Abstract
Various animal models for experimental osteochondral defect healing have been used in orthopaedic research. Two main defect locations were chosen: the patellar groove or the central part of the medial femoral condyles (MFC). To date, it is not clear whether both locations display similar patterns in critical size osteochondral defect healing. We retrospectively analysed both locations in our minipig model hypothesizing that they show similar healing pattern. Thirty-five defects were analysed after three or 12 months. Osteochondral defects were 10 mm deep and 6.3 mm (MFC,
The healing of cartilage defects is still a challenging problem in the clinical setting. Many operative procedures have been established, such as microfracturing, 1 osteochondral autograft transplantation 2 and autologous chondrocyte transplantation, 3 but each of them has shown distinct limitation in clinical experience. 4 Subsequently, there is a focus on tissue engineering-based strategies in current research to improve articular cartilage repair including stem cell, scaffold and growth factor-based technologies. For a detailed review the reader is referred to Hunziker. 5 As a matter of fact any new treatment protocol has to be evaluated in an appropriate animal model before being brought into clinical practice. For this purpose various animal models were employed to investigate experimental defect healing. The smallest animals used were mice 6 and rats, 7 being followed by rabbits. 8 For more advanced preclinical studies, large animal models such as dogs, 9 goats, 10 pigs 11 or horses 12 were used.
It has been shown that spontaneous healing of osteochondral defects is dependent on defect size, 13 depth, 14 geometry 15 and age 16 of the individual, but little is known about the influence of defect location on the natural healing response of osteochondral defects in the knee joint.
A larger animal used in our and other investigators’ work dealing with osteochondral articular cartilage defect repair is the Göttingen minipig (GMP) model. 11,17–22 In earlier studies investigating various scaffolds to improve repair tissue quality, we evaluated experimental osteochondral defect healing using two different localizations in the knee of miniature pigs: either in the central portion of the medial femoral condyle (MFC) or the medial facet of the trochlear groove (TG). This is the first report on different healing patterns of osteochondral defects created either in the MFC or the TG. To our knowledge there are only rare comparative studies available evaluating the impact of different defect localizations on osteochondral defect repair in one species. We retrospectively analysed the histological outcome of spontaneous osteochondral defect healing and hypothesized that both localizations will display a similar healing pattern.
Methods
Animals
All animal care, housing and treatment was performed according to the German Animal Welfare Act of 25 May 1998, after receiving written allowance for the experiments by the animal rights protection authorities in Baden Württemberg, Germany (Regierunspraesidium Karlsruhe, AZ: 35-9185.81/5/00, 35-9185.81/151/00).
The outcome parameters of 35 mature Göttingen minipigs (Elegaard Soro Landevej 302, DK-4261 Dalmose, Denmark) of either sex (12 male; 23 female) were retrospectively used for this study. The average age and weight of the minipigs were 32 months (22–49 months) and 34.1 kg (21–46 kg), respectively, at the beginning of the study. Animals were held in big indoor runs for at least one week for acclimatization with unrestricted movement of food and water
Defect localization and surgery
All animals were operated in aseptic conditions under general anaesthesia on both hind legs either at the MFC or the medial facet of the TG as described elsewhere, creating exactly one defect per knee joint. 18,20 In both locations the defect diameter was chosen to allow exact fitting in the area of the corresponding joint surface. One side was left empty, the other one was treated with various experimental procedures, reported elsewhere, see above.
To access the MFC, a medially-placed paramedian skin incision was set at each knee, followed by soft tissue preparation to the knee capsule which was opened with a 2 cm incision directly placed over the MFC. The location for the osteochondral lesion was chosen centrally and midline on the central weight-bearing portion of the medial condyle, 10 mm distal of the intercondylar notch. The cartilage was cut to the subchondral bone plate perpendicular to the joint surface with a circular cutter to avoid cartilage tearing. A diamond bone cutting system (Surgical Diamond Instruments [SDI]; MedArtis AG, Germany) was used to create a 6.3 mm wide and 10 mm deep cylindrical osteochondral defect. The diamond cutter was rinsed with 0.9% sterile saline to avoid heat damage to the adjacent bone.
