Abstract
Collagen-induced arthritis (CIA) in rats is a widely used preclinical animal model of rheumatoid arthritis (RA). However, CIA development in Sprague-Dawley (SD) rats is less severe in terms of inflammatory response compared with other strains. Therefore, a modified CIA model called MCIA, using N-acetylmuramyl dipeptide (MDP), has been developed in the less sensitive SD rat strains. This work was conducted to better understand the immunopathological role and contributions of the pro-inflammatory T-helper type 1 (Th-1) cytokines and inflammatory mediators, interleukin-1 (IL-1β), tumour necrosis factor-alpha (TNF-α) and nitric oxide (NO); the anti-inflammatory T-helper type 2 (Th-2) cytokine, IL-10 and autoantibodies such as rheumatoid factor (RF)-immunoglobulin M (IgM) in this newly developed RA model. TNF-α, NO and RF-IgM levels were significantly increased, while IL-1β levels were not affected in this MCIA rat model. The levels of IL-10 were lower than the baseline when compared with controls. In conclusion: (1) the immunological features represented in the MCIA rat model favour the Th-1 cytokine profile over Th-2 and (2) RF-IgM can be used as a diagnostic test in preclinical RA models.
The exact immunopathogenesis of rheumatoid arthritis (RA) is unknown; however, it is considered as an autoimmune disease in which autoantibodies known as rheumatoid factors (RF) such as immunoglobulin (Ig)M, IgG and IgA isotypes are produced and form immune complexes leading to complement activation. 1 The arthritis severity is usually associated with elevated levels of RF. For example, the increased serum levels of IgM-RF indicate active and ongoing inflammation, 1 while elevated levels of either IgA-RF or IgG-RF in the serum and synovial fluid (SF), respectively, reflect the presence of bone erosion. 2 Serum IgM level is an important biomarker in collagen-induced arthritis (CIA) rat model. 3 IgM-RF concentration in serum is often used in the diagnosis of RA in humans. 4 To the author's best knowledge, IgM-RF has not been evaluated as a potential diagnostic tool in preclinical RA models. There are several preclinical models of RA. However, the modified CIA model called MCIA, using N-acetylmuramyl dipeptide (MDP), used in this study is a newly developed RA model that offers advantages over other models in terms of the severity of the inflammatory response. 5 Therefore, IgM-RF was evaluated in this newly developed RA model.
The interaction between macrophages and T-helper type 1 cells (Th-1) stimulates the activation of macrophages, which in turn can infiltrate the synovium and secrete tumour necrosis factor-α (TNF-α) and interleukin (IL)-1β. 6,7 These major pro-inflammatory cytokines stimulate synovial fibroblasts and articular chondrocytes to produce matrix metalloproteinases, such as collagenase and other neutral proteases, that lead to tissue and cartilage damage accompanied with deterioration of the adjacent bone and periarticular tissues. 6 In contrast, Th-2 cytokines such as IL-4 and IL-10 favour the activation of humoral immunity, which that has been ascribed to carry a protective role for arthritis development. 8 For example, IL-10 has potent anti-inflammatory functions and is reported to reduce joint swelling, cellular infiltration, pro-inflammatory cytokine production and cartilage degradation in CIA in rats. 9 The contributions of both Th-1 and Th-2 to the rat MCIA have not been evaluated. Therefore, we investigated the roles of TNF-α, IL-1β and IL-10 in this model.
Nitric oxide (NO) is an important inflammatory mediator of RA in preclinical models.
10,11
NO activity of freshly isolated blood mononuclear cells correlated significantly with disease activity parameters such as tender and swollen joints.
12
In addition, systemic or local treatment with the NO inhibitors such as
This is the first investigative study on the immunopathogenesis and contributions of Th-1 and Th-2 cytokines in an RA model. In this study, we investigated, for the first time, TNF-α, IL-1β, IL-10, RF-IgM and NO in a newly developed rat MCIA model.
Materials and methods
Animals and experimental design
Sprague-Dawley male rats (BK1:SD), at the age between six and 10 weeks, were assigned into two experimental groups. The SD strain was originally purchased from B&K Universal Limited (Grimston, Aldbrough, UK). Rats for this work were obtained from a breeding colony maintained under controlled conditions at the biological centre of research in the Jordan University of Science and Technology. The animals were maintained under controlled environment at 24°C and an alternating 12 h light–dark cycle. Each rat was housed individually in a metallic cage containing wood shavings, fed the standard rodent chow and allowed tap water. Treatment and maintenance were in accordance with the Jordan University of Science and Technology Animal Care and Use Committee (JUST-ACUC), which follows the international animal care and use guidelines. 14 Therefore, under the proposed design, animals in group A (14 animals) were injected with MDP-collagen emulsion while animals in group B (10 animals) served as a negative control for arthritis and were injected with phosphate-buffer saline (PBS) containing 0.05 mmol/L acetic acid. Arthritic animals were monitored daily for the clinical signs manifestation. Careful consideration of the effect of arthritis severity on the animal behaviour and vital physiological signs were implemented as described by Morton and Griffith. 15 Animals were also housed individually to minimize pain and distress.
