Abstract
Analysis of gene expression is often used to evaluate the effects of experimental manipulations in laboratory animals. Blood is a rich source of potential biomarkers, including gene expression information, which may be obtained from whole blood. When compared with the end of a study, when whole blood samples can be easily obtained for gene expression measurements, the limiting volumes of whole blood obtainable from animals during the course of an experiment requires a method for RNA isolation from a minimal volume of whole blood. The PAXgene™ Blood RNA Extraction System originally designed for isolation of total RNA from 2.5 mL of human whole blood, was modified and successfully used to isolate high-integrity total RNA from as little as 50 μL of mouse whole blood. Fifty microlitres of mouse whole blood yielded an average of 2.3 μg highly intact total RNA, of sufficient quality and quantity allowing for multiple gene expression determinations. The utility of this method was demonstrated by confirming the time- and dose-dependent upregulation of haem oxygenase-1 (Hmox1) mRNA in response to a single injection of cobalt protoporphyrin. The successful isolation of total RNA from small volumes of mouse whole blood can allow for serial sampling on the same animals, thereby reducing the number of animals required for experimentation.
Analysis of whole blood is often used to evaluate the effects of experimental manipulations or potential therapeutics. Generally, whole blood is readily available and can be collected from humans and/or preclinical species using standard venepuncture. Analytes such as glucose, cholesterol and other components can easily be measured in the plasma derived from whole blood, and complete blood counting can provide information on the cell types present. In addition to these rather standard measures, in some cases it may be desirable to examine the level of expression for genes of interest. When gene expression information is added to the standard measures, a more complete physiological picture can be obtained.
There are several technical options for gene expression measurement including, but not limited to northern blot, quantitative RT-PCR, microarray profiling and RNase protection. All methods require isolation of intact transcripts representative of the actual gene expression pattern within the tissue at the time of sample collection. However, isolation of such an mRNA sample from whole blood is often compromised by degradation of RNA and/or changes in gene expression, which can occur when harvesting cells prior to RNA isolation. 1 When studies involve small preclinical species such as hamsters, rats or mice, it is quite difficult to obtain sufficient whole blood from individuals to allow isolation of the amount of RNA required for such analyses. This difficulty is further exaggerated in studies in which serial, rather than terminal blood samples are obtained, such as in time-course studies. Therefore, a method which efficiently enables isolation of RNA in sufficient quality and quantity from microvolumes of whole blood without collection-induced perturbations to the transcriptome is highly desired.
The PAXgene™ Blood RNA Kit (Qiagen, Valencia, CA, USA) is designed for the isolation of total RNA from a 2.5 mL sample of human whole blood, which is previously collected and stored in PAXgene™ Blood RNA tubes. According to the manufacturer, the PAXgene™ tube contains a proprietary reagent that immediately stabilizes intracellular RNA for days at room temperature and weeks at 4°C. Given these advantages, whether or not this reagent could be used for the isolation of RNA from whole blood of laboratory animals is of great interest. By scaling down and slightly modifying the manufacturer's procedure, we routinely obtained surprisingly high yields of total RNA from very small volumes of mouse whole blood (e.g. typical yield of 2 μg RNA from 50 μL of whole blood). RNA quality was excellent, as judged by typically high RNA integrity numbers determined following microcapillary electrophoretic RNA separation. 2 Using this method for RNA isolation, the dynamic upregulation of haem oxygenase 1 (Hmox1) gene expression by cobalt protoporphyrin (CoPP) was easily observed by RT-PCR. The results demonstrate that the method allows isolation of robust and intact RNA yields from quite small volumes of whole blood.
