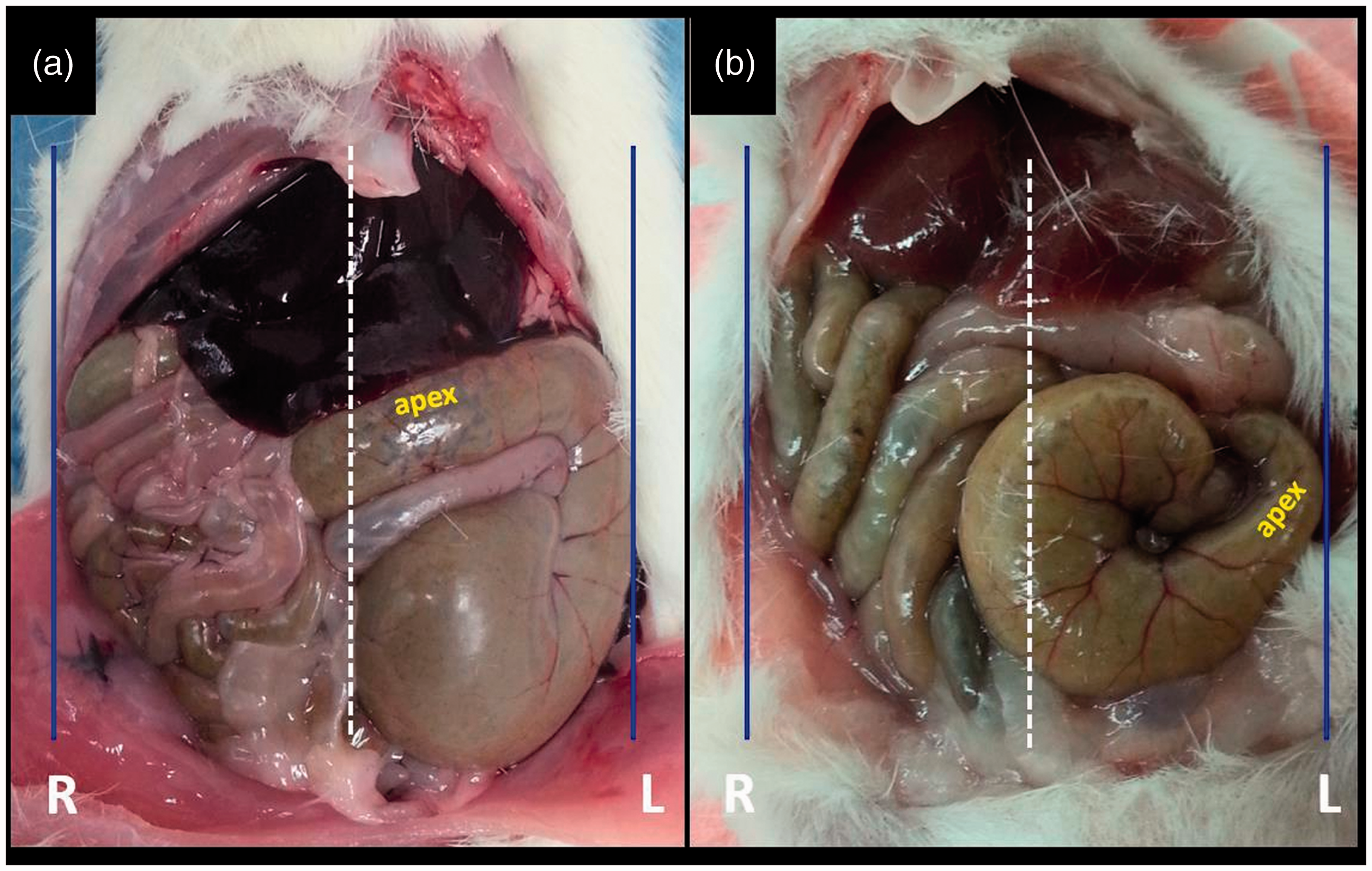
Discussion
The caecum is a blind sac-like organ located at the junction of the ileum and the proximal colon. Because rats and mice are herbivorous animals, the caecum in these animals provides storage for fibre-rich plant food while bacteria break down the cellulose. 20 As it is relatively large and almost always filled with dietary content, the caecum is susceptible to injury during i.p. injection. 11 Because millions of rats and mice are used experimentally, with at least that number of i.p. injections being performed worldwide every year, knowledge of the anatomy of the caecum and its actual location within the abdomen is important to researchers. 1 Our results confirm that the caecum is located in the left caudal quadrant of the abdominal cavity in the majority of rats and mice, and this is independent of their sex. Therefore, we consider that the most appropriate site for i.p. injection in these animals is the lower right quadrant.
Contrary to our results, classical textbooks state the caecum location as the lower right quadrant in laboratory animals.2,4 The main reason for this controversy is possibly because only one study investigated caecum location in rats, and no studies on this topic have been reported in mice. Therefore, the caecum is commonly assumed to be in the lower right quadrant in these animals, similar to what is normal in humans. However, Coria-Avila et al. have reported that the caecum was mostly located in the left caudal quadrant in their study of rats, 11 which is similar to our results.