For the TG defect localization, a median skin incision was placed between the tibial tuberositas and the distal patellar pole while extending the knee. After soft tissue preparation, the patellar tendon and the inferior patellar fat pad were split longitudinally, exposing the patellar groove. The articular cartilage on the medial trochlear facet was cut 10 mm above the intercondylar notch as described above with a 5.4 mm circular cutter, and the osteochondral defect of 5.4 mm × 8 mm was created with a bone wet grinding device. After extraction of the osteochondral plug in either localization, the defect was rinsed with saline solution and any bone or cartilage debris was removed carefully and the wound was closed in layers.
The animals of both groups were allowed immediate, full weight bearing postoperatively. Animals were followed-up closely during the first 10 days after operation, with special concern for postoperative pain levels and abnormal gait (limping). If necessary, intramuscular pain medication was administered with 0.01 mg/kg buprenorphine (Temgesic©, Essex Pharma GmbH, Germany). Animals were sacrificed by an intravenously administered overdose (40 mL) of barbiturate (Eunarcon®, Upjohn GmbH, Germany) either three or 12 months postoperatively and the hind legs were dissected to obtain the knees for further preparation.
Histological evaluation
Following defect preparation, a cube of approximately 2 × 2 × 2 cm was cut out of the MFC or the medial trochlea, respectively, including the repair zone in the centre. This cube was cut dividing the round repair zone into 2/3:1/3. Formalin-fixed specimens were decalcified in 0.5 mmol/L ethylenediaminetetraacetic acid (pH = 7.4) for four to six weeks followed by dehydration and paraffin embedding. Serial sections (5
Histological grading scale by Mainil-Varlet reaching a maximum of 34 points
Means of each subheading are given in brackets for the medial femoral condyle (MFC) and trochlear groove (TG) defects
Statistical analysis
