Abstract
Cryopreservation of mouse spermatozoa has been widely used; however, fertility of frozen spermatozoa in some strains, especially when inseminating cryopreserved oocytes, is low and may be improved by assisted fertilization techniques. The present study was performed to investigate the effect of partial zona pellucida (ZP) digestion on the in vitro fertilization (IVF) capacity of frozen mouse spermatozoa. Mouse oocytes were subjected to partial ZP digestion using acidic Tyrode's solution (pH 3.1). Fertilization rates in digestion groups (30 or 45 s) were higher (P < 0.05) than that of zona-intact control (78.3% or 86.3% vs. 52.5%). The recovery rate at 45 s was lower (P < 0.05) than that at 30 s (84.2% vs. 97.3%). Among vitrified oocytes, the fertilization rate in treatment group (digested for 30 s) was higher (P < 0.05) than that of zona-intact group (50.8% vs. 22.1%). After embryo transfer at the two-cell stage, 17.7% and 11.8% of transferred embryos derived from fresh and vitrified digested oocytes developed to term and showed no significant difference as compared with that from zona-intact oocytes (24.1%, P > 0.05). These results indicate that partial ZP digestion improves IVF efficiency of fresh and vitrified oocytes with frozen mouse spermatozoa, which can provide valuable information for in vitro assisted fertilization using cryopreserved gametes in the re-establishment of mouse colonies.
The mouse is one of the most widely used animal models in biological and medical research. With the development of gene manipulative techniques, increasing numbers of mutant mouse strains have been produced in laboratories and maintaining those strains would exhaust limited fund and housing space. This problem may be solved by the application of gamete and embryo cryopreservation techniques. 1 Cryopreserved mouse sperm can be used in in vitro fertilization (IVF) to generate large numbers of embryos in order to re-establish breeding colonies. 2 First experiments of successful cryopreservation of mouse spermatozoa were reported by Tada et al., 3 Okuyama et al. 4 and Yokoyama et al. 5 in 1990. Subsequent studies were performed to optimize the cryoprotectant formula 6–10 and freezing process 11–13 on various mouse strains. 14,15 However, results were not consistent from one strain to another. 2,16 In some mouse strains, such as BALB/c 17 and C57BL/6, 18 fertilization abilities of frozen spermatozoa were low and needed to be improved.
Assisted fertilization techniques reported in recent years include intracytoplasmic sperm injection (ICSI), 19 partial zona dissection (PZD), 18 partial zona incision by piezo-micromanipulator (ZIP) 20 and laser-assisted zona-drilling (LAZD). 21 ICSI, ZIP and LAZD procedures require special equipment and limited oocytes can be treated at one time in ICSI. The PZD methods are simple and easy to handle, but fertilized embryos with PZD must be cultured in vitro to morula or blastocyst stage to avoid blastomere loss from the relatively large zona slit, which can occur during the two-cell embryo transfer process. 20 Because large-scale oocytes should be fertilized in vitro during re-establishment of a mouse strain using cryopreserved spermatozoa, it is necessary to develop a simple and effective method for assisted fertilization.
Generally, partial zona pellucida (ZP) digestion has been used in assisted hatching of human IVF embryos to overcome zona hardening induced by culture in an artificial environment or cryopreservation of embryos. 22,23 The zona hardening also decreased fertilization capacity of cryopreserved mouse oocytes; 24 therefore, the fertilization rate was low when inseminated with frozen spermatozoa. 25,26 Mordel et al. 27 digested ZP of mouse oocytes partially using pronase and demonstrated that at low sperm concentration (500 cells/mL), the longer digestion time (5 min vs. 3 min) the better the fertilization. Compared with ZIP, LAZD and PZD, partial ZP digestion maintains the integrity of ZP, which acts as a major block against polyspermy 28 and bacterial and fungal infection 29 during IVF and embryo transfer. In the present study, acidic Tyrode's solution was used to partially digest the ZP in order to exactly control digestion time. The objective of this work was to investigate the effects of partial zone digestion on IVF using fresh and vitrified oocytes with frozen spermatozoa.