Materials and methods
Isolation of total RNA from microvolumes of whole blood
Prior to blood collection, the PAXgene™ RNA stabilizer solution was removed from the BD Vacutainer™ style PAXgene™ Blood RNA tubes. Venous blood obtained from the retro-orbital sinus of C57BL/6 mice under anaesthesia was collected into heparinized tubes according to a protocol approved by the GlaxoSmithKline Institutional Animal Care and Use Committee (IACUC). When larger volumes (>50 μL) of whole blood were used, these were obtained by cardiac puncture under an IACUC-approved protocol. Increasing blood volumes from 0.05 mL to 2.5 mL were added to the RNA stabilizer reagent in 1.5 mL Eppendorf tubes (for volumes <500 μL) or in 12 × 75 Falcon tubes (for volumes ≥500 μL). In all cases, the volume ratio of RNA stabilizer reagent to blood sample was kept at 2.76 according to the manufacturer's original protocol. When processing these samples on a single column, we found that the total RNA yield per starting volume of whole blood was reduced for volumes greater than 0.4 mL. Therefore, multiple columns were required to maximize yield. The number of columns used was simply determined by dividing the total harvested whole blood volume by 0.4 mL. For example, the number of columns required to isolate total RNA from 2.0 mL of whole blood was 2.0 mL/0.4 mL or five columns. Tubes were gently inverted several times and incubated at room temperature for 2–24 h or stored at −20°C before RNA isolation. Frozen samples were removed from storage and brought to room temperature for 45 min. They were inverted several times and then centrifuged at 5000 ×
RNA analyses and quantitative RT-PCR
RNA samples were quantitated using the Quant-iT™ RiboGreen® RNA Assay Kit (Molecular Probes; Invitrogen Corporation, Carlsbad, CA, USA) and then analysed for overall integrity using the Agilent 2100 Bioanalyser according to Agilent's RNA 6000 Nano Assay Protocol. RNA samples and Ambion's RNA 6000 ladder Cat No. 7152 were heat-denatured at 70°C for 2 min and chilled immediately on ice. Samples and RNA ladder were pulse-centrifuged just prior to loading 1 μL of each into the chip's designated sample and ladder wells. The chip was placed into the adapter of the vortex mixer (IKA Works Vortexer, model MS2-S9) and vortexed for 1 min at 2400 rpm. The chip was then inserted into the Agilent 2100 Bioanalyser for electrophoretic separation and analysis of samples. Subsequently, 5 μL of each sample corresponding to 0.075–1.00 μg total RNA was reverse-transcribed to cDNA using the high-capacity cDNA Archive Kit (Applied Biosystems, Forster City, CA, USA) according to the manufacturer's instructions. Realtime PCR reactions were performed on the ABI Prism 7700 Sequence Detector using TaqMan® Fast Universal PCR Master Mix (Applied Biosystems) with either murine Hmox1 TaqMan® Gene Expression Assay (Applied Biosystems, part no. Mm00516004_m1) or Rodent GAPDH Control reagents (Applied Biosystems, part no. 4308313).
The expression of Hmox1 for each treatment group was calculated as follows: for each sample, Hmox1 expression relative to the GAPDH housekeeping gene was calculated (ΔCt). The average ΔCt for the day 0 samples was subtracted from the ΔCt values of samples at all other timepoints (ΔΔCt), and the fold change in Hmox1 expression relative to day 0 was calculated from these ΔΔCt values by the relative Ct method as specified by Applied Biosystems. 3 Significance was calculated using Student's t-test.
Treatment of mice with cobalt protoporphyrin
On day 0, groups of five mice received either 2 mg/kg or 20 mg/kg CoPP by subcutaneous injection. On days 3 and 7 post-CoPP administration, 50 μL blood was obtained from each mouse under anaesthesia by retro-orbital bleeding. On day 14 post-CoPP administration, blood was obtained by cardiac puncture and exsanguinations of animals. Fifty microlitres of blood was combined with 138 μL PAXgene™ RNA stabilizer solution, and RNA was isolated as described above.
Results
Although the original PAXgene™ method is intended for isolation of total RNA from 2.5 mL of human blood, the scaled-down method clearly yielded total RNA from mouse whole blood, even for volumes as low as 50–300 μL (Figure 1a). For small volumes of mouse whole blood (≤300 μL) RNA yield was proportional to the volume of mouse whole blood processed, however for larger blood volumes, RNA yields from a single PAXgene™ column were reduced. However, yield of total RNA from these larger blood volumes was increased when samples were processed on multiple columns (Figure 1b). From the slopes of the lines in Figure 1, it can be estimated that approximately 50 μg of total RNA is obtained per millilitre of whole mouse blood and that the yield is proportional over a large range of blood volumes. In contrast, reported yields (4–20 μg) from 2.5 mL of human whole blood are typically lower. 4,5 The results in Figure 1b suggest that the low yield of total RNA from 2.5 mL of human blood samples compared with the yields from mouse whole blood may be mainly due to reduction in yield owing to saturation of the PAXgene™ silica column when processing larger volumes.
