Abstract
In many pharmacological and toxicological studies knowledge about the intestinal absorption, which is dependent upon the surface area of absorptive epithelia, is indispensible. Although mice are often used in such preclinical studies, very few quantitative data about their intestinal surface area are available. Especially for locally acting candidate drugs in development, this information is crucial for dose translation towards humans. Therefore, the surface area of the intestinal tract of CD-1™ IGS mice was assessed in the present study. The intestinal tracts of 12 mice were collected after euthanasia. From six animals, histological sections from the duodenum, jejunum, ileum, caecum and colon-rectum were made according to common stereological principles. Using these sections, the volumes and surface areas of each intestinal segment were estimated applying stereological counting procedures. In the other six animals, the density and surface area of the microvilli present in each intestinal segment were determined by means of scanning and transmission electron microscopy to assess the increase of the intestinal surface area attributable to the presence of microvilli. The mean total volume and surface area of the intestinal tract were 1.34 cm3 and 1.41 m2, respectively. The relative intestinal surface area (intestinal surface area divided by the body surface area) was 119. The relative intestinal surface area of mice is very similar to that of humans. The results of this study are important for the appropriate dose translation of candidate therapeutic compounds in drug development from mouse to humans.
To extrapolate the pharmacological characteristics of orally administered candidate therapeutic compounds between species, detailed knowledge about their absorption is of major importance. 1 This absorption is highly dependent upon the presence of absorptive epithelia. A primary factor influencing the absorption of orally administered compounds is the intestinal surface area. Although the mouse is the most commonly used laboratory animal, quantitative data about its intestinal tract are hardly available. 2 In contrast, the intestines of the rat have frequently been subjected to quantitative studies, but this rodent species is not always suitable in pharmacological and toxicological studies as there are significant differences among species regarding the absorptive epithelia. 1 Consequently, there is an obvious need for morphometric data of the murine intestinal tract.
In the present paper, the intestinal volume and surface area of the mouse are assessed. For this purpose, stereological calculations were combined with data obtained by scanning and transmission electron microscopy (SEM and TEM). Data relating to the intestinal surface area of humans will be presented and the murine intestinal surface area will be discussed in view of oral dose translation of therapeutics from mouse to humans.
Materials and Methods
Determination of the intestinal surface area at the light microscopical level
Animals and sample collection
Six 15-week-old CD-1™ IGS multipurpose mice (3 males and 3 females with an average weight of 41 and 33 g, respectively) (Charles River, Calco, Italy) were housed in the animalarium of ActoGeniX NV in type 3 cages with wire lids and in groups of 2–4 animals. Both the presence of sufficient feed and drinking water, provided ad libitum, as well as the welfare and health status of each individual animal were checked daily. The cages had a solid bottom covered with sufficient amounts of sawdust and were provided with nesting material to create a microclimate. Twenty-four hours before euthanasia, the animals were fasted to empty their gastrointestinal tracts. After they were euthanized by means of cervical dislocation, their abdomens were opened through a ventral midline incision and transitions between the duodenum and jejunum, and the jejunum and ileum were marked in situ according to the criteria stipulated by Hoffmann. 3 This enabled us to distinguish the several intestinal segments after excision of the intestinal tract, from the descending duodenum, just distal to the pylorus, unto the descending colon–rectum, just proximal to the anus. No distinction between the colon and rectum was made as the border between both segments is very faint in mice. 3 In order to remove any chyme and to reduce postmortal decay, the intestines were immediately and successively flushed with physiological saline solution and 3.5% buffered formaldehyde at room temperature.
Each intestinal segment was, according to their length, divided into 3–10 equal pieces. Within the first of these pieces a transversely cut sample of 1 mm in length was taken at random position. All transverse samples from the subsequent pieces were taken at exactly the same position within those pieces. As a result, a fixed interval T was present between each transverse sample (Figure 1) that was used for stereological volume estimation.
Schematic illustration of the sampling procedure for stereological volume estimation. Each intestinal segment is divided into a set of equal pieces. In this example six pieces (a–f) are obtained through five transverse cuts (1–5). Within the first of these pieces, a transversely cut sample, 1 mm in length, is taken at random position (1’). All transverse samples from the subsequent pieces are taken at exactly the same position within those pieces (2’-6’). As a result, a fixed interval T is present between all transverse samples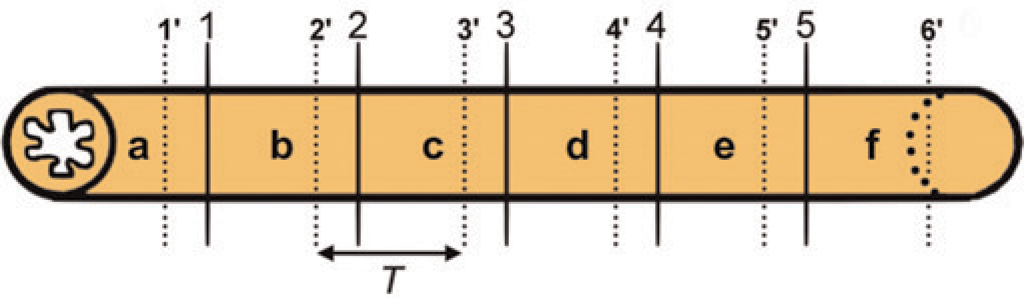
Additionally, a second transversely cut sample of 2 mm in length was collected adjacent to each 1 mm sample. They were opened at the antimesenterial side and their circumferences were measured. From each of these samples a round core, 2 mm in diameter, to be used for stereological surface area estimation was punched out using a small trocar. 4
Histological processing
After the samples were fixed for two days in 3.5% buffered formaldehyde at room temperature, they were dehydrated (Microm tissue processor STP420D, Prosan, Merelbeke, Belgium) and embedded in paraffin (Microm embedding station EC 350–1, Prosan). All cores that were punched out of the intestinal wall were, however, randomly rotated along their vertical axis before embedding. 4 An 8 μm thick transverse section (Microm microtome HM 360, Prosan) was made of each transverse sample at uniform random position (UR section) (Figure 2a) and of each core at random position within that core (vertical uniform random section, VUR) (Figure 2b). All sections were mounted on slides, stained with haematoxylin and eosin (Linear Stainer II, Sakura Finetek Europe BV, Zoeterwoude, The Netherlands), and examined with a motorized light microscope (Olympus BX 61, Olympus Belgium, Aartselaar, Belgium) linked to a digital camera (Olympus DP 50, Olympus Belgium).
