Abstract
Summary
Increased incidence of headless sperms (HS) was spontaneously observed in the urine of adolescent naïve male SPF/VAF Crl:CD(SD) rats. To clarify the factors contributing to this event, the HS incidence in urine and the epididymis was periodically examined in conjunction with measurements of testis and epididymis weights, motility and morphology of sperms and testosterone, transferrin or follicle-stimulating hormone (FSH) concentrations in serum and/or the testis. The urinary HS incidence was 61%, 69%, 44%, 30%, 14%, 9% and 7% in 100 sperms counted at ages 8, 9, 10, 11, 12, 13 and 14 weeks, respectively; namely, HS peaked at 9 weeks, gradually decreased from 10 weeks and became almost a plateau from 12 weeks onwards. The epididymal HS incidence, which was lower than that in urine, peaked at 8 weeks, decreased from 10 weeks and became almost zero from 12 weeks. By scanning electron microscopy of HS in the epididymis, a narrow gap between the sperm head and neck was clearly seen along with the posterior ring. Concentrations of testicular testosterone and transferrin, a marker for Sertoli cell maturation, reached mature animal levels at 12 weeks. In contrast, no change in serum FSH concentration was seen throughout the study period. These results demonstrate that a marked increase in urinary HS incidence in naïve rats at ages 8–11 weeks would be a physiological phenomenon seen in connection with the process of Sertoli cell maturation.
Keywords
Many chemicals have been reported to affect sperm morphology in the epididymis of rats; they include adriamycin (Kato et al. 2001), ethyl methanesulphonate (Takagi et al. 2000), nitrobenzene (Yamamoto et al. 2000), boric acid (Linder et al. 1990), gossypol (Swan et al. 1990), methyl chloride (Working et al. 1985), 1-bromopropane (Ichihara et al. 2000) and tri-o-cresyl phosphate (Somkuti et al. 1991). Headless sperms (HS) are normally classified as one type of abnormal sperm morphology in a traditional toxicology study with rats (Lock & Soares 1980). Practically, HS are more commonly observed in the urine of control male rats at urinalysis performed during various toxicity studies. As a general rule, these abnormal sperms in urine have been recorded as existing, but their toxicological significance was not explored in detail. For a case in which urinary HS incidence increased, especially in test article-treated groups, extensive studies would be required. In humans, HS have been observed in ejaculated semen (Baccetti et al. 1989, Toyama et al. 2000), and are regarded as one of the causes of sterility (Panidis et al. 2001).
Rat spermatogenesis is a process from spermatogonia to mature sperm, and it takes about 58 days. Spermatogenesis is divided into proliferative, meiotic and spermiogenic phases. In the proliferative phase, stem cell spermatogonia undergo successive divisions to proliferative spermatogonia and differentiating spermatogonia. In the meiotic phase, the spermatocyte goes through preleptotene, leptotene, zygotene, pachytene and diplotene stages, and reaches the first meiotic division. The secondary spermatocyte is divided into spermatid by the second meiotic division. In the spermatogenic phase, round spermatid transforms into elongate spermatid, and finally develops to mature sperm (Russell et al. 1990).
To the best of our knowledge, the HS incidence in urine and the epididymis has not been reported in naïve Sprague-Dawley (SD) rats so far. In the present study, age-related changes in the incidence of urinary HS were first investigated in naïve male SPF/VAF Crl:CD(SD) rats at ages 8–14 weeks. Afterward, factors contributing to this event were studied with regard to the testis and epididymis weights, motility and morphology of sperms, and testosterone, transferrin and follicle-stimulating hormone (FSH) concentrations in serum and/or the testis.
Materials and methods
Animals
Naïve male SPF/VAF Crl:CD(SD) rats at ages 8–14 weeks were purchased from Charles River Japan Inc (Yokohama, Japan) and used for the present investigation after quarantine and acclimatization to the environment for three days. All animals were individually housed in metal cages with wire-mesh floors in an air-conditioned room with a 12/12 h light/dark cycle, temperature of 23 ± 2°C, relative humidity of 55 ± 20% and 15 air changes per hour. Laboratory chow (F2, Funabashi Farms, Funabashi, Japan) and chlorinated tap water were given ad libitum. Rats at ages 8–14 weeks were chosen for this study, since the sacrifice age in the initial exploratory screening of new chemical entities has been 8–11 weeks.
All experimental procedures were performed in accordance with the ‘Law concerning the Protection and Control of Animals’ (Law No. 105, 1 October 1973, partially revised on 22 December 1999 and on 22 June 2005) and ‘Standards Relating to the Care and Management, etc. of Experimental Animals’ (Notification No. 6 of The Prime Minister's Office, Japan, 27 March 1980, partially revised on 28 May 2002).
Headless sperms incidence in urine at aging
Urine was collected from 30 rats at ages 8, 9, 10, 11, 12, 13 and 14 weeks for a 4 h period (09:00–13:00 h) at ambient temperature once weekly using individual metabolic cages. Urinary sediments were prepared by centrifugation (550 ×
Epididymis examinations
To compare the HS incidence in urine and the epididymis of identical rats, 10 males each at ages 8, 10, 12 and 14 weeks were used based on the above results. Immediately after the urine collection under the same experimental conditions as the aforementioned study, about 5 mL of blood was withdrawn from the interior vena cava of each animal under ether anaesthesia for the assay of FSH described in the following section of Testis examinations. Then, the animals were euthanized by exsanguination, and the epididymis was removed. The right caudal epididymis was weighed and shredded in 5 mL of HANKS′ solution (Nissui Pharmaceutical Co, Ltd, Tokyo, Japan), and the sperm suspension was prepared by holding the small tissue pieces at 37°C for 5 min to help epididymal sperms migrate into the solution.
