Abstract
The results of this study show that the oral administration of ivermectin (48 mg/L) repeatedly for 72 h used in accordance with the present protocol is a safe and highly effective treatment for Giardia spp. and Hymenolepis nana in laboratory rat colonies. The drug can be easily and safely administered using drinking water. This simple regimen should control pinworm infection (Syphacia muris), a problem that can be endemic in laboratory colonies. Experiments using healthy animals are likely to generate more consistent results, thereby requiring a reduced number of animals per group.
Laboratory animals must be kept in appropriate health conditions to ensure the higher efficiency of experimental studies.1–5 Parasitized animals are not suitable for experimental research since they may present nutritional, immunological and haematological differences compared with normal values.6–9 At least three parasites are common threats to rodent colonies: Syphacia spp., Giardia spp. and Hymenolepis nana. A common infection found in rat colonies is due to Syphacia spp., which has also been reported to infect humans. 6 In the host, these worms develop into adults from eggs in 4–7 days. New eggs are deposited in the host’s perianal region, which are easily dispersed in the environment, contaminating other animals. 10 Such infections are usually asymptomatic, but may be associated with rectal prolapse, intussusception, low weight gain, hypertension, irritability and immunological alteration.11–14 Giardia spp. is another common parasite genus found in rat colonies. The infecting cysts are maximally present in faeces 5–14 days after ingestion by the host. 15 Indeed such infections are asymptomatic, but unspecific clinical signals may be present such as weight loss, lower growing rate, abdominal distension and diarrhoea. 2 H. nana is a threat to both animals and humans. It has a direct cycle of between 14 and 16 days duration. Severe infection with H. nana leads to dehydration, abdominal pain, malnutrition, diarrhoea and weight loss. This is the only Taenia that infects humans without needing an intermediate host.9,16,17
Ivermectin is a macrocyclic lactone widely used as an antiparasitic agent either in veterinary or in human medicine. It targets a glutamate-gated chloride channel in some invertebrates as well as the chloride channel of gamma-aminobutyric acid type A (GABAA) receptors, promoting cellular hyperpolarization and worm paralysis. 18 Although generally considered secure and well-tolerated, ivermectin can cause toxic effects in the host depending on its concentration in the central nervous system (CNS), since it is freely permeable through the blood–brain barrier. 19 The aim of the present study was to investigate the occurrence of endoparasites in the intestinal tract of a conventional Wistar rat colony, and to evaluate the efficacy and security of ivermectin for the control of these natural infections.
Animals
Forty male and 50 female Wistar rats (Rattus norvegicus) from a conventional biotery (Florianópolis, UFSC, Brazil) weighing 200–250 and 150–200 g, respectively, were housed under standard conditions for laboratory animals in groups of five with a controlled 12 h light/dark cycle, temperature 20 ± 2℃ and ad libitum access to standard diet and water. All experimental procedures were conducted according to the European Convention for the Protection of Vertebrate Animals and were approved by the local ethical committee for animal research (CEUA–UFSC).
Materials and methods
Sample collections and laboratory techniques
Faecal sampling was obtained on three consecutive days before the treatment, and on another three consecutive days a week after the treatment. Faecal material was collected directly from the anus and stored in a thimerosal–iodine–formaldehyde solution at room temperature in a proportion of 1:10. Samples from 10 animals were pooled and submitted to the Hoffman–Pons–Janer (HPJ) technique, Willis technique, and also direct visualization using an optical microscope.
HPJ can detect the presence of helminth eggs and larvae, and protozoan cysts. This technique utilized spontaneous faecal sedimentation. 20 Briefly, faecal samples were diluted in water and filtered through a gauze strip into a conical sedimentation glass and the sediment was evaluated. The Willis technique is based on the floating effect of helminth eggs in a saturated sodium chloride solution (1.2 g/mL). 21 Using this technique, the saturated saline solution with the emulsified faeces was deposited in a round-bottomed flask and a meniscus was formed on the surface. Next, the flask was covered with a glass slide. After several minutes, the slide was removed and examined under a microscope.
Finally, a direct examination was performed using an optical microscope and Lugol staining. The results were expressed as number of eggs in the samples.
