Abstract
Intratracheal instillation (ITI) of a test compound is an alternative method to inhalation methods that require complex aerosol generation, exposure chambers and airflow monitoring instruments for exposing the lungs of animals to a test compound. For ITI in the rat, a laryngoscope is generally used for endotracheal intubation, and the procedure is difficult to perform. Therefore, we designed and constructed an automatic video instillator (AVI) for the accurate delivery of a dose of a test compound into the trachea of rats. The device has a videocamera probe for image guidance, and a liquid-crystal display for image display. These two items are used to visualize the larynx and trachea for intratracheal insertion of the tubing, and for placing the tip of the instillation tubing beyond the vocal cords for ITI of the test compound. After a 2 h training session on the use of the AVI in an anaesthetized rat, we assessed the utility of the device by ITI of 0.25% (w/v) solution of Evans Blue dye into the lungs of 30 isoflurane-anaesthetized rats. Necropsy examinations were performed on 20 rats immediately after the completion of the procedure, and on 10 rats three days after the procedure. Based on the results of these examinations, we concluded that the device could be used for rapid, reproducible and successful ITI of a test compound into the lungs of a rat by one operator.
The pulmonary toxicology of environmental toxins to assess their physicochemical characteristics, such as the effect of particle size and distribution, and the biological response that follows their exposure in humans is studied frequently by either inhalation or intratracheal instillation (ITI) of the toxin in rodents. 1 For pulmonary toxicity studies, specialized equipment that includes aerosol generators, airflow monitoring instruments, exposure chambers, exhaust systems and often analytical chemical support is required in order to deliver known amounts of the test compound into the lungs. Consequently, the need for this equipment increases the cost and technical complexity of such studies. 2
ITI has become a feasible alternative method for delivering a toxin into the lung of rodents,3–6 because the required equipment is less sophisticated than that required for inhalation toxicity studies.7–9 This method is also simpler and safer than the inhalation method: many dose levels of compounds with a wide range of physicochemical properties and toxicity can be introduced rapidly into the lungs with minimal risk to the operator. Furthermore, ITI is used often as a screen to determine an appropriate dose range for inhalation toxicity studies. Despite the advantages of ITI over inhalation, the use of ITI for the pulmonary toxicology studies in rodents does have limitations. 10 One such limitation is the resultant pulmonary distribution of the toxin. In inhalation studies, the distribution of the test compound in the right and left lungs is uniform, whereas it is non-uniform when using ITI. In addition, Lakatos et al. suggested that oropharyngeal aspiration should be considered as an alternative to ITI in order to overcome respiratory defence mechanisms when studying the pulmonary toxicity of silica and other particulates in rodents. 11
For ITI, the intratracheal site of instillation of the test compound should be beyond the vocal cords, and substantiated after its instillation. However, it is difficult to visualize and confirm the intratracheal site of instillation through the laryngeal opening of a rodent when using a laryngoscope because of its relatively large size when compared with the small dimensions of the rat oropharynx. In addition, the ITI procedure should not be injurious to the tissues. Therefore, when needles are used to deliver the test compound for ITI, their tips should be rounded so that they are non-injurious to the tissues, and can be passed easily into the lumen of the trachea. In order to identify the intratracheal site of instillation, the trachea can be transilluminated using an external light source instead of a laryngoscope. 12 Furthermore, training is required in order to become proficient in ITI.
With the objective of reducing the difficulty and complexity of the ITI procedure, we developed an automatic video instillator (AVI) for ITI in the rat. This device has a triggeractivated syringe pump for accurate delivery of a predetermined dose of a test compound, and a charged-coupled device (CCD) videocamera probe that is attached to the delivery tubing for direct visualization of the larynx and trachea. In this report, we describe its design and utility for ITI.
Materials and Methods
Animals and test compound
Thirty (15 female and 15 male and approximately 8-week-old) specific-pathogen-free Crj/Bgi:CD Sprague-Dawley rats, which were imported from Charles River Laboratory, Japan by Orient Bio Inc, Sungnam, Korea, were used in the study. The rats were group-housed and acclimatized for one week prior to the study. They were fed with a standard laboratory rodent chow (Lab Diet® #5002, PMI Nutrition International, Richmond, IN, USA), and had access to water ad libitum. The rats were maintained in an Association for Assessment and Accreditation of Laboratory Animal Care International accredited facility on a 12 h light/dark cycle and at 45-60% relative humidity.