A part of the sperm suspension was placed on a glass slide and mixed with 10% eosin Y to prepare a smear. The HS incidence was calculated from the number of HS per 100 sperms randomly counted in the smear under the microscope. Sperm motility in the suspension was measured with a sperm quality analyser (Medical Electronic System Ltd, Migdal Haemek, Israel) and the value obtained was expressed as the sperm motility index (SMI).
To examine the direct involvement of urine components in the HS incidence, each urine sample obtained from rats at ages 8, 10, 12 and 14 weeks was measured for volume (manual method), osmolality (Advanced Cryomatic Osmometer, Model 3C2, Advanced Instruments Inc, MA, USA) and pH (N-Multistix SG-L and Clinitek 200 +, Bayer Corporation, PA, USA). For the in vitro study, moreover, an aliquot (0.05 mL) of the epididymal sperm suspension from rats at 8 weeks was incubated (preserved) in 0.45 mL of urine supernatant of both rats at ages 8 and 14 weeks for 24 h at room temperature. The incidence of HS was calculated both before and after preservation.
To estimate the sperm morphology in detail, the sperm suspensions obtained from rats at ages 8, 10, 12 and 14 weeks were mixed with an equal volume of 2% glutaraldehyde in 0.1 mmol/L phosphate buffer solution. One drop of the mixture was placed on a poly-L-lysine-coated glass slide, washed with 8.2% sucrose buffer solution and fixed with 1% buffered osmic acid. After washing with the sucrose buffer, the specimens were stained with 1% tannic acid, washed and fixed once again. They were dehydrated with a graded ethanol series, freeze-dried, coated with palladium platinum, and observed under a scanning electron microscope (S-800, Hitachi Ltd, Tokyo, Japan).