Histological sections of the various transversely cut intestinal segments used for volume assessment (column a), and 2 mm diameter cores used for surface density assessment (column b). Notice the large Peyer's patch in the jejunum (arrow)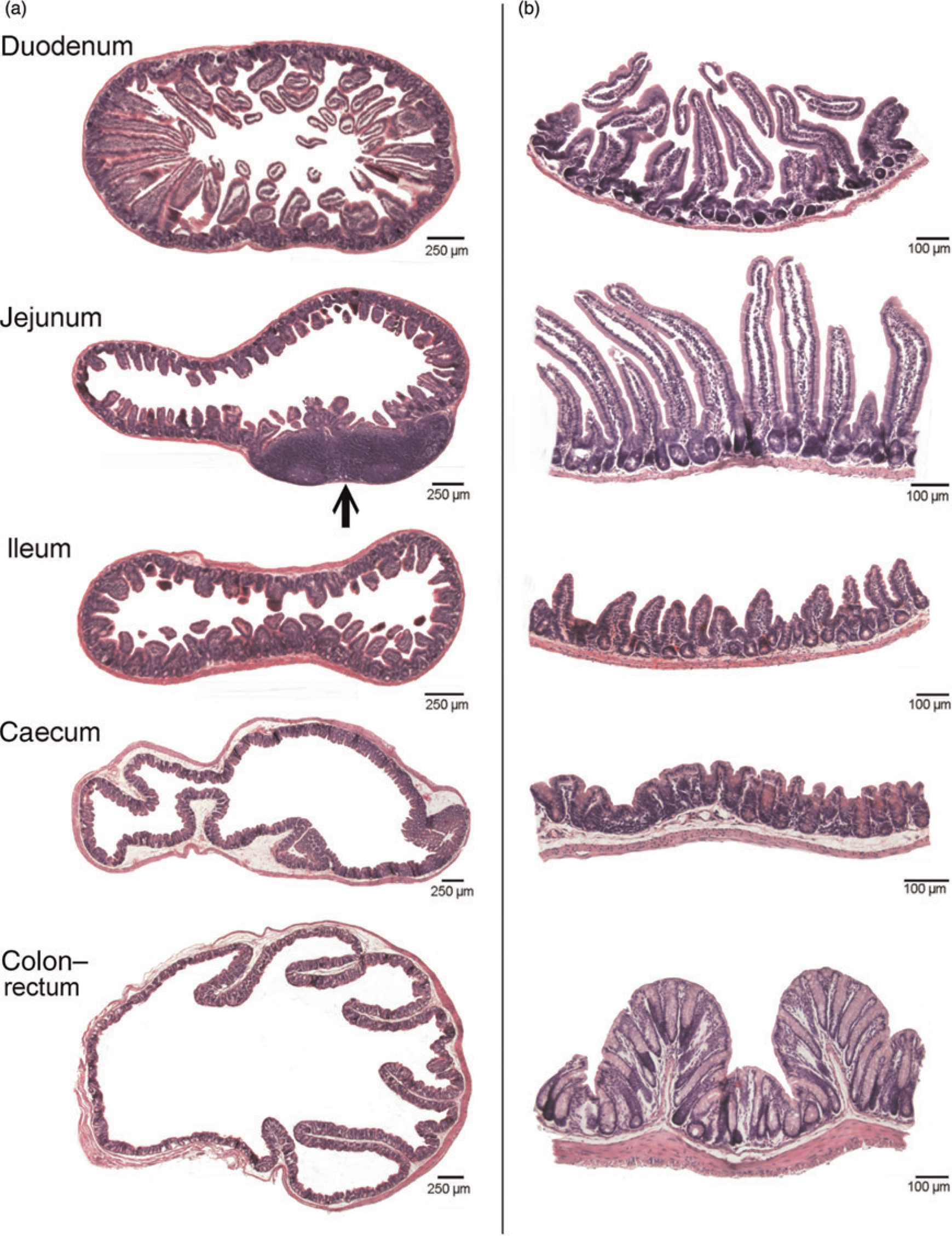
Stereological analysis
Example of a grid used for stereological volume assessment overlying a histological section of the murine duodenum. The crosses indicate the grid points (grid crossings) hitting the intestinal tissue. In this example, 33 points will be counted. Each grid point is associated with the area of the surrounding square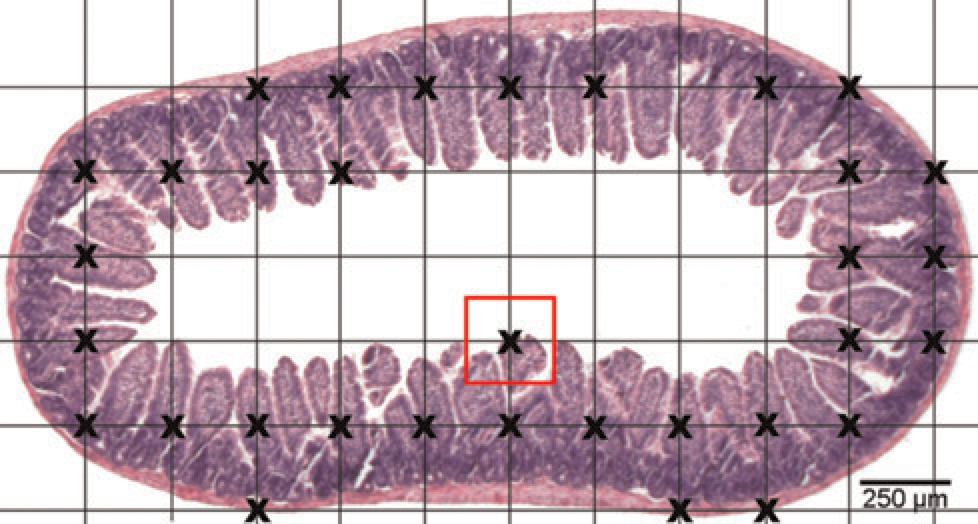
Cycloidal grid used for surface density calculation overlying a histological section of a 2 mm diameter core punched out of the murine caecum. The number of intersections of the cycloidal grid (wavy line) with the epithelial surface (small black dots) has to be counted as well as the number of grid points hitting the reference space (large grey dots)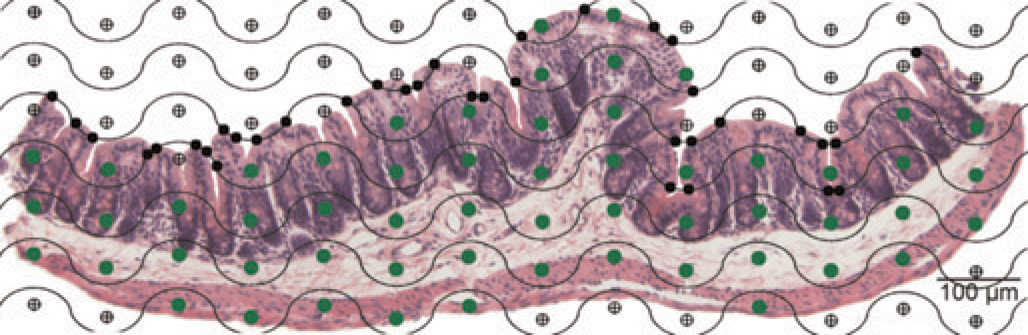
Correction for tissue shrinkage
When estimating volume and surface area based on histological sections, the effects of histological processing on fresh tissues have to be taken into account. Two assumptions to correct the data have, however, to be made. First, the various cell layers of the intestinal wall do not shrink differently. Second, shrinkage of the gut is identical in each direction (circumferentially, axially and radially). 11 Assessing the impact of tissue shrinkage was done by measuring the circumferences (2rπ) of the intestinal segments before and after tissue processing. The methodology of shrinkage calculation as described by Van Ginneken et al. 11 was applied.
Determination of the intestinal surface area attributable to the microvilli
To assess the increase of the intestinal surface area attributable to the presence of microvilli (microvillus amplification factor, MAF), the microvillus density (#/μm2), determined by SEM, was multiplied by the microvillus surface area (μm2), determined by TEM.
Animals and sample collection