Evans Blue dye (Aldrich-Sigma, St Louis, MO, USA) was used as the test compound for assessing the utility of the AVI for ITI. For this purpose, a 0.25% (w/v) solution of the dye was prepared by ultrasonic agitation of the dry powder in phosphate-buffered saline (pH 7.2) for 12 h.13,14
The use of animals and the experimental procedures were reviewed and approved by the Animal Care and Use Committee of the Korea Institute of Toxicology.
Design and construction of the AVI
TheAVI is composed of (a) fused silica tubing (inner diameter 0.5 mm, outer diameter 0.7 mm, Upchurch Scientific, Oak Harbor, WA, USA) for ITI of the dye; (b) a CCD videocamera probe (outer diameter 5 mm) for image guidance; 15 (c) a miniaturized light-emitting diode (LED) as a light source for illuminating the larynx and the vocal cords; (d) a liquid-crystal display (LCD) for image display; (e) a switch for triggering a syringe pump (KDS200, KD Scientific, Holliston, MA, USA) in order to instill the test compound; and (f) a 10 mL syringe (Hamilton, Reno, NY, USA) that is connected to the delivery tubing via a luer adapter (P-659, Upchurch Scientific) (Figures 1 and 2). The surface of the tip of the tubing was smoothed using a fine sandpaper (1000 mesh grade) in order to reduce the likelihood of the tip causing any trauma to the surrounding tissues during its intratracheal insertion.
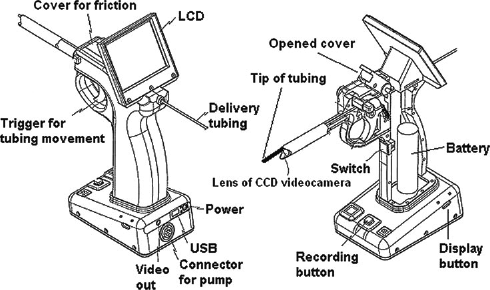
Schematic illustrations of the automatic video instillator. LCD = liquid-crystal display; CCD = charged-coupled device; USB = universal serial bus

Photograph of the tubing and cable connection between the automatic video instillator and the syringe pump
The miniaturized CCD videocamera probe was attached under the fused silica tubing in such a manner that the tip of the tubing was more advanced than the videocamera (Figure 1). For ITI, the thin bundle of the tubing and the videocamera probe was passed through the larynx as far as the vocal cords. When placed at this location, the vocal cords and the trachea are illuminated by the LED, and are seen on the LCD screen. The entire ITI procedure can be recorded and stored on a 2 GB secure digital memory card, and the stored data can then be transferred to a computer through a USB port (Figure 1). Using a wireless receiver and transmitter, the CCD images can be transferred also from the AVI to a computer with a TV tuner card for image display on the computer monitor. Therefore, the CCD video images can be displayed simultaneously on the LCD screen of the AVI and a computer monitor (Figure 3).

A photograph of a charged-coupled device video image that is displayed simultaneously on the liquid-crystal display screen of the automatic video instillator and on a computer monitor at the time of intratracheal instillation of a 0.25% (w/v) solution of Evans Blue dye
The AVI has a trigger for advancing the tubing, and for instilling the test compound intratracheally. Due to the fact that the tubing is sandwiched between the upper surface of the trigger and a cover (Figure 1), friction can be generated, and is used to advance the tubing only when the trigger is pulled. For this purpose, the cover is closed during the ITI procedure in order to generate the friction. When the trigger is released, the tubing is moved backwards. When the cover is open, it is possible to adjust the tubing length.
Using this trigger, the tip of the tubing is passed through the laryngeal opening into the trachea, and is then advanced in 5 mm steps to a position beyond the vocal cords so that the site of ITI is constant in each rat when many rats are used in a pulmonary toxicology study. Once the site of ITI is chosen, the injection switch is then triggered by the middle finger of the hand of the operator to instill the test compound intratracheally.
Training is required to use the AVI. In addition to learning howto use the device, training necessitated the use of an anaesthetized rat, and lasted about 2 h. During the training session on an isoflurane-anaesthetized rat, the operator is trained to pass the tubing through the laryngeal opening, advance the tubing and to activate the syringe pump for ITI so that these actions coincide with the end of the expiratory phase and the start of the inspiratory phase of the respiratory cycle. Upon completion of the training, the rat was killed humanely by an overdose of isoflurane.