Testis examinations
Following the removal of the epididymis from 10 males each at ages 8, 10, 12 and 14 weeks, the testis was excised and weighed. After dissection of the tunica albuginea, the right testis was placed in a test tube, distilled water was added up to a final volume of 12 mL, and then the tissue was dispersed for 10 s with a dispersion mixer (IKA-Labortechnik, Staufen, Germany) and sonicated for 3 min with a sonicator (Branson, CT, USA) for preparation of the sperm suspension.
The sperm head count (SHC), an index for the total number of sperms in all stages of spermatogenesis, was conducted according to the previously reported procedure (Meistrich 1989). An aliquot of the sperm suspension was placed in a Makler counting chamber (Sefi-Medical Instruments, Haifa, Israel) and sperm heads were counted under a phase contrast microscope (NIKON, Tokyo, Japan). The count of sperm heads in 100 squares of the field was multiplied by 1,200,000 to give the total sperm number in the testis.
A part of the testicular suspension prepared for SHC was centrifuged at 1200 ×
Statistical analysis
The data are expressed as the group mean ± standard deviation, and differences in the mean values between rats at 14 weeks and the other ages were statistically analysed. If the F-test indicated the variance to be homogeneous, the Student's t-test was conducted. If the F-test indicated the variance to be heterogeneous, the Aspin-Welch t-test was conducted. A P value of less than 5% was considered to be significant.