Materials and methods
Animals
All experimental protocols concerning the handling of mice were in accordance with requirements of the Institutional Animal Care and Use Committee at China Agricultural University. Kunming (KM) mice, an outbred strain originating from Swiss mice, were purchased from the Academy of Military Medical Sciences (Beijing, China) and raised in a temperature- and light-controlled room (20–25°C, 14 L:10 D). Sexually mature male mice at 10–15 weeks of age were sacrificed for epididymal spermatozoa collection. Female mice at 5–10 weeks of age were used for oocyte donors and embryo recipients.
Reagents and media
All chemicals and media were obtained from Sigma-Aldrich Chemical (St Louis, MO, USA) unless otherwise stated. Eighteen percent (w/v) raffinose and 30% (v/v) egg yolk were dissolved in distilled water at 60°C and centrifuged at 10,000
Freezing and thawing of spermatozoa
Procedures for sperm cryopreservation were performed as described by Nakagata and Takeshima 11 with some modifications. Two epididymides were removed from one male mouse and transferred into 200 μL of the cryopreservation solution in a 35 mm dish (Nunc, Roskilde, Denmark). The epididymides were cut with the edge of an injection needle and spermatozoa were allowed to disperse at room temperature (25 ± 1°C) by gently shaking the dish for 2 min. Approximately 10 μL of sperm suspension at a concentration of 1–2 × 108 cells/mL was drawn into a 0.25 mL plastic straw (IMV, L'Aigle, France), and the straw was heat-sealed. The straws were cooled at 4°C for 30 min in a refrigerator for equilibrium, then transferred into liquid nitrogen (LN2) vapour (2 cm above LN2 level) for 10 min before being plunged into LN2. Spermatozoa were frozen for at least one week before use. For thawing, the straw was removed from LN2 and directly immersed in a water bath at 37°C, and 1 μL of thawed sperm suspension was added into 100 μL of the human tubal fluid (HTF) medium. 31 The thawed spermatozoa were pre-incubated for 1–1.5 h in an incubator (37°C, 5% CO2) before IVF.
Oocyte collection
Female mice were superovulated by intraperitoneal injection of 10 units of pregnant mare's serum gonadotropin (PMSG, Ningbo Hormone Products, Jiangsu, China) and 10 units of human chorionic gonadotropin (hCG, Ningbo Hormone Products) given 48 h apart. Oocytes were collected from oviducts 14–15 h post-hCG injection. Cumulus cells were removed by the treatment with 300 units/mL hyaluronidase in M2 and oocytes were washed and kept in the HTF medium in an incubator before use.
Vitrifying and warming of oocytes
Vitrification procedures were previously described by Zhou et al. 30 The 0.25 mL plastic straws (IMV) were used for making open pulled straws (OPS). 32 The inner diameter at the narrow tip of pulled straws was 0.10–0.15 mm with a wall thickness of 0.05 mm. Manipulations of freezing and warming were conducted on a 37°C hot-plate (Wenesco, Chicago, USA) at ambient temperature of 25 ± 1°C. Oocytes were pretreated in 10% EG + 10% DMSO for 30 s, then transferred to the EDFS30 solution. Approximately, 10 oocytes were picked up by the narrow end of OPS within 25 s and the OPS was immediately plunged into LN2 and stored for at least one week. For thawing, the OPS was taken out from LN2, of which the narrow tip was immersed in prewarmed 1 mL of 0.5 mol/L sucrose. Oocytes were expelled from the OPS with a mouth pipette and immediately transferred into another drop of the same solution for 5 min to dilute cryoprotectants. Recovered oocytes were washed three times in PBS (supplemented with 3 mg/mL BSA) and evaluated under a stereomicroscope. Morphologically normal oocytes were cultured in HTF medium in an incubator for at least 1 h before IVF or zona digestion.
