Abstract
New Zealand White (NZW) rabbits (n = 34) received intravenous propofol (16 ± 5 mg/kg) for induction of anaesthesia followed by maintenance with sevoflurane (4.0 ± 0.5%) in oxygen. All animals underwent ovariohysterectomy. Heart rate, respiratory rate, haemoglobin oxygen saturation, end-tidal carbon dioxide concentration, end-tidal sevoflurane concentration and oesophageal temperature were monitored every 5 min. Time from induction of anaesthesia to tracheal extubation and sternal recumbency were recorded as was the quality of recovery. Direct arterial blood pressure values (mmHg) were recorded every 5 min from 19 rabbits and 22 arterial blood gases analyses were performed (11 postintubation and 11 at the time of recovery). Propofol produced smooth induction of anaesthesia without production of apnoea. Intubation was successfully performed in all but one rabbit in an average of 4 ± 3 min from the beginning of propofol administration. No ventilatory support was required during the anaesthetic period. Respiratory rate averaged 51 ± 8 bpm and end-tidal CO2 (kPa) was 4.0 ± 0.5 mmHg during anaesthesia. Blood gas values were maintained within normal limits and average mean arterial blood pressure was 73.4 ± 7.9 mmHg. Time to regain the swallowing reflex following discontinuation of sevoflurane was 2 ± 1 min and time to sternal recumbency was 8 ± 0.3 min. No anaesthetic-related mortality occurred and all animals recovered uneventfully. Propofol–sevoflurane anaesthesia produced a good quality of surgical anaesthesia for ovariohysterectomy and stable cardiopulmonary conditions. Propofol–sevoflurane anaesthesia in young healthy NZW rabbits appears to be an effective and practically useful method of anaesthesia.
Rabbits are widely used for research and have also gained popularity as companion animals. However, providing safe and effective induction of anaesthesia and a rapid, smooth recovery from anaesthesia remains a challenge. Inhaled anaesthetic agents have historically been the anaesthetic of choice for small mammals, but their use for induction of anaesthesia in rabbits is associated with complications. Rabbits hold their breath when exposed to even very low concentrations of volatile agents and this is accompanied by marked bradycardia, hypercapnia and hypoxia. 1–3 Unfortunately, these responses are not prevented by some commonly used sedatives or tranquillizers, although animals remain outwardly calm and do not struggle. 4
A study of anaesthetic mortality in veterinary practice by Brodbelt et al. 5 highlighted the particularly high risk of death when anaesthetizing rabbits compared with dogs and cats, with the postoperative period being associated with the highest incidence of mortality. This study reported that, in all species, 50% of anaesthetic-related deaths occurred within the first 3 h following termination of anaesthesia. Similar data for laboratory rabbits are not available, but it seems reasonable to suggest that anaesthetic regimens associated with a rapid recovery might facilitate a significant reduction in mortality.
The intravenous anaesthetic agent propofol demonstrates many desirable qualities in human beings, dogs, cats and other species, both as an induction agent and for maintenance of anaesthesia by incremental dosing or continuous infusions. Its rapid onset of action, short anaesthetic duration, smooth recovery and minimal accumulation after repeated doses has made it a popular agent. While long-term propofol anaesthesia in rabbits seems to be contraindicated because of profound cardiopulmonary effects, prolonged recoveries and increased incidence of death, 6 it may be suitable for use as an induction agent in this species. Data by Glen 7 suggested that rabbits tolerate propofol well despite requiring a higher dose for induction than other domestic species. However, they also reported that use of propofol alone to produce surgical anaesthesia resulted in respiratory arrest in some animals and so inhalant agents may be required for maintenance of anaesthesia.
Sevoflurane is a volatile, non-flammable anaesthetic with little or no pungent odour. Induction and recovery are rapid in comparison to many other volatile anesthetics. 8 The minimum alveolar concentration of sevoflurane in response to a noxious stimulus (tail clamp) has been reported to be 3.7% in rabbits. 9 The purpose of this study was to evaluate anaesthesia for ovariohysterectomy, with induction of anaesthesia with propofol, and maintenance with sevoflurane. The quality of anaesthetic induction and recovery from a surgical plane of anaesthesia was evaluated, and selected cardiopulmonary effects were recorded.