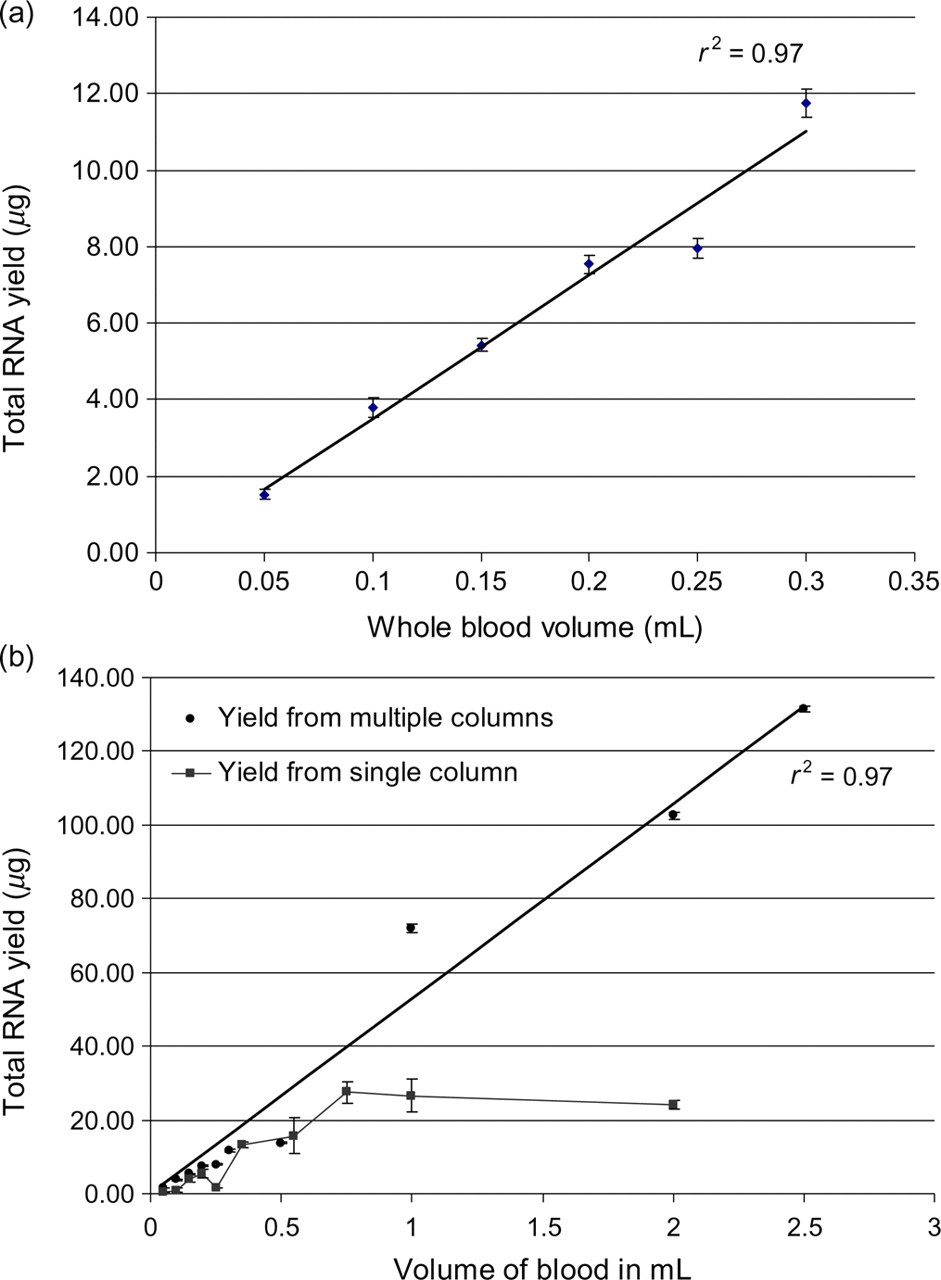
Total RNA yield per volume of mouse whole blood. (a) Linearity of total RNA yield from small volumes of mouse whole blood using a single PAXgene™ column per sample. Values represent the mean of four samples ± SEM. The indicated volumes of mouse whole blood were processed as described in Materials and Methods on a single PAXgene™ column. (b) Total RNA yield from larger volumes of mouse whole blood (>0.5 mL) can be increased by processing on multiple PAXgene™ columns. Values for the yield from multiple columns represent the mean total yield for two 1, 2 and 2.5 mL sample volumes each processed on five columns ± SEM. Values for the yield from single columns represent the mean of four samples ± SEM. The indicated volumes of mouse whole blood were processed as described in Materials and Methods on either a single (square symbols) or multiple (circles) PAXgene™ column
The total RNA isolated by the described microscale procedure is highly intact. The average 260/280 ratio was 2.00 ± 0.02 (n = 80). Figure 2 shows a microcapillary electropherogram of a typical 50 μL mouse whole blood total RNA sample. Characteristic 18S and 28S ribosomal RNAs are clearly evident. An objective and quantitative measure of RNA integrity is the RNA integrity number (RIN), which is calculated by the Agilent 2100 Bioanalyser software from the overall electropherogram, taking into account features such as 18S/28S ratio and indicators of RNA degradation such as presence of RNA migrating faster than 18S and/or between the 18S and 28S peaks. Fully intact RNA is assigned an RIN of 10 and highly degraded RNA an RIN of 1. 2 The average RIN ± SEM for the total RNA isolated by this procedure from 50 μL samples of mouse whole blood was 7.7 ± 0.133 (n = 12), consistent with reproducible isolation of largely intact RNA.