Zona pellucida digestion
Acidic Tyrode's solution 33 was prepared at 25 ± 1°C and adjusted to pH 2.5, 2.7, 2.9, 3.1, 3.3 and 3.5 with Analar HCl using a pH meter (Model 3320, Jenway, Dunmow, UK). Denuded oocytes were transferred individually into 20 μL droplets of the acidic Tyrode's solution under mineral oil at 25 ± 1°C and observed continuously using an inverted microscope. The time required for complete dissolution of ZP was visually recorded. When ZP was partially digested, 20–30 oocytes were transferred into 200 μL drop of the acidic Tyrode's solution covered with mineral oil. The digestion time was strictly controlled to avoid complete removal of ZP. After partial ZP digestion, the oocytes were taken out and washed immediately with 1–2 mL of M2 medium to dilute the digestion solution. Morphological images of zona-intact and partial ZP digested oocytes were photographed with a video camera (Leica DFC 300 FX, Wetzlar, Germany) mounted on an inverted microscope (Olympas IX70, Tokyo, Japan). The acquired images were processed with the LSM 5 Image Browser software (Version 3.2, Carl Zeiss, Jena, Germany) to measure the thickness of ZP. This measure was repeated four times on different places of ZP to limit a potential effect of thickness variation, and a mean was calculated for each oocyte.
In vitro fertilization, embryo culture and embryo transfer
The zona-intact or partial ZP digested oocytes were introduced into drops of HTF medium for IVF. During IVF, the final spermatozoa concentration was approximately 1–2 × 106 cells/mL. At 5 h after insemination, embryos were removed from fertilization drops, washed in HTF medium and cultured in 50 μL drops of HTF medium. At 24 h post-insemination, eggs were examined and those cleaved into two equal blastomeres were considered fertilized. The number of two-cell embryos was scored. Embryos were either surgically transferred into oviducts of pseudo-pregnant mice for the evaluation of in vivo development or cultured to the blastocyst stage for four days to assess in vitro development.
Statistical analysis
The thickness of the ZP and the time required for complete ZP removal were given as mean ± standard deviation (SD). Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) program. The thickness of ZP between zona-intact and digested oocyte groups and digestion time between fresh and vitrified were compared by t-test. The experiments of IVF were repeated at least three times, and the data from IVF and embryo transfer were analysed by χ 2 test. Values of P < 0.05 were considered statistical significance.
Results
Time for complete zona removal by acidic Tyrode's solutions at different pH values
When fresh-collected oocytes were transferred to acidic Tyrode's solution, ZP of oocytes began to dissolve from outside promptly until they disappear entirely. Time required for complete ZP removal became longer along with the increase in pH values of acidic Tyrode's solutions in the range of 2.5–3.5. In pH 2.5, 2.7, 2.9, 3.1, 3.3 and 3.5 acidic Tyrode's solutions, time required for complete ZP removal was 11.3 ± 0.8, 16.5 ± 1.4, 23.7 ± 2.6, 58.7 ± 3.9, 103.9 ± 7.6 and 214.5 ± 14.3 s (mean ± SD, Table 1), respectively.
Time required for complete removal of zona pellucida of mouse oocytes in acidic Tyrode's solutions with different pH values at 25°C
In vitro fertilization of partial zona digested fresh oocytes with cryopreserved spermatozoa
The pH 3.1 Tyrode's solution was used for the screening of appropriate partial digestion time. As shown in Table 2, fertilization rates of partial zona digested oocytes inseminated with frozen-thawed spermatozoa increased along with prolonged digestion time (15–45 s). When digestion time was at 30 or 45 s, fertilization rates in treatment groups were significantly higher (P < 0.05) than that of zona-intact control (78.3% or 86.3% vs. 52.5%), so were the blastocyst formation rates (56.6% or 61.5% vs. 39.4%). However, a low percentage of oocytes lost their ZP during digestion and washing handlings when digestion time was increased. The recovery rate of digested oocytes at 45 s was significantly lower (P < 0.05) than at 30 s (84.2% vs. 97.3%). Figure 1 shows thinned ZP of mouse oocytes digested for 30 s in pH 3.1 Tyrode's solution. The ZP thickness in treatment group (digested for 30 s) was significantly lower (P < 0.05) than that of zona-intact control (4.7 ± 0.4 μm vs. 7.4 ± 0.8 μm, n = 20).