Six 10-week-old CD-1™ IGS multipurpose mice (3 males and 3 females weighing 35 and 31 g, respectively) (Charles River, Italy) were housed and treated as mentioned above. Immediately after sacrifice by cervical dislocation, they were perfused via the abdominal aorta with a HEPES-buffered solution of 2% paraformaldehyde and 2.5% glutaraldehyde at room temperature. The intestinal tract was taken out and the proximal, middle and distal parts of each intestinal segment were opened at the mesenterial side. The chyme was washed out with the fixative and samples of 2 mm in diameter for SEM and TEM were punched out of the gut wall using a trocar (Figure 5).
Schematic representation of a murine intestinal segment (in this case the jejunum) showing the positions at which samples for scanning electron microscopy (SEM) and transmission electron microscopy (TEM) (visualized by the stereomicrographs) were taken
Scanning electron microscopy
The protocol used for SEM processing was analogous to that described by De Spiegelaere et al. 12 After fixation and post-fixation, the samples were dehydrated and dried at critical point (Balzers CPD 030, Sercolab, Merksem, Belgium), mounted on metal bases and coated with platinum (JFC 1300 Auto Fine Coater, Jeol, Zaventem, Belgium). They were examined with a JSM 5600 LV scanning electron microscope (Jeol) and micrographs (magnification 20,000 x) were taken from the middle region of the villi. The microvillus density was determined using image analysis software (Cell D, Olympus Belgium) (Figure 6).
Scanning electron microscope (SEM) image demonstrating the microvillus density counting procedure
Transmission electron microscopy
For TEM processing, a similar protocol as that described by De Spiegelaere et al. 12 was used. After the samples had been fixed, postfixed and dehydrated they were embedded in Spurr's resin. Sections of 60 nm were made (EM UC6, Leica Microsystems, Groot-Bijgaarden, Belgium) and post-stained in uranyl acetate and lead citrate (EM AC20, Leica Microsystems). For examination, a JEM 1010 transmission electron microscope (Jeol) was used, operating at 60 kV. Micrographs (magnification 20,000 x) of randomly selected enterocytes with longitudinally sectioned microvilli were taken. Using image analysis software (Cell D, Olympus Belgium) the microvillus length (l) from tip to base and the diameters (2r) of several microvilli were measured according to the criteria of Brown 13 (Figure 7). The microvillus surface area was calculated via 2rπl.