Results
Headless sperms incidence in urine at aging
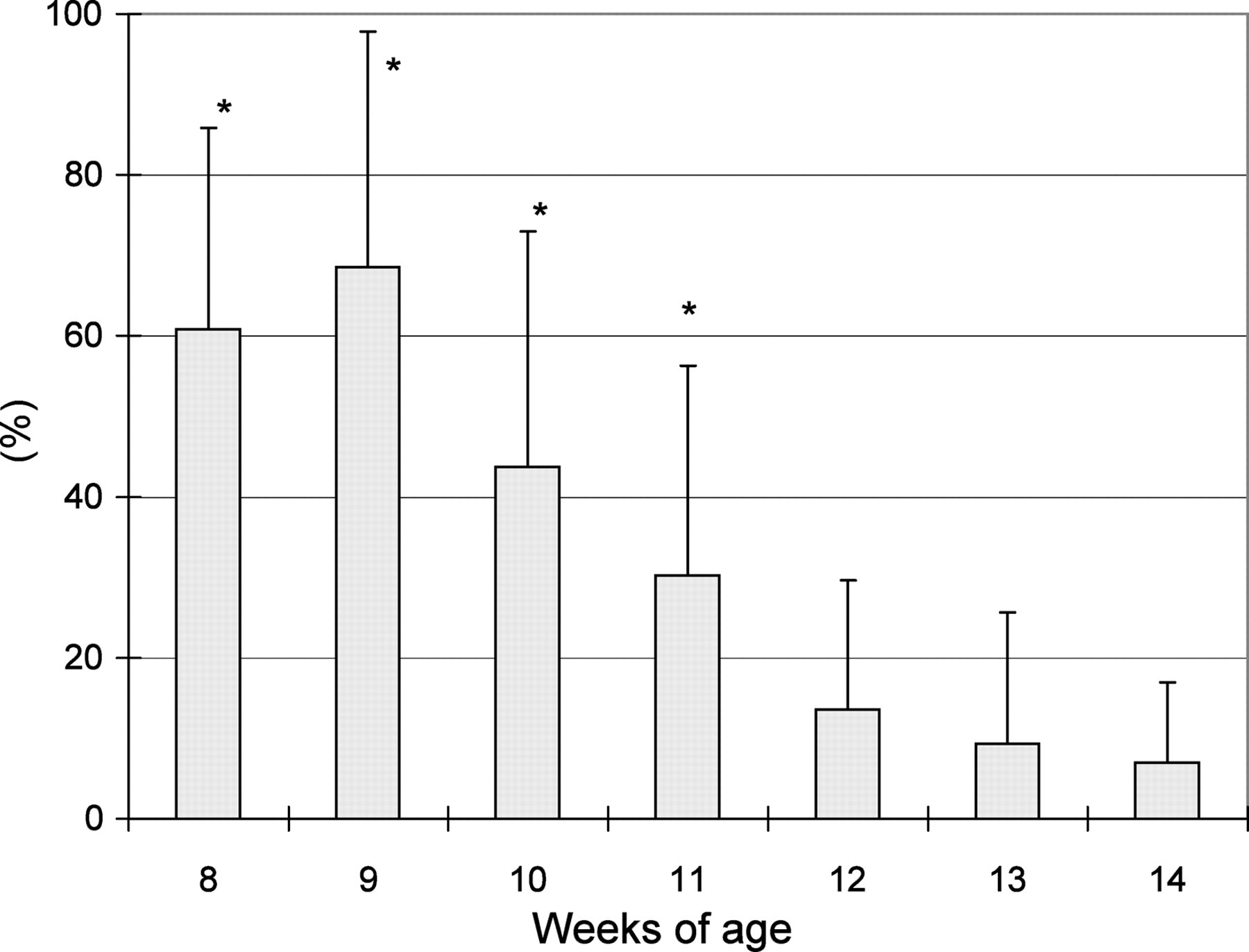
The presence of sperm in urine was observed in 12 (40%), 17 (57%), 25 (83%), 17 (57%), 23 (77%), 23 (77%) and 25 (83%) of 30 rats at ages 8, 9, 10, 11, 12, 13 and 14 weeks, respectively. HS were included in all rat urine except for one rat at 14 weeks. The HS incidence was 61 ± 25% (n = 12), 69 ± 29% (n = 17), 44 ± 29% (n = 25) and 30 ± 26% (n = 17) at ages 8, 9, 10 and 11 weeks (adolescent animals), respectively, and 14 ± 16% (n = 23), 9 ± 16% (n = 23) and 7 ± 10% (n = 24) at ages 12, 13 and 14 weeks (matured animals), respectively. The HS incidence at ages 8–11 weeks was significantly different from that at 14 weeks (Figure 1). Thus, the urinary HS incidence was markedly high at 9 weeks and almost a plateau from 12 weeks onward. However, sexual behaviour that would affect the sperm population in the epididymis was not seen throughout the experimental period.
Epididymis examinations
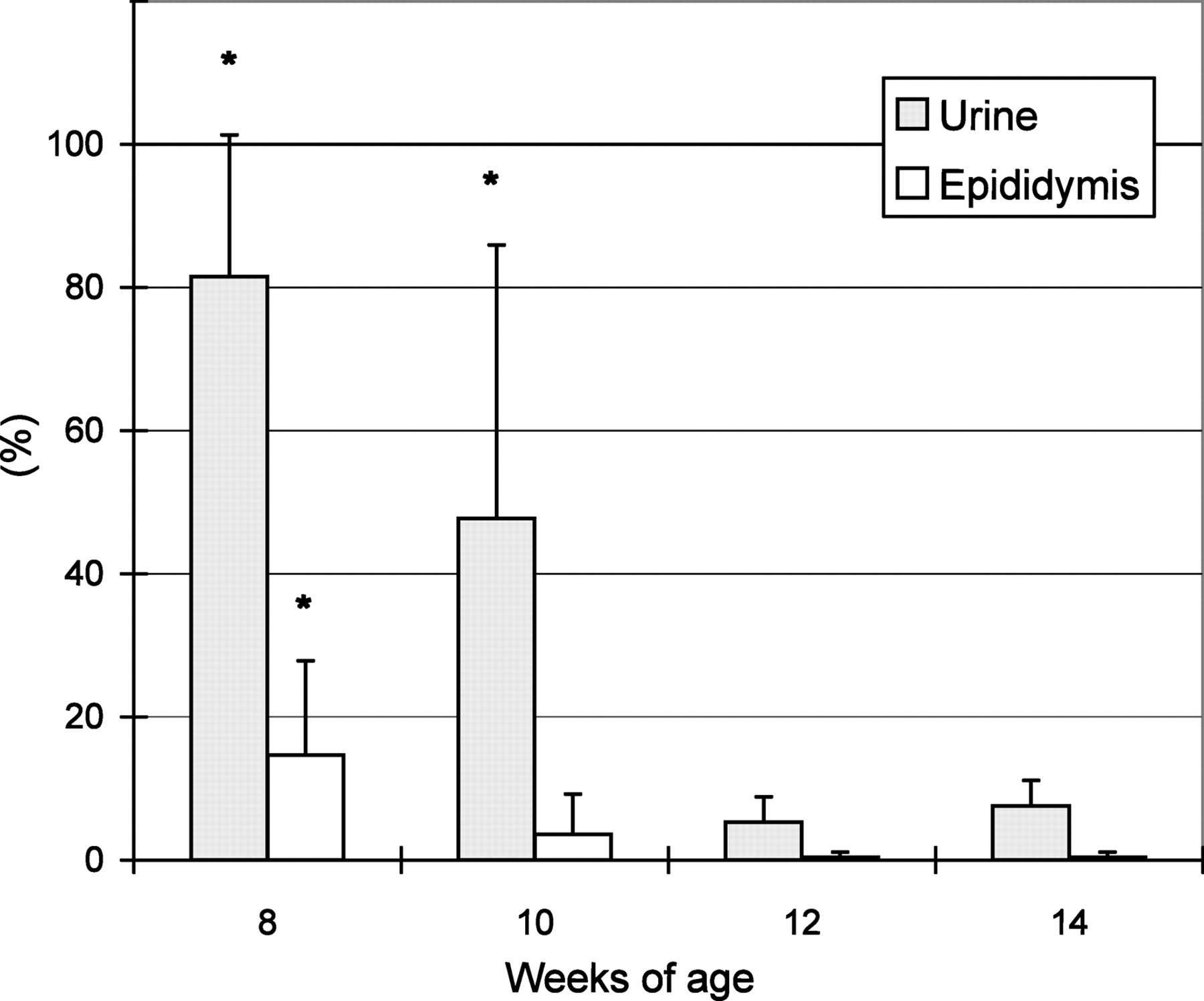
The epididymal HS incidence was 14.7 ± 13.1%, 3.6 ± 5.6%, 0.4 ± 0.7% and 0.4 ± 0.7% at ages 8, 10, 12 and 14 weeks, respectively. This HS incidence at 8 weeks was significantly higher than that at 14 weeks. As compared with the urinary HS incidence, those values in the epididymis were 5 to 20 times lower (Figure 2).
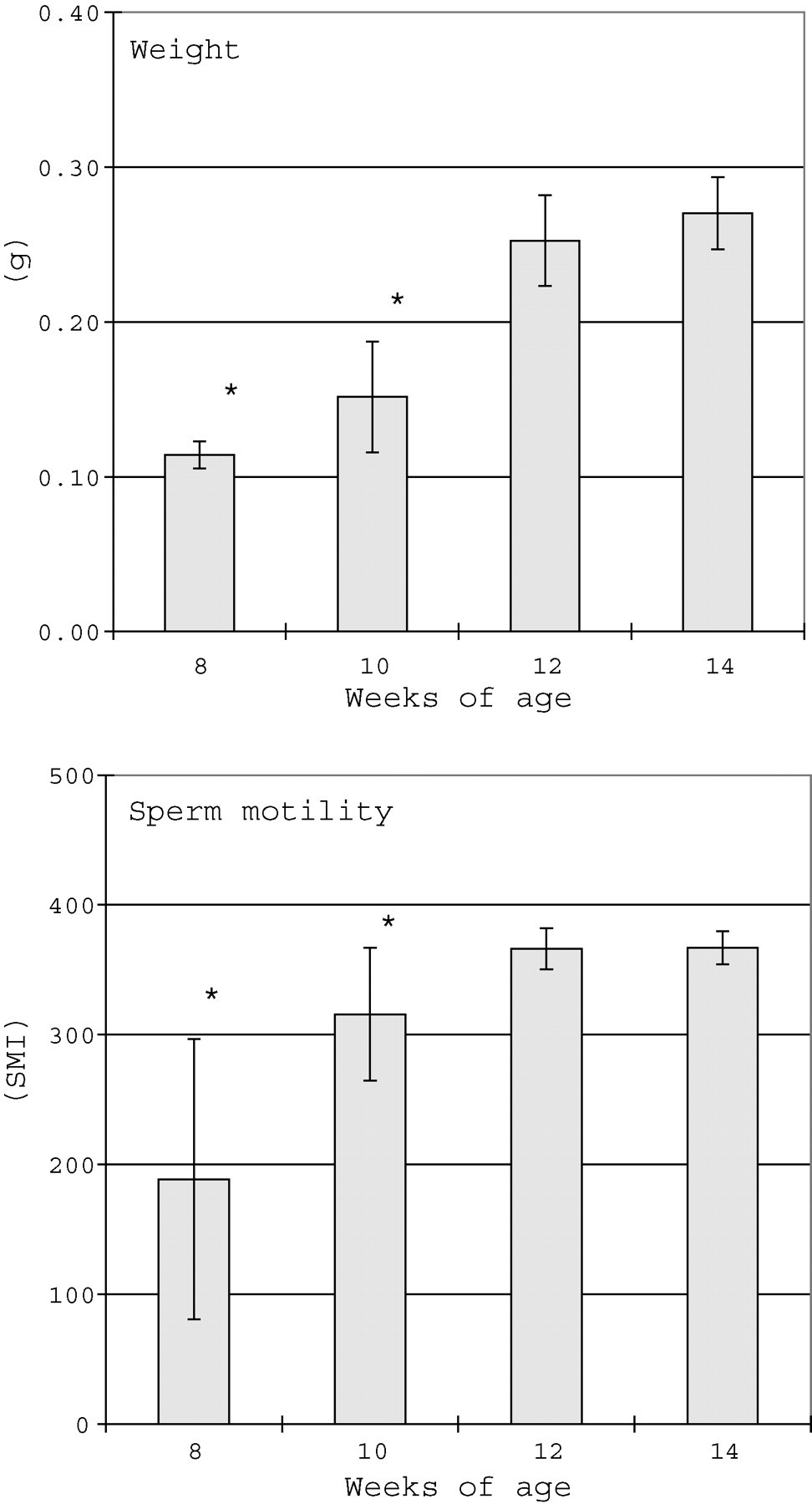
Both the weight and sperm motility of the caudal epididymis at ages 8 and 10 weeks were significantly lower than those at 14 weeks, but no difference in values was seen between 12 and 14 weeks (Figure 3).
