Abstract
Recently, a vast number of genetically-engineered mice have been created in various laboratories worldwide, all of which need to be effectively archived. The cryopreservation of mouse sperm provides a simple and economical means of storing the mice in mouse resource facilities. The current protocol for sperm cryopreservation using 18% raffinose pentahydrate and 3% skim milk (R18S3) has been adopted in most laboratories. In general, we can attain relatively high fertilization rates for frozen/thawed sperm in many inbred and F1 hybrid strains. However, the sperm of C57BL/6J mice shows an extremely low fertility rate after freezing and thawing (0–20%). In this study, we attempted to improve the low fertility of frozen/thawed C57BL/6J mouse sperm. Our results showed that a combination of R18S3 containing
Sperm cryopreservation is a useful tool for effectively archiving vast numbers of genetically-engineered mice, such as transgenes, targeted mutations and chemically-induced mutations, in a mouse repository and resource centre. 1–4 Now, the need for sperm cryopreservation is growing in recognition, so a great number of mice have been produced worldwide and some knockout mouse projects continue in Europe and America. 5
Most laboratories have adopted the protocol for sperm cryopreservation using cryoprotective agents (CPA) composed of 18% raffinose pentahydrate and 3% skim milk (R18S3). 6 In general, frozen/thawed sperm in many inbred and F1 hybrid strains show relatively high fertilization rates in in vitro fertilization (IVF). 6–8 However, the sperm from C57BL/6J mice, which is the main strain used for the production of transgenic mice, show a low level of fertility after freezing and thawing (0–20%). 6–10
Previously, we have demonstrated that methyl-β-cyclodextrin (MBCD) dramatically improved the fertility of frozen/thawed sperm in C57BL/6J mice by stimulating the cholesterol efflux from the plasma membrane. 11 MBCD is a methylated cyclic heptasaccharide consisting of α-(1-4)-glucopyranose units with a hydrophilic outer and lipophilic cavity at its centre, which can form inclusion complexes with many lipophilic agents by taking up a molecule. 12 It is well known that MBCD can remove cholesterol from the sperm membrane and induce sperm capacitation in mice. 11,13,14
On the other hand, we have found that cryopreserved C57BL/6J mouse sperm frequently suffer from cryo-induced cellular injuries during freezing and thawing. 10 Improving the conditions of sperm cryopreservation may further help to obtain a high and stable rate of fertilization from cryopreserved C57BL/6J mouse sperm.
There are some reports of
In this study, we tried to examine the combination effect of R18S3 containing
Material and method
Animals
C57BL/6J mice were purchased from CLEA Japan Inc (Tokyo, Japan) and used as sperm and oocyte donors. Female and male donors were an 8- to 10- and a 12- to 15-week-old, respectively. Mice used as recipients for the transfer of two-cell embryos were of the Jcl/ICR strain and were 8 to 16 weeks old. All animals were kept under a 12 h/12 h dark/light cycle (lights on: 07:00 to 19:00 h) at a constant temperature of 22 ± 1°C with free access to food and water. All animal experiments were carried out with the approval of the Animal Care and Use Committee of the Kumamoto University School of Medicine.
Media
Sperm cryopreservation solutions were used containing 18% raffinose pentahydrate and 3% skim milk (R18S3, 420 mOsm) or R18S3 with 100 mmol/L
A modified Krebs-Ringer bicarbonate solution (TYH) 22 with 4.0 mg/mL bovine serum albumin (BSA) or 0.75 mmol/L MBCD (Sigma, St Louis, MO, USA) with 1.0 mg/mL polyvinylalcohol (cold water soluble; Sigma) was used as a medium for sperm preincubation. 11 Human tubal fluid (HTF) 23 and modified Whitten's medium (mWM) 24 were used for IVF and the culture of two-cell embryos to the blastocyst stage.
Sperm freezing and thawing
The cryopreservation of sperm was performed as per our published method with a little modification.
