Abstract
This study evaluated the comparative plasma dispositions of ivermectin (IVM), doramectin (DRM) and moxidectin (MXD) following subcutaneous administration in rabbits. Fifteen New Zealand white rabbits were allocated into three groups of five animals each. The animals in each group received IVM, DRM or MXD by subcutaneous injection at a single dose of 0.3 mg/kg. Blood samples were collected at various times between 1 h and 40 days after treatment and the plasma samples were analysed by high-performance liquid chromatography using fluorescence detection. Moxidectin was absorbed faster from the injection site and reached the peak plasma concentration (C max) significantly earlier than IVM and DRM. There was no significant difference in C max values among the three molecules, whereas the area under the concentration–time curves of DRM (258.40 ng.d/mL) and IVM (191.62 ng.d/mL) was significantly higher than that of MXD (83.17 ng.d/mL). The mean plasma residence time and terminal half-life (t 1/2λz ) were longer for DRM (7.52 and 4.48 days, respectively) and MXD (8.97 and 8.16 days, respectively) compared with IVM (4.73 and 2.75 days, respectively). Considering the pharmacokinetic parameters for the parent molecules, the persistence of DRM and MXD are significantly longer than IVM and this may have a positive effect on their efficacy in rabbits following subcutaneous administration or utility relating to interdosing interval.
Avermectins and milbemycins have been used intensively to control parasites of animals, humans and crops worldwide. Both chemical groups are naturally derived 16-membered macrocyclic lactones 1 produced by the soil dwelling actinomycetes, Streptomyces spp. Milbemycins are structurally related to the avermectins and share physicochemical properties with avermectins. Avermectins and milbemycins have excellent activity against nematodes and have systemic activity against several pathogenic ectoparasites of domestic animals at low dosage rates. Due to the high activity of avermectins and milbemycins against both nematodes and arthropods, they are now classified as ‘endectocides’. 2 The pharmacokinetic behaviour of avermectins and milbemycins is significantly affected by route of administration, formulation of the drug, and interspecies and interindividual variation. 2 These anthelmintics are highly lipophilic substances, and are extensively distributed throughout the body and slowly eliminated through tissues such as liver and fat. 3 For this reason a larger volume of distribution (V d) may be obtained for these compounds compared with other anthelmintics. 4
Rabbits are widely used as laboratory animals for animal experiments. Endectocides are used extralabelly to control particularly ectoparasites, such as mite and mange infestations in rabbits. 5−12 Pharmacokinetic behaviours of endectocides have been extensively studied in different animal species. Although kinetic dispositions of ivermectin (IVM) have been reported previously, 6,13 very little information is available on the plasma pharmacokinetics of doramectin (DRM) and moxidectin (MXD) in rabbits following subcutaneous administration. However, extralabel use of endectocides for the treatment of endo- and ectoparasites in rabbits has been increasing recently. Therefore, this study aimed to provide scientific data on the comparative pharmacokinetics of endectocides for specialists including veterinarians with an interest in laboratory animal medicine. For this purpose the comparative plasma disposition of IVM, MXD and DRM was conducted over a 40-day period in rabbits following single subcutaneous administration at a dose rate of 0.3 mg/kg.
Material and methods
Animals and study design
A total of 15 six-month-old New Zealand white rabbits (nine males and six females and supplied from Animal Research Laboratory, Faculty of Art and Science, Kafkas University) with body weight (BW) ranging from 2.5 to 3.4 kg were used in this study. For the duration of the study, the animals in each group were housed in individual stainless-steel cages with wired bottom and rough hay at 20°C under a 12 h light/12 h dark cycle in the Faculty of Veterinary Medicine. The animals were fed with grass hay and feedlot starter pellets with lipid content of about 3–3.5% with free access to food and water. This study was approved by the Animal Ethic Committee of the University of Adnan Menderes.
The animals were allocated into three groups of five such that the mean BW and gender of animals in each group was similar. Group I (IVM), group II (DRM) and group III (MXD) received a subcutaneous injection of the commercially available formulation of IVM (Vilmectin, 1%, Vilsan, Ankara, Turkey), DRM (Dectomax, 1%, Pfizer, Istanbul, Turkey) or MXD (Cydectin, 1%, Fort Dodge, USA), respectively. Drugs were administered by subcutaneous injection in the lateral midline of the back as a single dose at 0.3 mg/kg BW.