Transmission electron microscope (TEM) image showing the method to measure microvillus length and diameter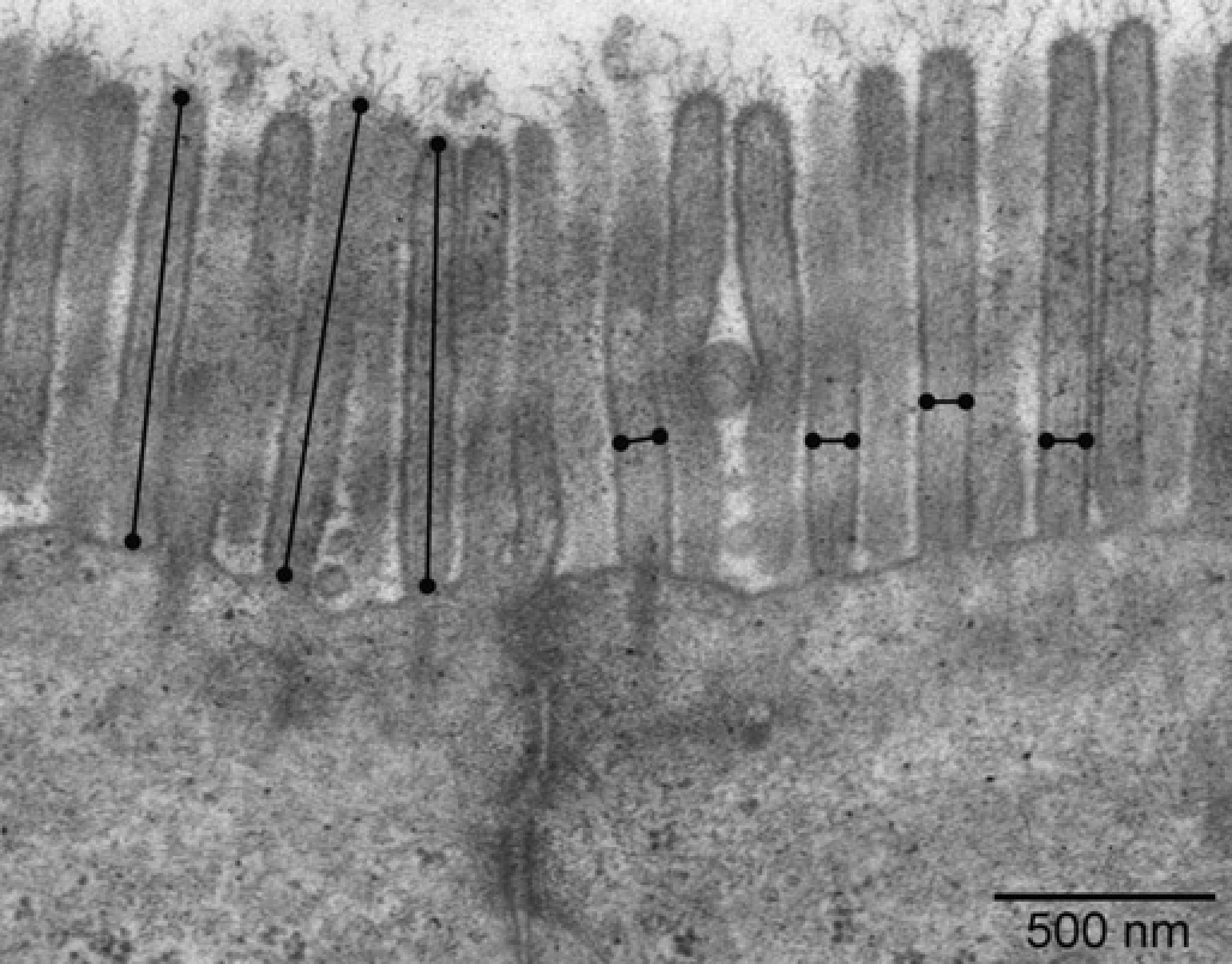
Calculations and statistics
For each intestinal segment a mean MAF was calculated from the values of the proximal, middle and distal parts of the segments. For both male and female mice the surface area of each intestinal segment was assessed by multiplying the surface area determined at the light microscopical level by their respective MAFs. By summing these values the surface area of the total intestinal tract was obtained.
The difference between male and female mice concerning the length, volume and surface area of the intestinal tract was analysed using a t-test. Statistical analyses were also performed on the MAFs. Comparisons between male and female mice were made using a t-test. Multiple comparisons between the various intestinal segments were performed using one-way analysis of variance applying the Bonferroni method. Differences were considered significant when P < 0.05. All statistical analyses were performed using S-Plus 8.0.
Results
Intestinal length, diameter and volume
The mean lengths, circumferences and diameters of the various intestinal segments of the six mice included in the light microscopical analysis are presented in Table 1. The mean murine intestinal length was 55.5 ± 1.8 cm. That of the male mice was 59.2 ± 0.8 cm and that of the females 51.8 ± 0.8 cm (females 12.5% significantly smaller than males; P = 0.0175).