6
After the male mice were sacrificed by cervical dislocation, two-tailed caudal epididymides were taken from one male mice (Figure 1a). Aliquots of 60 μL of the R18S3 with or without
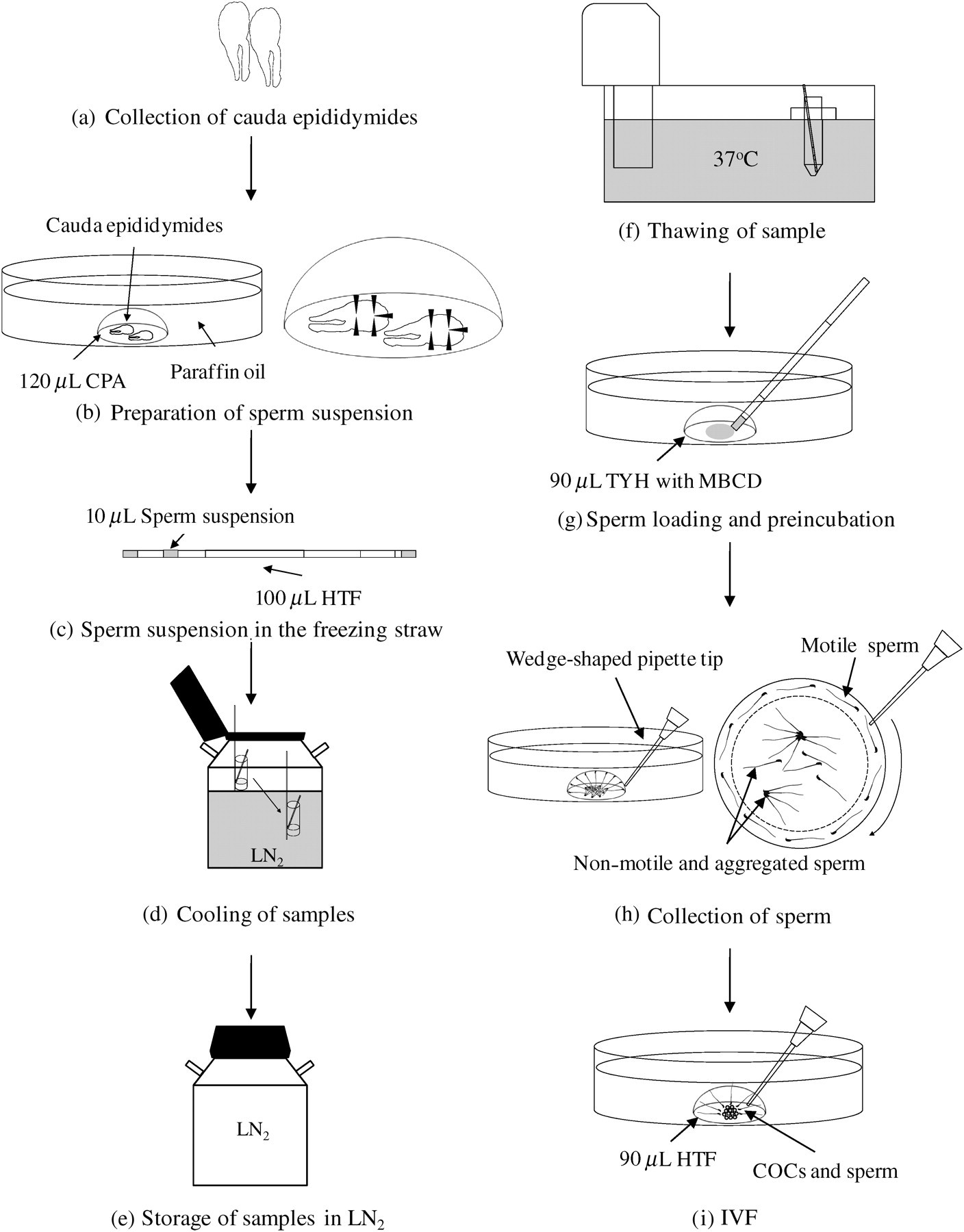
Schematic descriptions of mouse sperm cryopreservation and in vitro fertilization (IVF) protocol. Sperm cryopreservation was performed by following procedures (a–e). (a) One pair of cauda epididymides was collected from a male mouse. (b) The cauda epididymides were transferred into a 120 μL drop of cryoprotective agent (CPA) and cut into five portions as indicated by arrowheads. (c) Prepared sperm suspension was divided into 10 aliquots and packed into to a 0.25 mL plastic straw. (d) Plastic straws containing 10 μL sperm suspension were cooled in the liquid nitrogen (LN2) vapour for 10 min and were then plunged directly into LN2. (e) These samples were then stored in the LN2 before use. Thereafter, IVF using frozen/thawed sperm was performed following steps (f–i). (f) The stored samples were retrieved from LN2 and immediately soaked and warmed in the water bath at 37°C for 10 min (g) Aliquots of 10 μL sperm suspension were loaded and preincubated in a 90 μL drop of TYH with methyl-β-cyclodextrin (MBCD) for 30 min at 37°C. (h) An aliquot of 10 μL sperm suspension was collected from the peripheral part of the drop containing motile sperm using a wedge-shaped pipette tip in the direction shown by the arrow. (i) Sperm suspension was added to 90 μL human tubal fluid (HTF) containing cumulus-oocytes-complexes (COCs) and coincubated for 5–6 h. Twenty-four hours after insemination, two-cell embryos were obtained at 37°C with 5% CO2 in the air
In vitro fertilization
The procedures used for preincubation and IVF using fresh or frozen/thawed sperm were essentially the same as those described previously. 11,13 Mature female mice were superovulated by an intraperitoneal injection of 7.5 IU of equine chorionic gonadotropin (eCG) (ASKA Pharmaceutical Co Ltd, Tokyo, Japan) followed by 7.5 IU of human chorionic gonadotropin (hCG) (ASKA Pharmaceutical Co Ltd) 48 h later. At 14–15 h after the injection, the mice were sacrificed by cervical dislocation and their oviducts were removed. The four to five cumulus-oocytes-complexes (COCs) obtained from the ampulla of the fallopian tube were introduced in a 90 μL drop of HTF medium covered with paraffin oil.