Heparinized blood samples (1–2 mL) were collected by inserting a 20-gauge needle into the marginal ear vein one day prior to drug administration and 1, 2, 4, 8, 12, 24, 32, 48, 72, 96 h and 5, 6, 8, 10, 12, 15, 20 25, 30 and 40 days post-treatment. During the first 24 h sampling, a total of six withdrawals were performed. Approximately, 1 mL of blood sample was withdrawn at each time point (l, 2, 4, 8, 12 and 24 h). The total blood volume that was collected from each animal would be equal to about 6 mL for a 24 h period and about 19 mL for the first month. Blood samples were centrifuged at 2000
Analytical procedures
The parent compounds of IVM, DRM and MXD in plasma were analysed by high-performance liquid chromatography with a liquid–liquid phase extraction procedure, as previously described by Gokbulut et al. 14 Stock solutions (100 μg/mL) of pure standard of IVM (Sigma, St Louis, MO, USA), DRM (Pfizer Inc, Groton, USA) and MXD (American Cyanamid, Princeton, NJ, USA) were prepared using acetonitrile as the solvent. These were diluted to give 1, 5, 10, 100, 250, 500 ng/mL standard solutions for calibration as standard curves and to add to drug-free plasma samples for determination of recovery.
Drug-free plasma samples (0.25 mL) were spiked with IVM, DRM or MXD standards to reach the following final concentrations: 0.25, 0.5, 1, 10, 25 and 50 ng/mL. Doramectin (50 ng) was used as an internal standard for IVM and MXD studies, and IVM was used for DRM (50 ng) study. Acetonitrile (0.25 mL) was added, and after vortexing for 15 s, 3 mL trichloromethane was added. The tubes were shaken on a slow rotary mixer for 15 min. After centrifugation at 2000
The analytical method used for IVM, DRM and MXD in rabbit plasma was validated prior to the start of the studies. Recovery of the three molecules under study was measured by comparison of the peak areas from spiked plasma samples with the areas resulting from direct injections of standards. The inter- and intra-assay precision of the extraction and chromatography procedures were evaluated by processing replicate aliquots of initially drug-free rabbit plasma samples fortified with known amounts of the drugs on different days. Calibration graphs for IVM, DRM and MXD were prepared (linear range 0.25–50 ng/mL). The limits of quantification of the assay were defined as the lowest concentration that would have a coefficient of variation (CV) <20%.