Mean length, circumference and diameter with standard error of each murine intestinal segment (n = 6)
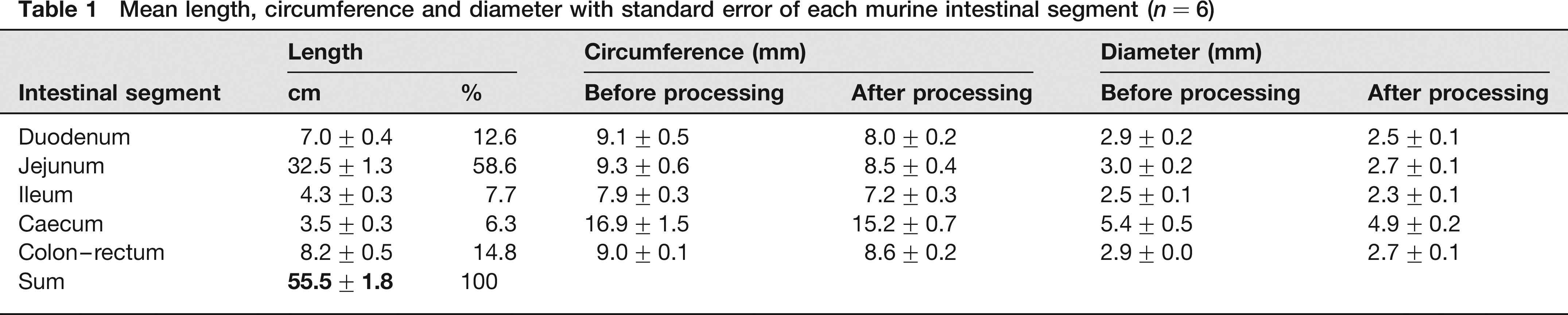
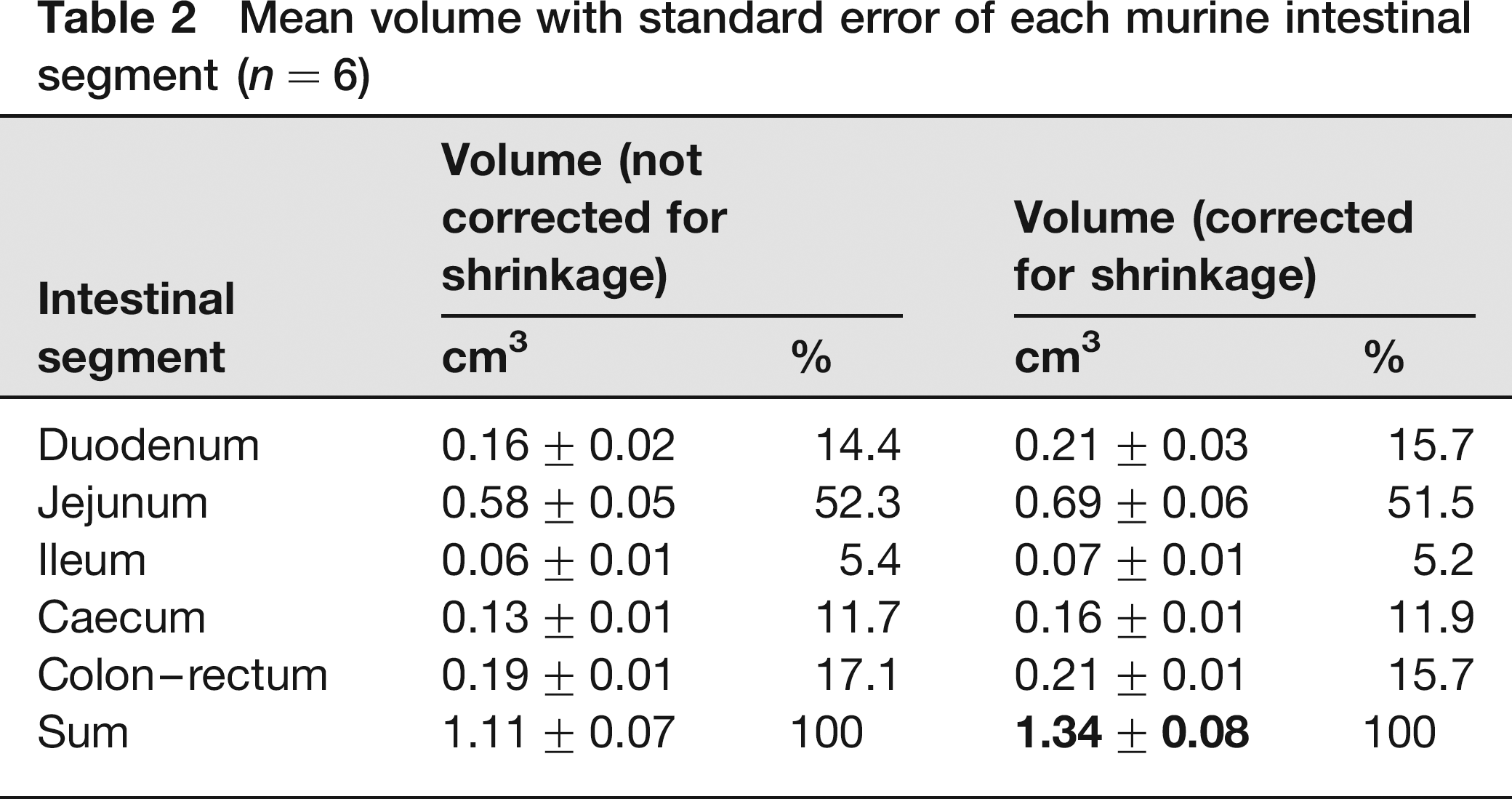
During tissue processing the mean circumferences (2rπ) of the duodenum, jejunum, ileum, caecum and colon-rectum shrank for 12.1%, 8.6%, 7.7%, 10.1% and 4.4%, respectively (linear shrinkage). As a result, the circumferences, and thus the radii r, after shrinkage were only 87.9%, 91.4%, 92.3%, 89.9% and 95.6% of the circumferences, or the radii r, of the fresh tissues. Using the formula of the surface area of a circle (r2π), it was calculated that the areal shrinkage of the transverse sections, used for volume estimation, amounted to 22.7%, 16.5%, 14.8%, 19.2% and 8.6%. This resulted in a similar underestimation of the volume (volumetric shrinkage) of each intestinal segment as the interval T was determined on fresh tissue. The mean uncorrected intestinal volume was 1.11 ± 0.07 cm3, whereas the mean corrected intestinal volume was 1.34 ± 0.08 cm3 (Table 2). The intestinal volume of the three male mice amounted to 1.46 ± 0.07 cm3, and that of the female mice to 1.22 ± 0.12 cm3 (volume 17% smaller in females, but not significant; P = 0.1508). The CE values amounted to 7%, 6%, 11%, 15% and 7% for the duodenum, jejunum, ileum, caecum and colon–rectum, respectively.