Induction of arthritis
Arthritis induction and MDP-collagen emulsion preparation were performed as previously described. 5 Briefly, each rat in group A was injected on day 0 with 0.2 mL (200 μg) of cold MDP-collagen emulsion with a Hamilton glass syringe subcutaneously at the base of the tail, 2–3 cm from the body within 2 h of preparation. To prepare MDP-collagen emulsion, the lyophilized bovine type II collagen extracted from the nasal septum cartilage (Sigma, St Louis, MO, USA) was dissolved in acetic acid at a concentration of 4 mg/mL by gentle stirring overnight at 4°C. Muramyl dipeptide (MDP) (Sigma) at 2.5 mg/mL was mixed with collagen at a ratio of 1:1 (v:v). The MDP-collagen solution was further emulsified with an equal volume of incomplete Freund's adjuvant (IFA) (Sigma) in a drop-wise fashion with continuous stirring with an electric homogenizer. A stable emulsion should remain, as a solid clump in water without dispersion. Rats were boosted subcutaneously by a second injection of 0.1 mL of the freshly prepared MDP-collagen emulsion also into the base of the tail 60 days after the first immunization.
Sample collection
Blood samples were obtained from the anaesthetized rats (ketamine 40–60 mg/kg intraperitoneally (Al Hikama Pharmaceuticals, Amman, Jordan) and xylazine 5 mg/kg (Laboratorios Calier, SA, Barcelona, Spain)) by cardiac puncture, 90 days after the first MDP-collagen injection. Serum samples were collected by centrifugation for 10 min at 3000 rpm. Serum aliquots were then frozen at −20°C until analysis. Anaesthetized animals were then euthanatized using CO2 inhalation, chamber method. 16
Spleens from each animal were removed and dispersed aseptically into a single-cell suspension, and then washed once with RPMI 1640 (Sigma). Spleen cells were cultured at 1 × 106/mL in complete medium (RPMI 1640 supplemented with 10% fetal bovine serum, 2 mmol/L
Cytokines measurement in splenocyte cell culture supernatant and serum samples
The DuoSet sandwich ELISA kits (R&D Systems Inc, Minneapolis, MN, USA) were used to measure IL-10, TNF-α and IL-1β concentrations in cell culture supernatant and serum samples. Briefly, Immulon II 96-well plates were coated with mouse anti-rat cytokine capture antibody and incubated overnight at room temperature. Wells were then blocked with 1% bovine serum albumin (BSA) in PBS for one hour at room temperature. Diluted samples (serum or cell culture supernatants) as well as recombinant cytokines standard were added to wells and incubated for 3 h at 37°C. Biotinylated goat anti-rat IL-10, TNF-α or IL-1β was added to all wells at a concentration of 100 ng/mL and then plates were incubated for 2 h at 37°C. Plates were then incubated with horseradish-peroxidase-conjugated streptavidin for 20 min in a dark place. Wells were washed three times after each incubation period to remove the non-specific binding of the reagents, and then colour was developed using horseradish-peroxidase substrate (R&D Systems Inc). Optical density (OD) values were measured at 450 nm wavelength using an automated enzyme-linked immunosorbent assay (ELISA)-plate reader (Dynatech MR5000, Guernsey, Channel Islands) to quantify colour changes. Concentrations of IL-10, TNF-α and IL-1β were estimated from standard curves generated using the serial dilution of the corresponding protein standards.
Measurement of IgM (RF)
Direct ELISA was developed to measure IgM-RF concentration in serum samples. Briefly, microtitre plates (Immulon II, Dynatech, Chantilly, VA, USA) were coated overnight at 4°C with purified rabbit IgG (AbD Serotec, Oxford, UK) diluted in carbonate/bicarbonate buffer (pH 9.6). The remaining protein-binding sites on the wells were then blocked with 2% BSA in carbonate/bicarbonate buffer for 2 h at 37°C. Diluted serum samples (1:50) were added to the wells and then incubated for 2 h at 37°C. Appropriate dilutions of the horseradish-peroxidase-conjugated mouse monoclonal anti-rat IgM (AbD Serotec) were then added and incubated for one hour at room temperature. Wells were washed three times with PBS containing 0.01% Tween-20 after each incubation period to remove the non-specific binding of the reagents. Samples and antibodies were diluted in sample buffer containing 0.1% Tween and 1% BSA, and then colour was developed using horseradish-peroxidase substrate (R&D Systems Inc). OD values were measured at 450 nm wavelengths using an automated ELISA-plate reader (Dynatech MR5000) to quantify the colour changes and wells without loading serum sample were regarded as blank.
NO measurement
The production of NO was determined by assaying culture supernatant for NO− 2, a stable reaction product of NO. Briefly, 100 μL of culture supernatant was mixed with an equal volume of Griess reagent (1% sulphanilamide and 0.1% N-[naphthyl] ethylene diamine dihydrochloride in 2.5% H3PO4) at room temperature for 10 min. Absorbance was measured at 550 nm in a microplate reader. Nitrite concentration was calculated from the standard NaNO2 curve.