Drug and method of administration
Ivermectin (Hebei Veyong Animal Pharmaceutical Co Ltd, Shijiazhuang, China) was dissolved in water (48 mg/L) and was offered alone ad libitum during a 72 h period. Control groups received tap water only.
Investigation of ivermectin’s depressive effect on the rats’ CNS
To study the potential depressive effect of ivermectin on the CNS, which could cause lasting changes and interfere with behaviour and the dynamics of memory formation, a contextual fear-conditioning protocol 22 was performed seven days after ivermectin treatment. Behavioural testing was always carried out under low intensity illumination (70 lux) during the diurnal phase (from 13:00 to 17:00 h). Fear conditioning was performed in a rectangular chamber (35 × 20 × 30 cm), with aluminium sidewalls and a front wall and ceiling made of Plexiglas. Its grid floor, made of stainless steel bars (3 mm diameter, spaced 9 mm apart centre-to-centre), was connected to a circuit board and a shock generator (Insight, Ribeirão Preto, SP, Brazil) to enable the delivery of controlled electrical foot shocks. In all the experiments, each animal was placed in this environment (Context A), and allowed to explore it freely for 3 min, as an initial familiarization session, and was then returned to its home cage. On the next day, the animal was again placed there for the training session, during which it received, after an initial 30 s delay (pre-shock period), the unconditioned stimulus (three electrical foot shocks of 0.7 mA, 60 Hz, for 3 s, with a 30 s inter-shock interval). The animal remained in this chamber for an additional 30 s (post-shock period) before returning to its home cage. In the test, the animal was re-exposed to this paired context for 3 min in the absence of unconditioned stimulus presentation. All sessions were spaced 24 h apart. After each behavioural session, both chambers were cleaned with paper tissue soaked with a 10% ethanol–water solution. The experimenter was unaware of the treatment condition in all the studies.
Freezing behaviour, a commonly used index of fear in rats, 23 and defined as a total absence of body and head movements except those associated with breathing, was continuously recorded during the experimental sessions by a video camera. The freezing time in each period was quantified (in seconds) by a trained observer (inter- and intra-rate reliabilities ≥ 90%) blind to the experimental groups, using a stopwatch, and expressed as the percentage of total session time.
Statistical analysis
Statistical analysis was carried out using GraphPad Prism 5.0® software (Graphpad Software Inc, La Jolla, CA, USA). Egg occurrence was presented as mean number of eggs/mm3 ± SEM. Comparisons were made using a paired Student’s t-test for simple means. For contextual fear-conditioning protocol the results were expressed as mean ± SEM. After ensuring the assumptions of normality and homogeneity of variance, the percentage of freezing time recorded during the test was submitted to an unpaired Student’s t-test. The statistical significance level was set at P < 0.05.
Results
In male rats, Syphacia eggs were found in 100% of the samples at 8.7 ± 1.2 eggs/mm3, Giardia eggs in 50% of samples at 2.5 ± 1.4 eggs/mm3 and Hymenolepis in 25% of samples at 1.2 ± 1.2 eggs/mm3. In female rats, Syphacia eggs were found in 50% of samples at 3.7 ± 2.4 eggs/mm3 and Giardia eggs in 25% of samples at 2.5 ± 1.4 eggs/mm3. Hymenolepis was not detected in female rats.
Ivermectin treatment eliminated the occurrence of Giardia eggs in males and females, and Hymenolepis in males. The drug reduced the incidence of infection from 100% to 50% in males, which caused a 94% decrease in the number of Syphacia eggs (0.5 ± 0.3 eggs). Similar rates were found in females, where the incidence of infection was reduced from 50% to 25%, with a mean number of eggs of 0.2 ± 0.2 eggs (Figure 1).