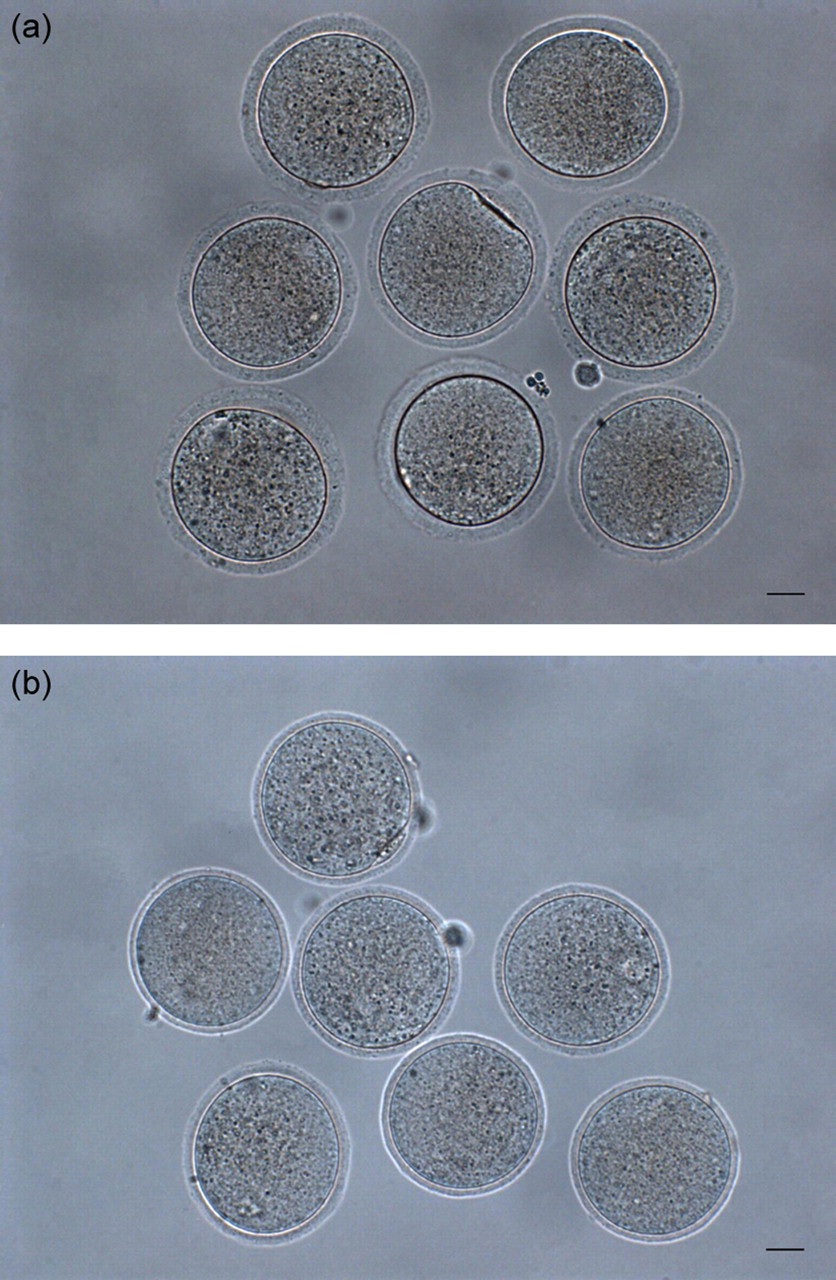
Partial zona pellucida (ZP) digestion of mouse oocytes. (a) Fresh-collected oocytes with intact ZP (zona thickness: 7.4 ± 0.8 μm, n = 20); (b) Partial zona digested oocytes with thinned ZP digested in pH 3.1 Tyrode's solution for 30 s at 25°C (zona thickness: 4.7 ± 0.4 μm, n = 20); scale bar = 20 μm
In vitro fertilization and subsequent development in vitro of partial zona digested fresh mouse oocytes fertilized with frozen-thawed spermatozoa
*The fertilization rate is expressed as number of two-cell embryos/number of oocytes recovered
†The blastocyst rate is expressed as number of blastocysts/number of oocytes recovered
‡§**Values with different superscripts in the same column differ significantly (P < 0.05)
In vitro fertilization of partial zona digested vitrified oocytes with cryopreserved spermatozoa
When vitrified-warmed oocytes were digested in pH 3.1 Tyrode's solution, the time required for complete ZP removal was 60.1 ± 4.2 s. No significant difference was found in digestion time between vitrified and fresh oocytes (P > 0.05, Figure 2). After partial zona digestion for 30 s, 96.0% of vitrified oocytes kept normal morphology (Table 3). The fertilization rate and blastocyst formation rate of vitrified zona-intact oocytes with frozen-thawed spermatozoa were significantly lower (P < 0.05) than those of fresh oocytes (22.1% vs. 52.5% and 14.4% vs. 39.4%). After partial zona digestion, the above data of vitrified oocytes increased to 50.8% and 30.8%, which showed no significant difference as compared with those of fresh oocytes (P > 0.05).