Statistical analysis
Statistical analyses were performed using one-way analysis of variance. The difference was considered significant at a value of P < 0.05.
Results
The incidence and severity of arthritis in the rat MCIA model
This rat MCIA model was evaluated previously for arthritis incidence and severity. 5 Data in the current work were generated from samples obtained from animals that were extensively evaluated clinically and histopathologically for arthritis development. 5 Briefly, animals developed arthritis as early as day 13 post collagen-MDP emulsion administration with a peak incidence reaching 66% on day 30. By day 60, 57% of the rats exhibited clinical signs of arthritis including swollen joints, redness, joint deformities, lameness and paw deformities. The severity of the clinical signs were evaluated in the previous work according to the arthritis index (AI) described previously by Marty et al. 17 Accordingly, all the animals that showed clinical arthritis (57%) had severe clinical lesions at the early stages (day 30). Then the severity for some of the animals started to decrease at the time of the second MDP/collagen/IFA emulsion injection where only 71.4% of the arthritic animals had severe lesions (high AI score). After the second injection of MDP/collagen/IFA emulsion, no new animals developed arthritic lesions, but the severity of the lesions in the animals that previously showed clinical arthritis increased where 83% of the arthritic animals had severe lesions (high AI score). In addition, histopathological changes were observed in 85% of the rats with 72.7% of those animals classified as having advanced stage of arthritis where animals prominently had new bone formation, bone destruction and extensive pannus formation causing ankylosing joints. 5 Therefore, some new cases were reported in the histopathological evaluations that were not apparent clinically. 5