Animals
Thirty-four healthy, 12-week-old, female, New Zealand White (NZW) rabbits (Oryctolagus cuniculi) (Hsdlf:NZW, supplied by Harlan UK Limited, Bicester, UK), weighing between 1.8 and 2.6 kg were studied. They were individually housed in adjacent floor pens (1 m × 1 m) on autoclaved pine shavings and sawdust (Lillico Biotechnology, Blechworth, UK). As enrichment to their habitat each rabbit received a cardboard tube, pine rabbit chew block and a litter tray. Food pellets (Rabma pellets, SDS, Whitham, Essex, UK) and water were provided ad libitum and not withdrawn before surgery. The animals were free from any common pathogens in accordance with the Federation of European Laboratory Animal Science Association Health Monitoring Recommendations. 10 The rabbits were acclimatized for a period of 14 days prior to the start of the study. This study was approved by the local Ethical Review Committee and the UK Home Office.
The rabbits were part of a study aimed at developing a pain assessment scheme for rabbits 11 following routine ovariohysterectomy, so animals received either the placebo (vehicle) (n − 8) or one of three doses of meloxicam one hour prior to surgery. Individuals that exhibited two or more of the following signs (abdominal writhing, belly pressing, back-arching, sustained contraction of the abdominal muscle) postoperatively, within a 5 min period, received intravenous buprenorphine (0.01 mg/kg) as rescue analgesia. Additionally, all rabbits received one dose of enrofloxacin (10 mg/kg subcutaneously, Baytril; Bayer plc, Newbury, Berkshire, UK) preoperatively.
Materials and methods
Prior to and during induction of anaesthesia, oxygen was administered by face mask at 3–4 L/min for 5 min. To facilitate auricular intravenous and arterial catheter placement, lidocaine and prilocaine (EMLA cream, EMLA, Astra Pharmaceuticals Ltd, King Langley, UK) was applied topically on a clipped area 30 min before catheterization. Immediately prior to anaesthetic induction, a 22 SWG, 1″; 0.9 × 25 mm over the needle catheter was placed percutaneously into a marginal ear vein and a T-connection was attached to it. In 19 out of 34 rabbits, a 22 SWG 25 mm over the needle catheter was inserted percutaneously into the central auricular artery.
General anaesthesia was then induced by intravenous injection of propofol (Propofol-®Lipuro 1%, Braun; B Braun Melsungen AG, Melsungen, Germany) via the preplaced venous catheter. The initial induction dose of 10 mg/kg was given by hand, over 60 s. Additional incremental doses (1–2 mg/kg) were given until sufficient relaxation occurred to allow intubation, assessed by noting the overall degree of relaxation or lack of chewing responses when the jaws were gently moved. When the animal appeared sufficiently anaesthetized, a 3 mm (ID) uncuffed endotracheal tube was placed using a capnograph-guided ‘blind technique’. 12 Endotracheal intubation was performed by one person, who was experienced in the technique. Ease of endotracheal intubation and quality of induction were recorded: excellent intubation (<2 attempts, time <2 min), good (<4 attempts, time <5 min), poor (>4 attempts, time >5 min). The main criteria for ease of intubation were how many attempts were necessary and if the intubation technique had to be modified to intubation via direct vision using an otoscope.
After induction of anaesthesia and endotracheal intubation, anaesthesia was maintained with sevoflurane (SevoFlo® Abbott Laboratories Ltd, Kent, UK) in oxygen (oxygen flow rate 150 mL/kg/min) using a non-rebreathing system (Bain circuit). The arterial catheter (placed in 19 rabbits) was connected, via a saline-filled high pressure tubing to an electrical pressure transducer positioned at the level of the right atrium (external landmark: humeroscapular joint). Heart rate, respiration rate, haemoglobin oxygen saturation, end-tidal carbon dioxide and end-tidal sevoflurane concentration (E'CO2, E'sevo; side-stream sampling at the endotracheal tube connector port, sample rate 100 mL/min [Kontron Colormon Plus; Charter Kontron, Milton Keynes, Bucks, UK]), arterial blood pressure (invasive) and core body temperature were measured every 5 min after anaesthesia induction. Haemoglobin oxygen saturation was measured continuously via a pulse oximeter (Nonin® 8600V, Nonin Medical, Inc, Plymouth, MN, USA) with a clip-on probe placed at the base of the ear. Arterial blood samples (approximately 0.5 mL) from 11 rabbits were collected into heparinized syringes for immediate blood gas analysis (PICO 50, Radiometer Medical ApS, Copenhagen, Denmark). The first sample was taken immediately after endotracheal intubation (endotracheally intubated with an FiO2 of 0.96) and the second sample in recovery (vapourizer turned off, endotracheally intubated and with an FiO2 of 0.96).
