Abstract
This paper describes the results of a study of the effects of modified housing conditions, conditioning and habituation on humans using a rabbit model for monitoring whole-cell pertussis vaccine (pWCV)-induced adverse effects. The study has been performed with reference to previous vaccine safety studies of pWCV in rabbits in which results were difficult to interpret due to the large variation in experimental outcome, especially in the key parameter deep-body temperature (T b). Certain stressful laboratory conditions, as well as procedures involving humans, e.g. blood sampling, inoculation and cage-cleaning, were hypothesized to cause this large variation. The results of this study show that under modified housing conditions rabbits have normal circadian body temperatures. This allowed discrimination of pWCV-induced adverse effects in which handled rabbits tended to show a dose-related increase in temperature after inoculation with little variance, whereas non-handled rabbits did not. Effects of experimental and routine procedures on body temperature were significantly reduced under modified conditions and were within the normal T b range. Handled animals reacted less strongly and with less variance to experimental procedures, such as blood sampling, injection and cage-cleaning, than non-handled rabbits. Overall, handling had a positive effect on the behaviour of the animals. Data show that the housing modifications have provided a more robust model for monitoring pWCV adverse effects. Furthermore, conditioning and habituation of rabbits to humans reduce the variation in experimental outcome, which might allow for a reduction in the number of animals used. In addition, this also reduces distress and thus contributes to refining this animal model.
Keywords
The rabbit is used as the animal of choice in the evaluation of pertussis whole-cell vaccine (pWCV) safety studies, as the weight and sensitivity of rabbits for endotoxins are comparable with those of infants. Vaccination against whooping cough with pWCV is, and will be, part of vaccination programmes in many countries throughout the world. 1 However, incidentally, vaccination against whooping cough with pWCV might cause acute adverse effects in babies, such as high fever and persistent crying, but permanent disabilities have also been reported. 2 The adverse effects and the re-emerging of pertussis in well-vaccinated populations indicate the need for improving the current pertussis vaccines with fewer adverse effects. 1,2 Although nowadays an acellular pertussis vaccine, which induces less adverse effects, is available, it is expensive and worldwide demand cannot be met. Consequently, there is a growing demand for an improved pWCV with fewer adverse effects. 2 Endotoxin or lipopolysaccharide (LPS), a heat-stable toxin associated with the outer membranes of certain Gram-negative bacteria, and pertussis toxin (PT) are considered the major factors for adverse reactions. PT upsets the balance in immune responses and induces leukocytosis. LPS mainly induces fever. Therefore, studies into pWCVs with different amounts of pertussis antigen have been performed in rabbits.
Unfortunately, results of these studies were difficult to interpret due to the large variation in experimental outcome. Especially, the high variation in body temperature increase after inoculation made it impossible to detect vaccine-related responses. Housing conditions as well as experimental and routine procedures were identified as the major stress factors that might have contributed to this large variation. This was based on the observations of the animal caretakers and biotechnicians who worked with the rabbits during these procedures. Based on the rabbits' behaviour it was determined that catching the animals out of the pen, blood sampling preceding inoculation and inoculation itself was very stressful for the animals.
In order to provide a more robust animal model for pWCV safety testing, housing conditions in the present study were modified compared with the earlier studies. These modifications were based on the natural requirements and biological rhythms of rabbits. The following changes were made: lowering of room temperature (from 21°C to 19°C), reduction of group size (from 20 to 10 rabbits per pen), introduction of dusk and dawn periods, lowering of light intensities (from 90 lux to ±60 lux) 3 and restricted feeding. Furthermore, stress-reducing measures were implemented, such as conditioning to persons entering the animal room. Finally, half the number of animals were habituated to humans by handling from birth onwards. This handling was performed as an additional stress-reducing measure as evidence was accumulating that the experimenter might have been considered a predator by the animals, which caused high levels of anxiety, which might have confounded the reliability and validity of the results. 4 Positive interactions between animal caretakers and research animals are believed to minimize stress (responses) and therefore reduce bias in experimental outcome. 4–6
In order to investigate whether these modifications and stress-reducing measures were refinements in the housing and husbandry of rabbits, and whether these modifications resulted in a more robust animal model for the pWCV safety testing, the immunization assay in the earlier studies was used as a case study in the present study. We used the DKTP-Hib (diphtheria-pertussis-tetanus-polio-haemophilus influenzae type b) vaccine (Netherlands Vaccine Institute [NVI]) with purpose-made dilutions of pWCV as a test compound to study the effects of the modified conditions and the effect of handling on the variation in the test results. The parameters measured were body temperature, serological and haematological parameters and changes in body weight.
Materials and methods
Animals, housing and husbandry
In the present study, at the age of four weeks, 20 handled and 20 non-handled specific pathogen-free (SPF), male New Zealand White outbred rabbits (Harlan, Horst, The Netherlands) were weaned and transported in a climate-conditioned van to the animal facilities of the NVI. The handled animals were housed in a room different from the non-handled rabbits. The animals were housed under SPF conditions in groups of 10 animals per pen (2.5 × 1.4 × 1 m; length × width × height of the border). Each pen was enriched with a removable and height adjustable shelf (1.00 × 0.50 m) that functioned as a hiding and look-out place. The solid floors were covered with autoclaved beech-wood cubic grain (grain size 2.5–3.5 mm; Lignocel; J Rettenmaier & Söhne GmbH + Co, Rosenberg, Germany). The pens, two per room, were situated in temperature-controlled rooms (19 ± 0.7°C with 50–60% relative humidity). As rabbits are crepuscular/nocturnal animals, they were held under a 12:12 h dark (5–8 lux; intensity measured at animal level)/light (60–65 lux; intensity measured at animal level) cycle with incorporated dusk and dawn periods of 30 min that preceded the onset of, respectively, the dark and light period at 18:00 h and 06:00 h. The animals were fed pellets and hay. To ensure that the animals would not have to undergo a rapid dietary change on top of the change in environment upon arrival at the NVI, it was decided to gradually (2 weeks' time period) switch from pellets (Teklad Global Diet pellets, Harlan, UK) that were fed to the rabbits during their nursing period at the breeder's facility to pellets fed at the NVI (Hopefarms, Woerden, The Netherlands). Tap water was available ad libitum and was being refreshed while animals were given their daily amount of food. Pens were cleaned out when necessary, as the degree of soiling depends on age and body weight of the rabbits. All animals were visually inspected in the morning and in the afternoon during feeding. At times of weighing, at least once a week, the animals were submitted to clinical examination.
In order not to disturb the rabbits' physiological circadian rhythm, feeding, water refreshment and all experimental procedures were performed in the afternoon prior to the onset of dusk. All animals were conditioned and habituated to persons entering the animal room for routine (daily visual controls, cleaning, feeding and water refreshing) and experimental (immunization, blood sampling and weighing) procedures by knocking on the door before entering the animal room. While entering the room, the rabbits were talked to in a low soothing tone of voice. 7 All persons who had visual or physical contact with the rabbits were involved in both pleasant, such as feeding, and unpleasant, such as blood sampling, procedures and wore white laboratory coats and latex gloves.
Differences in animals, housing and care between the previous studies and this study are shown in Table 1. From hereon the laboratory and experimental conditions under which the animals were held in the earlier studies will be referred to as reference conditions. The conditions the animals were held under in this study will be referred to as modified conditions.