Urinary volume had a tendency toward a gradual increase at ages 12 and 14 weeks, but no changes in osmotic pressure and pH were noted throughout the study period (Table 1).
The values represent the group mean ± standard deviation. *P < 0.05 significant difference from rats at 14 weeks
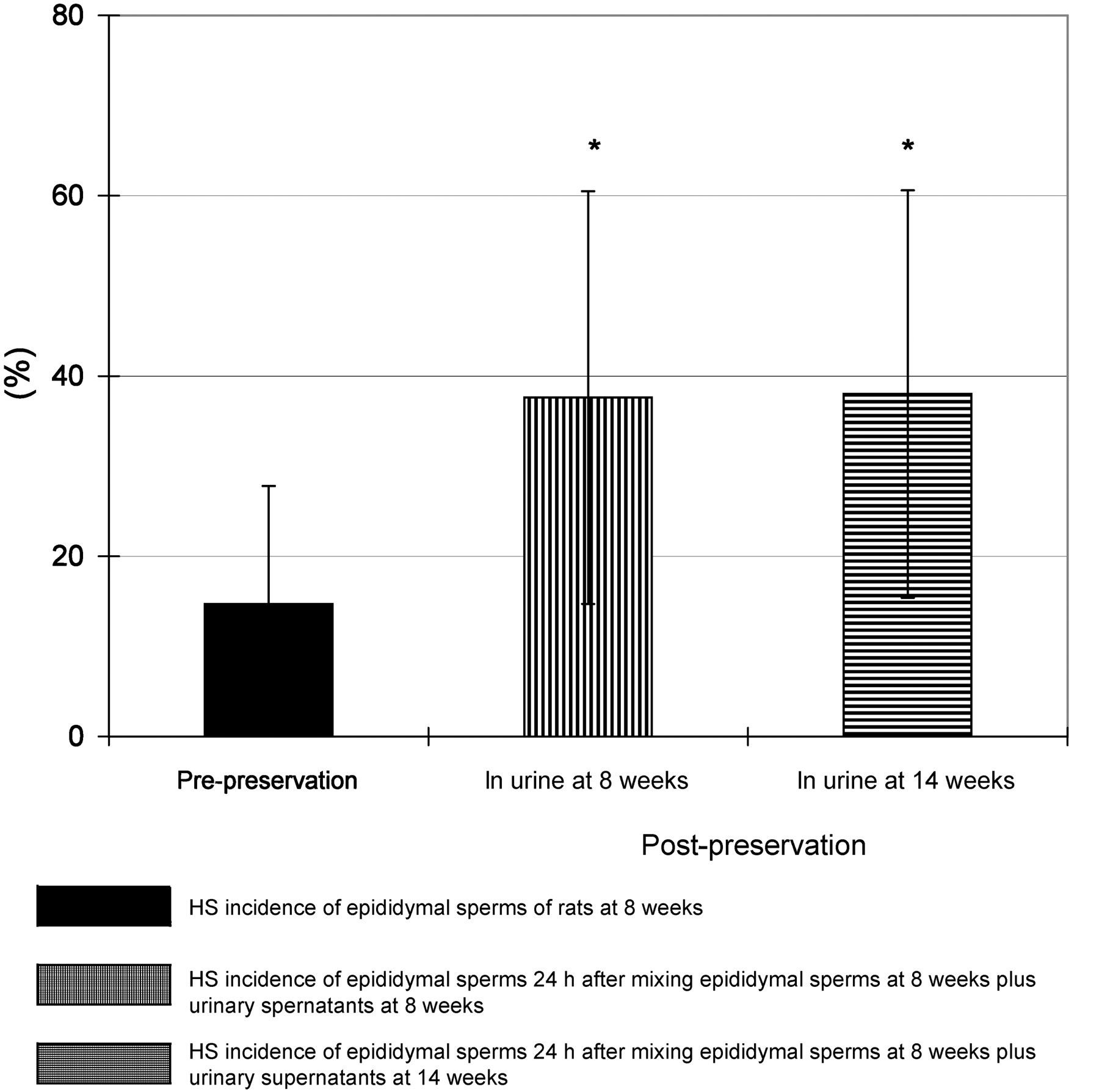
In epididymal sperms placed in urinary supernatant obtained from rats at the same or different ages for 24 h in the in vitro system (Figure 4), the HS incidence at post-preservation increased to 38% over that (15%) at pre-preservation. In brief, approximately 23% of sperm with normal morphology became HS following preservation in the urine supernatant of both ages 8 and 14 weeks.