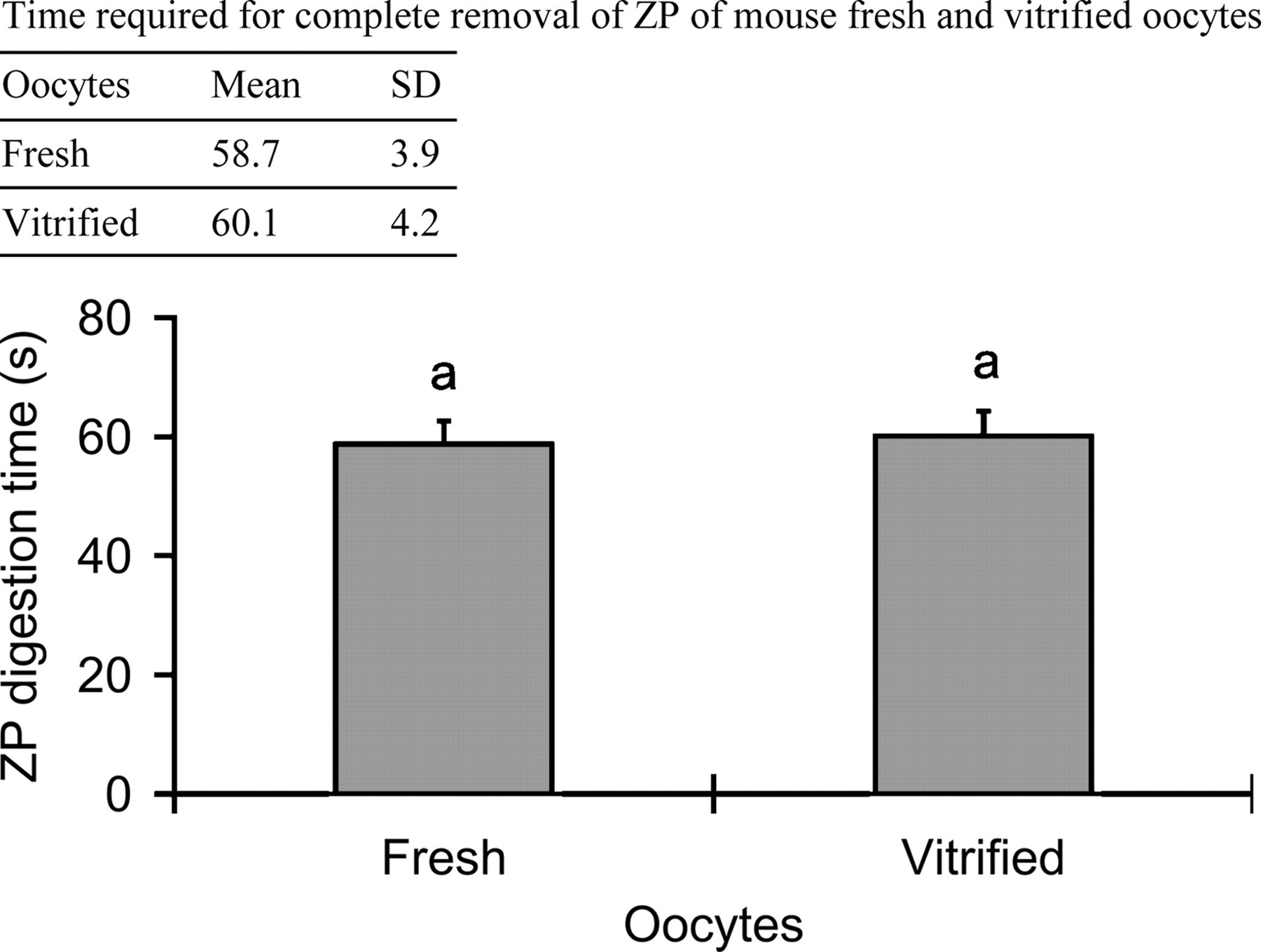
Time required for the complete removal of zona pellucida (ZP) of fresh and vitrified mouse oocytes (n = 20, respectively) in pH 3.1 Tyrode's solution at 25°C. aValues with same superscripts showed no significant difference (P > 0.05)
In vitro fertilization and subsequent development in vitro of partial zona digested vitrified mouse oocytes fertilized with frozen-thawed spermatozoa
*The fertilization rate is expressed as number of two-cell embryos/number of oocytes recovered after digestion
†The blastocyst rate is expressed as number of blastocysts/number of oocytes recovered after digestion
‡Data were the same as that in Table 2
§**Values with different superscripts in the same column differ significantly (P < 0.05)
In vivo development of two-cell embryos derived from partial zona digested oocytes
As shown in Table 4, when frozen spermatozoa were used, pregnancy rate of transferred embryos derived from fresh oocytes with or without digestion was 40.0% (4/10), and the pregnancy rate of vitrified oocytes was 27.3% (3/11). Furthermore, live birth rates of fresh and vitrified digested oocytes were 17.7% and 11.8%, respectively, which were similar to those of fresh zona-intact oocytes (24.1%, P > 0.05).
The effect of partial zona pellucida digestion by the acidic Tyrode's solution on transfer of two-cell stage embryos derived from mouse oocytes fertilized with frozen-thawed spermatozoa
*Number of recipients delivered/number of recipients transferred