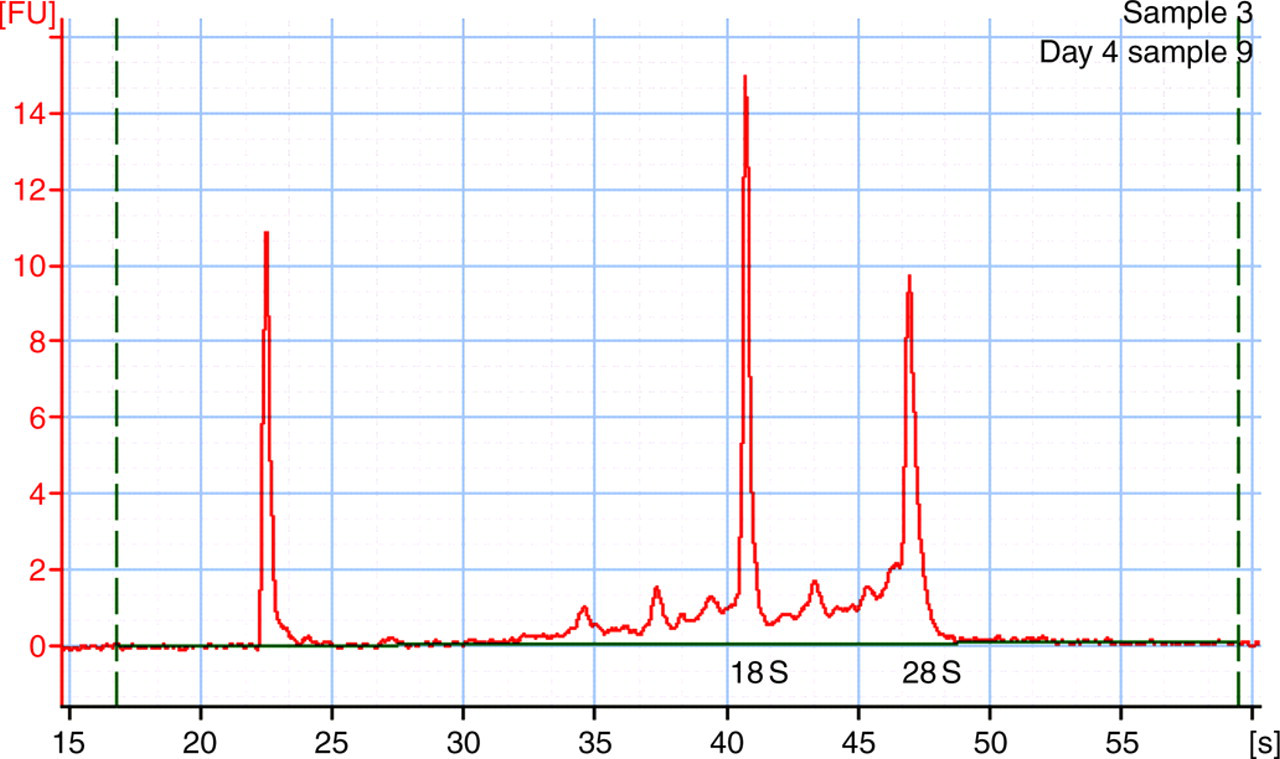
Typical microcapillary electropherogram of total RNA sample from 50 μL of mouse whole blood (RNA integrity number = 8.0). Positions of 18S and 28S rRNA are shown. The leftmost peak is the reference marker. RNA samples were analysed for overall integrity using an Agilent 2100 Bioanalyzer as described in Materials and Methods
The total RNA isolated by the microscale PAXgene™ procedure was perfectly functional, as shown in the typical realtime RT-PCR amplification plot for the invariant gene, GAPDH (Figure 3). The GAPDH expression level and variability as determined by cycle threshold (Ct) values measured among our samples are consistent with those reported using the PAXgene™ system to process 2.5 mL samples of human whole blood. 