Ivermectin is effective in combating Giardia spp. and Hymenolepis nana. Also, it reduces the prevalence of Syphacia spp. A week before and after ivermectin treatment (4.8 mg/L in drinking water for 72 h), parasitological analysis was performed. Values are expressed as mean ± SEM of eggs/mm3. Asterisks indicate a significant difference (P < 0.05) from pre-ivermectin treatment (paired Student’s t-test).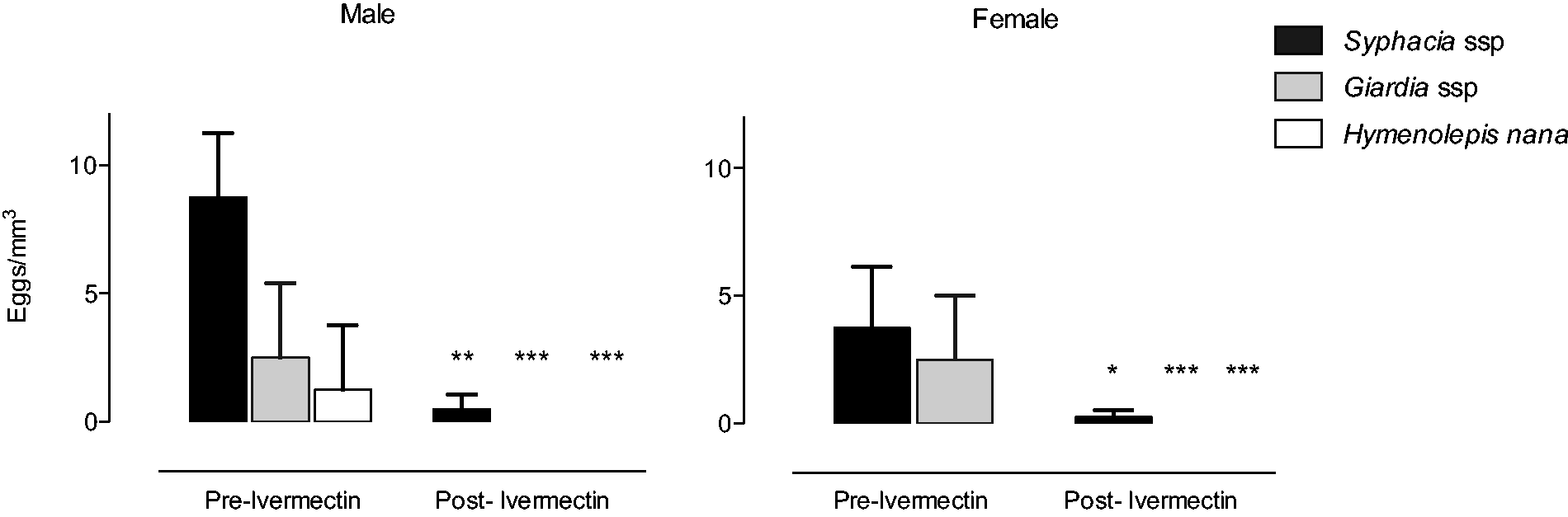
Ten male rats were randomly allocated to two independent groups (n = 5/group) based on the previous treatment, which consisted of vehicle or ivermectin (48 mg/L in drinking water for 72 h), given a week before fear conditioning. As can be observed in Figure 2, there was no difference between treatment condition during re-exposure to the paired context (t8 = 0.004; P = 0.98), evidenced by the lack of residual or long-lasting effects of the drug on memory processing (Figure 2).