†Embryos transferred to recipients that were pregnant/embryos transferred to all recipients
‡Percentages of offspring were calculated based on the number of embryos transferred to recipients that were pregnant
§Values with same superscripts had no significant difference (P > 0.05)
Discussion
An important problem associated with cryopreservation of mouse spermatozoa is that widely used cryopreservation protocols may not generate similar fertilization rates in some mouse strains, as spermatozoa collected from these strains are sensitive to cryopreservation. 2,16 In this case, assisted fertilization techniques applied on oocytes are needed to compensate the outcome of reduction in fertilization capacity because of sperm injury. Naito et al. 34 demonstrated that zona-free oocyte could be fertilized normally and develop to term. Furthermore, mouse spermatozoa that could not penetrate the ZP retained the fertilizing ability if the sperm was attached to the surface of the ooplasm. 35 These results suggested that overcoming the obstacle from ZP could increase fertilization rate of sperm with restricted ability to penetrate the ZP. In ICSI, the whole sperm or sperm head is injected into the ooplasm in order to directly bypass ZP and oolemma. 19 The purpose of zona dissection or zona drilling is to let fewer motile sperm penetrate ZP from the incision and fertilize the oocyte. 36 In the present study, the method of partial ZP digestion was utilized to thin or soften ZP in favour of sperm penetration.