Mean volume with standard error of each murine intestinal segment (n = 6)
Intestinal surface area at the light microscopical level
The mean intestinal surface area of the six mice, determined at the light microscopical level using stereology, is shown in Table 3.
Mean surface area with standard error of each murine intestinal segment (n = 6)
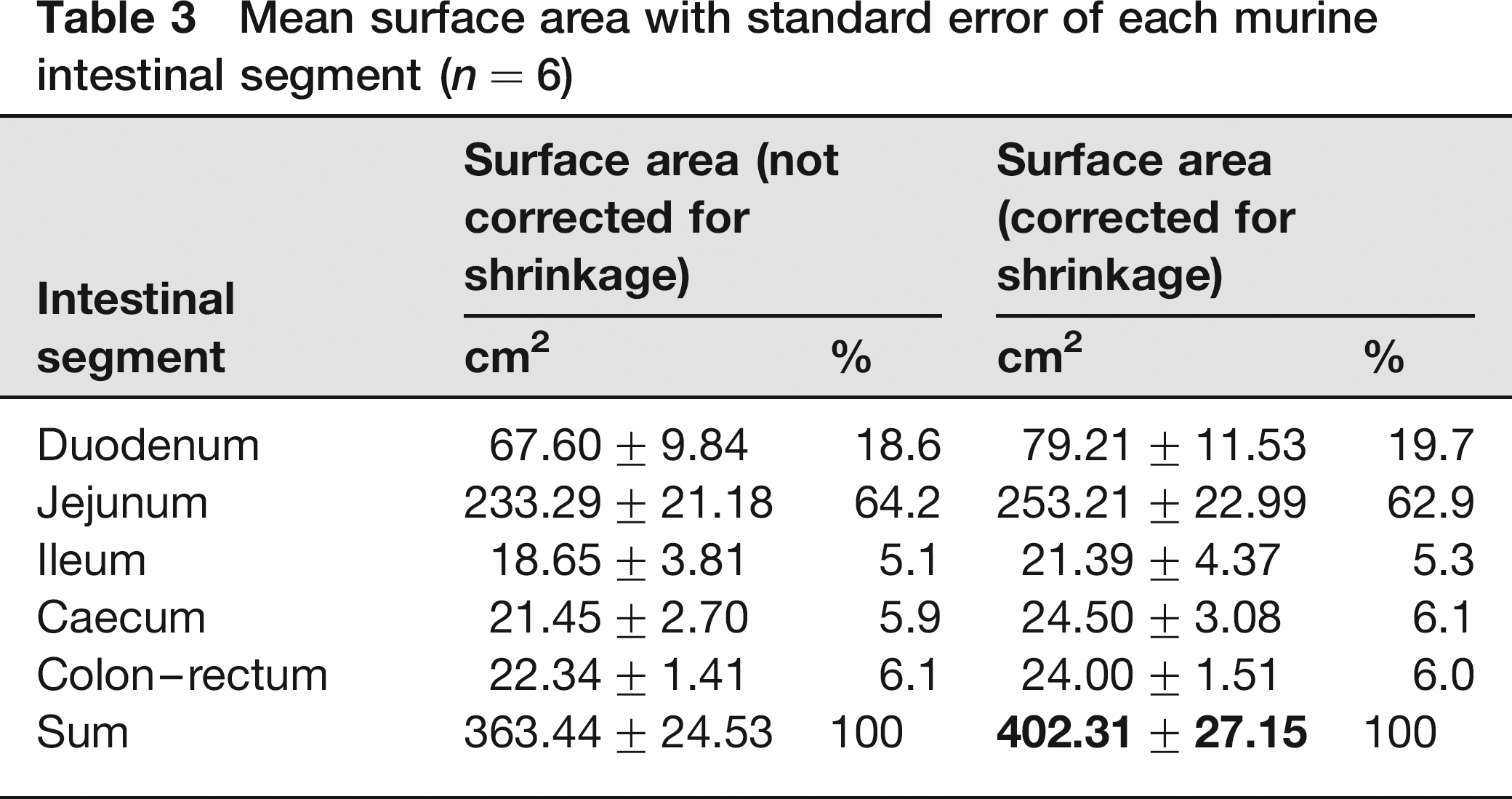
The mean correction for shrinkage of each intestinal segment amounted to 14.6%, 7.9%, 12.8%, 12.4% and 6.9%. The mean corrected intestinal surface area of the three male mice was 434.22 ± 22.38 cm2 and that of the female mice 370.40 ± 46.55 cm2 (surface area 15% smaller in females, but not significant; P = 0.2843). The CE values for the duodenum, jejunum, ileum, caecum and colon-rectum were 10%, 5%, 9%, 11% and 10%, respectively.
Increase of the intestinal surface area attributable to the microvilli
The mean MAF for each intestinal segment of the six mice included in the ultramicroscopical analysis can be found in Table 4. The values for the microvillus density, length, diameter and surface area are additionally shown.