The lack of ivermectin-induced impairments in memory encoding or expression in rats. A week after ivermectin treatment (4.8 mg/L in drinking water for 72 h), animals underwent a familiarization session and then entered Context A paired with three foot shocks (US). When re-exposed to the paired context a day later (Test A), groups spent the same amount of time freezing. Bars represent the percentage of total time spent freezing. Values are expressed as mean ± SEM.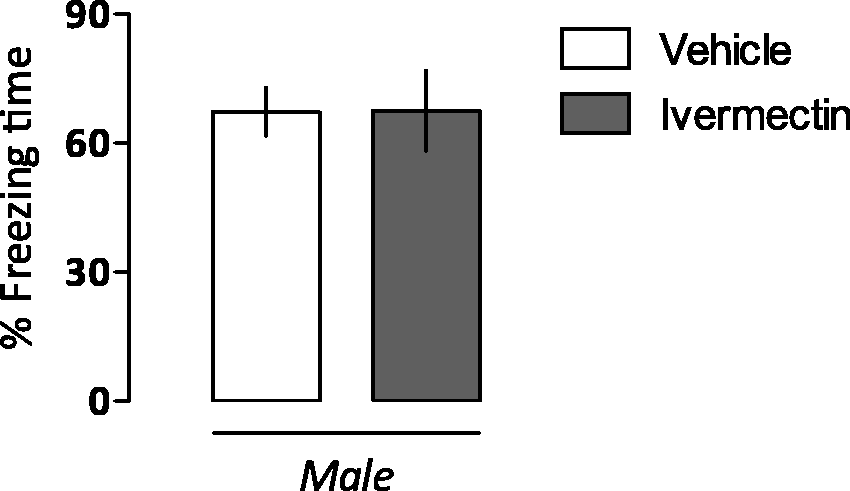
Discussion
The endoparasites encountered in our animals were the same as those normally described in laboratory animals.2,24 There are several treatments and protocols adopted for the control and eradication of these infecting parasites, and the efficacy of ivermectin has been demonstrated in several species. In hamsters, a dose of 300 µg/kg subcutaneously produces an eradication rate of 99.1% and 99.5% in the experimental acute and chronic Giardia infections, respectively. 25 However, a dose of 200 µg/kg intravenously eliminates a natural Giardia infection. 26 Ivermectin doses necessary to control Giardia infection in rats have not yet been described, thus the present study has provided an important reference for a practical treatment in experimental colonies.
In humans, ivermectin (0.2 mg/kg) eliminates 50% of the hymenolepiasis. 27 On the other hand, in hamsters, this drug is ineffective at an oral dose of 0.5 mg/kg. 28 However, an allometric extrapolation 29 suggests that the dose used in hamsters should be threefold lower than the effective one used in humans. Ivermectin is effective in eliminating Hymenolepis eggs in rats. Allometric extrapolation indicates that the above human dose would be equivalent to 1.2 mg/kg in rats. Taking into account the daily water ingestion by rats (100 mL/kg), it is conceivable that the total ivermectin dose was about 5 mg/kg, which may explain the effectiveness against hymenolepiasis.
The efficacy of ivermectin against pinworm (Syphacia muris) in several studies has been variable, probably due to differences in dose regimen, time of treatment and method of administration. One common factor in such studies that were successful in the elimination of the parasite was the repetition of the treatment at four- to nine-day intervals.30,31 The control of S. muris is difficult, since cross infections occur easily after egg depositions. Although ivermectin is effective against adult and larval forms of Syphacia, it has no effect on eggs. Since eggs are light and resistant, they spread out in the environment, being found intact even in ventilation ducts.10,32 For these reasons, disinfection protocols must be applied concomitantly to pharmacological treatment, until the complete elimination of the eggs is ensured. 33
Since ivermectin acts by enhancing GABAA-mediated transmission, the main inhibitory system in the nervous system, high doses of this drug can be associated with general neuronal depression.19,34 Despite the high dose of ivermectin administered here (5 mg/kg) and the length of treatment (three days), the rats did not show altered performance in the fear-conditioning test. This is in line with a previous report in which it was observed that doses of up to 7.5 mg/kg did not produce visible CNS depression in rats. 19 Memory encoding, a process dependent on brain circuitry integrity, is highly modulated by the GABAergic system and, therefore, highly sensitive to interventions in this inhibitory system.35–38 However, as was observed in the present study, the oral treatment with ivermectin did not impair the encoding of this aversive memory, as no differences in freezing behaviour could be observed between groups.
Overall, the present antiparasitic protocol may be considered to be effective against threatening worms in rat colonies, and also secures the subsequent use of such animals for at least some studies in neuroscience. The collective treatment regimen in drinking water is thought to ease its implementation, reducing the sources of animal distress such as physical restraints and invasive administration. Furthermore, we can assume that this prophylactic measure can even target other parasites, beyond those focused on in the present study, since ivermectin is also effective in ectoparasites, thereby providing better sanitary conditions and homeostasis. Such physiological balance is also critical in reducing individual variability in experimental studies and, consequently, in reducing the number of animals required for generating statistically relevant data.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research was funded by Brazilian government funding agencies CAPES and FAPESC-CNPq/PRONEX.