6 Measurement of gene expression in whole blood can provide biomarkers or surrogates for the transcriptional consequences of pharmacological, genetic or other treatments. To validate the utility of the microscale RNA isolation method, mice were injected subcutaneously with CoPP, a known inducer of Hmox1 in rodents, 7,8 and the level of expression of the Hmox1 gene in whole blood was examined at days 0, 3, 7 and 14 postinjection. Hmox1 expression in mouse whole blood was robustly upregulated three and seven days following injection with 20 mg/kg CoPP (Figure 4). In contrast, 2 mg/kg CoPP did not upregulate Hmox1 at any timepoint. Thus, the observed upregulation of Hmox1 by CoPP was both time- and dose-dependent.
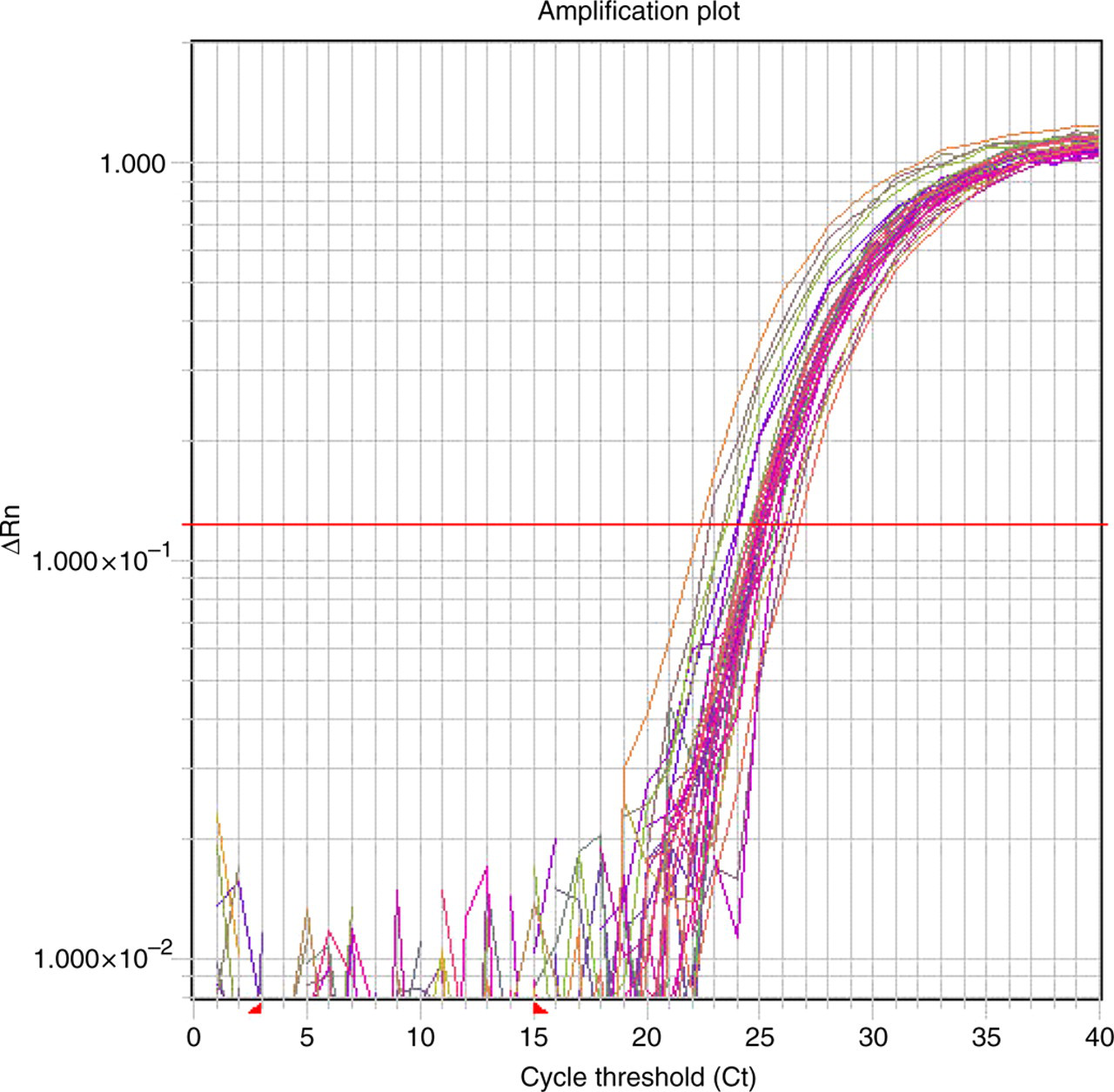
Typical realtime RT-PCR GAPDH amplification plot obtained from total RNA isolated from 50 μL samples of mouse whole blood. Variability in cycle thresholds (Cts) reflect different amounts of input RNA reverse-transcribed
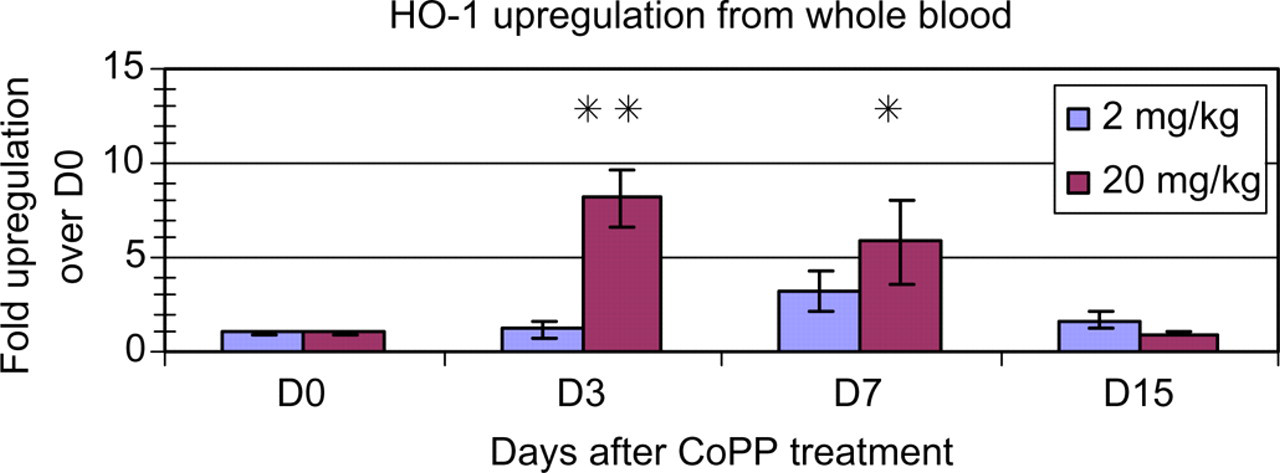
Total RNA isolated from mouse whole blood by the PAXgene™ method shows the expected dynamic and dose-dependent changes in haem oxygenase-1 (Hmox1) gene expression. Groups of mice were injected on day 0 with 2 or 20 mg/kg cobalt protoporphyrin (CoPP), and total RNA was isolated from 50 μL samples of whole blood taken from individual mice at days 0, 3, 7 and 15 postinjection. Hmox1 expression was measured by quantitative RT-PCR as described in Materials and Methods. (**P < 0.01, *P < 0.05) compared with day 0
In the experiment described in Figure 4, the average yield of total RNA from 50 μL of mouse whole blood was 2.284 ± 0.123 μg (n = 37) when quantitated using the Quant-iT™ RiboGreen® RNA Assay Kit (Invitrogen). Similar yields were calculated when RNA samples were quantitated by microcapillary electrophoresis in the Agilent 2100 Bioanalyser (1.710 ± 0.236 μg, n = 12).