Since the variance of the evaluated groups was significantly different, non-parametric tests were utilized: the Mann-Whitney
Data analysis was performed using SPSS for windows 10.0 (SPSS Inc, Chicago, IL, USA).
Results
After three and 12 months, respectively, nine and 10 MFC defects were available, whereas nine and seven TG defects could be examined.
Clinical observation revealed that animals with osteochondral defects in the MFC showed prolonged postoperative recovery in terms of higher pain levels with limping up to 7–10 days, whereas only 3–5 days of limping was seen in the TG group.
Semi-quantitative histological evaluation with the modified O'Driscoll score did not show any statistical differences in the three-month group (median/interquartile range [IQR] 13/8.5 in the MFC vs. 14/6.5 in the TG). After 12 months, the modified O'Driscoll score values improved for both defect localizations showing significance only for the MFC group (
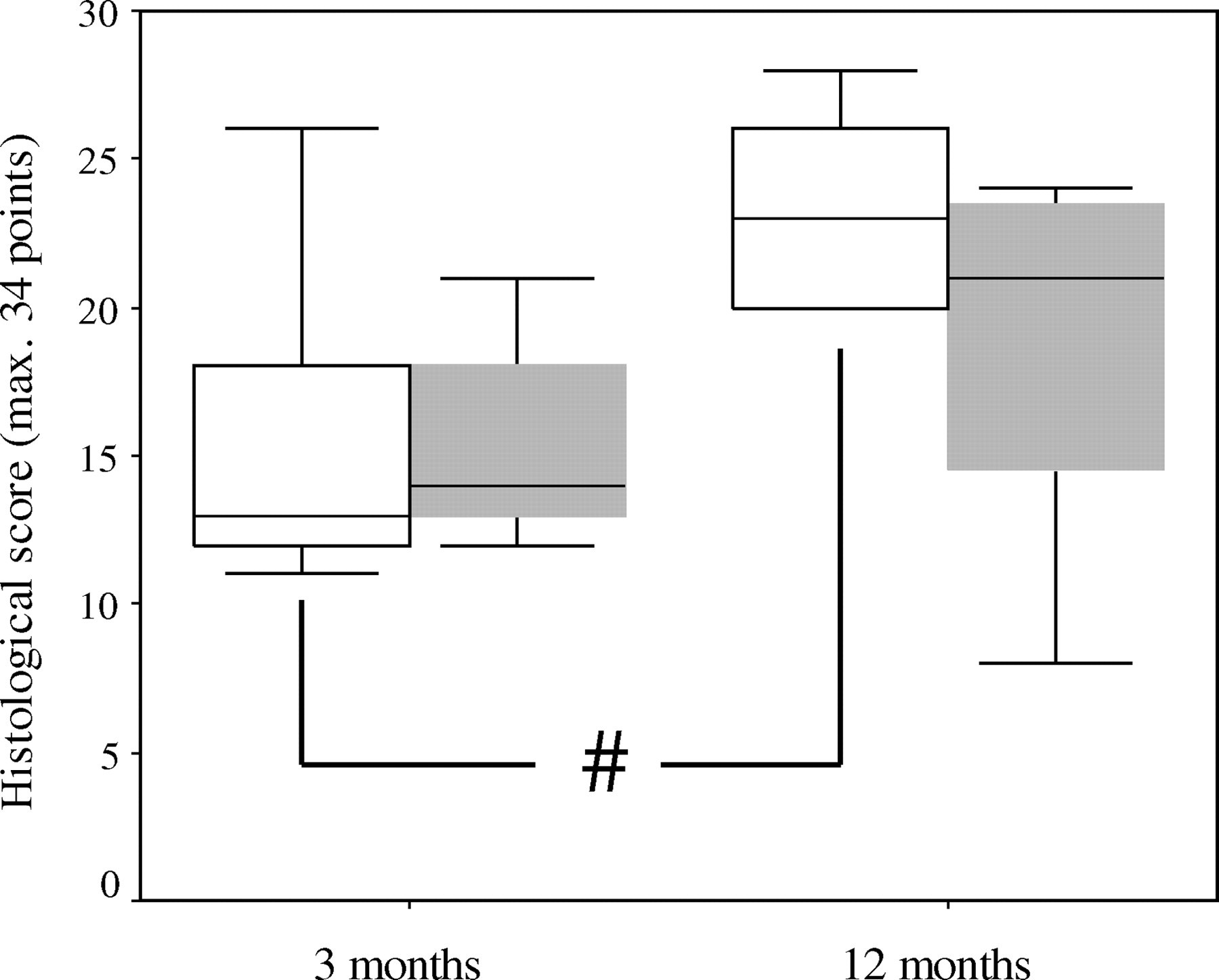
Box plot showing median, 25 and 75 quartiles of the histopathological scoring revealing no significant differences between the two defect localizations after either three or 12 months. A significant change of the histopathological score values was found between the three and 12 month interval in the femoral condyle group (#
New trabecular bone formation was conducted by enchondral ossification beginning at the defect edges in the three-month group. This process started to ‘bridge’ the osteochondral defect in the upper third of the defect immediately after three months in the MFC group and allowed neo-cartilage tissue formation towards the joint cavity (Figure 2A). Anyhow up to three months no defect was ‘bridged’ completely. Complete restoration of the subchondral bone plate after 12 months resulted in subchondral bone cyst formation, which did not regenerate until 12 months postoperatively (Figure 3).
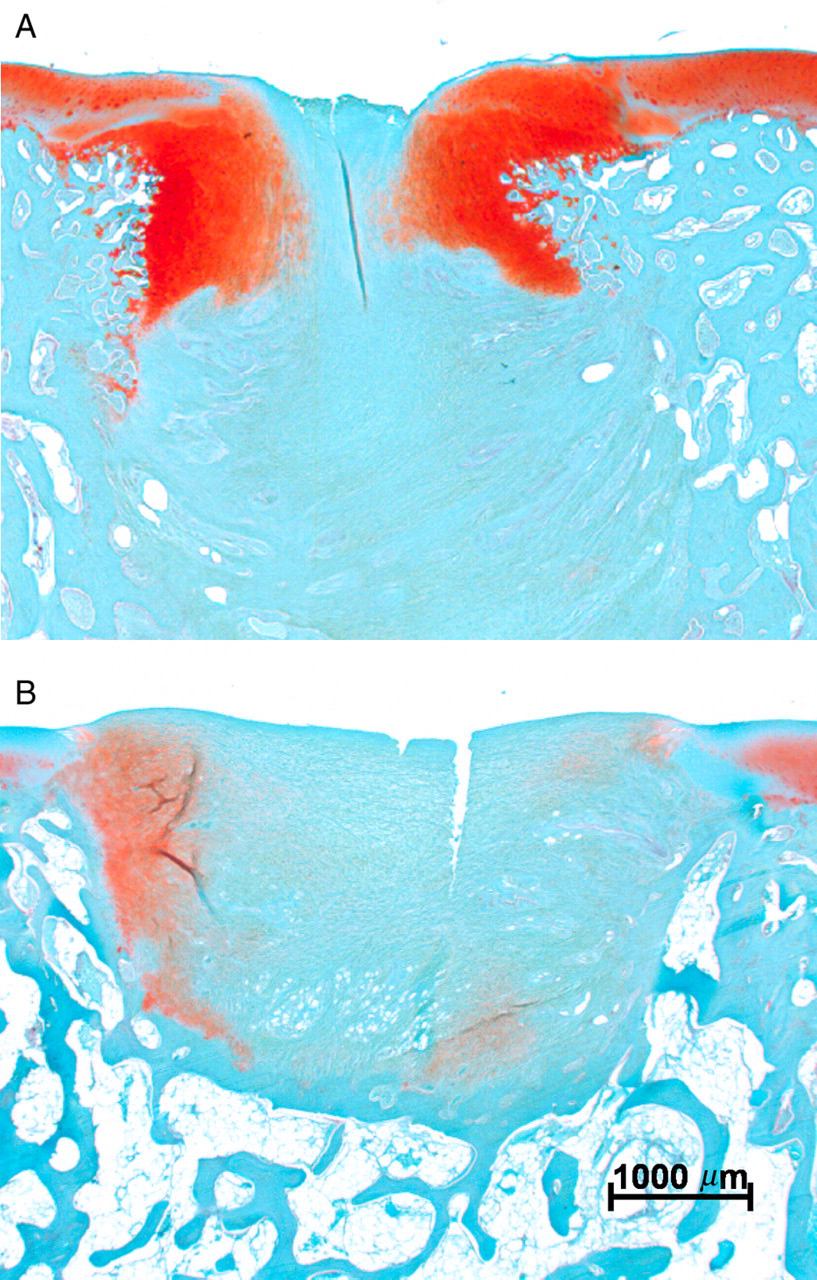