The reason that the majority of rats and mice have their caecum on the left side is not clear. According to Coria-Avila et al., the main reason that the caecum has been observed in different regions is that no ligament holds the caecum in place. However, this hypothesis does not explain why the caecum is mostly observed on the left. Another hypothesis that was put forward by the same researchers is that the caecum location is affected by the intra-abdominal fat content. 11 We hypothesize that it is more frequently located on the left because the transverse colon influences caecum location. Although the proximal colon, distal colon, and caecum are i.p. organs, they do not possess any mesentery connecting them to the dorsal abdominal wall. By contrast, the small intestine is attached to the distal colon via a mesentery that is formed by two sheets of the peritoneum. The distal colon is in a continuum with the rectum; therefore, it has a tendency to stay on the midline. The transverse colon is attached to the dorsal abdominal wall via a peritoneal connection that extends towards the hepatic port. Because the proximal and distal colons are mobile but the transverse colon is fixed to the dorsal abdominal wall, extension of the transverse colon toward either the left or right influences the location of the intestinal organs. As the transverse colon extends toward the right side, the proximal colon and caecum, which are in a continuum, are located in the right caudal quadrant of the abdominal cavity, whereas the remaining right caudal portion of the abdominal cavity is filled with the small intestine. The transverse colon may be found toward the left in the majority of rats and mice, which is why the caecum is mostly observed on the left side in these animals.
In our study and in the study by Coria-Avila et al., the caeca of some animals were located in the middle of the abdominal cavity. 11 The most likely reason for this could be the displacement of the caecum, which is normally located on either the left or right side, toward the midline due to pressure exerted on it by the proximal or distal colon as it fills with faecal content. In support of this hypothesis, we observed that in the majority of rats and in all mice with their caeca located in the middle, the proximal colon or distal colon was filled with faecal content.
In our study, to examine whether the caecum location changed postmortem, a group of animals were examined using CT, and their caecum location was determined prior to euthanasia. According to our findings, the caecum did not appear to have changed location postmortem. Although we did not use micro-CT in this study, determining the caecum location was quite easy because it is a large organ and is filled with gas. Nevertheless because our study is the first to use CT for the determination of caecum location, further studies that use CT or micro-CT are necessary to confirm our findings.
A few studies that investigated the macroscopic anatomy of the caecum have reported that the caecum had the appearance of a comma.21,22 In addition to that, we also observed round and S-shaped caecum types in our study. The parameters that we used for categorizing the caecum were position of the apex relative to the ileocaecal junction and the angle between the apex and the body. According to this, the most common type was that resembling a comma, and the least common type was that resembling the letter ‘S’. In addition to morphological typing of the caecum, we also determined the position of the caecal apex relative to the midline. Depending on its own orientation, whenever the apex moves away from the midline and projects toward the lateral abdominal wall, the body and the base of the caecum are observed to be localized close to the midline. Possibly, this position makes the caecum more vulnerable to puncture with the tip of the needle during i.p. injections. Many textbooks recommend inserting the needle just next to the midline during i.p. injections, in which case the probability of puncture of the caecum with the tip of the needle would be high if the body and the base of the caecum are located close to the midline, as these parts are much larger than the apex.16,18 Therefore, due to the high risk of perforation of both the urinary bladder and the body and the base of the caecum during injections to the midline, we recommend making the injection at the midpoint of the distance between the midline and the lateral abdominal wall.
Intraperitoneal injection is the most commonly used route of parenteral drug administration to laboratory animals. 4 It is preferred over i.v. application, as it is an easier procedure. 2 Although it provides some practicality, injury to the intra-abdominal organs and complications that may even lead to sepsis can occur, especially after injections made by inexperienced staff. Therefore, knowledge of the appropriate injection site is important. However, classical textbooks do not provide clear information about caecum location, and this can lead to confusion about the correct site for performing i.p. injections. In our study, the caecum was located on the left side in 77% of male and 69% of female rats. Coria-Avila et al. also reported that the caecum was primarily located on the left side; however, they gave the same rates of 59% and 73%, respectively. 11 No significant differences were observed for caecum location between the two studies in female rats; however, the frequency of a left-sided caecum in male rats was higher in our study. A possible explanation for this difference is that the rats were produced in breeding colonies, and therefore, differences between laboratories at a genetic level would have an influence on the caecum location. For that matter, units that produce laboratory animals should carry out their own preliminary studies to determine caecum location, and they should inform researchers about caecum location and the correct site for i.p. injections. In this study, we evaluated the caecum localization of the BALB/c mouse, which is a widely used inbred strain of laboratory mice. However, other inbred strains (e.g. C57BL/6 and DBA/2 J) are also frequently used in laboratory experiments. Therefore, a need exists for further studies concerning a possible difference between BALB/c and other inbred species in terms of caecum localization, which will affect the abdominal quadrant selected for i.p. injections in inbred species.
Conclusion
In our study, we observed that the caecum was primarily located on the animal’s left side, and this location did not change postmortem. According to these findings, the most appropriate site for i.p. injection would be the right caudal quadrant of the ventral abdominal wall. Furthermore, we believe i.p. injection should not be made near the midline because there is a higher risk of puncturing the caecum if the apex is projected laterally and a higher risk of puncturing the urinary bladder if it is full.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