Mean microvillus density, length, diameter, surface area and amplification factor with standard error of each murine intestinal segment (n = 6)
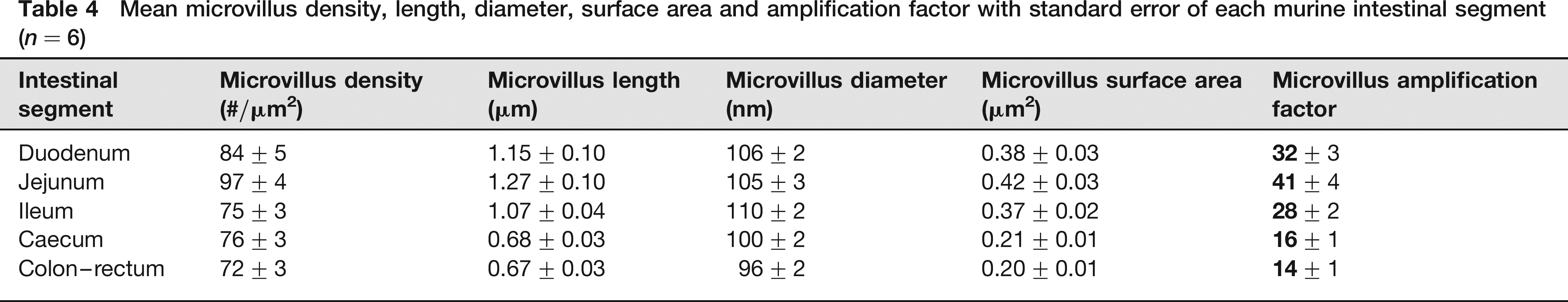
No significant difference between male and female mice was observed for the MAF of each intestinal segment (P values of 0.0956, 0.7343, 0.2852, 0.9394 and 0.4908 for the duodenum, jejunum, ileum, caecum and colon–rectum, respectively). The MAF differed, however, significantly between the duodenum and jejunum (P = 0.0439), duodenum and caecum (P = 0), duodenum and colon–rectum (P = 0.0001), jejunum and ileum (P = 0.0036), jejunum and caecum (P = 0), jejunum and colon–rectum (P = 0), ileum and caecum (P = 0) and ileum and colon–rectum (P = 0). Only between the duodenum and ileum, and between the caecum and colon–rectum the difference of the MAF was not significant (P values of 0.4680 and 0.4735, respectively).
Overall murine intestinal surface area
Table 5 shows the mean surface areas of each intestinal segment of male and female mice. The mean overall intestinal surface area for both gender amounts to more than 1.4 m2.
Mean intestinal surface area with standard error of each intestinal segment for male (n = 3) and female (n = 3) mice
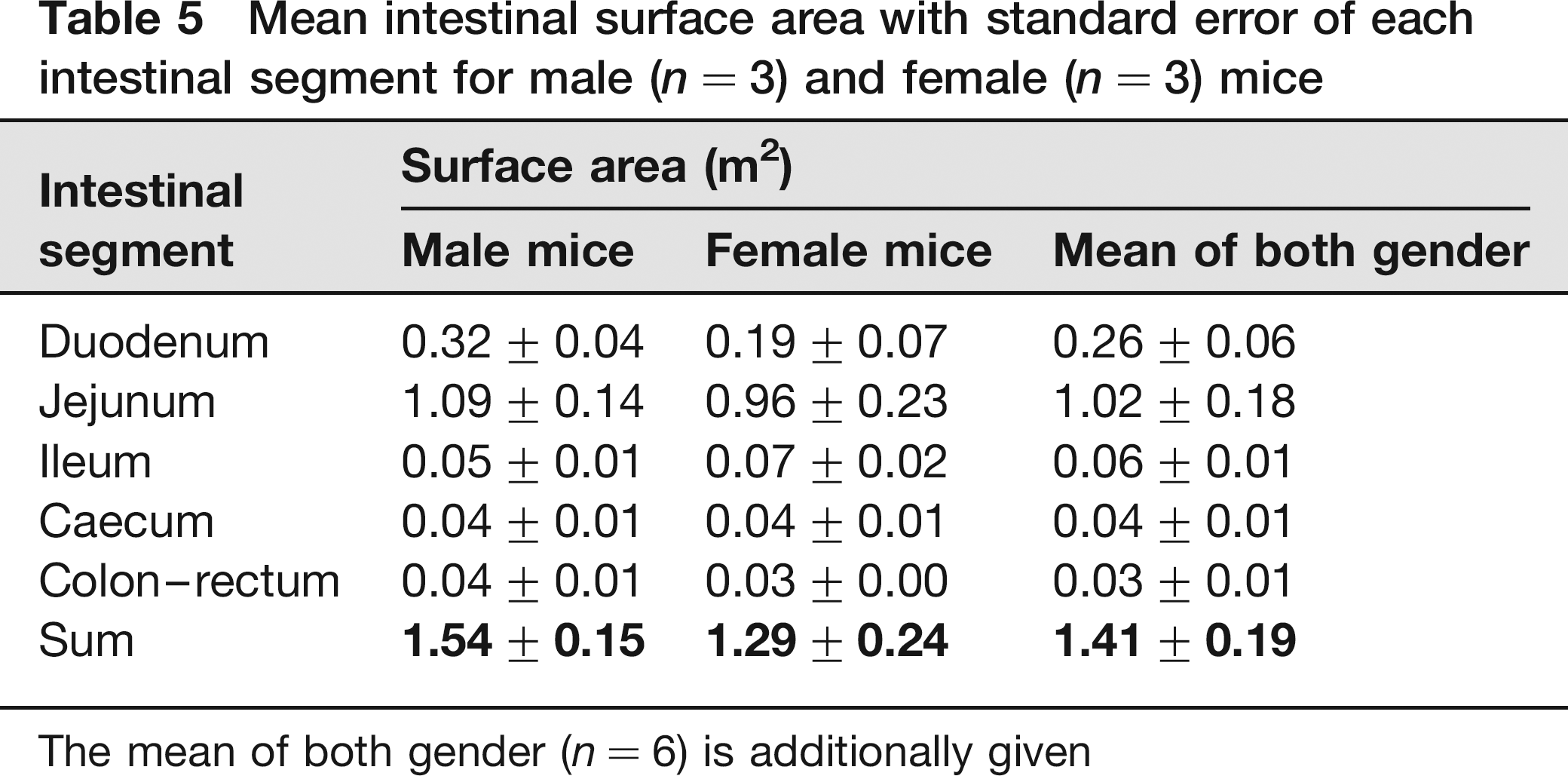
The mean of both gender (n = 6) is additionally given
Discussion
The length of the intestinal tract is a crucial factor when assessing its volume and surface area. While it has been suggested that loss of muscle tone after death could lengthen the intestines, 14 Choi and Chiou 15 have demonstrated that death has no significant effect on the intestinal length. Measuring this length must of course be performed with care and under standardized conditions as stretching may lead to an overestimation. 16 In the present light microscopical study, the mean lengths of the small and large murine intestines were 43.8 and 11.7 cm, respectively. In humans, the length of the small and large intestines are approximately 7 and 1.5 m, respectively.17,18 The human jejunum and ileum have an equal length of approximately 3 m. 17 In contrast, the murine jejunum is much longer than the ileum since the latter segment is defined as the terminal transverse part of the murine small intestine. 3 The relative proportions of the small and large intestines are, however, similar in humans and mice (approximately 80% and 20%).