Overview of differentiations in animals, housing and care between reference and modified conditions
For the immunization assay, the animals had to reach a minimum body weight of 2.5 kg, which is comparable with the weight of babies upon immunization. By the time the animals had reached this weight, i.e. at the age of 12 weeks, they started to become sexually active. In order to maintain group housing throughout the entire experiment, the rabbits were castrated at this age by removing the testicles and epididymis. The rabbits were sedated using a combination of ketamine 10% (Ketamine®, Alfasan, Woerden, The Netherlands) and medetomidine hydrochloride (Domitor®, Pfizer, Capelle a/d Ijssel, The Netherlands). This was injected intramuscularly in the Musculus triceps brachii using a 26G × 0.5″ needle on a 1 mL Terumo® syringe; Terumo Europe NV, Leuven, Belgium. In the handled rabbits, it took longer for the anaesthetics to become effective. Therefore, the handled and non-handled rabbits were injected with 0.25 mg/kg and 0.2 mg/kg body weight of both ketamine 10% (Ketamine®; Alfasan, Woerden, The Netherlands) and medetomidine hydrochloride (Domitor®, Pfizer, Capelle a/d Ijssel, The Netherlands), respectively. This phenomenon might be due to the fact that the non-handled animals were more active, which may be related to higher stress levels. This activity may have resulted in a better circulation of the anaesthetics through the body. In order to anaesthetize the animals only once, the telemetry transponder (for details see Van der Hel et al. 8 and Jansen et al. 9 ) was implanted into the peritoneal cavity by means of laparotomy directly after orchidectomy for the continuous body temperature measurements.
The surgical procedures were performed following the Guidelines of Rodent Survival Surgery (
According to Dutch legislation, the institutional Animal Ethical Review Committee approved the entire experiment.
Experimental design and procedures
Handling procedure
Sixteen timed-pregnant does, housed at the breeder's facility, were used for this experiment. All does gave birth within one or two days from each other. To prevent doe-related and litter size-related factors from interfering with the experiment, part of the litter of one doe was exchanged with part of the litter of another doe. This resulted in each doe having more or less the same litter size consisting of some pups of its own and some of another. After this exchange, litters were randomly assigned (randomizer at
The pre-weaning handling treatment followed the procedure of Bilkó and Altbäcker 4 and consisted of removing the entire litter, transporting it in a Makrolon cage with sawdust bedding, measuring the weight of the rabbits one by one on an electric scale and then returning the pups back into the nest. This way, handling was standardized, as all pups were caught four times, and the time spent out of the nest was also more or less the same for each litter, in general not more than 5 min. This procedure was performed six times a week from birth until weaning at the age of four weeks. The non-handled pups were weighed once a week following the same procedure.
At four weeks of age the pups were weaned and separated by gender. Thereafter, 20 handled and 20 non-handled male pups were randomly assigned (randomizer at
Behavioural tests were also performed in order to study whether modifying laboratory conditions and experimental and routine procedures, as well as conditioning and habituation to humans, will improve animal welfare. The results of these tests will be published in a separate manuscript.
Immunization assay
In order to investigate whether refinements in the housing and husbandry of the rabbits resulted in a more robust animal model for pWCV safety testing, the immunization assay in the previous studies was used as a case study in the present study. We used the DKTP-Hib vaccine (NVI) with two dilutions of pWCV, i.e. 8 and 16 International Opacity Units (IOU)/mL. Control animals were immunized with sterile saline. Together with the handling treatment, this resulted in the experimental groups shown in Figure 1. Animals with higher and lower body weights were equally assigned to the different product concentrations in order to have a similar mean body weight per product group and thus to overcome effects of body weight on the responses to the different products.
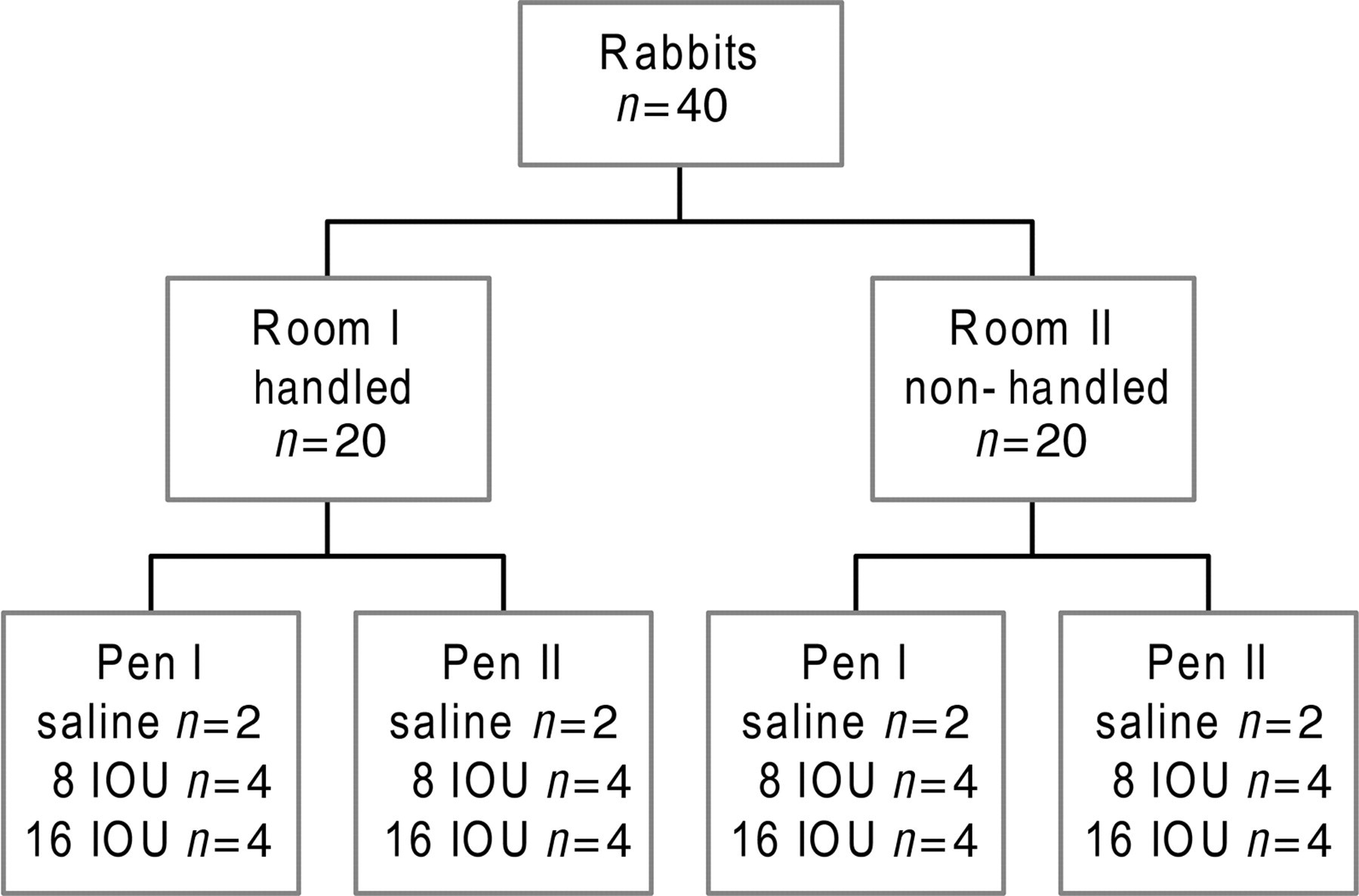
Experimental design showing the groups studied under modified conditions. n = number of animals
Each animal was immunized three times with a four-week interval in the Musculus vastus lateralis of the hind leg, which equals the muscle in the thigh of babies where the vaccine is injected. This muscle is located at the front side of the thigh and together with the rectus femoris, vastus medialis and vastus intermedius belong to the quadriceps femoris. This muscle was selected as it is easy to fixate and thus one can be sure of an intramuscular injection, which is not always the case when injecting in the mass of thigh muscles (manuscript in preparation). The volume used was 1 mL, which is the same volume as that used in babies (information leaflet DKTP-Hib vaccine, NVI, February 2003).