(A) Enchondral ossification three months postoperatively at the defect rim of femoral condyle group: an ossification front is closing the upper part of the defect leaving chondrocytes at the defect surface, restoring the chondral part of the defect. (B) Nearly no enchondral ossification was seen three months postoperatively in the trochlea groove group
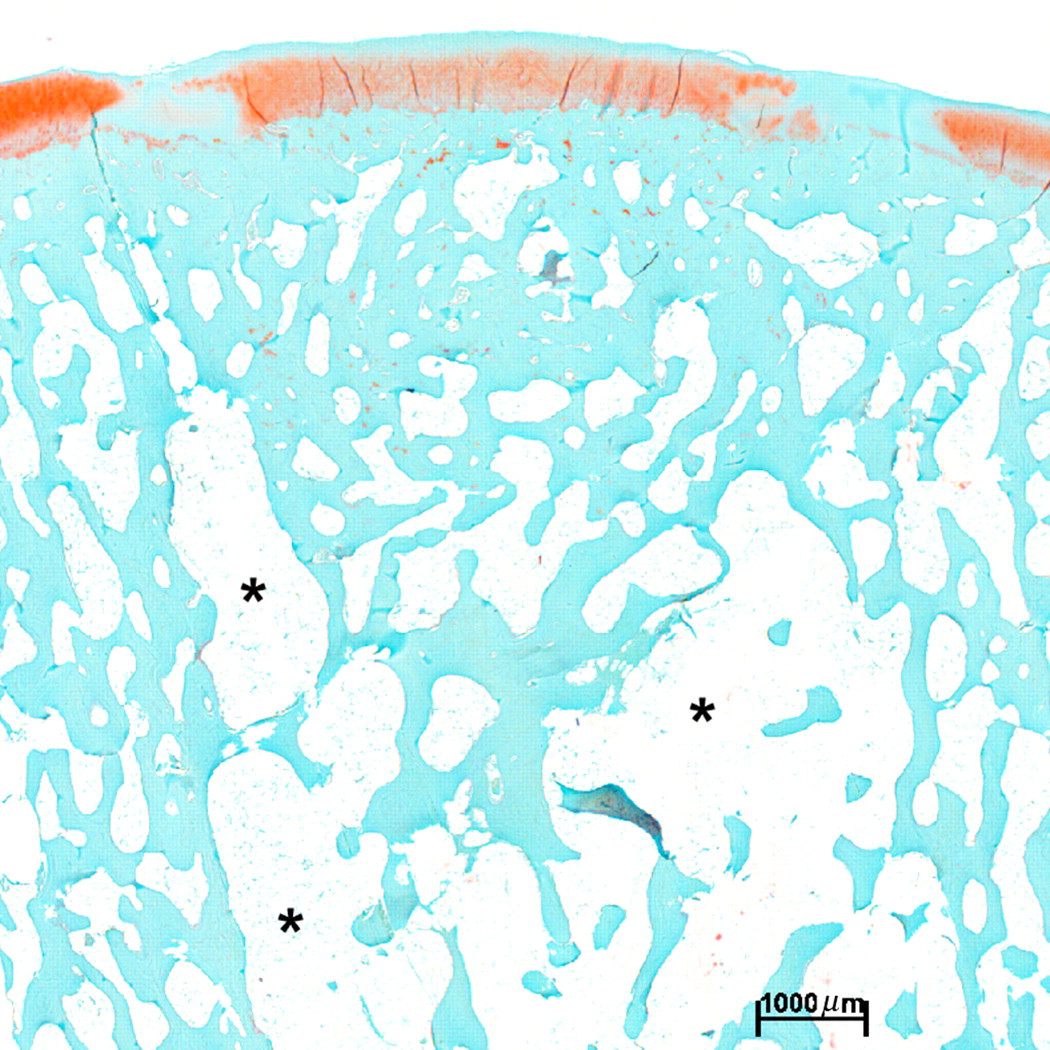
Defect healing by enchondral ossification in the femoral condyle group was showing nearly almost complete restoration of the subchondral bone after one year, even though a bone cyst (see asterisks) resulted in the deeper parts of the defect
In TG defects enchondral bone formation at the upper defect edges was also found after three months, but on a much lesser extent compared with MFC defects (Figure 2B). In contrast, complete closure of the subchondral bone cavity by new bone formation creating a subchondral bone cyst was rarely seen at three and 12 months. Going along with this finding, less glycosaminoglycan (GAG)-positive regeneration tissue formation in the TG group was seen. Statistical analyses revealed bone cyst formation significantly more often after 12 months in MFC defects when compared with TG defects (70% in MFC vs. 14.3% in TG defects,
Seven out of 10 specimens with cyst formation in the MFC group showed nearly complete subchondral bone repair in the upper third of the defect. The regenerated bone was covered with cartilagineous repair tissue (in 8 out of 10 defects), with round or oval chondrocyte-like cells within lacunae surrounded by a GAG-rich extracellular matrix (ECM) (Figure 4A). ECM appeared to be hyaline-like, without prominent collagen bundles and in three specimens columnar radial orientation of the cells similar to native articular cartilage (Figure 4B) was found. The regeneration tissue of TG defects displayed a more fibrocartilaginous quality (in 5 out of 7) with no radial orientation of the chondrocyte-like cells and less GAG deposition in the ECM (Figures 4C and D).
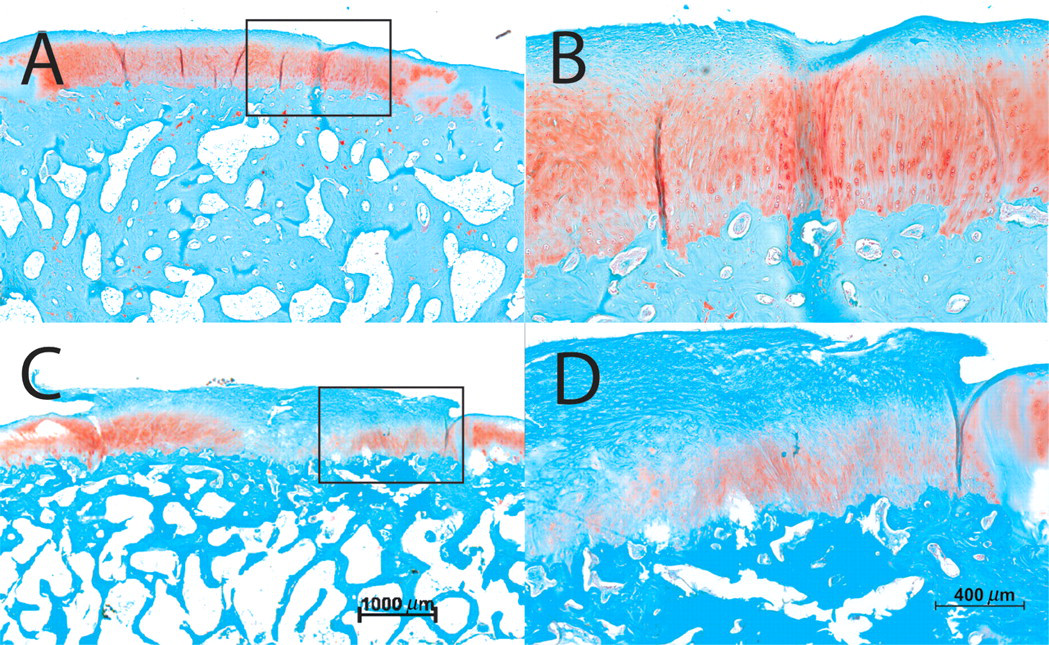
