Abstract
The aim of this review is to evaluate the suitability of rat molar teeth in preclinical evaluation of medical devices for direct pulp capping. The ISO standard 7405 states clearly that only non-rodent mammals are suitable species for animal research in dentistry. Furthermore, without clear justification a considerable number of researchers previously rejected results of animal experiments concerning preclinical evaluation of the biocompatibility of dental materials undertaken in rat molar teeth. However, in the past 50 years about 70 studies have been published using rat molar teeth in order to evaluate direct pulp capping, pulpotomies and tissue reactions after pulp exposure. Numerous studies showed that the healing of rat molar pulp tissue after direct pulp capping is histologically comparable with humans and other animal species pulp tissue. Rat molar teeth, including pulp tissue, can be seen anatomically, histologically, biologically, and physiologically as miniature human molar teeth. Hence, the essential biological reactions of the pulp tissue and the interaction during the different stages of wound healing of rat molar teeth are comparable with that of other mammals. Rat molar teeth are a valid study model in order to provide valuable data concerning pulp tissue reaction after direct pulp capping and related questions in dentistry. Therefore, the use of rats may significantly reduce the number of currently used higher animals in research. Tests in higher developed animals should be limited to experiments which clarify inconsistent results. However, some technical difficulties, like the small size of rat molar teeth must be dealt with before undertaking any research.
International Organization for Standardization (ISO) standard 7405 determines test methods for dental material and preclinical evaluation of biocompatibility of medical devices used in dentistry. 1 The purpose of this ISO standard is the evaluation of biological effects of dental devices and dental materials, e.g. restorative filling materials, prosthetic materials for crowns and bridge work, dentures and root canal filling material including pharmacological agents used in dentistry. All these materials and devices may get in contact with the facial skin, oral mucosa, dental pulp or bone tissue as well as dental hard tissues such as enamel, dentine and cementum. New materials and devices and modified materials and devices have to be evaluated or re-evaluated to establish their biocompatibility. The aim of this evaluation process is to ensure the safe use of the tested materials for humans.
Three test groups are defined in the ISO standard 7405. Group I comprises in vitro tests of cytotoxicity such as agar diffusion tests, filter diffusion tests, direct contact or extract tests and dentine barrier tests. Group II evaluates the acute systemic toxicity by oral application, acute systemic toxicity by inhalation, subchronic systemic toxicity by oral application, skin irritation and intracutaneous reactivity, sensitization, subchronic systemic toxicity by inhalation, genotoxicity and local effect after implantation. Finally, group III comprises dental pulp and dentine usage tests, dental pulp capping tests and endodontic usage tests.
Chapters 6.3 and 6.4 of this ISO standard deal with dental pulp and dentine usage tests and dental pulp capping tests, respectively. The ISO standard states clearly that only mammals, e.g. monkeys, dogs, ferrets or miniature pigs, are suitable species for animal research in order to assess the biocompatibility of dental materials when in direct contact with dentine and dental pulp tissue. Rodents are explicitly not included in this list of suitable animals. As a consequence, primates and other highly developed animals are recommended for quite basic biocompatibility testing where only tooth filling or pulp capping materials (e.g. calcium hydroxide preparations, cements or composite resins) get in direct contact with vital dental pulp tissue.
Although rats are not listed as suitable animals in ISO 7405, about 70 studies have been published in dental research journals using rat molar teeth in order to evaluate direct pulp capping, pulpotomies and tissue reactions after pulp exposure in the past 50 years.2–71
The histologically observed healing of dental pulp tissue after direct pulp cappingwith calcium hydroxide (a gold standard in dentistry) in rats is comparable with the healing process described in humans3,7,8,10,16,21–28,30,32,41–43,45–47,49,50,53,55–59,61,62,65–67,69 or was even denoted as to be identical. 13 The first sequence of the cellular reorganization after direct pulp capping with calcium hydroxide was found to be identical in rats, primates and humans. 72
Therefore, it can be argued that rat molar teeth are a suitable research model for these kinds of studies. It needs to be investigated how results in rat molar teeth compare with results found in humans. The aim of this review of the current literature is to evaluate the suitability of rat molar teeth in the preclinical evaluation of medical devices for direct pulp capping.
Suggested test methods for dental materials
In 1974, Autian 73 presented a testing programme for dental materials including three varieties of tests, which was later refined by Langeland and Cotton 74 in 1980. This led to the publication of ISO 7405 ‘Dentistry – Preclinical evaluation of biocompatibility of medical devices used in dentistry – Test methods for dental material’ in 1997. 1
Then, in 2005 Gängler 75 proposed a three-stage model for testing dental materials in order to evaluate their biocompatibility:
Screening tests of unspecific toxicity (e.g. cytotoxicity tests in cell lines; tests to evaluate growth inhibition of microorganisms, testing of genotoxicity; mutagenicity and carcinogenicity in bacteria; systemic toxicity testing by oral ingestion or respiratory uptake in mice or rats; sensibility testing and skin irritation testing in mice or rats);
Usage tests of specific toxicity in animal experiments on rats, pigs and monkeys (e.g. implantation in connective tissue and bone; histopathological and histobacteriological dental pulp tests);
Clinical controlled studies in humans.