Two control blood samples of 2 mL each were taken 20 days and seven days before the first immunization to check for abnormalities, e.g. present antibody titres against pertussis, and to establish baseline levels of the relevant serological and haematological parameters. In this way the animals were also familiarized with the blood sampling procedure. One day before each immunization, and seven days as well as 28 days after each immunization, blood was sampled from the central ear artery for serology and haematology. 11 In order to measure the effect of the product on body weight, animals were weighed one day before, and three days as well as seven days after immunization. Whenever possible this was combined with blood sampling. The effect of the product on body temperature was continuously measured by means of telemetry.
Haematology
Two millilitres of blood were dispensed into ethylenediaminetetraacetic acid tubes (Greiner Bio-One GmbH, Kremsmünster, Austria) for haematology and processed directly after the samples had been taken. Analyses were done using automated procedures. A Coulter Counter (Beckman Coulter Nederland BV, Mijdrecht, The Netherlands) was used to determine white blood cells (WBC), red blood cells (RBC), haemoglobin (Hg), haematocrit (Ht), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), lymphocyte counts (lymph) and the percentage of lymphocytes in blood (lymph %).
Serology
Two millilitres of blood were dispensed into serum tubes (Becton-Dickinson Vacutainer®, Plymouth, UK). Serum samples were allowed to clot overnight at a temperature of 7°C and were then centrifuged for 10 min at a speed of 3500 rotations per minute (rpm). Serum was stored in cups at −20°C until assayed on pertussis antibodies by means of a whole-cell enzyme-linked immunosorbent assay (WCE). The WCE was carried out as described previously by Van der Ark and colleagues. 12 Some modifications were made for this study.
Flat bottom 96-well microtitre plates (Immulon 2HB; Becton-Dickinson) were coated with 100 μL/well of an 18,323 whole-cell pertussis suspension (0.25 IOU) in phosphate-buffered saline (PBS) until evaporation was complete (overnight at 37°C). The plates were washed with water containing 0.03% Tween-80 and then incubated for one hour at 37°C with the serum samples and two control sera (negative rabbit serum 517 pre-serum facilitated by Marcel Hijnen and a positive mouse serum; pool group 1 200400279 imm. 1.6 IOU whole cells Tohama) serially diluted in eight steps of three-fold serial dilutions in PBS containing 0.1% Tween-80, starting at a 1:50 dilution. After incubation the plates were washed again followed by a second incubation for one hour at 37°C with goat anti-rabbit IgG (H + L chain-specific) horseradish peroxidase conjugate (SBA-4050-05), 1:5000 diluted in PBS containing 0.1% Tween-80 and 0.5% skimmed milk powder (Protifar, Nutricia, The Netherlands). Plates were washed and 100 μL of TMB substrate (0.1 mg/mL 3,3',5,5'-tetramethylbenzidine, 0.012% H2O2 in 0.1 mmol/L sodium acetate buffer pH 5.5) was added. After incubation for 10 min at room temperature, the reaction was stopped by adding 100 μL of 2 mmol/L H2SO4 (SVM, Bilthoven, The Netherlands). The optical density (OD) of the yellow colour that was formed was measured at 450 nm and IgG antibody titres were expressed as the serum dilution, giving an OD 50% of the maximum at OD450 (=0.8).
Data analyses
The main parameter, body temperature, was measured continuously by means of telemetry. The transponders measured the deep-body temperature of rabbits to an accuracy of 0.1°C. 8,9 The sequence table had a time interval of 9 min. Thus, the body temperature of each animal was recorded within this time period of 24 h a day.
To measure the effect of experimental procedures on body temperature, the circadian rhythm of body temperature per animal was established by continuously monitoring the body temperature during the experimental period. The days on which experimental procedures and cage-cleaning were performed were left out. The data of the remaining days (4 months) were averaged per rabbit per hour of day (Figure 2a). The changes in body temperature related to experimental and routine procedures were corrected for the normal circadian body temperature: the latter values were subtracted from the procedure-induced changes. The maximum increase in body temperature and the maximum duration of it as a reaction to experimental and routine procedures were calculated by means of SigmaPlot (version 8.0, Systat Software GmbH, Erkrath, Germany). These parameters, along with the haematology parameters and antibody titre, were further analysed with the Statistical Package for Social Sciences (SPSS, version 12.0; SPSS Benelux BV, Gorinchem, The Netherlands).
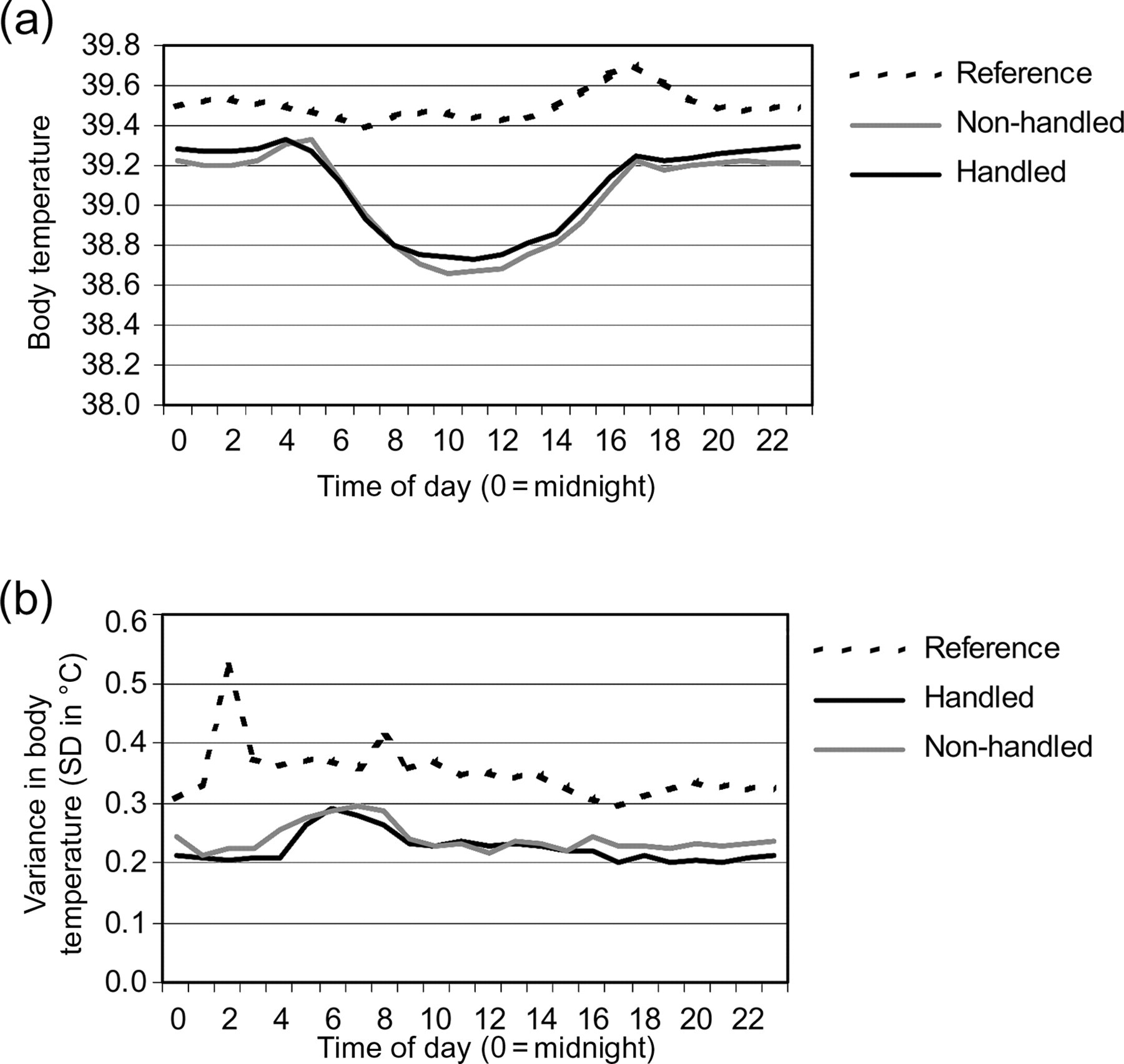
(a) Normal circadian body temperature of rabbits during the experimental period. The figure shows the daily mean T b of rabbits per experimental condition. Values per time of day are means of pooled data of days on which no experimental procedures or cage-cleaning were performed. (b) Variance in the normal circadian body temperature of rabbits during the experimental period. The figure shows the variance in the daily mean T b of rabbits per experimental condition. Values per time of day are means of the variance of pooled data of days on which no experimental procedures or cage-cleaning were performed
The Kolmogorov-Smirnov test of normality and the Levene's test of homogeneity of variance were run for all parameters. The majority of the parameters demonstrated normality and equal variances between groups, which made parametric tests preferable. The significance of the differences between groups per time-point was calculated by means of analysis of variance (ANOVA). For all parameters handling and vaccine were taken as fixed factors and body weight as covariate. Repeated measures were performed to analyse the effects over time. The within-subject factors were the different time-points of the within-subject variables blood sampling and immunization. Handling and vaccine were used as between-subject factors.