(A) Cartilaginous repair tissue was seen in eight out of 10 specimens after one year in the femoral condyle group. Radial orientation of the chondrocyte-like cells was noted in some specimen (B = magnification of A). (C) In trochlear groove defects, fibrocartilaginous repair tissue was found with no radial orientation of the cells (D = magnification of C). A and C: bar = 1000
Discussion
Clinical repair of osteochondral defects is still a challenging problem. Various defect models in different species have been used recently to examine the natural healing response and to evaluate the benefit of new treatment modalities. The central part of the MFC and the TG have been extensively used in several larger experimental animal models including goats, 10,25 dogs, 9 sheep 26 and pigs. 19,21,27–29 Surprisingly, only a few studies are published comparing the effect of the defect location on the natural course of osteochondral healing in the knee joint in a larger animal model. 30,31
In the presented work we retrospectively analysed our data when both defect locations were used for experimental osteochondral defect repair in the GMP model. A remarkable finding was that osteochondral defects located in the MFC did show a significantly more frequent cystic subchondral bone cavity after complete restoration of the subchondral bone plate. In addition, articular cartilage repair tissue quality seemed to improve with time and at its best resulted in cartilage-like tissue in MFC defects compared with fibrocartilaginous tissue in TG defects. Jackson
Several factors have been identified to predominantly influence the natural healing response of artificial osteochondral defects. Among these are defect size,
13
depth,
14
geometry
15
and the age of the individual animal.
16
There are also published data indicating that the mechanical environment of the defect location does play an important role for spontaneous defect repair. Duda
Based on earlier published data, the tibiofemoral and patellofemoral joint compartments have to be considered as fully weight-bearing in humans and larger animals with a similar amount of mechanical load. Kuster