Ovariohysterectomy was performed by the same research worker via a ventral midline incision. Anaesthetic gas was turned off after the last intradermal sutures were placed. Oxygen (FiO2 of 0.96) was administered until the swallowing reflex returned and the rabbit's trachea was then extubated. Quality of recovery from anaesthesia was recorded as well as time from disconnection of sevoflurane to extubation and sternal recumbency. Recovery was defined as excellent (no excitement, calm), good (some excitement, but calm) and poor (overexcitement, struggling).
Once in sternal recumbency the animal was placed in an incubator at 30°C for approximately 30 min and then returned to its holding pen. Close observation was continued for the next 48 h, and these data have been reported separately. 11
Results
All results are expressed as mean ± 1 SD. Animals lost consciousness with the initial induction dose of propofol, but all required incremental doses to allow intubation (mean total dose 16 ± 5 mg/kg of propofol). Most rabbits maintained a reactive airway (e.g. coughing and bucking on the endotracheal tube). No animals became apnoeic during induction of anaesthesia and intubation. Quality of endotracheal intubation was considered excellent in 16 rabbits, good in 15 rabbits and poor in three rabbits. Reasons for three poor endotracheal intubations were because one rabbit could not be endotracheally intubated and was maintained on a face mask and two rabbits had to be endotracheally intubated under direct vision using an otoscope and introducer. Time to successful intubation (in 33 rabbits) was 4 ± 3 min from the start of propofol administration. No ventilatory support was required during the anaesthetic period as all animals maintained end-tidal CO2 within the normal range (3.8–5.3 kPa). Respiratory rate averaged 51 ± 8 bpm (n = 34, Figure 1) and end-tidal CO2 (kPa) was 4.0 ± 0.5 mmHg (n = 33) during anaesthesia. An E'sevo of 4 ± 0.5% (n = 33) was required to prevent movement responses during ovariohysterectomy. Blood pressure in 19 rabbits instrumented with an auricular arterial catheter showed that most of the rabbits maintained mean arterial blood pressure >70 mmHg (mean 73 ± 8 mmHg, Figure 2) throughout surgery. Hypotension occurred in one rabbit after induction (mean arterial pressure of 56 mmHg) but improved after decreasing the sevoflurane concentration and administering 10 mL/kg of crystalloid fluids intravenously. No inotropic support was required. Heart rate remained relatively stable throughout the period of anaesthesia, with a mean rate of 290 ± 24 (Figure 1). All but one rabbit remained well oxygenated while in dorsal recumbency (Table 1). This animal had a PaO2 of 11.45 kPa and a PaCO2 of 5.54 kPa after induction, and PaO2 13.92 kPa and PaCO2 of 4.87 kPa in recovery.