A rejection criterion of 0.05 was set for all statistical tests. When the ANOVA showed statistically significant effects of handling, the group means were further compared using the unpaired Student's t-test. If the ANOVA showed statistically significant effects of product, the group means were further compared using the Bonferonni post hoc test. Based on the results of earlier experiments, no direction could be assumed for the qualitative and/or quantitative nature of these effects. Therefore, all statistics were two-tailed.
Results
Body temperature of rabbits
In order to allow discrimination of the effects of experimental and routine procedures on the body temperature (T b) of rabbits, the circadian body temperature was determined for each individual rabbit. Figure 2a gives an overview of the mean circadian rhythm in T b per experimental group. Figure 2b shows the variance (expressed in standard deviation of the population) in T b per experimental group. From these figures it can be concluded that the modification of the housing conditions resulted in the characteristic sinusoidal waveform of deep-body temperature with only little variance in T b per hour of day. Handling had no effect on the sinusoidal waveform, temperature range or variance in T b.
Effect of blood sampling on body temperature
In the earlier studies, using non-handled rabbits, the effect of blood sampling on T b interfered very strongly with the inoculation response, thereby not allowing significant differences between the different products to be detected (Figure 3). The figure illustrates the results of the blood sampling preceding the first immunization. Under the modified conditions blood was sampled the day preceding the immunization, to prevent interference of blood sampling on the inoculation response. From Figures 4 and 5 it may be concluded that blood sampling seems to have minimal effects on the normal T b of rabbits, in which handled animals show the smallest effect with only little variance. In Table 2, the effect of blood sampling on body temperature is shown. From Table 2 it can be concluded that under the modified conditions the maximum increase and variance in T b due to blood sampling is much lower than in the reference study. Overall, handled animals showed less variance in T b increase and duration of the response than non-handled animals (Table 2). Surprisingly, the handled animals showed an increase in T b to blood sampling over the experimental period, whereas non-handled animals showed a decrease, just as the rabbits did in the reference study (Table 2). Yet, the variance in blood sampling in the handled animals decreased over the experimental period, whereas the variance in the non-handled animals increased (Table 2). During the experimental period, the duration of response increased for the handled animals, whereas it decreased for the non-handled animals (Table 2).
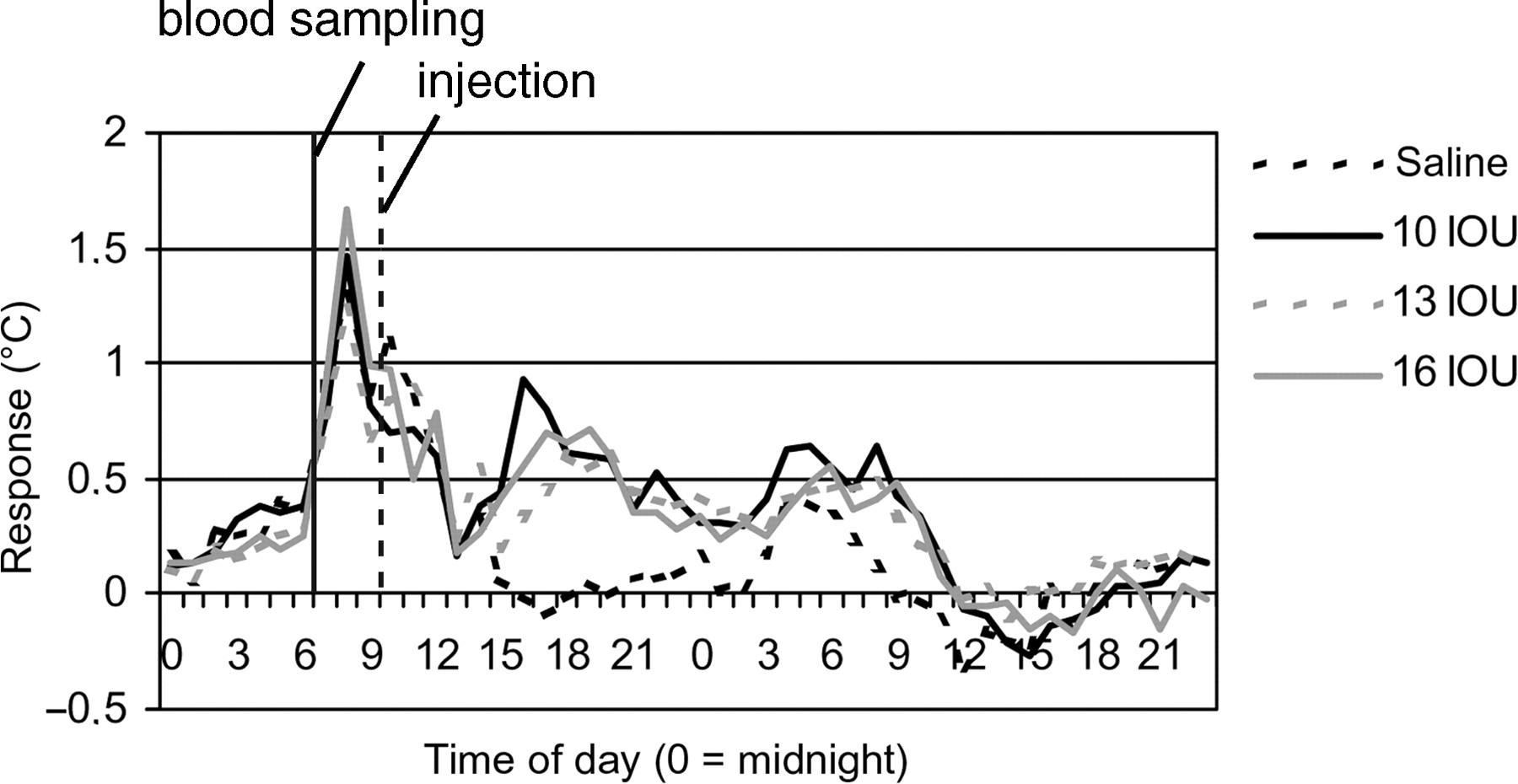