An aliquot of 10 μL thawed suspension was added to the centre of the drop of preincubation medium (90 μL) covered with paraffin oil (Figure 1g). Two kinds of preincubation media, such as TYH with BSA or TYH with MBCD, were used as sperm preincubation in experiment 1, whereas TYH with MBCD was used as sperm preincubation in experiment 2. The thawed sperm were preincubated in the TYH with BSA for 60 min or in the TYH with MBCD for 30 min at 37°C with 5% CO2 in the air. After preincubation, an aliquot of 10 μL sperm suspension was collected from the peripheral part of the drop containing motile sperm using a wedge-shaped pipette tip (0.5–10 μL, Quality Scientific Plastics, Petaluma, CA, USA; Figure 1h). The sperm suspension was carefully transferred to an IVF drop containing COCs and incubated at 37°C with 5% CO2 in the air (final motile sperm concentration = 200–400/μL; Figure 1i). After 5 to 6 h, the inseminated oocytes were washed three times in a drop of 100 μL HTF covered with paraffin oil and were then cultured at 37°C with 5% CO2 in the air. Twenty-four hours after insemination, the fertilization rates were calculated as the total number of two-cell embryos divided by the total number of inseminated oocytes × 100.
Embryo culture and transfer
After IVF using fresh or frozen/thawed sperm, the fertilized oocytes that had developed to the two-cell stage within 24 h of insemination were divided into two groups (experiment 3). One group of two-cell embryos was transferred and washed three times in a 100 μL drop of mWM and further cultured for 72 h. At this time, the development rates of blastocyst stage embryos were calculated by the number of blastocyst stage embryos divided by the number of two-cell embryos × 100. In the other group, 20 of the two-cell embryos were transferred into the oviducts of each pseudopregnant Jcl/ICR female on the day a vaginal plug was found (day 1 of pseudopregnancy). After 19 days, the number of offspring was recorded.
Statistical analysis
Statistical analysis was performed using Prism version 3.0 (GraphPad, San Diego, CA, USA). Data are given as the mean ± SD. Comparison of the differences between the means for each treatment were carried out using analysis of variance after arcsine transformation of the percentage data. Differences between the means were considered to be significant when P < 0.05 was achieved.