Intratracheal instillation using the AVI
For assessing the utility of the AVI for ITI, each rat was anaesthetized by placing the rat in a 1000 mL glass jar with a wire-mesh floor that contained 1 mL of isoflurane for approximately 60 s. Once anaesthetized, the rat was removed from the jar, and then placed on its back in a head-up position on a slant board whose angle was approximately 30° from the horizontal. 16 For this purpose, the upper incisors of the anaesthetized rat were held by a rubber band, which was fixed by two pins on the slant board. The tongue was gently moved to one side of the oral cavity, and then fully extended with small blunt forceps (Figure 3). The AVI was then used to visualize the larynx, and the trigger on the AVI was used to advance the tubing through the larynx and vocal cords (Figure 4).
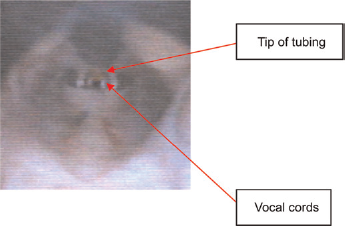
Charged-coupled device video image of the tip of the tubing while passing it over the vocal cords of an anaesthetized rat for intratracheal instillation using the automatic video instillator (see also the video in the online supplementary material at http://la.rsmjournals.com/cgi/content/full/la.2009.009003/DC1)
After confirming the intratracheal position of the tip of the delivery tubing on the LCD screen, 60 μL of 0.25% Evans Blue dye solution was instilled intratracheally by triggering the syringe pump whose flow rate was set at 3600 μL/min. It takes about one minute to perform endotracheal intubation and ITI using the AVI.
After completion of ITI, the tubing was withdrawn from the trachea, and the rat's tongue was then released gently. The rat was held in the operator's hand in a head-up position for 2 min until the rat recovered from anaesthesia and the procedure. The duration of the entire procedure, which includes anaesthesia and positioning of the rat for ITI, the ITI procedure and recovery from anaesthesia, lasts approximately 5 min. A full-motion video that shows the use of the AVI for ITI is in the online supplementary material at http://la.rsmjournals.com/cgi/content/full/la.2009.009003/DC1
After completion of the ITI procedure, necropsy examinationswere performed in 20 rats in order to confirm the presence of dye in the lungs. For this purpose, the rats were killed by plunging them into a saturated isoflurane environment 5 min after their recovery from anaesthesia. The remaining 10 rats were monitored clinically for signs of trauma to the oropharynx, such as bleeding from the mouth, and/or swelling of the pharynx, as well as their eating and drinking ability for three days. After three days, the rats were then killed by plunging them into a saturated isoflurane environment, and necropsy examinationswere then performed in order to determine whether the procedure had caused any trauma and/or haemorrhage in the pharynx, vocal cords and trachea.
Results
The trained operator needed only one attempt to successfully intubate 30 isoflurane-anaesthetized rats for ITI using the AVI.
On necropsy examination, we found the dye in the lungs of the 20 rats that were killed 5 min after their recovery from anaesthesia and the ITI procedure. Figure 5 shows the typical uneven pulmonary distribution of Evans Blue dye following ITI in an isoflurane-anaesthetized rat using the AVI. The dye was also found in the stomach of two rats. The amount of the dye in the stomach of one of the rats was lesser than that found in the stomach of the other rat. We presume that its gastric presence was not due to its direct instillation, but possibly due to its backflow from the trachea and subsequent swallowing.
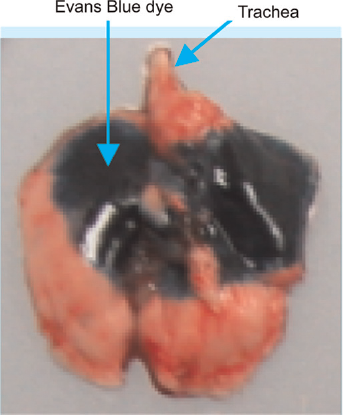
Dorsoventral photograph of the lungs of one of the 20 rats that was killed 5 min after its recovery from anaesthesia and the intratracheal instillation (ITI) procedure. The arrows point to the uneven pulmonary distribution of Evans Blue dye in the left and right lungs after its ITI
Of the 10 animals that were monitored clinically for three days after ITI, and then killed humanely, we found the evidence of haemorrhage in the region of the vocal cords in one rat at the necropsy examination. From these results, we concluded that the success rate of ITI using AVI is at least 90%, and that the AVI is non-injurious to the rats.