In both localizations incomplete filling of the articular cartilage defect void with fibrous and fibrocartilage-like repair tissue was present. Semi-quantitative scoring did not show any differences in mean total score values, whereas qualitative histomorphological analysis revealed cartilage-like repair tissue in MFC defects more frequently after one year. Tissue formation is a subtle and complex process during defect restoration and the process of bone and cartilage formation is linked to each other. A possible explanation for the better chondral regeneration in the MFC group may be the fact that the MFC defects display an earlier closure of the bony defect with a full restoration of the subchondral bone plate starting from the defect rim. As reported by Huang
In conclusion both defect localizations are principally suitable for experimental osteochondral defect repair in the GMP model. In experimental cartilage healing, low or even no self-repair capacity is desirable, in order to increase the differences between potentially beneficial cartilage-healing protocols and the empty defects. The somewhat better histomorphological results in cartilage repair and the longer limping in the MFC group have to be considered therefore as a major drawback. Since osteochondral defects in the patellar groove displayed a more limited capacity for regenerating a cartilage-like repair tissue with reduced pain levels, this defect localization seems to be more favourable for experimental osteochondral defect repair. Exact differences in defect rim stress and shear forces at these two defect localizations are not evaluated so far. Further experiments in that direction could enlighten the role of the biomechanical load in the spontaneous repair of osteochondral defects however.
Footnotes
ACKNOWLEDGEMENTS
We would like to thank Dr vet. Helga Lorenz for her expertise in animal anaesthesia and animal care. This study was funded partly by the Research Fund of the Department of Orthopaedic Surgery, University of Heidelberg, Germany.