Effect of blood sampling and vaccine-related dose–response on body temperature in the reference study. The figure shows the changes in T b related to blood sampling and vaccine dose-dependent response at the first immunization (means per dose group). Values for T b on the day of blood sampling and immunization were subtracted with values of the normal circadian body temperature
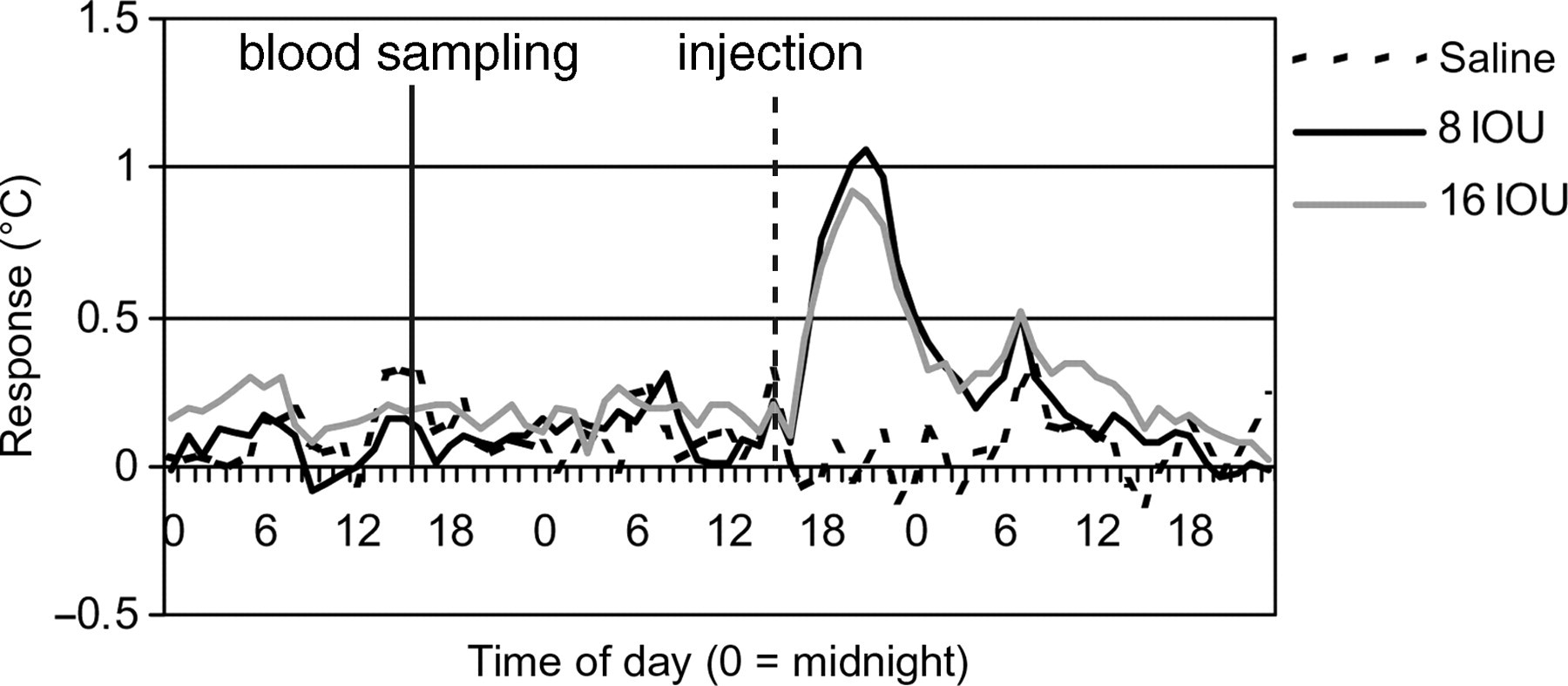
Effect of blood sampling and vaccine-related dose–response on body temperature under modified housing conditions for non-handled rabbits. The figure shows the changes in T b of non-handled rabbits towards blood sampling and vaccine dose-dependent response at the first immunization (means per dose group). Values for T b on the day of blood sampling and immunization were substracted with values of the normal circadian body temperature
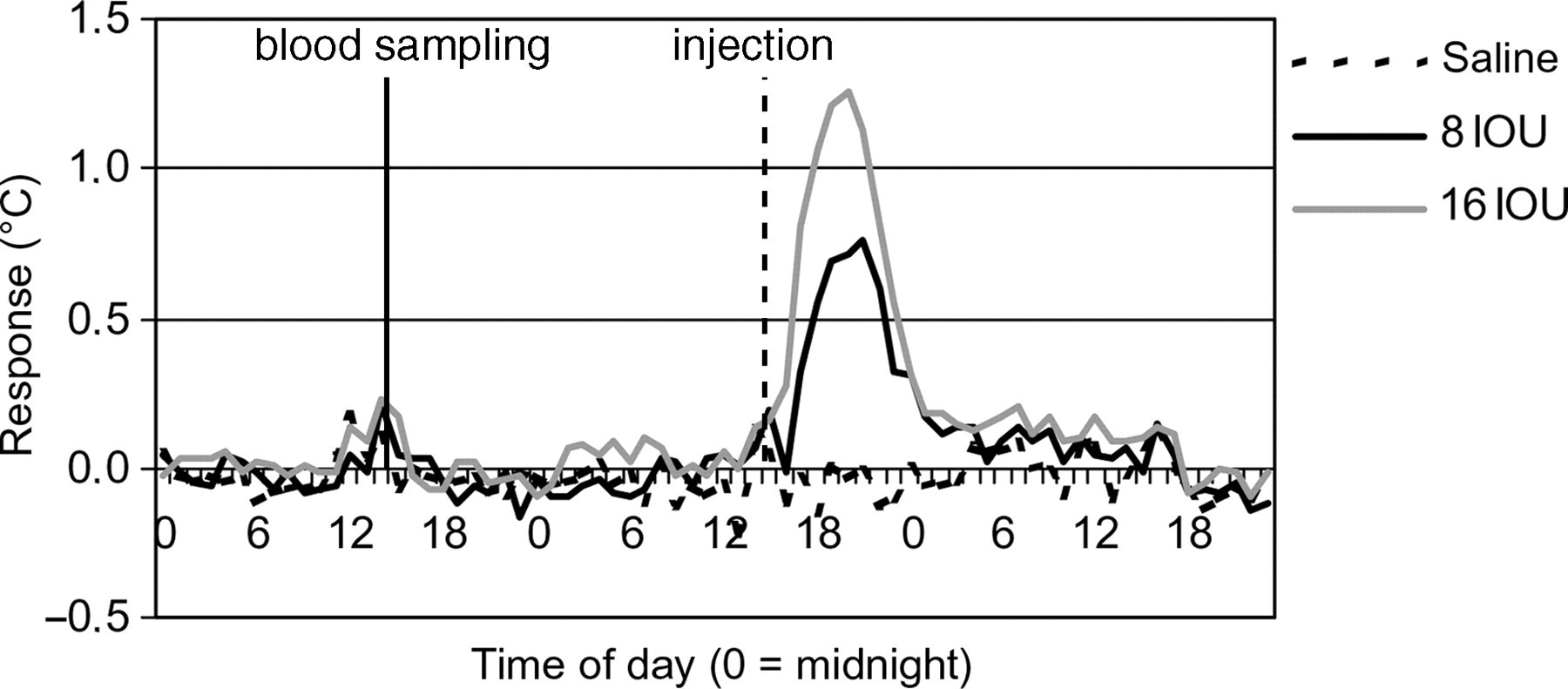
Effect of blood sampling and vaccine-related dose–response on body temperature under modified housing conditions for handled rabbits. The figure shows the changes in T b of handled rabbits towards blood sampling and vaccine dose-dependent response at the first immunization (means per dose group). Values for T b on the day of blood sampling and immunization were subtracted with values of the normal circadian body temperature
Effect of blood sampling on body temperature increase and duration of response under modified conditions compared with historic reference data from our laboratory
The effect of blood sampling on body temperature measured as mean ± SD (minimum/maximum) for maximum increase and duration of response (pooled data over the product groups as no significant interaction was observed with the product). n = number of animals, Control 1 = first control sample, Control 2 = second control sample, 1 = sample taken preceding first immunization, 2 = sample preceding second immunization, 3 = sample preceding third immunization
*Significant difference (P ≤ 0.05) in maximum increase over the experimental period between handled and non-handled rabbits
†Significant difference (P ≤ 0.05) in duration of increase over the period between handled and non-handled rabbits
‡Significant difference (P ≤ 0.05) in maximum increase over the experimental period within standard conditions
§Significant difference (P ≤ 0.05) in maximum increase between handled and non-handled rabbits
**Significant difference (P ≤ 0.05) in duration of increase between handled and non-handled rabbits
Effect of immunization with pWCV on body temperature
In the reference study, the animals were immunized at a time-point at which the T b was still elevated in response to blood sampling (Figure 3). Although the T bs of the animals increased after inoculation, no dose-dependent responses were observed (Table 3). Figure 3 and Table 3 are constructed using historic reference data from an earlier study performed at our laboratory for comparison with data obtained in the present study using the modified conditions.