To empty the intestines, food was withheld from the mice used in our study for 24 h. Fasting can, however, have an effect on the mucosal surface area. 19 In 24 h fasted rats, a significant reduction of the intestinal length is present. 20 Such a length reduction seems, however, not to be present in our mice since their measured intestinal lengths were definitely not smaller than the values found in the literature. 21 The villus and MAFs also decrease when rats are fasted longer than 24 h.19,22,23 From this we may conclude that fasting during one day had no influence on the intestinal surface areas of the mice used in the present study.
Another factor that can influence the MAF is the location of the microvilli on the villi. The microvillus surface area and the number of microvilli increase as enterocytes migrate from the crypts to the tips of the villi. The microvilli in the crypts are thus shorter, broader and more widely spaced than those at the tip. In our study the microvilli dimensions and densities were determined on microvilli that were located in the middle regions of the villi. This strategy probably rendered a good representation of the mean microvillus characteristics.
Due to critical point drying, which preserves morphology with a degree of shrinkage comparable to that obtained by routine histological processing, 24 our measurements of the number of microvilli per μm2 overestimate the in vivo situation as we used the scale bar of the SEM micrographs to define 1 μm2. On the other hand, the measurements of the microvillus lengths and diameters on the TEM micrographs probably underestimate the in vivo dimensions due to shrinkage during TEM processing. Since both shrinkage effects could more or less compensate each other, the MAF determined in the present study should be a fair estimate of the in vivo situation.
The data concerning the murine microvilli shown in the present study might be very valuable since very few morphometric studies on murine microvilli have been performed. In contrast, abundant data about rat microvilli are available.22,25–27 Smith et al. 2 mention that murine microvilli of the small intestine increase from 0.15 to 1.09 μm from villus crypt to tip, with the depth of a crypt and the height of a villus being 80 and 346 μm, respectively. The mean length of the microvilli present on the middle regions of the villi of the small intestine was 1.16 μm in our study. This value is thus somewhat larger than that obtained by Smith et al. 2
The value of the overall intestinal surface area of the mouse is of major importance for the appropriate dose translation of newly developed therapeutic compounds from experimental animals to humans. Literature data about the human intestinal surface area vary, however, widely. The most recent data have been generated by Niess and Reinecker, 28 who estimate that the human intestinal surface area is approximately the size of a tennis court (260–300 m2). Ritschel29 and Snyder et al. 17 report values of 120–200 m2 for the small intestine and of 0.3 m2 for the large intestine. The difference between humans and mice regarding the proportions of the surface area of the small and large intestines is striking. In humans, the surface area of the large intestine is approximately 400 times smaller than that of the small intestine. This is only 18 times in mice.
Although final therapeutic doses are often expressed per kg body weight, starting dose calculations in humans as extrapolated from animal studies should be expressed per kg metabolic weight, which is directly correlated to body surface area (BSA). 30 By dividing the intestinal surface area by the BSA, the relative intestinal surface area is obtained. 1 Using the formula to calculate the murine BSA, 31 the relative intestinal surface area of the mice used in the present study was 119, being quite similar to that of humans, which is 111. 1 As a result, an equal absorptive capacity in terms of unit BSA seems to exist in mice and humans. 32 However, a similar degree of dose dependency in absorption has also been found in rats and humans, although the relative intestinal surface area of the rat is only 26. The pharmacological characteristics of a compound are thus not only determined by the mucosal surface area but also by other parameters such as the vascularization of the intestines, transit time of the feed, gut motility and enterohepatic recirculation. 1 Notwithstanding the potential influence of these factors, the results obtained in the present study are obviously a prerequisite for dose extrapolation from mice to humans.
Footnotes
Acknowledgements
The authors would like to thank L De Bels, B De Pauw, L Standaert and P Vervaet for their technical assistance.
This work was financed by BOF Grant 05B01906 of Ghent University and by ActoGeniX NV.
C Casteleyn was involved in drafting the manuscript, sample collection and analyses, and study set-up. A Rekecki assisted in the sample collection. A Van der Aa was responsible for the study set-up. P Simoens and W Van den Broeck critically reviewed the manuscript and assisted in the study set-up.