The use of ratmolar teeth is recommended at stage 2 in order to determine the acute and short-term toxicity of dentalmaterials prior to long-term examination in pigs and monkeys. 75
The results of animal experiments in direct pulp capping, pulp infection or inflammation and related problems undertaken in rat molar teeth are without clear justification rejected by a considerable number of dental researchers. 76 One of the most important and fundamental work about the bacterial infection of pulp tissue was conducted more than 40 years ago by Kakehashi et al. 6 in rat molar teeth. Stanley emphasized that the use of rat molar teeth is a reliable method and valid model in order to evaluate histologically the outcome of direct pulp capping. 76
Animal models for pulp capping studies
The rat dentition consists of two incisor teeth and three molar teeth divided by a distinct diastema in both, the mandible and the maxilla arch (Figure 1). The incisors are typically shaped for rodents (permanently growing, with a wide open apex) and cannot be compared with human teeth. By contrast, the rat molar teeth, including the pulp tissue, can be considered as a downsized human molar tooth with similar anatomical, histological, biological and physiological features.2,4,5,47,77,78 Their development is analogous to that of human molar teeth. Rat molar teeth exhibit the same structural characteristics of the pulp chamber, pulp tissue, root and apical delta with foramina apicalia minores (Stratmann U, personal communication, 2005) (Figure 2). Hence, the essential biological reactions of the pulp tissue and the interaction during the different stages of wound healing of rat molar teeth are comparable to that of other mammals. Thus, after direct pulp capping with calcium hydroxide, Jean et al. 45 observed no difference in the healing process between pigs, rats and dogs in an observation period from 21 days to four months. In contrast, Watts and Paterson 25 found differences in pulp healing between dogs and rats. Twenty-eight days after direct pulp capping with hard setting calcium hydroxide cement, hard tissue formation was detectable in 83% of the rat molar teeth. However, only 60% of molar teeth in dogs showed hard tissue formation after six weeks. 25 In addition, the authors 25 referred to a study by Nyborg, 79 who compared pulp tissue reactions after direct pulp capping with calcium hydroxide in dogs and humans. Nyborg's 79 study demonstrated that capping human pulp tissue showed higher success rates than capping dog pulp tissue. Furthermore, the success rate of human pulp tissue found in Nyborg's 79 study was comparable with the success rates in rat molar pulps found in Watts and Patersons' 25 study. All this may lead to the conclusion that the results of studies concerning direct pulp capping conducted in rat molar teeth are better comparable with humans than the results obtained from experiments in dogs. The differences in the results between rats and dogs might be due to the fact that ingredients of the capping agent migrate more often into the dogs’ pulp tissue than into the rats’ pulp tissue. This migration of capping material may lead to an inflammatory foreign body reaction of the pulp tissue. 28
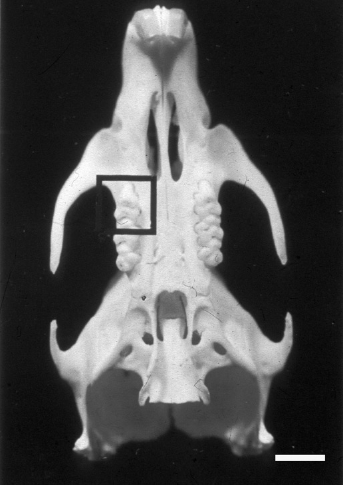
Inferior view of a rat skull and maxilla. Box indicates the first upper molar tooth (see Figure 2). Bar represents 3 mm, original magnification ×2

Light micrograph of an upper rat molar tooth. Toluidine blue staining. Bar represents 500 μm, original magnification ×40
Another commonly used animal in these kinds of experiments are monkeys. However, Brännström 80 disapproved the use of monkeys in direct pulp capping studies, because of differences in pulpal responses between human and monkey teeth. The production of reparative dentine (as a sign of tissue healing after pulp injury) is different in monkeys than it is in humans. 80 Even though monkeys are evolutionarily closer to humans than to dogs or rats, this may not be true for pulp tissue reactions.