Acidic Tyrode's solution is usually used for the removal of ZP of embryos with little negative effects on subsequent development. 37 In the present study, it was used for digesting ZP of mouse oocytes. Our results showed that time required for complete zona removal of mouse oocytes prolonged with an increase in pH values of Tyrode's solutions in the range of 2.5–3.5 (Table 1). Therefore, digestion time can be controlled precisely by adjusting the pH values of Tyrode's solutions. In the commonly used pH 2.5 Tyrode's solution, 33 the time for complete zona removal of KM mouse oocytes was 11.3 ± 0.8 s. It would be difficult to finish operations including partial digestion, oocyte transfer and washing during this time period. The pH 3.1 Tyrode's solution, with the time of 58.7 ± 3.9 s, was chosen to digest mouse ZP because during this time period, the screening of partial-digestion time could be performed.
Our results demonstrated that partial ZP digested mouse oocytes by acidic Tyrode's solution can fertilize with frozen-thawed spermatozoa. In the mouse, the ZP is composed of three sulfated glycoproteins (ZP1, ZP2 and ZP3), and the ZP3 is mainly responsible for mediating sperm bonding to ZP and inducting acrosome reaction. 38 The model of mouse ZP proposed by Wassarman 39 suggested that ZP was filamentous in its basic structure and filaments were consisted of repeated ZP2/ZP3 heterodimer units cross-linked by ZP1. Moreover, El-Mestrah et al. 40 proved that ZP1, ZP2 and ZP3 distributed throughout the entire thickness of ZP. So, it is reasonable to presume that even if the outer layer of ZP is dissolved by acidic Tyrode's solution, glycoproteins in remnants of ZP can normally interact with sperm. In addition, fertilization rates of partial zona digested oocytes increased with an increase of digestion time (Table 2). This finding agrees with that reported by Mordel et al. 27 who showed that when partially digested mouse oocytes were inseminated with low-concentration fresh sperm (500 cells/mL), the longer pronase treatment (5 min vs. 3 min) was more efficient in enhancing fertilization. Though the highest fertilization rate could be obtained when oocytes were digested for 45 s, the recovery rate of digested oocytes at 45 s was lower than that at 30 s (Table 2). Due to individual variation in ZP thickness, digestion at 45 s would lead to the removal of thin ZP during digestion or washing manipulations. Thus, it was suggested that optimal digestion time for mouse oocytes in pH 3.1 Tyrode's solution was limited in 30 s.
As the time for complete ZP removal in vitrified mouse oocytes was similar to that in fresh oocytes (Figure 2), results of digestion time screening for fresh oocytes were also fit for the vitrified. The fertilization rate of vitrified oocytes with frozen-thawed spermatozoa was lower than that of fresh oocytes (Table 3), indicating that OPS vitrification resulted in decreased fertilizing ability in mouse oocytes. Oocyte vitrification procedures could induce premature release of cortical granule contents, which modified the ZP through a process of ZP hardening to disturb the entry of sperm and fertilization. 41,42 When partial ZP digestion was used, there was a significant increase in fertilization rates of vitrified oocytes, indicating that partial ZP digestion may be an alternative method to overcome the effect of zona hardening related to oocyte cryopreservation.
The ZP plays an essential role in tubal transport of mouse embryos by preventing blastomere loss before the beginning of compaction. 20 In the PZD technique, blastomeres easily escaped from the relatively large slit of ZP when embryos were transferred at the two-cell stage, which led to a low success rate of development to term. 20 However, the birth rate of two-cell embryos with partial zona digestion after transfer was comparable with that for zona-intact embryos (Table 4), implying that the remnants of ZP after digestion can provide sufficient protection against blastomere loss.
In conclusion, our experiments demonstrated that partial ZP digestion can improve fertilization efficiency of fresh and vitrified oocytes inseminated with cryopreserved mouse spermatozoa, while no negative effect on developmental potential of digested oocytes is observed. The best results can be achieved by digesting for 30 s in pH 3.1 Tyrode's solution. As a simple time-saving method, partial ZP digestion can be used for routine IVF to generate mice using cryopreserved gametes. The results of this study can also provide some valuable information for in vitro assisted fertilization of other animals and humans.
Footnotes
ACKNOWLEDGEMENT
This work was supported in part by the Research Fund for the Doctoral Program of Higher Education (No. 20060019031) from China Ministry of Education.