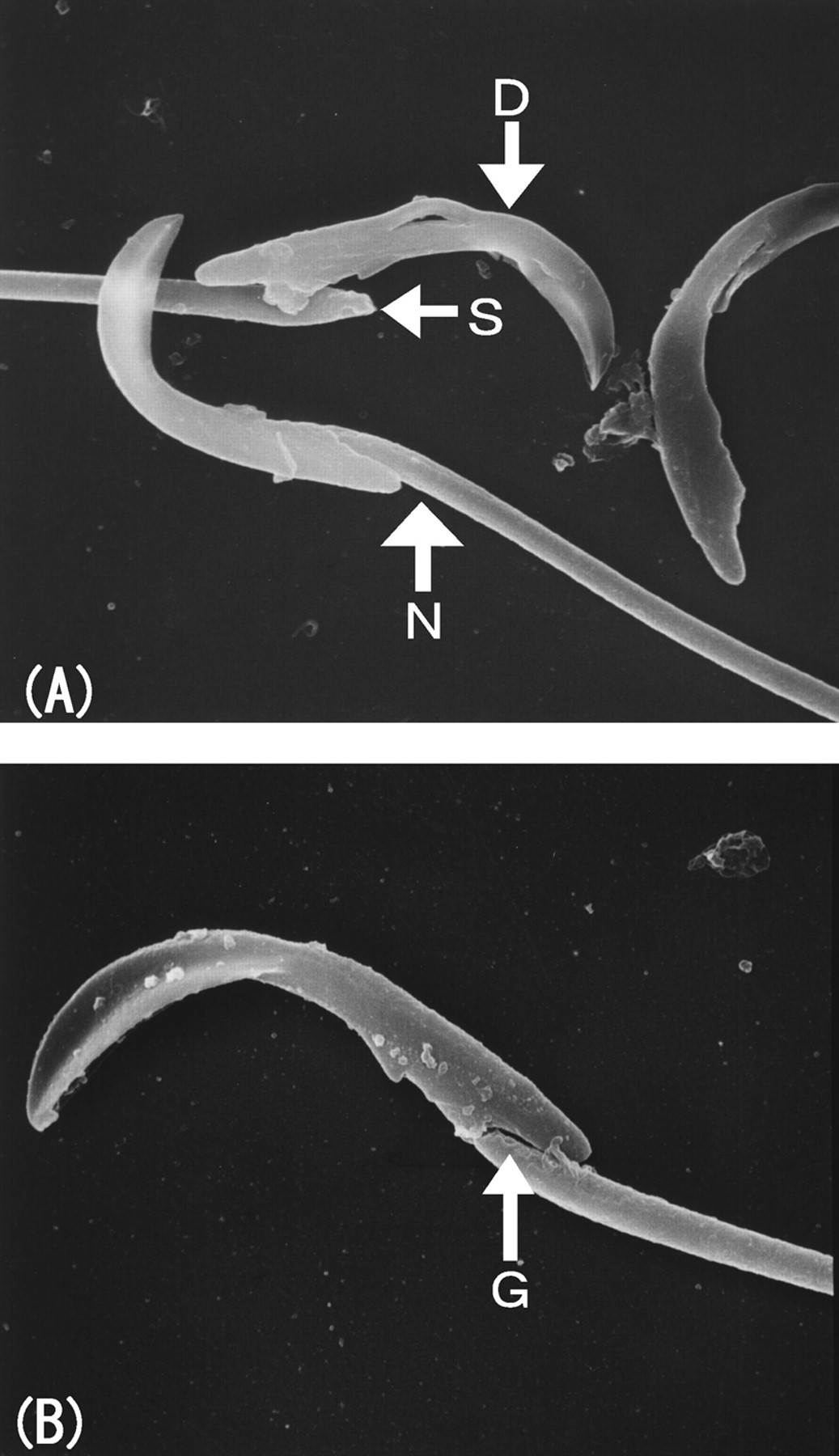
In scanning electron microscopic findings of epididymal sperms (Figure 5), sperms with normal morphology had tightly connected head and tail, whereas abnormal sperms showed a narrow gap between the head and neck along with the posterior ring, and a moderately or markedly widened angle between the neck and posterior end of the head. Further, detached heads were occasionally attached to a structure appearing as part of the neck.