Discussion
Visualizing the laryngeal opening is important in order to perform successful ITI in the rat and reduce the time of the procedure. There are two published reports on the success rate of endotracheal intubation in rats. Stark et al. 17 reported that the success rate for blind oral tracheal intubation was less than 90%. This success rate is lower than that reported by Vergari et al., 18 who used a fibreoptic arthroscope for orotracheal intubation. Vergari et al. reported 100% success rate, and the duration of the procedure was less than 5 min. We presume that this time is shorter than the time required for blind oral tracheal intubation. When using the AVI for ITI, the duration of the entire procedure is about 5 min, which is comparable to the duration of procedure by Vergari et al. Endotracheal intubation for ITI in the rat is carried out usually by two people: one to hold the rat's tongue and the other to intubate the animal using a laryngoscope, and generally involves several attempts to intubate the animal for successful ITI. In addition, training is required to become proficient in the procedure, and the duration of this training is generally longer than the 2 h that is required to become proficient in ITI using the AVI. When using the AVI for ITI, a single operator, who had undergone a 2 h training session, needed one attempt only to perform successful ITI in an isoflurane-anaesthetized rat. Moreover, the success rate of the operator was at least 90%, and this rate is comparable to that reported by Vergari et al. When compared with other ITI methods in the rat, we have demonstrated that ITI using the AVI is rapid, and can be done easily with a high success rate by one person.
Many devices that include fibreoptic illuminators, otoscopes and laryngoscopes can facilitate reliable endotracheal intubation in the rats.19,20 When using a laryngoscope for endotracheal intubation in the rat, it may obstruct the operator's view of the laryngeal cavity when attempting to insert the endotracheal tube into the trachea. When using the AVI for ITI, the CCD videocamera and the LED light source do not obstruct the operator's view of the oropharynx, and the vocal cords and epiglottis can be observed in their entirety on the LCD monitor. The CCD video images can be transferred from the AVI to a computer with a TV tuner card using awireless receiver and transmitter while the procedure is being done. In addition, the CCD video images can be stored in the memory card of the AVI, and then transferred to a computer for later viewing. As a result, the video recording of ITI can be used to review the entire procedure in a pulmonary toxicological study.
In their comprehensive and critical review of ITI, Driscoll et al. 10 compared the differences between the distribution, clearance and retention of test compounds when they are administered by ITI or inhalation. They commented that the most consistent disparity between inhalation and ITI relates to the intrapulmonary distribution of the test compound. Inhalation results in a relatively uniform distribution of the test compound throughout the lungs, whereas ITI generally results in a non-uniform distribution of the test compound with the frequent occurrence of high content pulmonary foci. Therefore, our finding of an uneven pulmonary distribution of Evans Blue dye after ITI using our AVI is not surprising because it is to be expected.
We foresee that the AVI can be developed for continuous ITI of volumes greater than 60 μL. We think also that the design of the existing device can be improved so that the triggering of the syringe pump can be synchronized automatically with the end of the expiratory phase and the start of the inspiratory phase of the respiratory cycle by a computerized system. Another future development will be to adapt the AVI for ITI in the mouse because the use of mice is becoming increasingly popular in many laboratories that conduct studies in pulmonary toxicology. 21 This development will utilize a very thin optical fibre for image guidance and narrow tubing,8,22 whose combined diameter will be considerably smaller than that of the bundle of the CCD videocamera probe and tubing in the AVI for the rat. In addition, the length of the trigger-activated step to advance the tubing will need to be made shorter because of the length of the mouse trachea.
In summary, we have shown that ITI using the AVI is a rapid, reproducible and non-injurious method for visualized administration of a test compound into the lungs of a rat by one operator. Furthermore, the device is easy to use, and its operator requires minimal training on its use. Although the AVI cannot be used in studies in which large numbers of animals are necessitated, or when repeat dosing in a single animal is required because of the need for repeated anaesthesia, we believe that the AVI will be useful for screening and making pharmacokinetic/toxicokinetic comparisons of environmental toxins in pulmonary toxicological studies in the rat.23,24
Footnotes
Acknowledgements
We appreciate the efforts of Dr Arieh Bomzon for reviewing our manuscript with constructive comments. This study was supported by a Ministry of Science and Technology for 2007 General Project grant to establish the Center for the International Inhalation Toxicology Evaluation Technology at the Korea Institute of Toxicology.