IL-1β, TNF-α, IL-10, RF-IgM levels in the serum and cytokine production in non-stimulated spleen cell culture supernatants
The analysis of IL-1β and TNF-α in the non-stimulated spleen cell culture supernatants showed no statistically significant differences between the groups (P > 0.05) (Figures 1a and b). In contrast, a statistically significant decrease in the level of IL-10 in the non-stimulated spleen cell culture supernatant was reported in the MCIA group compared with controls (P < 0.05) (Figure 4b).
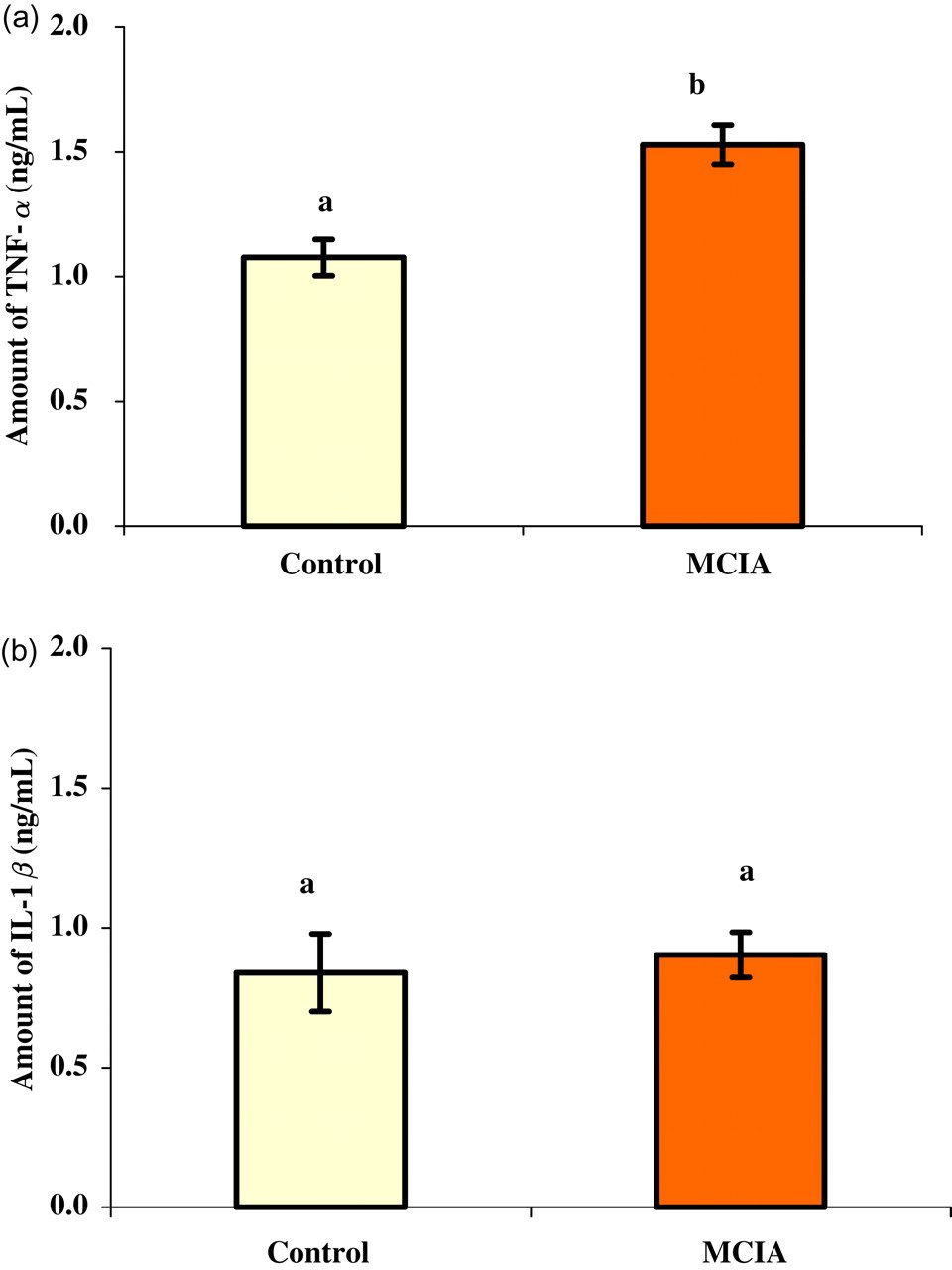
Level of TNF-α (a) and IL-1β (b) in non-stimulated spleen cell culture supernatant collected 90 days after the first collagen-MDP-IFA emulsion injection. Supernatants were collected from control rats (n = 10) and MCIA rat group (n = 14). Each value represents the mean value for each group (±SEM). Different letters represent a significant difference with a P value less than 0.05. TNF-α = tumour necrosis factor alpha; IL = interleukin; MDP = N-acetylmuramyl dipeptide; IFA = incomplete Freund's adjuvant; MCIA = modified collagen-induced arthritis model
The IL-1β level in the serum from the MCIA group was not different from the control group (P = 0.07) (Figure 2a), while serum from animals in the MCIA group had a statistically significant increase in TNF-α compared with control rats (P = 0.0005; Figure 2b). The analysis of IL-10 level in the rat serum obtained from the MCIA group was significantly lower, at least two folds, than the IL-10 level in the control group (P < 0.05; Figure 4a).