Results
Experiment 1. In vitro fertilization of frozen/thawed C57BL/6J mouse sperm using various media for cryopreservation and preincubation
Effects of sperm freezing media with
Sperm cryopreservation was performed using a cryopreservation media of R18S3 or R18S3 containing
Frozen/thawed sperm were preincubated in TYH or in TYH with MBCD before in vitro fertilization
Each percentage value represents mean ± SD (n = 5). Values are significantly different compared with controls of R18S3 and TYH group at *P < 0.05, **P < 0.01
Experiment 2. Individual variability of fertilization rates in frozen/thawed C57BL/6J mouse sperm
Using a novel protocol for the IVF using frozen/thawed C57BL/6J sperm, the fertilization rates were relatively high for each mouse (Table 2). Fertilization rates of frozen/thawed sperm showed to be a little lower than those of fresh control; however, relatively high and stable rates were obtained for the cryopreserved sperm from individual males using the novel protocol combined with R18S3 containing
Fertilization rate of fresh and frozen/thawed sperm in individual C57BL/6J mice
In vitro fertilization was performed using fresh and frozen/thawed sperm
Each percentage values in the lowest row of fresh and frozen/thawed group represent mean ± SD (fresh sperm: n = 5, frozen/thawed sperm: n = 10)
*Values are significantly different compared with fresh controls at P < 0.05
Experiment 3. In vitro and in vivo development of embryos derived from fresh and frozen/thawed C57BL/6J mouse sperm
The rate at which two-cell embryos derived from frozen/thawed sperm developed into blastocysts or live young is shown in Table 3. In vitro, 81% of two-cell embryos developed into blastocysts from the frozen/thawed group. In vivo, 52% of transferred embryos were born from the frozen/thawed group. There was no significant difference in the developmental ability of two-cell embryos of the fresh and frozen/thawed groups.
In vitro and in vivo development of two-cell embryos produced from fresh and frozen/thawed sperm
Two-cell embryos derived from fresh and frozen/thawed sperm were divided two groups: one group was incubated in the modified Whitten's medium (mWM) for 72 h or the other group was transferred into oviducts of pseudopregnant mice (20 embryos/female)
Development rates were calculated by number of blastocysts/number of examined two-cell embryos × 100 (in vitro development) or number of live young/number of two-cell embryos transferred × 100 (in vivo development)
Each percentage value represents mean ± SD (fresh sperm: n = 5, frozen/thawed sperm: n = 10)
*Values are significantly different compared with fresh controls at P < 0.05
Discussion
In the present study, we demonstrated that the application of R18S3 with
Our previous findings demonstrated that the treatment of frozen/thawed C57BL/6J mouse sperm with MBCD lead to a dramatic activation in their fertility in vitro. 11 The period of preincubation affects sperm fertility, so different preincubation conditions were for BSA and MBCD to gain the highest possible fertilization rate. The highest rate of fertilization using BSA was obtained at 60–120 min, whereas that of MBCD was acquired at 30 min. Therefore, in this study, we adapted the preincubation period for these media in experiment 1. As a result, MBCD has high potential for capacitating sperm and can greatly enhance the fertilization rate compared with BSA.
On the other hand, most of the C57BL/6J mouse sperm remain in a cryo-induced damaged state after freezing and thawing.
10
In this study, we tried to overcome the problem by modifying the R18S3 with the addition of
Moreover,
Previously, Liu et al.
26
showed that R18S3 with amino acids increased the fertility of frozen/thawed sperm in three substrains of C57BL/6J mice.
Recently, there have been some reports of mouse sperm cryopreservation using modified R18S3. 27,28 Yildiz et al. 27 reported membrane permeating polyols such as glycerol and fructose have protective effects for sperm cryopreservation when added to the modified R18S3. Ostermeier et al. 28 showed that a reducing reagent of alpha-monothioglycerol added to R18S3 protects the frozen/thawed sperm. These reports demonstrated that modified R18S3 tends to enhance the fertilizing ability of frozen/thawed C57BL/6J mouse sperm. However, they did not investigate the individual variability of fertilization rate in each male.
In this study, we compared the fertilizing ability of frozen/thawed C57BL/6J mouse sperm in 10 males. As a result, we obtained stable and relatively high rates of fertilization (between 35.1% and 82.0%) in all males. Therefore, our novel system will become practical and available for sperm cryopreservation and IVF of numerous genetically-engineered mice strains with a C57BL/6J background.
In conclusion, we suggest that
Footnotes
Acknowledgements
We wish to thank T Kondo, K Fukumoto, Y Nakagawa, Y Takeshita and Y Nakamuta for excellent technical assistance and the Center for Animal Resources and Development, Kumamoto University for its important contributions to the experiments.