Testis examinations
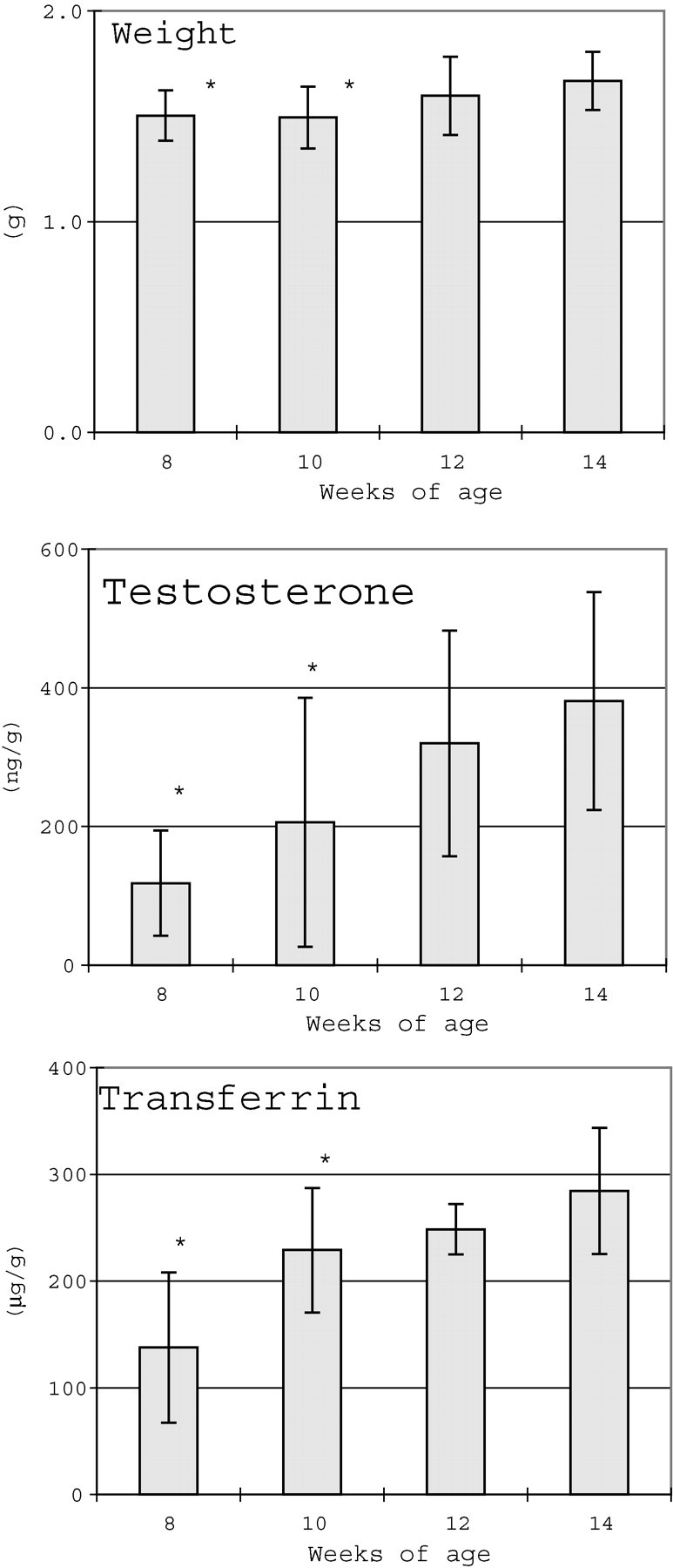
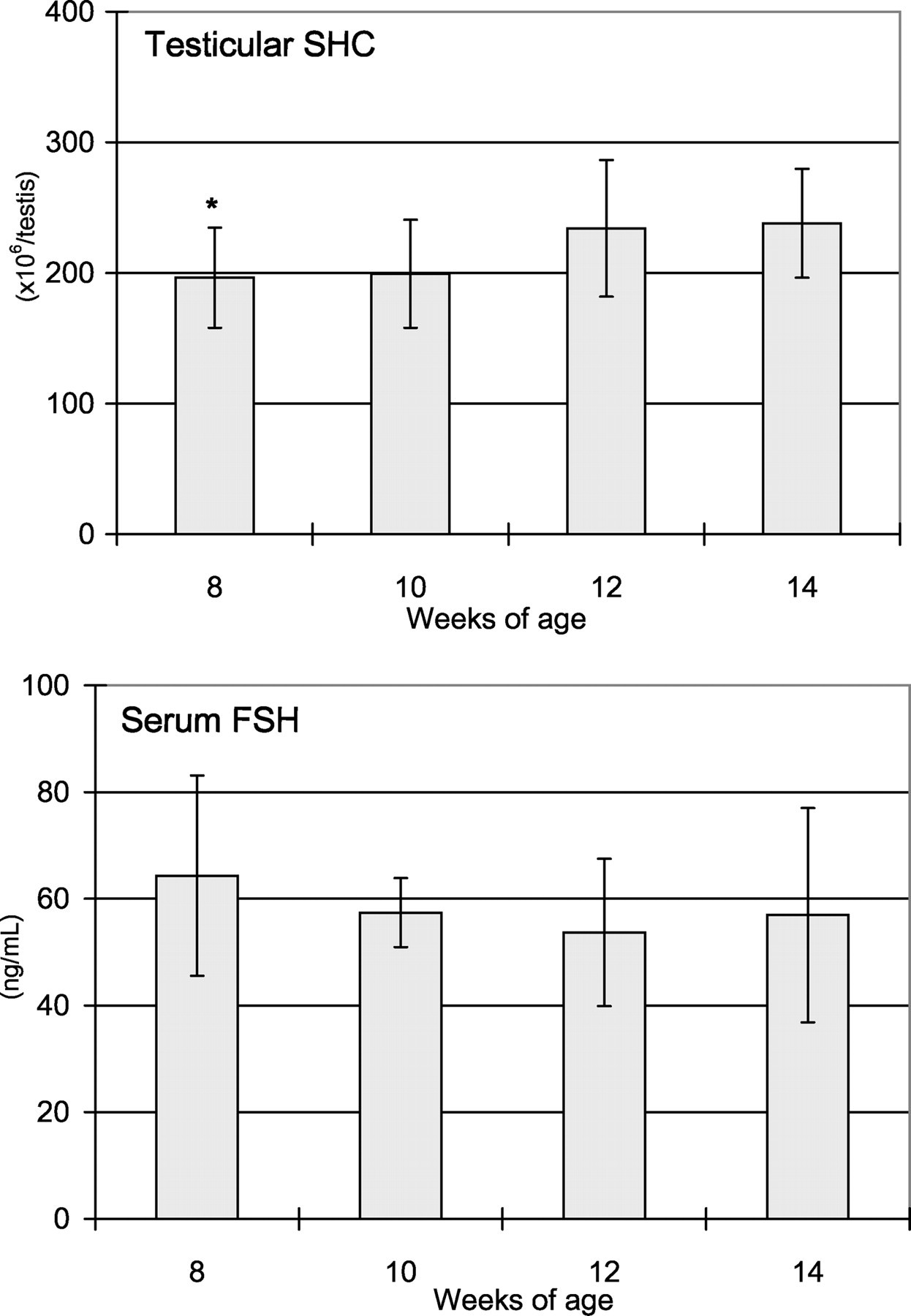
Testicular weights and testosterone, and transferrin concentrations at ages 8 and 10 weeks were significantly lower than those at 14 weeks (Figure 6). The SHC at 8 weeks was also lower than that at 14 weeks; however, no changes in serum FSH were noted throughout the study period (Figure 7).