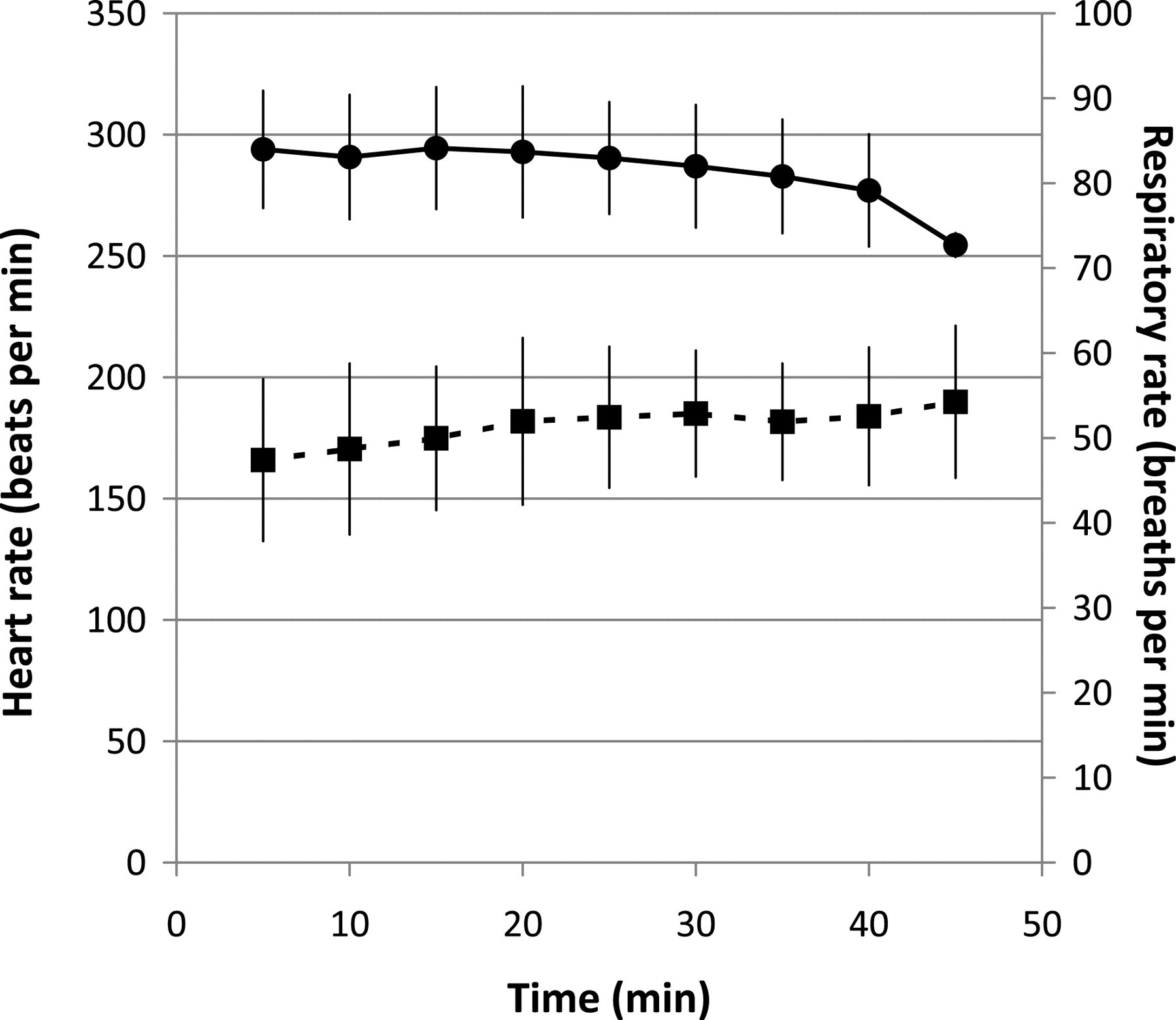
Heart rate (solid circles) and respiratory rate (solid squares) in 34 rabbits anaesthetized with propofol and sevoflurane and undergoing ovariohysterectomy. Values are mean ± 1SD
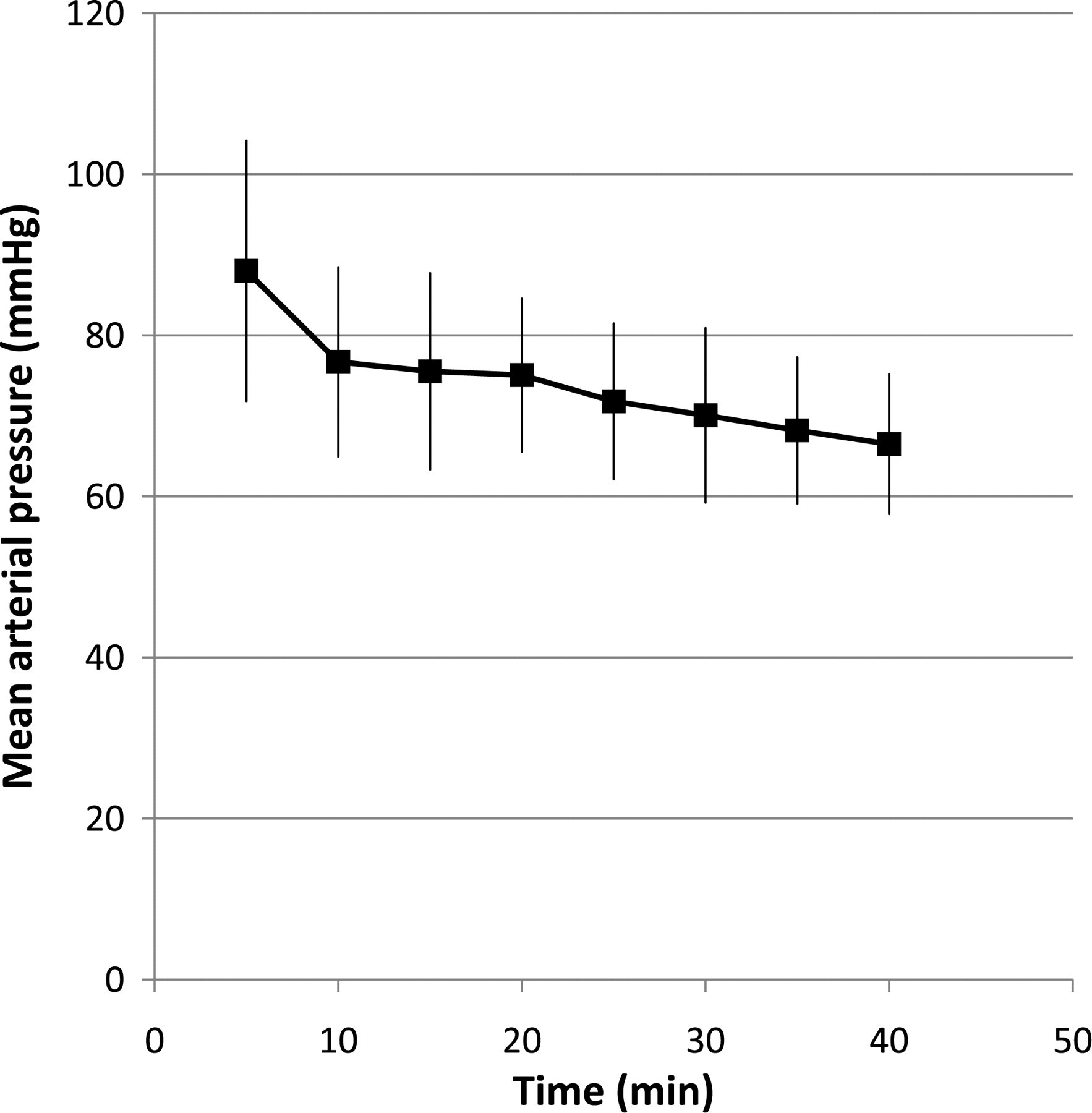
Mean arterial blood pressure recorded via the auricular artery in 19 rabbits anaesthetized with propofol and sevoflurane and undergoing ovariohysterectomy. Values are mean ± 1SD
Arterial blood gas values of 11 rabbits after induction of anaesthesia and at recovery (mean ± SD)
Anaesthesia duration recorded for each animal was less than 30 min. Body temperature was well maintained (38.77 ± 0.3°C 5 min after induction of anaesthesia, 38.44 ± 0.4°C at the end of anaesthesia, Figure 3). Recovery was classed as excellent in all rabbits. The animals could be extubated at 2.0 ± 1 (n = 34) min following termination of sevoflurane, when chewing returned or coughing was noted. Rabbits regained their righting reflexes at 8.0 ± 0.3 min (n = 34) and remained quietly in sternal recumbency unless manually stimulated. No rabbits required rescue analgesia during the subsequent 48 h.