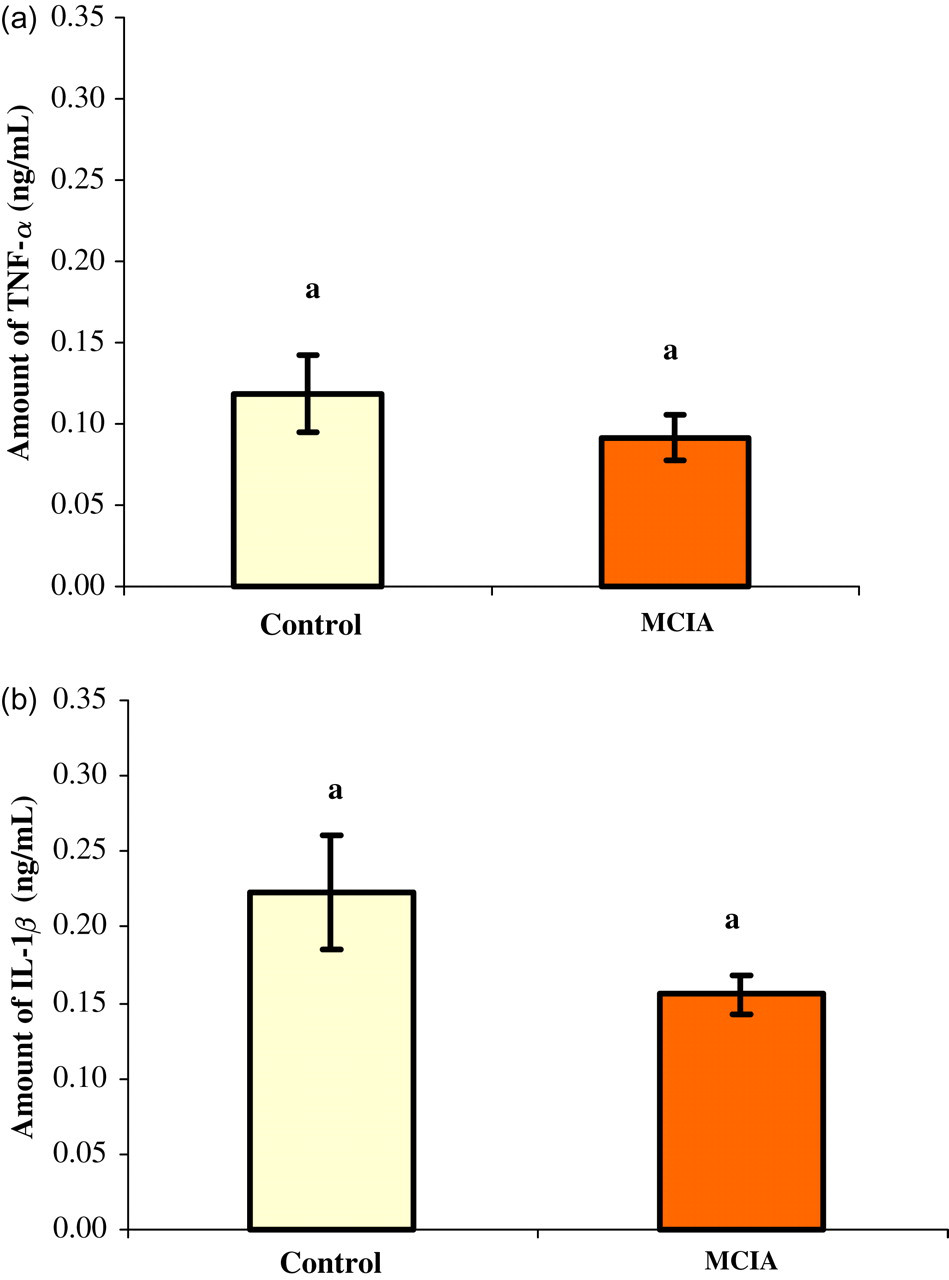
Level of TNF-α (a), IL-1β (b) in serum collected 90 days after the first collagen-MDP-IFA emulsion injection. Supernatants were collected from the control rat group (n = 10) and MCIA rat group (n = 14). Each value represents the mean value for each group (±SEM). Different letters represent a significant difference with a P value less than 0.05. See Figure 1 for abbreviations
Serum IgM levels were elevated in the rat MCIA group on day 90. The analysis of IgM-RF in the serum of the MCIA group showed a statistically significant increase compared with controls (P < 0.05; Figure 3). It is important to mention that there were no significant differences reported when comparing the cytokine values as well as the IgM-RF in the animals that had arthritic lesions with the animals that had no apparent clinical arthritis in the same MCIA group.
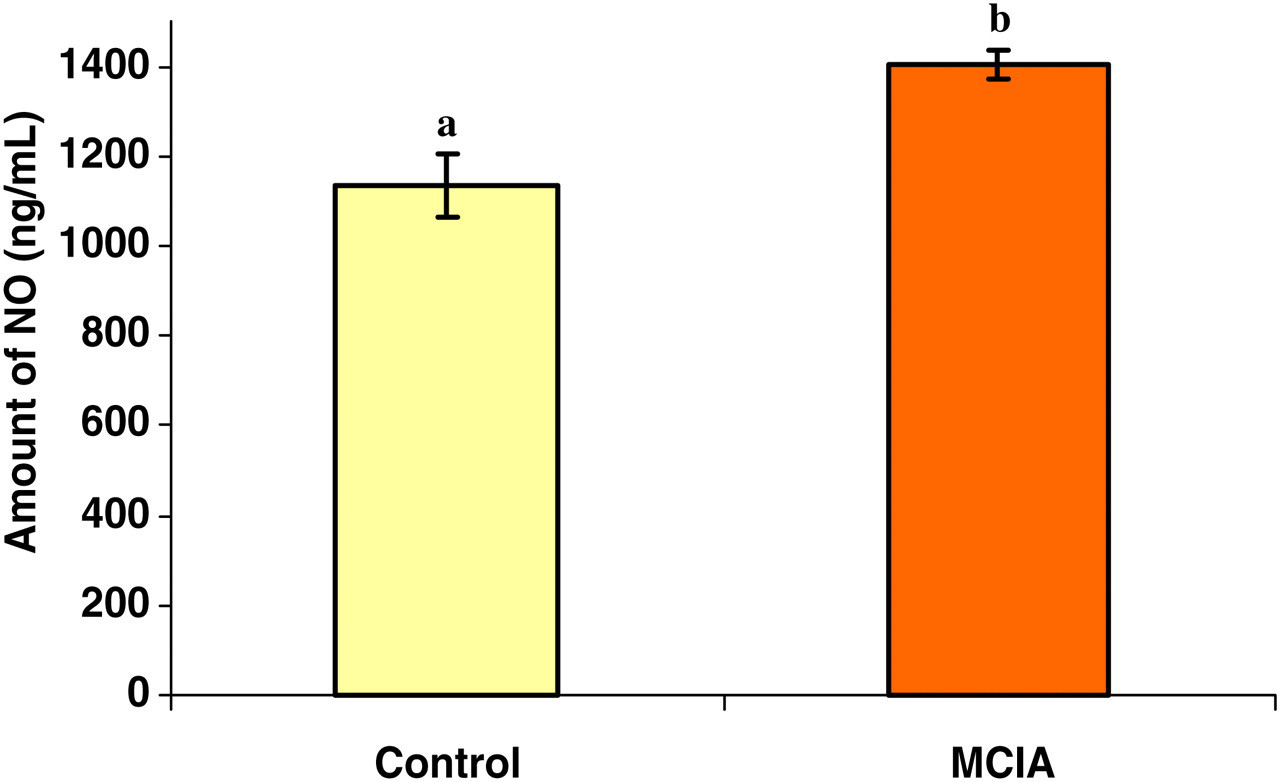
Level of nitric oxide (NO) in non-stimulated spleen cell culture supernatant collected 90 days after the first collagen-MDP-IFA emulsion injection. Samples were collected from the control rat group (n = 10) and MCIA rat group (n = 14). Each value represents the mean value for each group (±SEM). Different letters represent a significant difference with a P value less than 0.05. See Figure 1 for abbreviations
NO production in the MCIA model
NO production in splenocyte cell cultures of rats in the MCIA group increased when compared with the control group (Figure 3). There was no significant difference in the level of NO production between the animals that had arthritic lesions and the animals that had no apparent clinical arthritis in the same MCIA group.