The inoculation response on body temperature in the reference study
The inoculation response on body temperature in the reference study measured as mean ± SD (minimum/maximum) for maximum increase and duration of response. n = number of animals
*Significant difference between the saline-treated and pWCV-treated animals. No distinction between products could be made based on maximum increase and duration in response
Under the modified conditions, sampling the animals the day preceding immunization did not interfere with the vaccine-related responses. From Figures 4 and 5 it can be concluded that clear inoculation responses appeared after immunization. Table 4 shows the increase of body temperature as well as the duration of the response for the different immunizations. From Table 4 it can be concluded that handled animals showed a vaccine-related dose–response after all immunizations, whereas the non-handled animals did not. Rather, the non-handled animals showed an inverted dose–response relationship.
The inoculation response on body temperature under modified conditions
The inoculation response on body temperature under modified housing conditions measured as mean ± SD (minimum/maximum) for maximum increase and duration of response. n = number of animals
*Significant difference (P ≤ 0.05) between the saline-treated and pWCV-treated animals independent of handling
†Significant difference (P ≤ 0.05) between the handled eight and 16 IOU-treated animals
→Significant interaction (P ≤ 0.05) of immunization × handling × product in which the duration in the handled animals increases over the period, while over the period the duration decreases for the non-handled animals
Effect of routine procedures on body temperature under modified housing conditions
To assess the effect of routine procedures on body temperature, the time of day at which a procedure started and ended was denoted, and body temperatures analysed. Table 5 shows the data for the different procedures. Only the maximum increase in T b was analysed, as the duration in response largely depends on the duration of the procedures themselves. Handling only had a significant effect (P ≤ 0.01) on weighing: the non-handled animals showed lower increases in T b than the handled animals. The handling treatment itself, i.e. stroking the animals on the handler's lap, increased the T b with a small increase of 0.1–0.2°C (data not shown).
Effect of routine procedures on body temperature under modified housing conditions
The effect of accompanying procedures on body temperature under modified housing conditions measured as mean ± SD (minimum/maximum) for maximum increase in response
*Significant difference (P ≤ 0.05) in the response on weighing between the handled and non-handled animals
Body temperature of rabbits at the end of the experiment
After all the experimental procedures had been performed, the T b of rabbits was followed for another week to assess a final baseline. In Figure 6a, the daily means of T b of non-handled and handled animals are shown. Compared with the normal body temperature before experimental procedures (Figure 2a), the body temperature range of the non-handled animals were lower than before. Again handling had no effect on the variance in T b (Figure 6b).
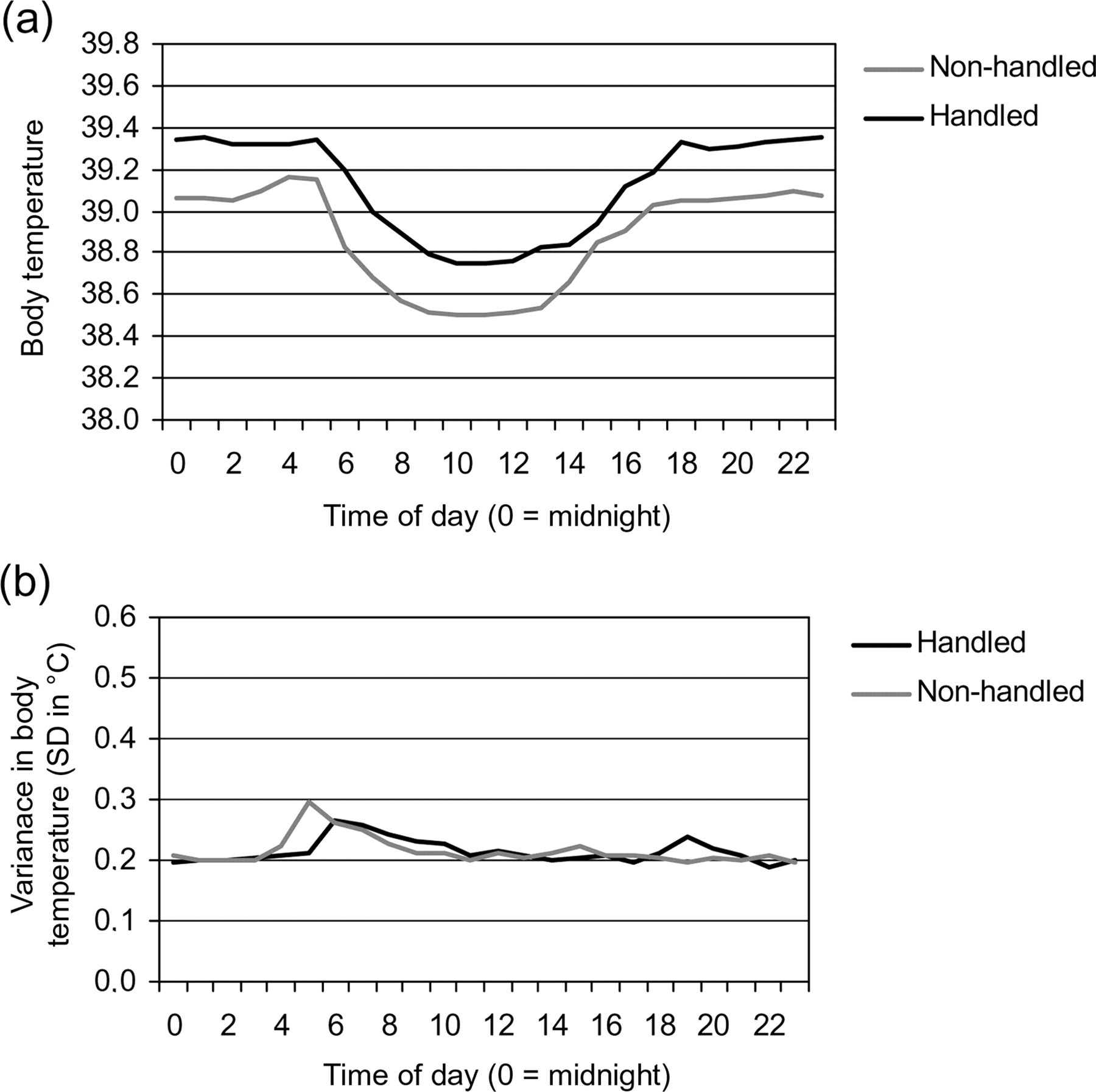
(a) Normal circadian body temperature of rabbits at the end of the experimental period under modified conditions. The figure shows the daily mean T b of rabbits per experimental condition. Values per time of day are means of pooled data of seven days after the last experimental procedure had been performed under modified conditions. (b) Variance in the normal circadian body temperature of rabbits at the end of the experimental period under modified conditions. The figure shows the variance in the daily mean T b of rabbits per experimental condition. Values per time of day are means of the variance of pooled data of days on which no experimental procedures or cage-cleaning were performed
Effect of handling on antibody titre
From Figure 7 it can be concluded that overall handled animals have higher antibody titres than non-handled animals. From Figure 7 it may also be concluded that handled animals tend to show a dose-dependent increase in antibody titre, whereas non-handled animals do not.