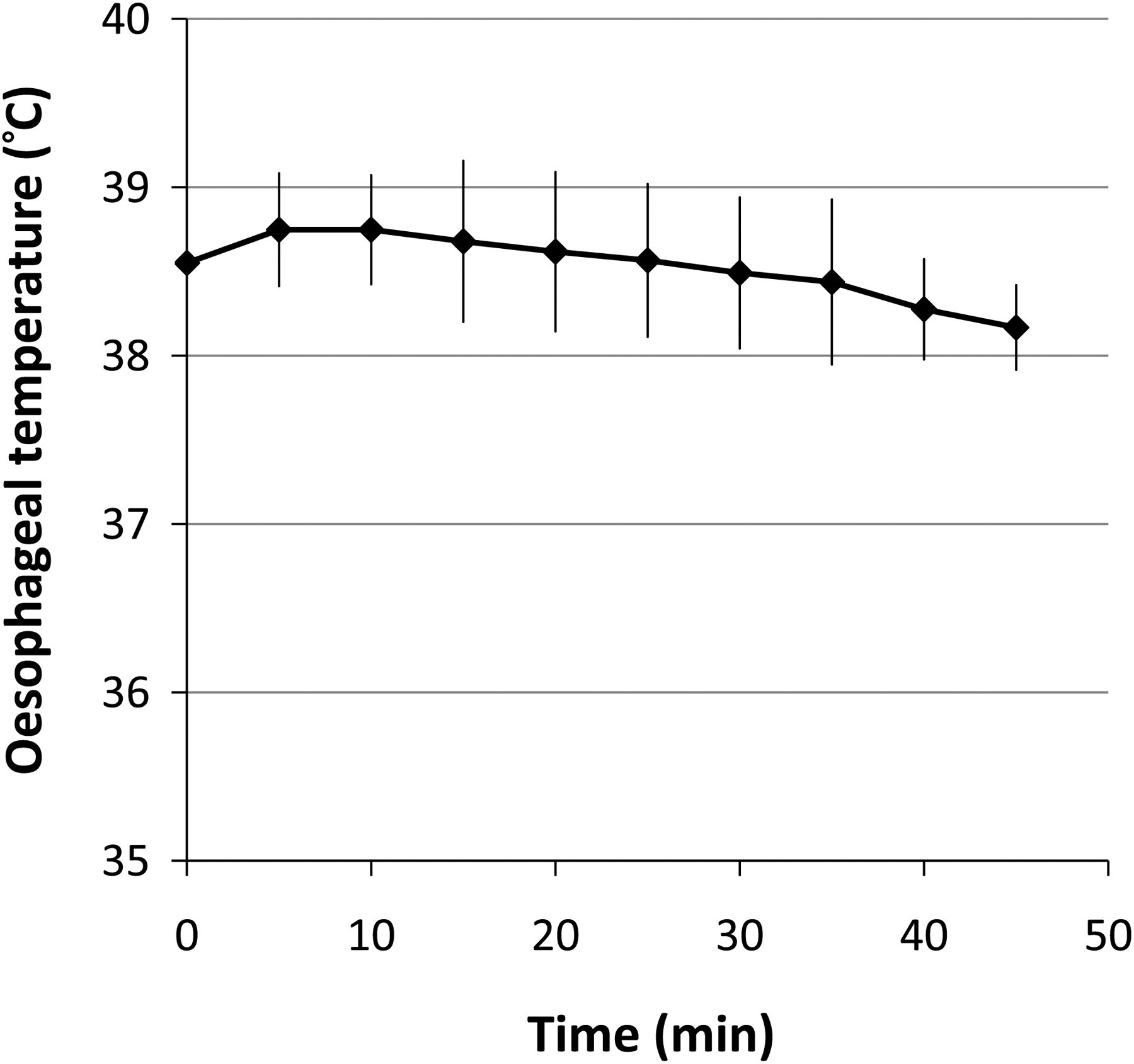
Oesophageal temperature in 34 rabbits anaesthetized with propofol and sevoflurane and undergoing ovariohysterectomy. Values are mean ± 1SD
Discussion
This study indicates that induction of anaesthesia with propofol followed by maintenance with sevoflurane is a suitable technique for use in healthy rabbits undergoing ovariohysterectomy. The propofol dose used is higher than that reported previously for induction of anaesthesia in NZW rabbits 13 and close to the dose reported to produce apnoea in rabbits 7 (20 mg/kg). This may be due to differences in the strain or age of animals used, or may be related to the speed of administration of propofol. Earlier studies with propofol, administered the agent at 0.25–0.5 mg/kg/s, 7 or 0.33 mg/kg/min 13 compared with 0.17 mg/kg/s in the present study. In other species, including people, rapid administration of propofol frequently results in apnoea, whereas slower infusion avoids this. 14,15 Although the slower administration rate may have reduced the incidence of apnoea, it is not likely to have significantly altered the dose of propofol required. 14,15 A more accurate rate of administration could have been achieved by using an infusion pump to deliver the required dose. The age, strain, sex and weights of the animals used by Glen 7 were not specified. The animals in the Aeschbacher and Webb study 6,13 were NZW rabbits of both sexes, with body weights ranging from 1.76 to 6.32 kg. Since this strain frequently becomes markedly obese when adult, this may account for the difference in induction dose required.