Pharmacokinetic and statistical analysis of data
The plasma concentration versus time curves obtained after each treatment in individual animals were fitted with the WinNonlin software program (Version 4.1, Pharsight Corporation, Mountain View, CA, USA). Pharmacokinetic parameters for each animal were analysed using non-compartmental model analysis with extravascular input. The C
max and t
max were obtained from the plotted concentration–time curve of each drug in each animal. The linear trapezoidal rule was used to calculate the area under the concentration–time curves (AUC):
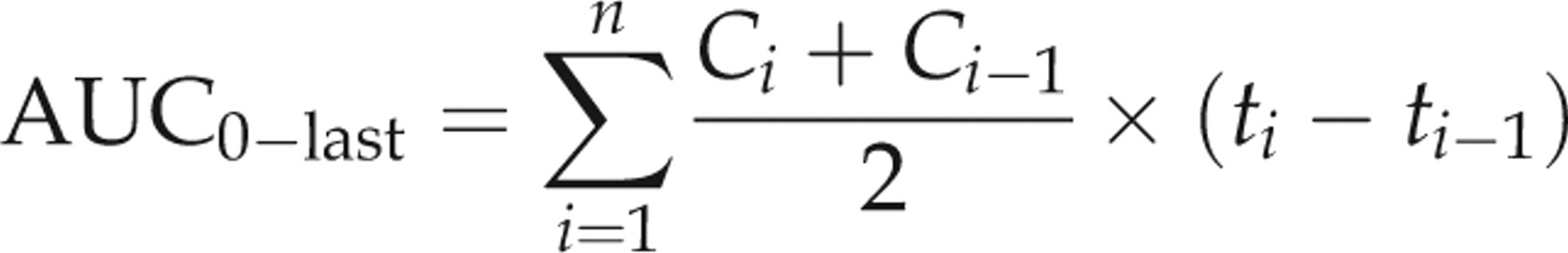
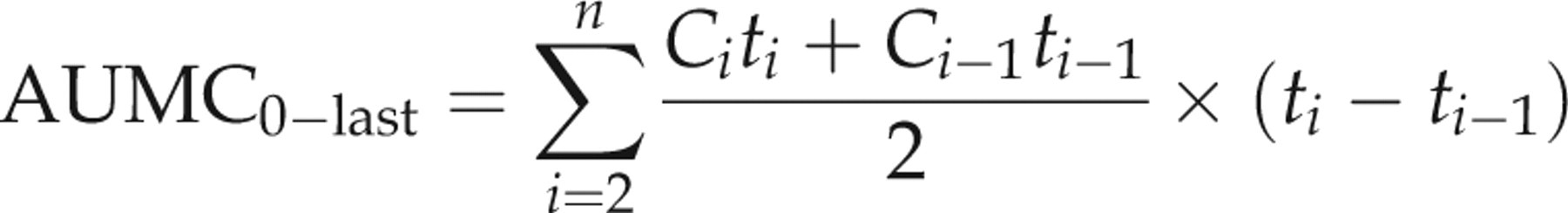
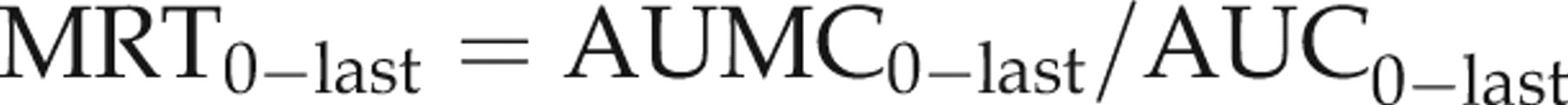
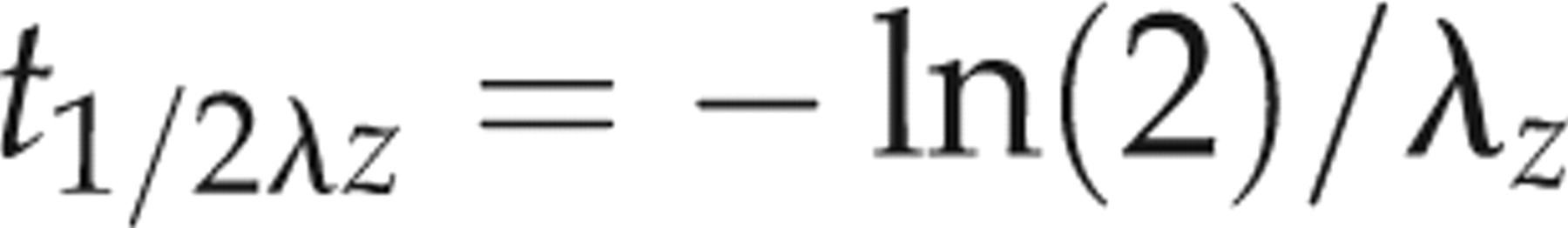
The pharmacokinetic parameters are reported as mean ± SD. The pharmacokinetic parameters of each group were statistically compared by a one-way analysis of variance. The mean values were considered significantly different at P < 0.05.
Results
No adverse response was observed clinically for any of the treatments during the study. The analytical method used to extract and quantify the plasma concentration of IVM, DRM and MXD by chromatographic analysis using fluorescence detector was validated. The validation parameters are summarized in Table 1.
Validation of analytical method used for determination of ivermectin (IVM), doramectin (DRM) and moxidectin (MXD) in rabbit plasma
Pharmacokinetic parameters of IVM, DRM and MXD are shown in Table 2 with the plasma concentration versus time curves (Figure 1). The parent molecule of IVM was detected in plasma between 1 h and 20 days, whereas the last detectable plasma concentration of DRM and MXD extended to 30 days following subcutaneous administration. The kinetic parameters were not significantly different between male and female rabbits in the same treatment group. There was no difference in C max values, but the AUC of DRM (258.40 ng.d/mL) and IVM (191.62 ng.d/mL were significantly higher than that of MXD (83.17 ng.d/mL). The mean plasma residence times (MRT) and terminal half-lives (t 1/2λz ) were longer for DRM (7.52 days and 4.48 days, respectively) and MXD (8.97 days and 8.16 days, respectively) compared to IVM (4.73 days and 2.75 days, respectively).