After pulp exposure in rat molar teeth odontoblast-like cells form a typical reparative dentine – identical to humans 81 (Figure 3). If the roof of the pulp chamber has been removed in rat molar teeth and the vital pulp tissue is in contact with the oral cavity, the resulting inflammatory reaction is identical to the reaction in humans, hamsters and other animal species. 82 However, the biological response process progresses faster in rats than in humans. 83 One month in a lifetime of a rat is equivalent to 30 months in humans. 84
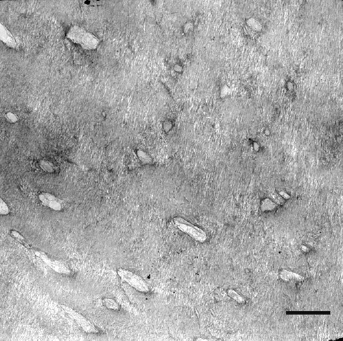
Electron micrograph of a rat molar tooth 70 days after direct pulp capping with calcium hydroxide. Reparative dentine with dentinal tubules and intact odontoblast processes is visible. Bar represents 3 μm, original magnification ×3300
Apart from the possible chemical toxicity of filling materials, the most relevant danger for dental pulp tissue after direct pulp capping is a bacterial infection and/or an infiltration with bacterial toxins. Awide variation of bacterial flora and subsequent toxin production exist between different animal species. 80 In rats the oral bacterial flora is more comparable to humans than to other commonly used species in research, such as dogs.46,85–87 The bacterial infection of vital pulp tissue will lead to its necrosis and a bacterial contamination of the root canals. Stashenko et al. 88 showed that with respect to root canal microbiology the rat model is very similar to that of primates and humans (Figure 4).
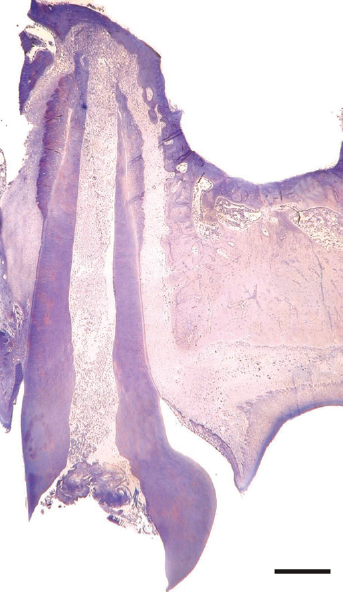
Light micrograph of an infected mesial root canal of an upper rat molar tooth 14 days after the pulp has been removed. The tooth was left open during this time. Toluidine blue staining. Bar represents 500 μm, original magnification ×36
However, rat molar pulps demonstrate an exceptional resilience and healing capacity, which must be taken into account when interpreting experimental results. 89 Due to a specific immune defence mechanism, rats are less susceptible to postoperative infection than many other animals 90 and the pulp tissue is more reactive than human pulp tissue. 91 Even in the absence of any pulp capping agent, spontaneous calcospherite formation (a layer of spheroidale calcium salts as a result of degenerative cell alterations) has been observed. This calcospherite formation leads to the deposition of an osteodentin-like matrix interspersed with demineralized pulp remnants. Generally, inflammatory processes will have subsided within one week, during which progenitor cell recruitment for reparative dentinogenesis will have occurred. Reparative dentinogenesis is initiated within two weeks, and – in the presence of calcium hydroxide as a pulp capping agent – a thick dentine bridge will form within the following two weeks. 89
Technical aspects when treating rat molar teeth
Some technical problems have to be solved before rat molar teeth can be employed as an experimental tooth for cavity preparation, direct pulp capping and subsequent filling: the small size of the teeth requires the use of suitably small instruments (e.g. only burs with a maximum size of ISO 008 should be used) and magnifying glasses or a microscope are needed. The anatomic position of the molar teeth posterior to a large diastema, which separates them from the incisor teeth, makes it difficult to access the treatment area. Soft tissues, such as tongue and cheeks, may reduce considerably visual access to the operating area as well. Therefore, proper positioning of the rat's head, with open visual access to the oral pharynx is a cornerstone in order to carry out any procedure successfully. Complete relaxation of the animal through deep anaesthesia must be attained. 2 Because of these specific requirements when undertaking research in rat molar teeth, appropriate training beforehand is an important precondition for a successful outcome. These requirements are in strong contrast to the preparation needed for animals used in dental research, such as pigs or monkeys. The size of the molar teeth in monkeys is comparable with human teeth, and therefore dentists are used to treating these kinds of teeth with ease.
Despite the small size of rat molar teeth and the associated technical difficulties, rat molar teeth have considerable advantages concerning ethical and economical considerations, as they are capable of providing valuable insight into pulp tissue reactions after direct pulp capping. 89 In vivo studies performed in rat molar teeth may reduce the number of studies in other animals, which may otherwise be necessary to verify found results. 61
Conclusion
Rat molar teeth are a valid model for pulp and dentine usage test of dental material and preclinical evaluation of biocompatibility of medical devices used in dentistry. The results of this review demonstrate that research undertaken in rat molar teeth is transferable to humans and other animal species. Overall, rat molar teeth are suitable for biological testing of local toxicity of pulp capping agents and of tissue regeneration after direct pulp capping.19,20,21,58,59
Therefore, ISO standard 7405 should be amended and the use of rat molar teeth should be explicitly approved as a useful model in direct pulp capping tests. The use of rats can significantly reduce the number of currently used higher animals, like monkeys, pigs or dogs for dental material testing and preclinical evaluation of biocompatibility of medical devices in dentistry. Tests in higher developed animals should be limited to experiments that clarify inconsistent results.
Footnotes
Acknowledgement
I am grateful to Dr Darius Sagheri, Department of Orthodontics, University of Cologne, Germany for kindly reviewing the manuscript.