Discussion
The present study demonstrated a relatively high incidence (40–83%) of urinary sperms accompanied by an age-related occurrence of HS in urine of naïve rats at ages 8–14 weeks. Interestingly, the urinary HS incidence (30–69%) at ages 8–11 weeks was evidently higher than that (7%) at 14 weeks. The HS incidence peaked at 9 weeks, gradually decreased from 10 weeks, and became almost a plateau from 12 weeks onwards. Likewise, the epididymal HS incidence among the animals tested peaked at 8 weeks, decreased from 10 weeks, and reached about zero from 12 weeks onwards. The HS incidence in urine, however, was 5–20 times higher than that in the epididymis, suggesting that sperms with normal morphology in the epididymis would become HS after excretion from the epididymis. To clarify a contributing factor to the appearance of urinary HS, the physico-chemical effect of urine on sperms was investigated. Urinary volume showed a tendency toward an increase at ages 12 and 14 weeks, presumably resulting from body weight gain with age, but no changes in osmotic pressure or pH were seen. When sperm prepared from the epididymis of rats at 8 weeks was preserved for 24 h in the urine supernatant from rats at ages 8 and 14 weeks to see its vulnerability in urine, the HS incidence obtained at post-preservation was obviously increased relative to that at prepreservation, but there was no difference in HS between urine from rats at 8 weeks versus 14 weeks. In a further study, although the epididymal sperm from rats at 14 weeks was preserved in the urine supernatant from rats at ages 8 and 14 weeks, no overt data were obtained because of an extremely lower epididymal HS incidence in rats at 14 weeks (data not shown). Based on these results, it was suggested that sperms of adolescent rats at 8 weeks or so were fragile, and their heads were easily detached in urine. Additionally, since the HS incidence was not affected by urinary composition or its preservative conditions, evidence obtained in our work also demonstrated the vulnerability of sperms passing through the vas deferens and urethra, rather than the effects of increased contamination (bacteria) in urine. However, consideration should be given that spermatozoa in urine are represented as aged sperm being shed from the epididymal reservoir while epididymal sperm generated after suspension of shredded epididymis would be a mixture of sperm populations of different ages.
By scanning electron microscopy, a narrow gap was observed between the sperm head and neck along the posterior ring in an epididymal abnormal sperm, and this was considered to be an early change in detachment of the head. This finding was thought to be followed by a widening of the angle between the neck and posterior end of the head, explaining the process of head detachment.
The relationship was investigated between the urinary HS incidence and changes in associated hormonal parameters for the epididymis and testis in terms of sexual maturation. The HS incidence in urine and the epididymis, epididymal weights and increased epididymal sperm motility were almost stable at 12 weeks. The increased motility at this age was well consistent with the morphological features, including developed tight connection of the sperm head and neck to prevent detachment. Similarly, the testicular weight and testosterone concentration reached a plateau at 12 weeks, suggesting sexual maturation. In the other study, when the histopathological examination in the testis was conducted in naïve rats at 8 weeks or older, no difference in the morphology between 8 and 14 weeks was observed (in-house data). Consequently, the HS incidence in urine and the epididymis was shown to decrease in parallel with sexual maturation.
The blood–testis barrier has been recognized to be formed by the tight-junction of Sertoli cells and germ cells (Dym & Fawcett 1970). During spermatogenesis in the testis, Sertoli cells keep contact with germ cells after the preleptotene stage, and secrete androgen-binding protein (ABP). However, FSH stimulates Sertoli cells to produce cAMP and secretes ABP into the seminiferous tubules, and eventually ABP binding testosterone maintains spermatogenesis (Hagenas et al. 1975). Sertoli cells also contribute to germ cell development by the secretion of transferrin, an iron transporter (Skinner et al. 1984), ceruloplasmine-like protein (Skinner & Griswold 1983) and plasminogen activator (Lacroix et al. 1977), together with a supply of pyruvic acid and lactic acid to germ cells as an energy source (Jutte et al. 1983). Therefore, the testis transferrin concentration has been reported as a marker for Sertoli cell maturation (Anthony et al. 1991). In the present study, the testis transferrin concentration reached mature animal levels (249 µg/g) at 12 weeks, indicating maturation of Sertoli cells at this age. Since testicular sperms received sufficient proteins, pyruvic acid and lactic acid originating from the mature Sertoli cells at 12 weeks, they were considered not to become HS. This notion could be supported by the fact that HS decreased to about 10% and almost zero in urine and the epididymis, respectively, at 12 weeks or older, and by the report that the epididymal HS increased in testicular toxicant-injured Sertoli cells (Somkuti et al. 1991, Yamamoto et al. 1999). In conventional toxicity studies with rats, when maturation was delayed due to ill-condition and decreased body weights, HS incidence may be increased only in the treatment groups, especially at an age of around 12 weeks. In these cases, it is crucial to discriminate Sertoli cell injuries from growth retardation, taking into consideration the basal HS fluctuations with age.
In conclusion, a marked increase in urinary HS incidence in naïve rats at ages 8–11 weeks would be a physiological phenomenon seen in connection with the process of Sertoli cell maturation.