Discussion
The immunopathogenesis of CIA has become an area of interest of many researchers who are debating the role of TNF-α and IL-1β in the inflammatory process and their destructive features for this experimental model. Both cytokines were found at high levels in the SF and tissue of RA patients. 18,19 Immunohistochemical studies have shown the presence of these cytokines in the synovial lining and sublining cells including type A synoviocytes. 20 TNF-α is mainly proposed to be responsible for the severity of the arthritis, while IL-1β is associated with increase in the incidence of the disease. 7,21–23
In the MCIA rat model, the incidence of the disease in the arthritic group remained constant after the re-challenge with the second injection of MDP-collagen IFA emulsion while the clinical assessment of the lesion severity increased. 5 The cytokines profile in the MCIA group showed a significant increase in the serum levels of the Th-1 cytokine, TNF-α. This implies that TNF-α has an important role in increasing the severity of the arthritic lesion. More interestingly, the incidence of the disease in the MCIA group remained constant after re-challenge with the second injection. Such finding implies that the elevation of TNF-α did not lead to the appearance of new cases of arthritis. These findings are in agreement with a previous study which reported that the treatment of CII-immunized DBA/1 with anti-TNF-α/β mAb before or after the onset of CIA reduced the paw swelling and the histological severity of the disease without reducing the arthritis incidence. 5 Contrary to TNF-α, the level of IL-1β in the MCIA group was similar to that in the control group, which clearly indicated that this cytokine has no effect on the severity of the disease and emphasized that IL-1β increase is often associated with an appearance of new arthritic cases. These observations are consistent with a previous reported observation that showed a significant increase in the level of TNF-α in the serum of CIA rats 33 days after immunization, while the level of IL-1β was similar to the normal rats due to the possible increase of IL-1 inhibitors in the synovium. 5,24
TNF-α contributes to the ongoing local inflammatory reactions by upregulating the expression of pro-inflammatory genes such as cyclooxygenase 2 and NO synthase, thereby increasing the production of inflammatory mediators such as prostaglandins and NO. 25 DBA/1 mice immunized with type II collagen and treated with TNFR1-IgG1 fusion protein shortly before the onset of clinical disease did not develop arthritis, suggesting that TNF-α is a crucial mediator in the late initiation phase of the arthritic process. 23
The differences in TNF-α production between the two experimental groups were only evident in the serum and not in the supernatants obtained from non-stimulated spleen cell culture. This might indicate that the source of high TNF-α produced in the serum is not the spleen cells, but instead it might be produced locally in the swollen joints and then reach the circulation. Therefore, further evaluation of these cytokines in this newly developed MCIA model needs to be conducted in future studies.
A high level of NO production, demonstrated in the non-stimulated splenocyte culture supernatant, emphasized the importance of this inflammatory mediator in RA . NO is one of the mediators involved in cartilage dysfunction in arthritis. 26 In patients with RA, nitrite and nitrate levels were elevated in serum, urine and SF. 27–29 This high NO concentration was related to disease activity parameters. 30 Its production by chondrocytes in the SF of arthritic patients was related to cytokine stimulation. 31 Interferon (IFN)-gamma causes an upregulation of NO production, but requires TNF-α as a co-signal for NO induction. 32 Therefore, NO production in macrophages is induced by inflammatory cytokines or bacterial products such as IFN-γ LPS or TNF-α. 33 Similarly, the increase in NO presented in this study is associated with an increase in the systematic TNF-α production.
The previous clinical, pathological and pro-inflammatory cytokine profiles correlated with the serum level of IgM-RF as well as with the serum and spleen cell culture supernatant level of IL-10 (Figure 4). The low serum level of IL-10 present in the experimental group compared with the control group is in agreement with the reported downregulatory effect of this cytokine at the sites of local inflammation or in the peripheral blood. 34 Constantly, studies had reported that in RA patients, the serum level of IL-10 had either been unaffected due to arthritis compared with healthy donors 35 or had a significant decrease compared with healthy controls. 34 Therefore, the pro-inflammatory and anti-inflammatory cytokine features support the well-known relationship between these two types of cytokines where anti-inflammatory cytokine, IL-10, elevation can inhibit Th-1 cell proliferation and downregulate their activity and vice versa. 36 Accordingly, the protective role that has been ascribed to Th-2 cells is their ability to express a broad range of anti-inflammatory cytokines. 37 For example, IL-10 has potent anti-inflammatory functions and is reported to reduce swelling, cellular infiltration, pro-inflammatory cytokine production and cartilage degradation in CIA in rats' joints.