The most striking feature of this anaesthetic regimen was the very rapid and smooth recovery. In contrast, recovery times for ketamine/medetomidine have been reported between 86 and 120 min. 16 If the effects of medetomidine were reversed by using the specific antagonist (atipamezole), righting reflex returned within 5 min, but sedation lasted for over an hour (Hedenqvist, personal communication). More rapid recovery was noted in rabbits receiving a lower dose of ketamine/medetomidine, or ketamine/midazolam, followed by maintenance with isoflurane (2–43 min and 2–65 min to head lift, respectively 17 ) but this is still considerably slower than that seen with the present regimen. Induction doses of alfaxalone followed by isoflurane were also associated with slower recovery times than propofol/sevoflurane (22.5 min to sternal 18 ). Fentanyl/fluanisone/midazolam produces prolonged recoveries from anaesthesia (duration of sedation for 120 min) and respiratory depression (over 90 min after administration of the compounds intramuscularly). 19 Comparisons between these different studies must be made with caution, since as discussed earlier, the strain, age, sex and health and nutritional status of the animals studied could all influence their response to anaesthesia.
The more rapid recovery seen in the present study may also have been influenced by the use of sevoflurane rather than isoflurane for maintenance. Although this has not been noted to have a significant effect on recovery time in dogs and cats, it may have advantages in rabbits, and a direct comparison of the two agents would be of interest.
Respiratory depression and apnoea are the most common complications encountered with propofol in dogs and cats. 20 Glen 7 found similar depression of ventilation with a propofol dose of 20 mg/kg proving lethal in rabbits. While we did not observe respiratory depression, as previously reported, we would recommend tracheal intubation. The advantage of having an endotracheal tube in place in case of respiratory failure cannot be overestimated. Oxygenation was well maintained in all but one rabbit, and it is not clear why this animal's oxygenation was compromised, but physiological shunt during general anaesthesia in dorsal recumbency could be one reason. Although endotracheal intubation is recommended, it is important to provide oxygen for 1–2 min prior to attempts to pass the endotracheal tube, to avoid periods of hypoxia if intubation takes longer than anticipated. For example in the present study intubation took up to 5 min in three animals. The use of local anaesthetic could have prevented the laryngeal reactions seen during endotracheal intubation. 12 It should be noted that propofol and sevoflurane have no specific analgesic effects, so analgesia should be provided, preferably preoperatively, and adopting a multimodal approach (e.g. with meloxicam and buprenorphine). If an opioid analgesic is given preoperatively, then the dose of propofol for induction of anaesthesia, and the concentration of sevoflurane required for maintenance, may need to be reduced.
The rabbits anaesthetized in this study were part of a separate investigation aimed at developing a pain assessment scheme for rabbits, and to evaluate the use of meloxicam as an analgesic. Since there are no pain scoring schemes for rabbits, and the only information on pain behaviour is anecdotal, a placebo control group was included, as has been done in other species. 21 To reduce the likelihood of animals experiencing more than mild–moderate pain, a veterinarian, blinded to the treatments, observed the animals, and was instructed to withdraw any animal from the study he/she believed to be in more than mild pain, and administer buprenorphine as rescue analgesia. No animals were withdrawn from the study. Full results of this have been published elsewhere, 11 but in summary, although detailed behavioural analysis demonstrated behavioural changes, these were not detected on routine clinical observation. This confirmed our current poor ability to detect pain in this species, and reinforces the need for placebo-controlled, blinded studies to assess analgesic efficacy.
The main advantage of propofol over other hypnotic agents, in addition to its rapid metabolism, is the fact that no excitation is seen either during induction or during recovery in humans. 22 Our study confirmed these findings in rabbits and demonstrates a high degree of predictability in achieving a rapid induction of anaesthesia and adequate depth for intubation, together with a low incidence of untoward side-effects in the immediate postoperative period and a zero mortality. Further studies are necessary to determine the safety and effectiveness of this anaesthetic protocol in different strains, ages and weights of rabbits. However, it seems likely that the rapid recovery, associated with rapid return to normal activity will not only reduce the risk of complications in the postoperative period but will represent a useful refinement of rabbit anaesthesia.
Footnotes
Acknowledgements
The authors thank Ms Caroline Fox, Denise Reid and Sandra Hogg for assistance with animal care and the anaesthetic and surgical procedures, and Dr Khursheed Mama, Colorado State University, for assistance with preparation of the manuscript. Dr S Allweiler was supported by a grant from Laboratory Animals Ltd.