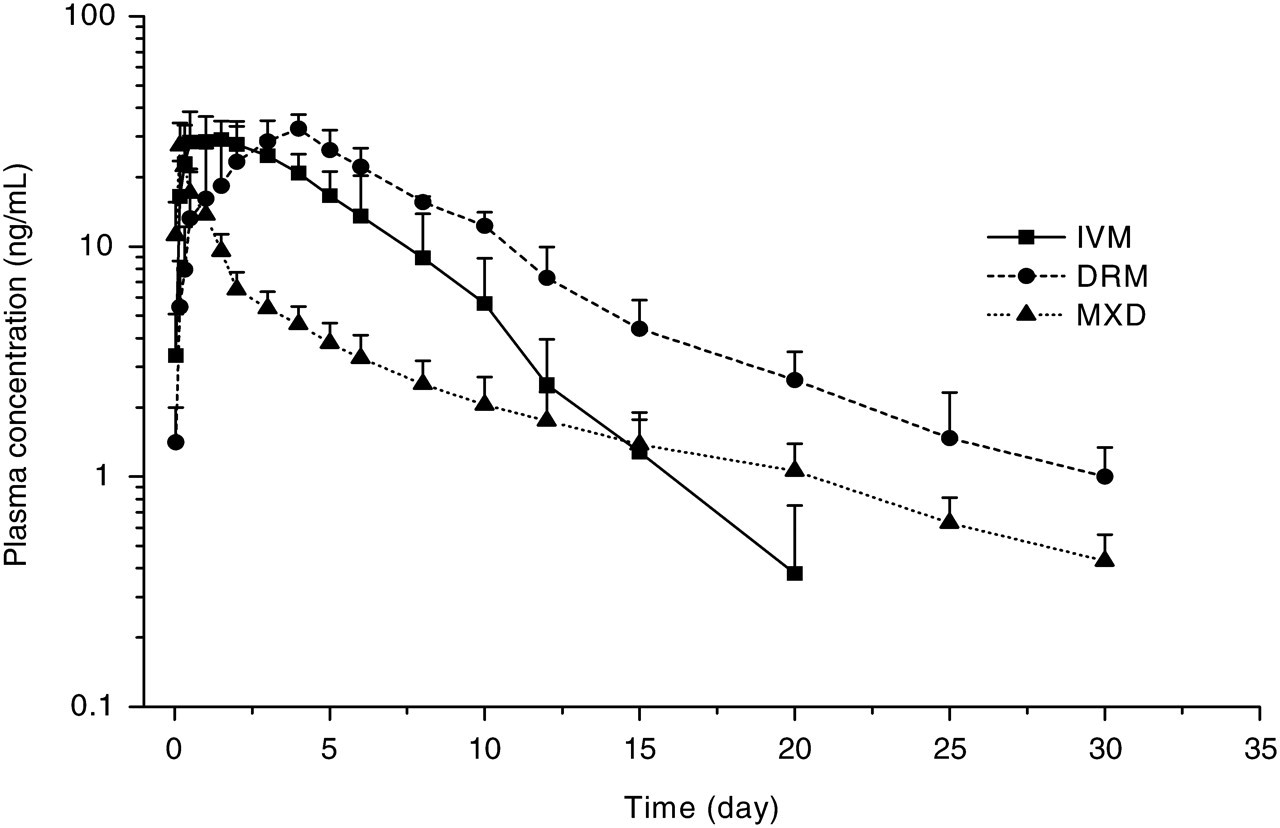
Mean (±SD) plasma concentration of ivermectin (IVM), doramectin (DRM) and moxidectin (MXD) following subcutaneous administration to rabbits at a dose rate of 0.3 mg/kg (n = 5)
Mean (±SD) pharmacokinetic parameters of ivermectin (IVM), doramectin (DRM) and moxidectin (MXD) following subcutaneous administration to rabbits at a dose rate of 0.3 mg/kg (n = 5)
t max: time to reach peak plasma concentration; C max: peak plasma concentration; AUClast: area under the (zero moment) curve from time 0 to the last detectable concentration; t 1/2λz : terminal half-life; AUMClast: area under the moment curve from time 0 to the last detectable concentration; MRTlast: mean residence time
Mean kinetic parameters of IVM are significantly different from DRM (¥ P < 0.05) and MXD (*P < 0.05, **P < 0.01)
Mean kinetic parameters of DRM are significantly different († P < 0.05, †† P < 0.01) from MXD
Discussion
This article reports the first study in rabbits on the comparative pharmacokinetics of IVM, DRM and MXD following subcutaneous injection. Pharmacokinetic parameter of IVM has been reported in rabbits previously following subcutaneous administration. 6,13 The results of the present study differ from those previously reported for IVM in rabbits. 6 In the previous study IVM was administered at 0.4 mg/kg whereas in this study it was administered at 0.3 mg/kg. Nevertheless the C max of 42.0 ng/mL obtained by McKellar et al. 6 was higher, whereas the AUC (147.6 versus 191.6 ng.d/mL) was smaller than that obtained in the present study. Differences in the two studies are probably associated with the terminal half-lives since IVM given at a higher dosage rate of 0.4 mg/kg in the previous study displayed earlier last detectable plasma concentration compared to the present study (12.5 versus 20 days). These differences may in part be due to differences in methodology. Parasitism could have had an effect since the rabbits used by McKellar et al. 6 were known to be infected with the ear mite Psoroptes cuniculi. Another likely factor affecting the pharmacokinetics of IVM was the size of the animals used, since Shoop et al. 15 indicated that there was a high correlation (r 2 = 0.922) between BW and time until MXD is no longer detectable in sheep. McKellar et al. 6 used animals weighing 2.2–5.0 kg, whereas animals weighing between 2.5 and 3.4 kg were used in the present study.