Discussion
The PAXgene™ Blood RNA Kit is designed for the isolation of total RNA from a 2.5 mL sample of human whole blood. As part of ongoing drug discovery efforts, we have previously used this method to successfully isolate total RNA from 2.5 mL of whole blood from large preclinical species such as cynomolgous monkeys. However, the prevalent use of smaller preclinical species – particularly mice – in biomedical research necessitates a method for the isolation of total RNA from limiting amounts of whole blood. The current study demonstrates the utility of a modified PAXgene™ method that isolates surprisingly good yields (2–15 μg) of high-integrity (RIN = 7–9) total RNA from microlitre volumes (50–300 μL) of mouse whole blood. This range of total RNA can potentially allow multiple gene expression determinations to be made using array platforms such as the TaqMan® low-density array (TLDA), or Affymetrix Genechip® systems. 9–12 Some of our mouse total RNA samples had RINs lower than the range (6.95–9.30) of those reported from human whole blood using the standard PAXgene™ method. 13,14 Although an RIN >8 is preferred particularly for microarray-based profiling, the minimum RIN required for successful and reliable real-time RT-PCR is only 5. 14 The method works equally well for other small animals, such as rat and hamster (unpublished).
Various commercially available reagent systems that allow isolation of total RNA from larger volumes (1–2 mL) of whole blood have been described. 1,5,14,15 For smaller volumes, the SV total RNA isolation system (Promega, Madison, WI, USA) and the ZR whole blood total RNA Kit™ (Zymo Research, Orange, CA, USA) are designed for a blood volume of 200 μL, while the Mouse RiboPure™-Blood RNA Isolation Kit (Ambion, Applied Biosystems) is designed for 0.1–0.5 mL of mouse or rat whole blood. 16–19 The latter system employs an acidic phenol/chloroform extraction step, which is both inconvenient and limits throughput. Whether the Promega or the Zymo Research reagent systems can be scaled down to allow successful isolation of RNA from 50 μL whole blood remains to be determined.
Of note, our modified PAXgene™ method for isolation of total RNA from as little as 50 μL of whole blood can easily be adapted to a 96-well format when used in conjunction with the RNeasy® 96 Kit (Qiagen) (data not shown). More recently, the 96 Blood RNA Kit (Qiagen) was introduced to allow high-throughput isolation of total RNA from 2.5 mL of human whole blood collected into the same PAXgene™ RNA stabilizer solution as used in the method described herein. Therefore, it should be possible to combine the use of our microscale method with either the RNeasy® 96 or the 96 Blood RNA to further increase the throughput of total RNA isolation from as little as 50 μL of whole blood from small preclinical species. However, even without the use of 96-well filter plates, the current protocol clearly has utility for gene expression measurement from limiting volumes of whole blood that can be obtained from small, living preclinical species. As demonstrated in Figure 4, it was possible to demonstrate the temporal and dose-dependent upregulation of Hmox1 using RNA isolated from serial 50 μL of whole blood samples obtained from the retro-orbital sinus of mice, without any animal sacrifice.
The PAXgene™ system does not remove globin mRNA, which can constitute up to 70% of the mRNA mass in whole blood total RNA. The presence of large amounts of globin mRNA can reduce the sensitivity of array-based gene expression profiling. 5 Compared with the standard PAXgene™ method, total RNA isolated from whole blood using Tempus™ Blood RNA tubes (Applied Biosystems) has reduced globin-mRNA content and is suitable for microarray applications. 5 Alternatively, globin mRNA may be removed from RNA samples isolated by the PAXgene™ method by hybridization 20 using GLOBINclear™ reagent (Ambion). According to the manufacturer, GLOBINclear™ is capable of removing mouse, rat and human globin mRNAs, and therefore could be used downstream of our microscale PAXgene™ method for total RNA from these species, if so desired.
Blood is the most accessible tissue for biomarker discovery and analysis. The ability to measure gene expression in whole blood provides a convenient biomarker that can be correlated with disease states and therapeutic interventions. In drug discovery, small preclinical species such as mice are often used as models of human disease. The ability to measure gene expression in mouse whole blood therefore provides the potential to discover and/or monitor gene expression biomarkers in blood that may extrapolate to the clinic. In this paper, we demonstrate that the PAXgene™ system, which has been extensively used for isolation of total RNA from human whole blood, can be scaled down for the successful isolation of total RNA from much smaller volumes of mouse whole blood. The small volume required (such as the 50 μL we obtained by retro-orbital bleeding for the results shown in Figure 4) allows for serial sampling and obviates the need for animal sacrifice. As such, compared with studies that utilize different animals at each timepoint, the method may reduce the variability in gene expression results and also reduce the number of animals per study.