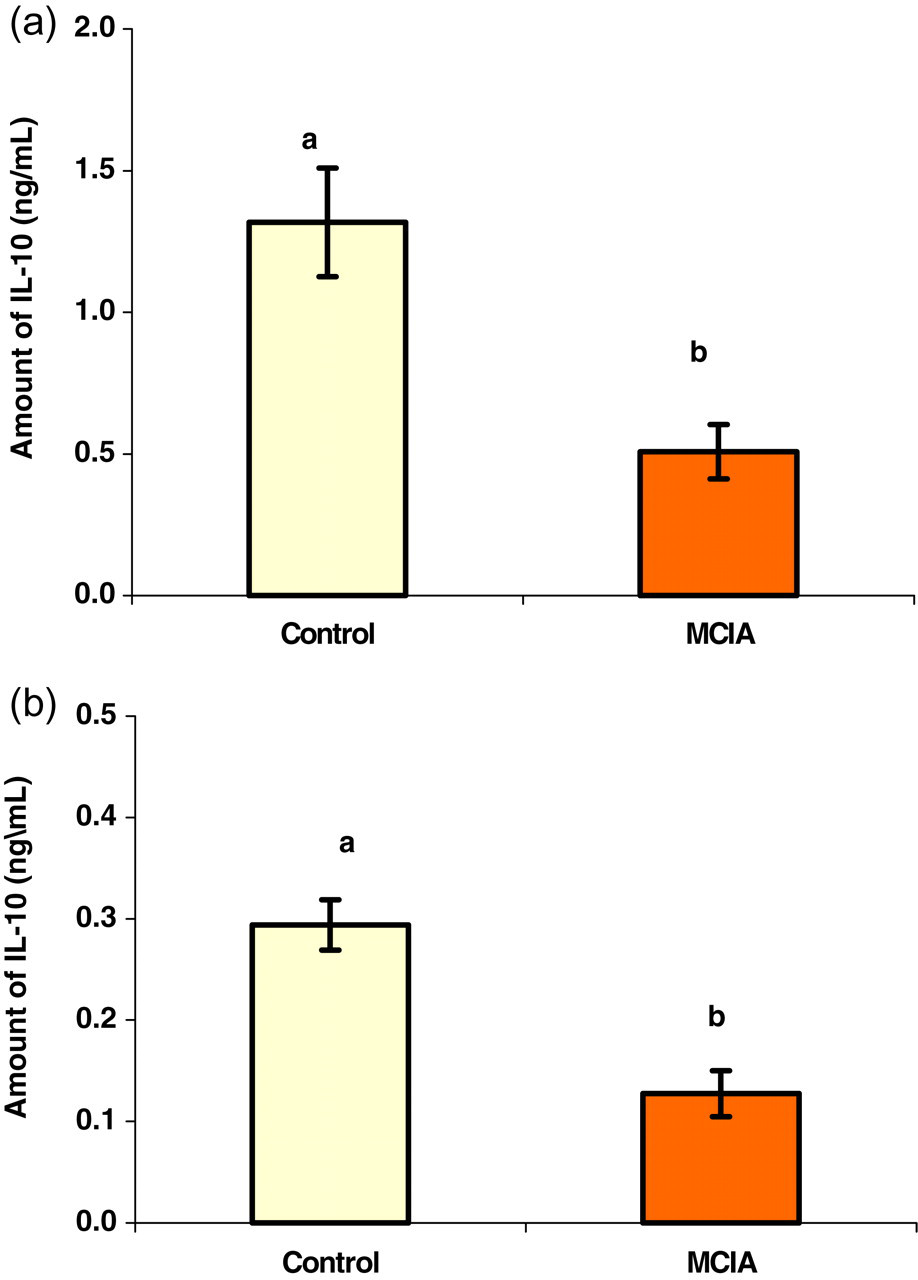
Level of IL-10 in serum (a) and in non-stimulated spleen cell culture supernatant (b) collected 90 days after the first collagen-MDP-IFA emulsion injection. Samples were collected from the control rat group (n = 10) and MCIA rat group (n = 14). Each value represents the mean value for each group (±SEM). Different letters represent a significant difference with a P value less than 0.05. See Figure 1 for abbreviations
IL-10 acts mainly as a switching factor that converts IgM either to IgG or to IgA. 38,39 Therefore, this might explain the accumulation of IgM-RF in this group (Figure 5). An increased IgM-RF level is correlated with the RA ongoing inflammation or disease activity, which is also apparent in this experimental group. Similarly, the IgM levels were dramatically increased and achieved an OD level range from 1.4 to 1 at 20 days and 45 days, respectively, in Lewis rats subjected to CIA. 3 Usually the elevated severity of arthritis is associated with elevated levels of RF. 2 In inflammatory polyarthritis, the severity of radiographic damage will be increased if higher titre of RFs is predicted. 1
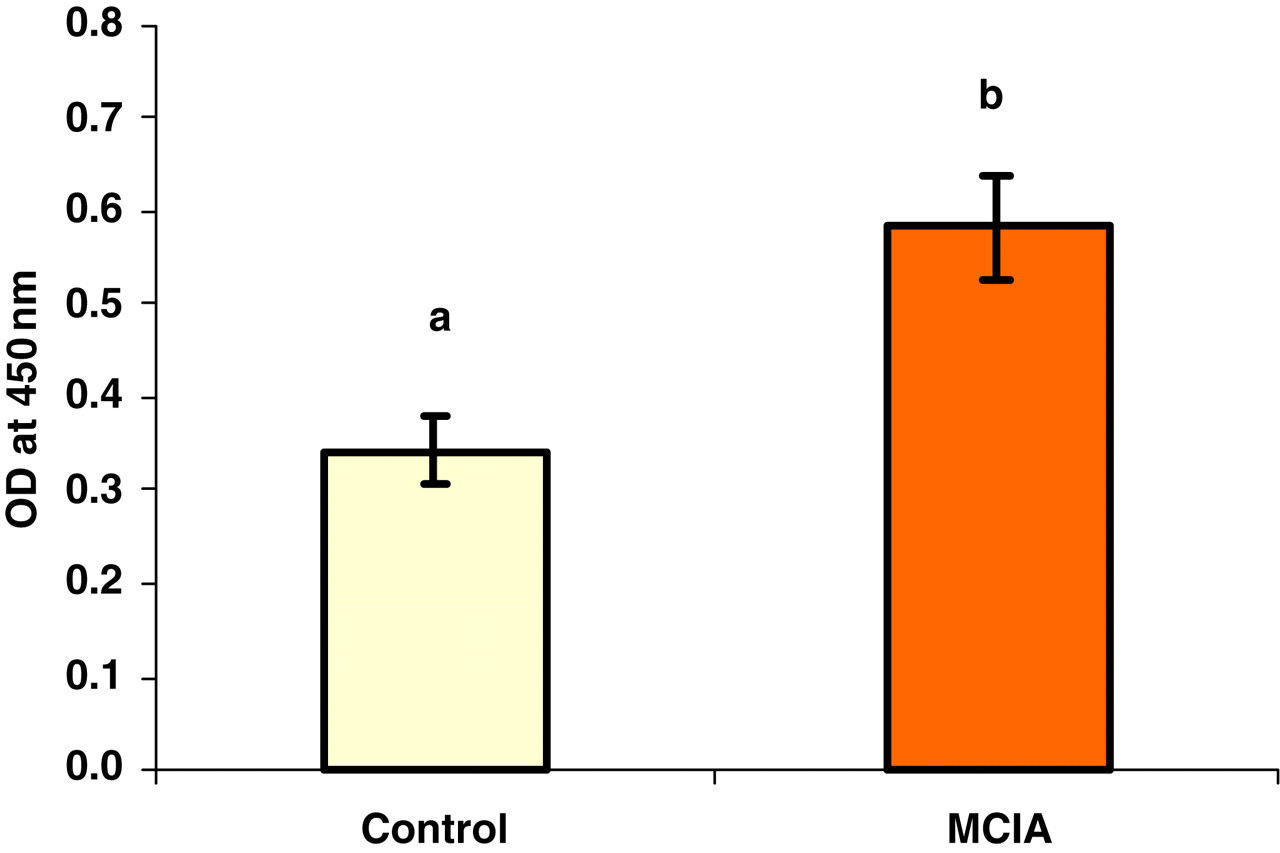
Level of IgM-RF in serum collected after 90 days of the first collagen-MDP-IFA emulsion injection and represented as optical density values (OD). Serums were collected from the control rat group (n = 10) and MCIA rat group (n = 14). Each value represents the mean value for each group (±SEM). Different letters represent a significant difference with a P value less than 0.05. RF = rheumatoid factor; Igm = immunoglobulin M; MDP = N-acetylmuramyl dipeptide; IFA = incomplete Freund's adjuvant; MCIA = modified collagen-induced arthritis model
The absence of significant differences in the parameters assessed in this study in animals that had arthritic lesion with the animals that had no apparent clinical arthritis in the MCIA group might be due to the fact that some animals that had no obvious clinical signs revealed histopathological changes that were not possible to be manifested clinically. 5 Therefore, analysing the laboratory parameters would represent the ongoing inflammation even if it is not apparent clinically or even histopathologically. In addition, this study demonstrated the potential use of serum IgM-RF as an indicator for arthritis progression and severity. In other words, the presentation of the clinical and histopathological evaluation of MCIA rat model along with the laboratory diagnostic parameters that reflect the arthritic disease progress and severity in serum samples in the current study is suitable to be employed for the evaluation of such disease models across the course of arthritis with minimal need to euthanize animals. Finally, as the previous work emphasized on describing the clinical and pathological changes of the MCIA model, 5 the current work closely investigated some of the key mediators involved in arthritis development in this model that have been usually targeted in RA therapy and diagnosis.
Footnotes
Acknowledgements
This work was supported by a grant from the Deanship of Research, Jordan University of Science and Technology. The authors acknowledge the technical help of Mr Mohammad Damreh at the Biological Center of Research (animal house).