The results for IVM in the present study also differ from those previously reported in another study in rabbits at the same dose rates. 16 The C max and AUC obtained in this study were higher (32.02 versus 20.82 ng/mL) and the AUC much larger (191.6 versus 73.52 ng.d/mL) compared with the previous study reported by Bassissi et al. 16 Although no significant difference was observed in the kinetic parameters between male and female rabbits in all three treatment groups of the present study because of the small animal numbers, the differences in the two studies may be associated with the gender of the animals used in the studies. Three male and two female rabbits were used in the each group in the present study, whereas all animals were male in the previous study reported by Bassissi et al. 16 There may be species and sex specificity regarding the macrocyclic lactone kinetics or metabolism. No sex difference was observed in vivo in cattle for IVM metabolism 17 and lack of sex-influence on the in vitro metabolism of IVM by hepatic microsomal preparations from cattle. Moreover, a gender effect for kinetic disposition of selamectin 18 and MXD 19 has been observed in dogs. It has been recently reported that significantly larger (68.7%) AUC of IVM was observed in female sheep compared with male sheep after subcutaneous administration. 20
In a previous study, the effect of lipid ingestion on the kinetic disposition of MXD has been investigated in rabbits following oral administration at a dose rate of 0.3 mg/kg. 13 This study suggested that lipid administration increases the systemic availability of oral MXD. However, the plasma availability of MXD after oral administration in the previous study was much lower (C max: 7.44 ng/mL, AUC: 8.62 ng.d/mL, t 1/2λz : 3.85 days, MRT: 1.52 days) compared with the present study (C max: 27.42 ng/mL, AUC: 83.17 ng.d/mL, t 1/2λz : 8.16 days, MRT: 8.97 days) following subcutaneous administration at same dose rates. This difference suggests that the bioavailability of MXD is remarkably lower after oral administration compared with the subcutaneous route in rabbits.
Previous studies have shown that the plasma disposition of endectocides in different animal species is significantly affected by the solvent vehicle of the drug formulation. The vehicle in the formulation may influence the absorption process and thus drug disposition in the circulation 21,22 and at the target site where parasites are located. 23 The formulations of IVM, DRM and MXD are different. IVM is formulated in propylene glycol/glycerol formal (60:40); DRM contains sesame oil/ethyl oleate (90:10) whereas MXD is formulated in aqueous solution in a vehicle containing benzyl alcohol (4.00% w/v), butylated hydroxytoluene (0.25% w/v) and disodium edetate (0.027% w/v). The absorption of MXD from the injection side was significantly faster compared with those of IVM and DRM. MXD reached the plasma peak concentration significantly earlier (0.17 day) than those of IVM (1.10 days) and DRM (3.10 days). However, the plasma decline of MXD was initially faster in comparison to IVM and DRM following subcutaneous administration in the current study (Figure 1). In contrast, the MRT value of MXD (8.97 days) is longer than the values for DRM (7.52 days) and IVM (4.73 days). These results may also be associated with greater proportions of MXD accumulating in fat tissue than IVM and DRM, since it has been demonstrated that the concentration of MXD in fat tissue was 90-fold higher than that detected in plasma 28 days following treatment in cattle. 24 The higher fat tissue reservoir of MXD may explain the extended persistence of that molecule compared with that of the avermectins. Moreover, it has been reported that MXD is 100 times more lipophilic than IVM 25 and that the water solubility of MXD (4.3 mg/L) 26 is much higher than that of IVM (0.006–0.009 mg/L). 27 The water solubility and lipophilicity of MXD are unusual for a drug since increased water solubility is usually directly associated with decreased lipophilicity.
In conclusion, the results from this study indicate that the persistence of DRM and MXD are significantly longer than IVM and this may have a positive effect on their efficacy in rabbits following subcutaneous administration at a dose rate of 0.3 mg/kg. Moreover, two doses of IVM with a 15–20-day interval between doses could be necessary for treatment of ectoparasites whereas single administration of DRM and MXD probably provide longer persistence efficacy after subcutaneous administration in rabbits.