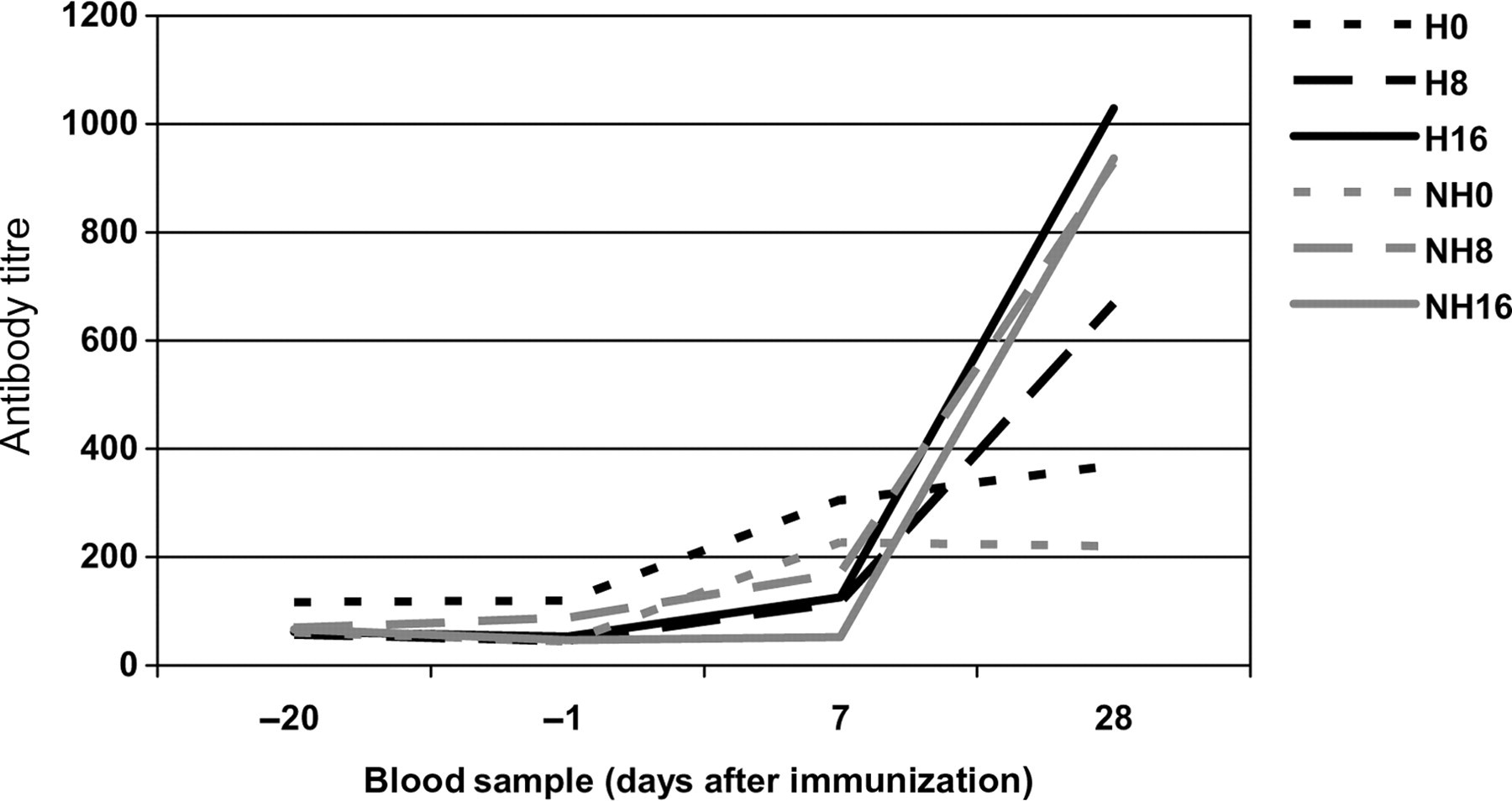
Effect of immunization with pWCV on antibody titre under modified housing conditions. H = handled rabbits; NH = non-handled rabbits
Effect of handling on body weight
As handling from birth onwards had been performed through weighing, the growth curve of the animals could be monitored closely. In order to investigate whether this frequent handling had an effect on growth, non-handled animals were weighed once a week. Pre-weaning handling had no effect on body weight at any time-point of the experiment (manuscript in preparation), except one week after the third immunization. At this time-point non-handled rabbits lost weight, whereas the handled animals gained weight substantially (P ≤ 0.0001; Figure 8). Within the handled and non-handled animals no significant vaccine-related effects or pen effects were observed (data not shown).
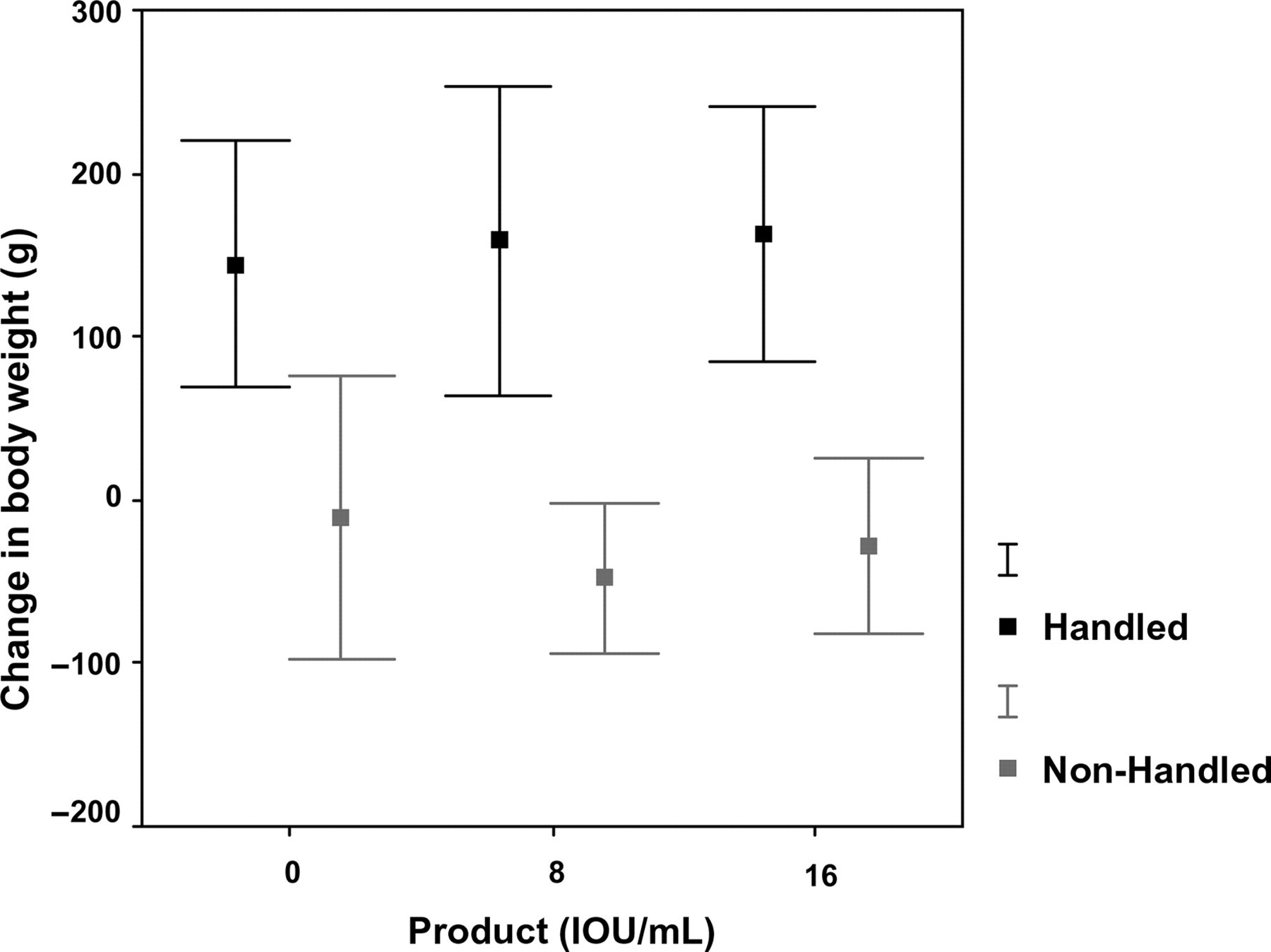
Change in body weight after the third immunization. The figure shows the change in body weight presented as mean ± 2 SD per experimental group. Independent of the product, non-handled rabbits grow significantly (P ≤ 0.0001) less than the handled animals
Discussion and conclusions
In the earlier vaccine safety studies of pWCV performed at NVI, the results were difficult to interpret due to the large variation in experimental outcome, especially in the key parameter deep-body temperature. Stressful standard laboratory conditions, as well as stressful experimental procedures, were identified as being the main reasons for this large variation. Stress-reducing measures, such as modified housing conditions and handling from birth onwards, have been implemented in the model in order to reduce variation in experimental outcome and to improve animal welfare.
The key parameter in this study, deep-body temperature, normally exhibits a diurnal pattern in which the light:dark cycle is mostly used as the external Zeitgeber. 13–15 In the reference study, under a 12:12 h light:dark cycle no circadian rhythm in body temperature was observed. As rabbits are crepuscular or nocturnal their rhythm in activity and rest, and thereby also the body temperature rhythm, is predominantly determined by dusk and dawn. 13 Other factors that influence the diurnal rhythm in body temperature are feeding and light intensity. 14,16,17 By introducing dusk and dawn periods, we have created a more natural situation that allowed the animals to prepare and respond to the changing light:dark situation. The periods of rest and activity have been taken into consideration by performing experimental procedures, cage-cleaning, feeding and refreshing of water only in the afternoon, a number of hours before dusk sets in, when the rabbits become active. For the reason that ad libitum feeding of animals can diminish the diurnal rhythm of physiological and behavioural processes, the rabbits were fed restrictedly. 16 The light intensity was lowered to approximately 60 lux, which is the maximum light intensity for albino animals. These modifications in housing conditions have resulted in a circadian body temperature rhythm with the characteristic sinusoidal waveform of deep-body temperature of homoeothermic animals.
Next to changes in the circadian rhythm of body temperature, we also observed that the temperature range of rabbits under modified conditions was lowered, i.e. between 38.6°C and 39.4°C. This range is more comparable with other homoeothermic animals that maintain their deep-body temperatures within relatively narrow limits (Mount 1979 in Van der Hel et al. 8 ). This narrow range is probably due to the lowered environmental temperature (from 21°C to 19°C), as it is also known that environmental temperature can have an effect on body temperature. 18,19 The daily constant body temperature of 39.5°C of the rabbits in the reference study seems to be in accordance with findings by Kuhnen 20 in which golden hamsters housed under chronic stress conditions (small barren cages), showed higher baseline body temperatures than hamsters housed in large enriched cages, i.e. under more stress-free conditions. This may thus suggest that the housing conditions of the reference study were stressful and that the modifications in the housing conditions are indeed stress reducing and, therefore, refinements for the model.
The modifications in the housing conditions allowed for determining the basal diurnal body temperature for each individual rabbit. This allowed discrimination of effects of routine and experimental procedures on this parameter at any given time-point. In this study, we were mainly interested in the effect of blood sampling, as in the reference study blood sampling preceding the immunization increased the body temperature to such an extent and duration that it interfered with and exceeded the inoculation response. Although under the modified conditions blood sampling was performed the day preceding immunization, to overcome interference with the inoculation response, this would not have been necessary per se as blood sampling had only minor effects, i.e. within the normal range of variance of body temperature. Routine procedures, such as weighing, marking and cage-cleaning, also had little effect on body temperature, i.e. the procedure-induced changes fell within the normal range of variance. The common denominator for handled and non-handled animals is conditioning to persons entering the animal room. So, it seems that conditioning indeed prepared the animals for possible events that were upcoming, thereby lowering the initial ‘negative acute stress’ response. 21 Compartmentalizing the pen for cage-cleaning and catching the animals out of the pen have probably also contributed to the small effects of these procedures on body temperature. This is in contrast to the significant increase in core body temperature in hamsters while their cage was being cleaned. 22 According to the animal caretakers, the rabbits in the present study seemed less stressed and were less disturbed, as it was easier to catch the rabbits out of the pen and cage-cleaning was performed without having to move the rabbits to another area, which is a very disturbing experience for rodents (various authors in Reinhardt and Reinhardt 17 ).
Next to the refinements accomplished by the modified housing conditions and conditioning on upcoming events, handling had some additional effects leading to further refinement and a more robust model. Handling had positive effects on the blood sampling procedure as measured by a smaller effect and a shorter duration of the effect on body temperature compared with the effects in the non-handled animals. Handled animals also reacted less strongly to the routine procedures, such as cage-cleaning, except for the effect of weighing, which was significantly lower for the non-handled rabbits than for the handled ones. This may be due to the fact that non-handled rabbits froze during this procedure and literally lowered their body temperatures. Handled rabbits, on the other hand, became more active as they started to explore the surroundings in accordance with the results from handled rabbits in an open-field and approach test 10,23 with a concomitant increase in body temperature.
Inoculation responses in body temperature became apparent for the different units of pWCV in both handled and non-handled animals. Although in the handled animals the response to the 16 IOU pWCV appeared to be stronger than that to the 8 IOU pWCV, the opposite was true for the non-handled rabbits. The underlying reason for the reversed dose–response relationship may be chronic stress. In situations of chronic stress, the continuous release of corticosteroids by the adrenal gland causes a depression of the immune system.
24
This may also be the explanation for the fact that non-handled rabbits show down-regulation of their antibody titre at the end of the experiment. In future studies, cortisol will be measured to verify this. The lowered body temperature of non-handled rabbits at the end of the experiment may well be the result of prolonged stress, as studies of rabbits indicate that several changes may take place under prolonged stress including a drop in body temperature (
Our animals were handled by weighing. Some researchers advise keeping handling within limits during the post-arrival period, as too much or too little handling may have adverse effects on the animal's body weight 26,27 (Lawlor et al. 1975 referred in Rowan 28 ). In this study handling had no influence on body weight, except after the third immunization: handled rabbits gained weight, whereas non-handled rabbits lost weight. As no differences in body weight gain were found earlier in the experiment, we hypothesized that this difference might be due to chronic stress in the non-handled animals, as it is known that chronic stress causes an increase in the basal metabolic rate leading to weight loss. 29
In summary, the modifications in housing condition and conditioning to humans have resulted in normal circadian body temperature profiles and little effects of experimental procedures on body temperature. In addition, handling tends to improve the pWCV-induced body temperature profiles, which makes it possible to differentiate body temperature responses between vaccine doses used. Handling leads to more robust dose–response curves in body temperature and in antibody titre induced by vaccines. Overall, handling decreases variance in the investigated parameters and has a positive effect on the behaviour of the animals, which makes them easier to work with. It can be concluded that the modifications provide a more robust model for monitoring pWCV adverse effects. Furthermore, conditioning and habituation of rabbits to humans reduce the variation in experimental outcome, which might allow for a reduction in the number of animals used. In addition, they also reduce distress and thus contribute to refining animal models.
